Abstract
Kawasaki disease is a febrile disease of childhood that is associated with increased inflammatory cytokines and immunoregulatory abnormalities. While the serum concentrations of soluble IL-2 receptor can change under such pathologies, the relevance of the soluble IL-2 receptor concentration in patients with Kawasaki disease has not been specified. We aimed to summarize the existing studies that reported the soluble IL-2 receptor concentrations in patients with Kawasaki disease. Original articles that were published up to July 2016 were collected using a PubMed/Medline-based search engine. A total of nine articles that reported the serum soluble IL-2 receptor concentrations in acute-phase Kawasaki disease were eligible. All of the articles described a high soluble IL-2 receptor concentration in patients with Kawasaki disease relative to the level of controls or the reference range. Two of five articles on patients with coronary artery aneurysms described a significantly higher soluble IL-2 receptor concentration in patients with coronary artery aneurysms than patients without. Two articles on patients with intravenous immunoglobulin therapy described a significant decrease of the soluble IL-2 receptor concentration after the therapy. Accordingly, the serum soluble IL-2 receptor can be a potent marker of disease activity and therapeutic effects in patients with Kawasaki disease; further studies are thus warranted for its use in the clinical setting.
Keywords
Introduction
Kawasaki disease (KD) is a febrile disease of childhood, which predominantly occurs in males and is sometimes complicated by coronary lesions. 1 The incidence of KD, which is more commonly seen in Asian countries than in other countries, is increasing.2,3 Although the causes of KD have not been fully elucidated, organism infection is thought to be a cause of the disease. 4 With immunoregulatory disorders, inflammatory conditions with the enhancement of cytokine molecules, such as interleukin (IL)-1, IL-6, and tumor necrosis factor (TNF)-α, accompany the disease.4–6
Of note, IL-2 is one of the inflammatory cytokines. While immunoregulatory disorders, including the activation of T-cells, with the enhancement of cytokines arise in patients with KD, 7 the activation of T cells leads to appearance of IL-2 receptors on the cell surface, and then its α-chain (CD25), an IL-2 receptor component, is released into circulation by protease.8,9 A fragment of CD25 is known as soluble IL-2 receptor (sIL-2R).8,9 Although the measurement of IL-2 itself is difficult due to its short-term behaviour, the concentration of sIL-2R can be measured in blood samples.
Currently, the serum sIL-2R is often used as a marker for diagnosis and treatment of malignant lymphoma in daily practice. 9 In paediatric patients, the sIL-2R concentration is also included in the diagnostic guidelines for haemophagocytic lymphohistiocytosis. 10 Basically, sIL-2R is a non-specific marker. Can the serum concentration be useful for considering the disease states of patients with KD? The present study aimed to summarize the existing studies that reported on the sIL-2R concentrations of KD patients in order to characterize the utility of sIL-2R as a marker in KD.
Methods
We used a PubMed/Medline-based search engine to perform a search of the relevant literature. The following search formula was used: soluble [All Fields] AND (‘receptors, interleukin-2’ [MeSH Terms] OR ‘receptors’ [All Fields] AND ‘interleukin-2’ [All Fields]) OR ‘interleukin-2 receptors’ [All Fields] OR ‘interleukin 2 receptor’ [All Fields] OR ‘interleukin-2 receptor alpha subunit’ [MeSH Terms]) OR (‘interleukin-2’ [All Fields] AND ‘receptor’ [All Fields] AND ‘alpha’ [All Fields] AND ‘subunit’ [All Fields]) OR ‘interleukin-2 receptor alpha subunit’ [All Fields]) AND (‘mucocutaneous lymph node syndrome’ [MeSH Terms] OR ‘mucocutaneous’ [All Fields] AND ‘lymph’ [All Fields] AND ‘node’ [All Fields] AND ‘syndrome’ [All Fields]) OR ‘mucocutaneous lymph node syndrome’ [All Fields]) OR (‘Kawasaki’ [All Fields] AND ‘disease’ [All Fields]) OR ‘Kawasaki disease’ [All Fields]). Original articles, which were published up to July 2016 and were written in English, were included in our search.
The search yielded a total of 15 articles. After our review of their contents, six articles were omitted because four articles were simply written by an author with the name ‘Kawasaki’ (not related to research on KD) and two articles were omitted because they did not focus on the sIL-2R concentration (e.g. cutaneous findings 11 and superantigens in relation to T-cell activation 12 ).
Results
The serum sIL-2R concentrations in patients with Kawasaki disease.
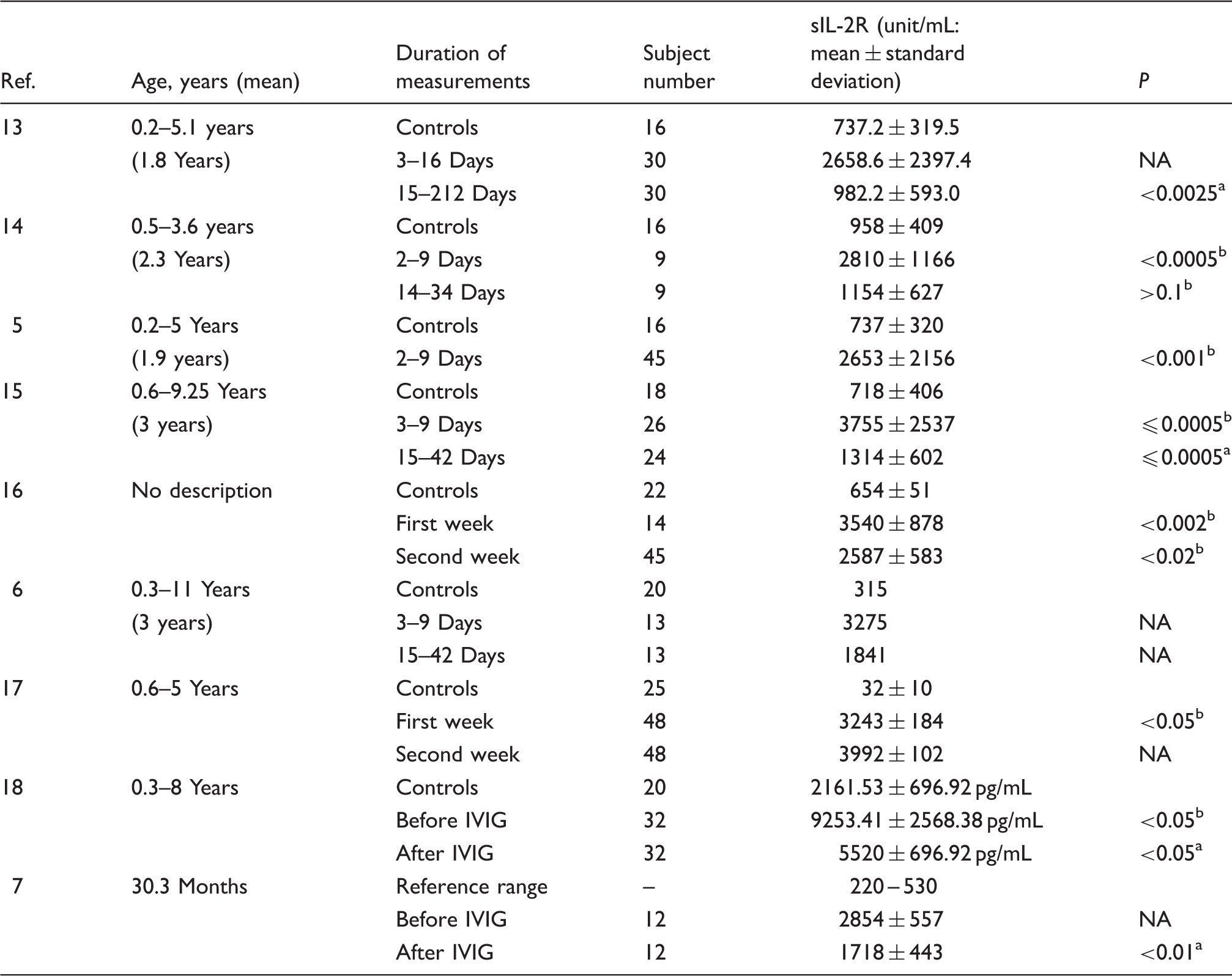
NA: not available, IVIG: intravenous immunoglobulin.
Comparison with acute phase.
Comparison with controls.
Almost of the articles described the serum sIL-2R concentrations in both controls and patients with acute-phase KD, while one article 7 described the reference range instead of the level of controls. The sIL-2R concentrations of controls ranged from 32 to 958 unit/mL, while those of patients with KD ranged from 2653 to 3755 unit/mL (in one article, 16 the used unit to measure sIL-2R differed from that of other articles). All of the articles showed that the sIL-2R concentrations in patients with KD were high relative to those in controls5,6,13–18 and the reference range, 7 while statistical analysis comparing between patients with KD and without was not necessarily carried out in one article. 6
The serum sIL-2R concentrations and CCAs.
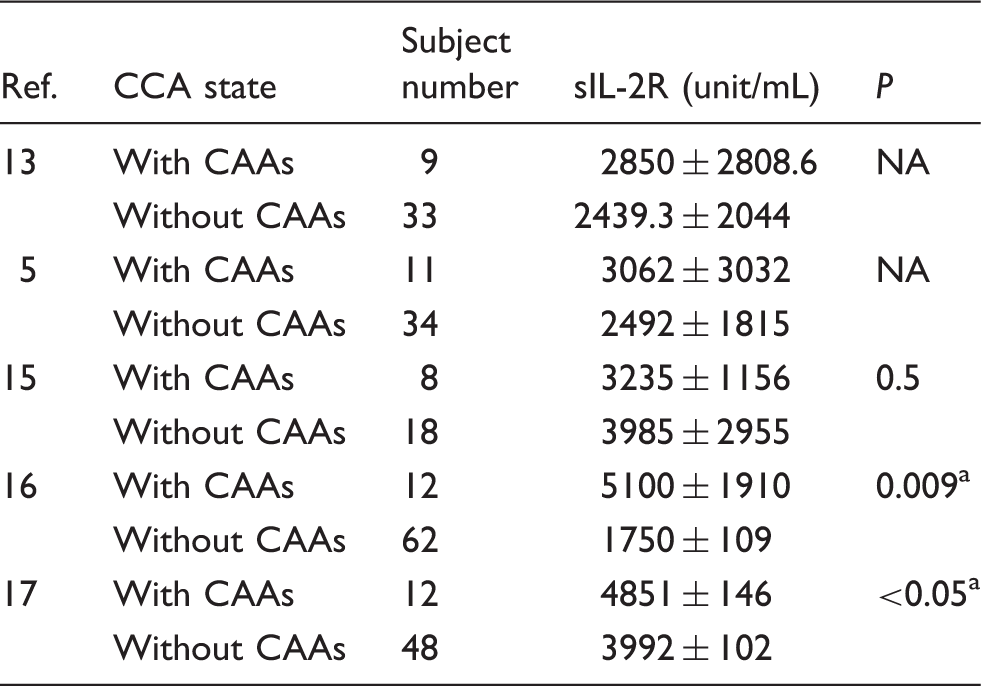
CCAs: coronary artery aneurysms, NA: not available.
Comparison between patients with and without CAAs.
Three articles reported the sIL-2R concentrations in the acute and convalescent phases of KD13,14,17 and two articles showed that the level was significantly higher in the acute phase.13,17 Furthermore, two articles (one cross-sectional study 7 and one interventional study 16 ) compared the sIL-2R concentrations in patients before and after intravenous immunoglobulin (IVIG) therapy.7,16 The sIL-2R concentration was found to be significantly decreased after therapy.7,16 On the other hand, the concentration after IVIG therapy remained significantly higher than that in controls, 16 and the concentration in IVIG-resistant patients (15 of 27 patients) was reported to be significantly higher than that in the patients who responded to IVIG. 7
Discussion
The serum sIL-2R concentration was found to be 3–100 times higher in patients with KD than in controls. While it was only suggested that the sIL-2R concentration in patients with CAAs might be high relative to patients without CAAs, the sIL-2R concentration could be reduced after IVIG therapy. Although the sIL-2R concentrations in patients with KD remain to be characterized, the serum sIL-2R concentration may be a potent marker of disease activity and therapeutic effects in these patients.
By activating T cells, CD25 (α-chain) is overexpressed in the IL-2 receptors and is released into circulation as sIL-2R.8,9 Blood T cells are activated in acute-phase KD7,12; thus, the serum sIL-2R concentration can be increased. In addition, the activation of T-cells in KD is assumed to be related to coronary artery damage.7,12 As this review did not consistently demonstrate that a high sIL-2R concentration might predict CAAs in patients with KD, the predictive value of sIL-2R on CCA formation should be explored in future studies.
Currently, IVIG therapy is used to treat patients with KD. 4 The IVIG therapy inhibits both the activation of nuclear factor kappa B (NF-κB) and the release of cytokine molecules, including IL-2 (which are induced by CD14+ monocytes/macrophages and CD3+ T cells), in patients with KD. 19 Thus, the therapeutic mechanism may partly explain the decrease of the sIL-2R concentration that is observed in patients who were treated with IVIG in our review.7,16 On the other hand, a still-high sIL-2R concentration was observed after therapy and/or in IVIG-resistance.7,16 The reason for this remains a topic for future research. Recently, cyclosporine, infliximab and prednisolone have been used to treat IVIG-resistant patients.20–24 Cyclosporine inhibits the dephosphorylation of nuclear factor of activated T cells and inhibits the release of cytokine molecules, especially IL-2, from activated T cells.20–24 It should be determined whether recently developed therapies reduce the sIL-2R concentrations.
This study is associated with several limitations. The number of articles reviewed was limited, and the number of patients who were enrolled in the respective articles was small. The assays for measuring the serum sIL-2R concentrations could not be completely unified. 16 Additionally, sIL-2R is reported to be present not only in the blood but also in the synovial/cerebrospinal fluid and urine.25,26 Understanding the behaviour of sIL-2R in other specimens, in relation to serum sIL-2R, could yield information on the pathophysiology of KD and related complications, such as systemic vasculitis. Prospective long-term follow-up studies and interventional therapeutic studies to investigate the coronary outcomes, using the sIL-2R concentration, must be conducted. Further studies are thus required to draw any definitive conclusions.
Conclusions
The present study showed that the serum concentration of sIL-2R in patients with KD was high in comparison to controls. While it was only suggested that a high sIL-2R concentration might be seen in patients with CAAs, the sIL-2R concentration could be reduced after IVIG therapy. The serum sIL-2R may be a potent marker reflective of disease activity and therapeutic effects in patients with KD. Further studies should be performed for the use of sIL-2R as a marker of KD.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by Jichi Medical University.
Ethical approval
Not applicable.
Guarantor
KK.
Contributorship
HT participated in the design of the study, searched the literature, and wrote the draft of the manuscript. KK, TM, and OS participated in the design of the study and wrote the manuscript. TT and EK interpreted the findings and reviewed the manuscript. All authors approved the final version of the manuscript.
