Abstract
Background
Compelling evidence indicate that traumatic brain injury is highly related to accelerated bone fracture repair, but the underlying mechanism still remains elusive. Fracture repair process relies greatly on the formation of new blood vessels in fracture site, and angiogenic factors have been confirmed to be essential for the initiation and maintenance of the fracture healing. Hypoxia-inducible factor-1α was demonstrated to be a critical regulator of angiogenic–osteogenic coupling during bone development and regeneration. The aim of the present study was to investigate the local and circulating concentrations of hypoxia-inducible factor-1α in patients with long-bone fractures and concomitant traumatic brain injury and to determine the potential role of hypoxia-inducible factor-1α in fracture healing.
Methods
Twenty-five patients with a long-bone fracture and concomitant traumatic brain injury (FT group) and 33 without a brain injury (Fr group) were enrolled in this study. Healthy subjects donated serum samples as control. Serum samples were collected over a period of six months, following a standardized time schedule. Hypoxia-inducible factor-1α concentrations were measured in fracture haematoma and serum of patients in both groups using enzyme-linked immunosorbent assay.
Results
Patients in FT group had a short time to union. Serum hypoxia-inducible factor-1α concentrations elevated in the early healing period and reached the maximum level during intramembranous bone formation phase in both groups. Thereafter, it decreased continuously and approached to the minimum levels until the end of the observation period. Serum hypoxia-inducible factor-1α concentrations in both groups were significantly higher compared with controls and hypoxia-inducible factor-1α concentrations in both serum and fracture haematoma were higher in FT group than that in Fr group. Fracture haematoma contained significantly higher hypoxia-inducible factor-1α concentrations compared with hypoxia-inducible factor-1α concentrations in serum. Serum hypoxia-inducible factor-1α concentrations had a positive correlation with hypoxia-inducible factor-1α concentrations in fracture haematoma in patients with fractures.
Conclusions
These findings suggest the local and systemic involvement of hypoxia-inducible factor-1α in fracture healing and the accelerated fracture repair in patients with traumatic brain injury might be associated with elevated hypoxia-inducible factor-1α concentrations in fracture haematoma and serum.
Introduction
In our clinical experience, we have noticed that patients with traumatic brain injury (TBI) always have an accelerated speed of fracture healing. Besides, TBI is also related to the rapid callus formation and heterotopic ossification, and the incidence was reported to be over 20%.1,2 However, the underlying mechanism of this phenomenon was still poorly understood till now. During the past few decades, extensive investigations have been performed to elucidate the responsible mechanism. Bidner et al. 3 firstly demonstrated that increased osteogenesis in patients with TBI is associated with elevated concentrations of circulating growth factors or circulating factors that stimulate local release of growth factors, providing a humoral mechanism for enhanced fracture healing of TBI. Since then, increasing attentions have been attracted to the pivotal role of humoral factors in promoting fracture healing in patients with TBI.
During bone fractures, the local vasculatures are also immediately severed, causing a haemodynamic change and the formation of a haematoma, which isolates the injury site from perfusion, makes a decreased oxygen tension and regional hypoxia microenvironmental around the fracture site.4,5 Therefore, the subsequent bone reparative and remodelling processes, especially the early inflammatory phase, are achieved under this hostile condition, by up-regulating a diverse set of genes, including haeme oxygenase-1 (HO-1), vascular endothelial growth factor (VEGF), inducible nitric oxide synthetase (iNOS) 6 and leptin, 7 which are response for anaerobic energy metabolism and matrix synthesis of fracture repair process. These target genes are under strict control of a newly identified transcription factor, hypoxia-inducible factor-1 (HIF-1), which belongs to the family of basic helix–loop–helix (bHLH)-containing PER–ARNT–SIM (PAS) domain transcription factors. HIF-1 is a heterodimer composed of two subunits, HIF-1α and HIF-1β. 8 The α-subunit is the primary and active component of HIF-1, mediating adaptive response to hypoxia, whereas the β-subunit, also named aryl hydrocarbon receptor nuclear translocator (ARNT), is constitutively expressed and invariable. HIF-1α, as a major oxygen homeostasis modulator, is tightly regulated by the ubiquitin-proteasome system by interacting with the von Hippel-Lindau (VHL) tumour suppressor through hydroxylation of two proline residues and acetylation of a lysine residue in the oxygen-dependent degradation domain (ODD) of HIF-1α under normoxic conditions,9,10 whereas under hypoxic condition, HIF-1α will not be degraded and translocate to the nucleus, functioning as a complex with the β-subunit to initiate transcription of hypoxic-associated genes subsequently. 11
As a angiogenesis- and growth-related transcription factor, HIF-1α was found to be widely expressed in numerous cell types involved in fracture healing such as osteoblasts, 12 osteocytes 13 and chondrocytes14,15 and are therefore of critical importance to revascularization and osteogenesis to optimize bone growth in the fracture and callus site. Hadjiargyrou et al. 16 demonstrated for the first time that the expression levels of HIF-1α was significantly elevated in rat fracture callus with microassay analysis. Their subsequent study further demonstrated that both transcriptional and translational levels of HIF-1α, together with its target genes, VEGF, HO-1 and iNOS were significantly increased in rat fracture callus. 6 Besides, immunohistochemistry analysis further revealed that HIF-1α was entirely localized to chondrocytes in the periosteal callus and osteoblasts throughout the regions of newly formed woven bone, 6 interpreting that chondrocytes and osteoblasts may depend on the adaptive functions of HIF-1α to maintain anaerobic metabolism and matrix synthesis for cellular survival during fracture repair. Previous studies demonstrated that hypoxic preconditioning (a way to activate HIF-1α) human bone marrow stem cells (hBMSCs) causes an enhanced tissue regeneration property in vivo. 17 Transfecting MSCs with HIF-1α resulted in increased cell adhesion and migration and contributed to significant improvement in terms of cardiac function, angiogenesis and cardiomyocyte proliferation, namely promoting cardiac healing. 18 Loading engineered bone grafts with HIF-1α expressing hBMSCs leads to a remarkably acceleration of bone defect repair. 19 A recent study by Agarwal et al. 20 demonstrated that inhibition of HIF-1α either by pharmacologic inhibitor (PX-478 or rapamycin) or using lineage-specific Hif1α knockout methods prevented both trauma-induced and genetic heterotopic ossification. All of these findings suggested that HIF-1α may have a crucial role in the process of fracture healing.
Although investigations concerning the correlation of circulating levels of cytokines and growth factors and accelerated osteogenesis of TBI have been extensively carried out, to our knowledge, there have been no detailed data on the association of local and systemic concentrations of HIF-1α with fracture healing in patients with fracture and concomitant TBI thus far. Therefore, the present study aimed to determine the local and circulating levels of HIF-1α in patients with bone fracture and concomitant TBI for a better understanding of the molecular mechanism and functional role of HIF-1α in the process of fracture healing.
Materials and methods
Study subjects
This study was approved by the Ethical Review Committee of the Qilu Hospital of Shandong University and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all subjects prior to their participation in the study. Fifty-eight patients with meta-/diaphyseal fractures of long bone (humerus, femur or tibia) admitted to Department of Emergency Surgery, Qilu Hospital of Shandong University, between March 2010 and September 2013, were consecutively enrolled in this study. Among the 58 patients, 25 confirmed with a long-bone fracture and concomitant TBI were designated as FT group. Thirty-three patients with an isolated shaft fracture of long bone without concomitant TBI were designated as Fr group. On admission, the two groups were both imaged by conventional X-rays to the long-bone fractures and evaluated with use of the Glasgow Coma Scale (GCS) system. BTI was diagnosed with computerized tomography and confirmed with a GCS score of less than 8 points, while Fr group sustained no TBI and had a GCS score of more than 13 points. The fractures in all patients were treated operatively. Participants were excluded on the basis of having any forms of prior nervous system or bone-related diseases, open fractures type III according to the Gustilo classification, multiple fractures, malignant disease, autoimmune disease, rheumatoid arthritis, diabetes, other chronic inflammation diseases as well as history of long time steroid, immunosuppressant, non-steroidal anti-inflammatory drugs or bisphosphonate therapy or survived no more than one year after surgery.
Among the two groups, 10 pairs of patients (one in Fr group pairs to another in FT group) with matched fracture type and location, extent of soft tissue damage and fixation pattern were further investigated. The fracture type was evaluated according to AO/ASIF classification system, and the extent of soft tissue damage was determined according to Gustilo’s classification. Thirty-five age- and sex-matched healthy subjects undergoing physical examination were contemporaneously selected in a large member of individuals and designated as controls.
Assessment of fracture healing
All patients were followed up for at least six months after the operation. The follow-up examination was based on clinical and radiological examination at 1, 2, 3, 4, 8, 12, 24 weeks after trauma. The diagnosis of bony consolidates or delayed union was evaluated according to exercise-induced pain and conventional X-ray radiographs. The time to union, defined as the time interval from treatment to bone bridging of both cortices on the radiographs, was independently assessed by two radiologists, who were blinded to the presence or absence of TBI. Delayed union was defined as failed fracture healing without radiological signs of bony consolidation after four months postoperatively. Non-union was defined as the absence of complete consolidation at six months after surgery.
Samples collection
Fresh fracture haematoma was obtained from all patients intraoperatively, before any manipulation to avoid blood contamination in the operating field and centrifuged to remove cells and joint debris. The supernatants were aspirated and stored at −80℃ until utilized. Venous blood samples were collected from all patients immediately after hospital admission (always within 8 h after trauma), at 3, 5, 7, 10 days and 2, 3, 4, 8, 12, 24 weeks after injury and centrifuged at 1600 × g for 15 min, and the resulting serum samples were stored immediately at −80℃ until measurement.
Measurement of HIF-1α concentrations
Double-blind quantitative measurement of HIF-1α concentrations in serum and haematoma was analysed with a commercially available enzyme-linked immunosorbent assay (ELISA) kit (RayBiotech, Inc., Norcross, GA, USA) according to manufacturer’s instructions. Briefly, standards of HIF-1 α, serum and fracture haematoma samples were added to 96-well microtitre plates precoated with mouse monoclonal antibody against human HIF-1α and incubated for 2 h at room temperature (RT). Wells were then washed four times with washing buffer and incubated for another 2 h at RT with a horseradish peroxidase-conjugated monoclonal antibody against HIF-1α. After four washes, substrate solution was added to each well, and the plate was incubated for 30 min at RT in the dark. Finally, the reaction was stopped with the stop solution, and absorbance was measured at wavelength of 450 nm using an automated microplate reader (Infinite® M200 PRO, Tecan Group Ltd, Männedorf, Switzerland). The manufacturer-reported precision was <10% for intra-assay coefficient of variation (CV) and <12% for inter-assay CV. The sensitivity (minimum detectable dose) of the assay was 61 pg/mL.
Statistical analysis
Statistical analysis was performed with the Statistical Package for Social Sciences software, version 16.0 for Windows (SPSS Inc.; Chicago, IL, USA). All data were presented as mean ± standard deviation of the mean (SD) or median (interquartile range). Kolmogorov-Smirnov test was performed to analyse the data normality and unpaired t-test, Mann-Whitney U test or Chi-square test was used to assess significance in clinical characteristics between patients with fracture only and concomitant TBI, as appropriate. Comparisons between groups of continuous variables were performed by using non-parametric ANOVA (Wilcoxon rank-sum test for two variables or Kruskal-Wallis-Test for more than two variables). For statistical comparison of serum concentrations of HIF-1α at a certain time point between the Fr group and the matched FT group, non-parametric Mann–Whitney U test for unpaired samples was used. Differences between HIF-1α concentrations in serum and fracture haematoma were analysed using the Wilcoxon signed-rank test for paired samples. Spearman’s rank correlation was used to determine the association between the HIF-1α concentrations in serum and fracture haematoma. P < 0.05 was considered to be statistically significant for differences and correlations. The power of the study was estimated with the published data in the literature and then calculated with Power and Precision software (Biostat, Englewood, NJ, USA), with results reaching over 80%.
Results
Baseline characteristics of the study groups
The baseline clinical parameters of patients with fracture alone and concomitant TBI.
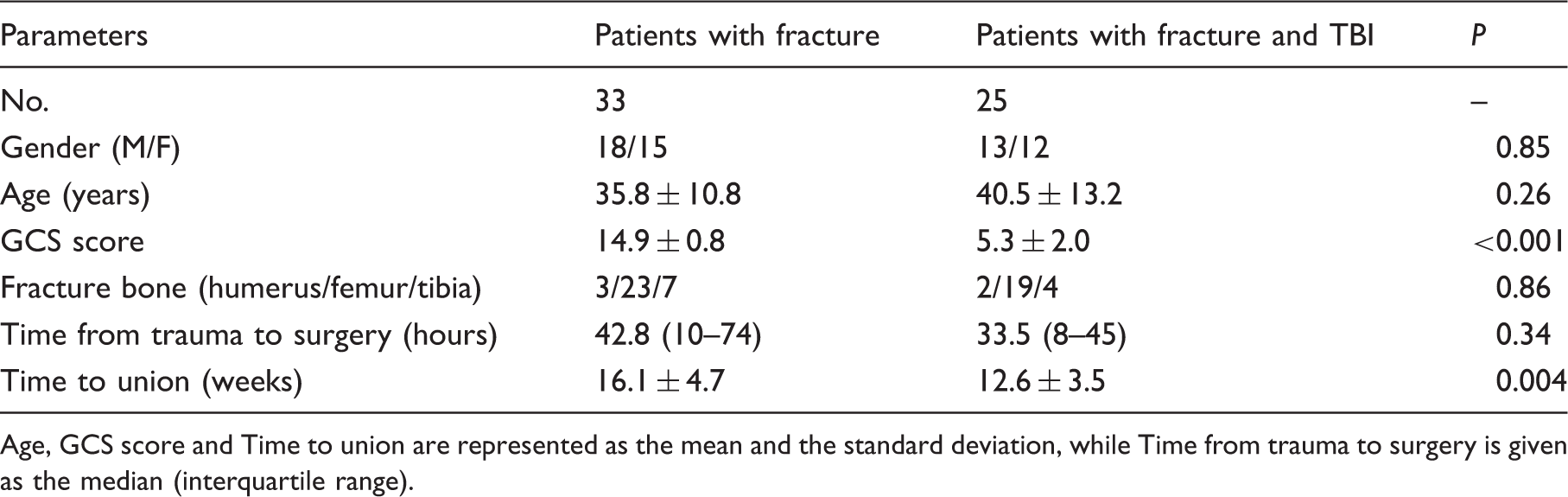
Age, GCS score and Time to union are represented as the mean and the standard deviation, while Time from trauma to surgery is given as the median (interquartile range).
Matching criteria and demographics of the matched patients from both groups.
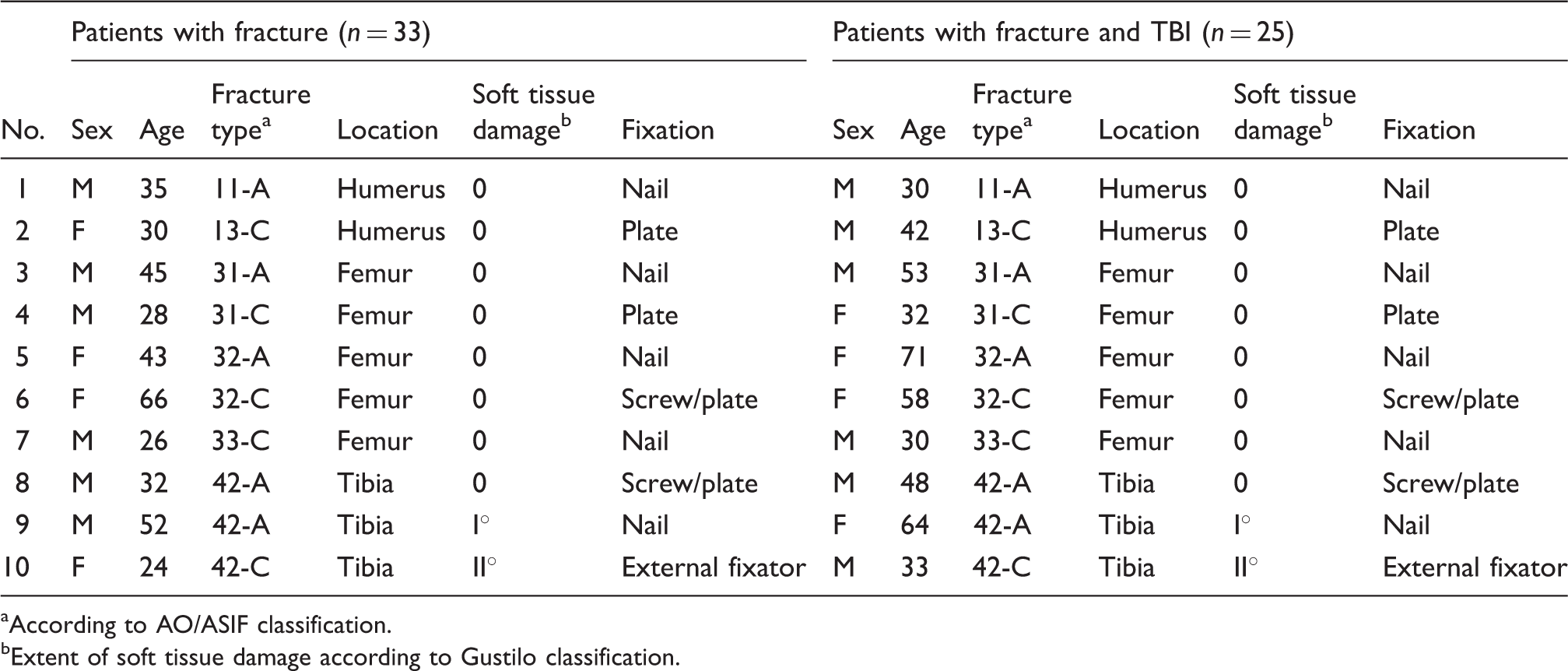
According to AO/ASIF classification.
Extent of soft tissue damage according to Gustilo classification.
Serum concentrations of HIF-1α in patients with fracture alone and concomitant TBI
In patients with fracture alone, serum HIF-1α concentration was 283.16 ± 58.69 pg/mL immediately after admission and increased gradually to reach the maximum concentration of 2568.56 ± 628.12 pg/mL at week 2 after injury. HIF-1α concentration decreased dramatically and continuously after week 2 and reached the minimum concentration (123.45 ± 38.48 pg/mL) at the end of the observation period at week 24 after trauma (Figure 1). Similar to patients in Fr group, serum HIF-1α concentration in FT group was 354.23 ± 46.45 pg/mL immediately after admission and increased to reach the maximum concentration at day 10 (2646.15 ± 645.43 pg/mL), which was followed by a decline at week 2 after injury (2346.49 ± 585.13 pg/mL) and a continuous approach to the minimum concentrations until the end of the observation period at week 24 (148.27 ± 36.04 pg/mL) after trauma. In the whole observation period, the serum HIF-1α concentrations in both groups were significantly higher than controls (113.45 ± 26.23 pg/mL). The serum HIF-1α concentrations in FT group were significantly higher than that in Fr group at days 3 and 10 after surgery (645.33 ± 146.50 pg/mL vs. 367.46 ± 93.52 pg/mL, P < 0.05 and 2646.15 ± 645.43 pg/mL vs. 1468.17 ± 498.20 pg/mL, P < 0.05, respectively) and the peak time in FT group was earlier than patients in Fr group (day 10 vs. week 2 after injury). The concentrations of HIF-1α were higher in FT group compared with Fr group at days 0, 5, 7 and weeks 3, 4, 8 and 12 after injury, but no statistically significant differences were observed (Figure 1).
Serum HIF-1α concentrations in controls, patients with long-bone fractures alone (fracture) and patients with long-bone fracture and concomitant TBI (fracture + TBI). HIF-1α: hypoxia-inducible factor-1α. *P < 0.05 vs. fracture group.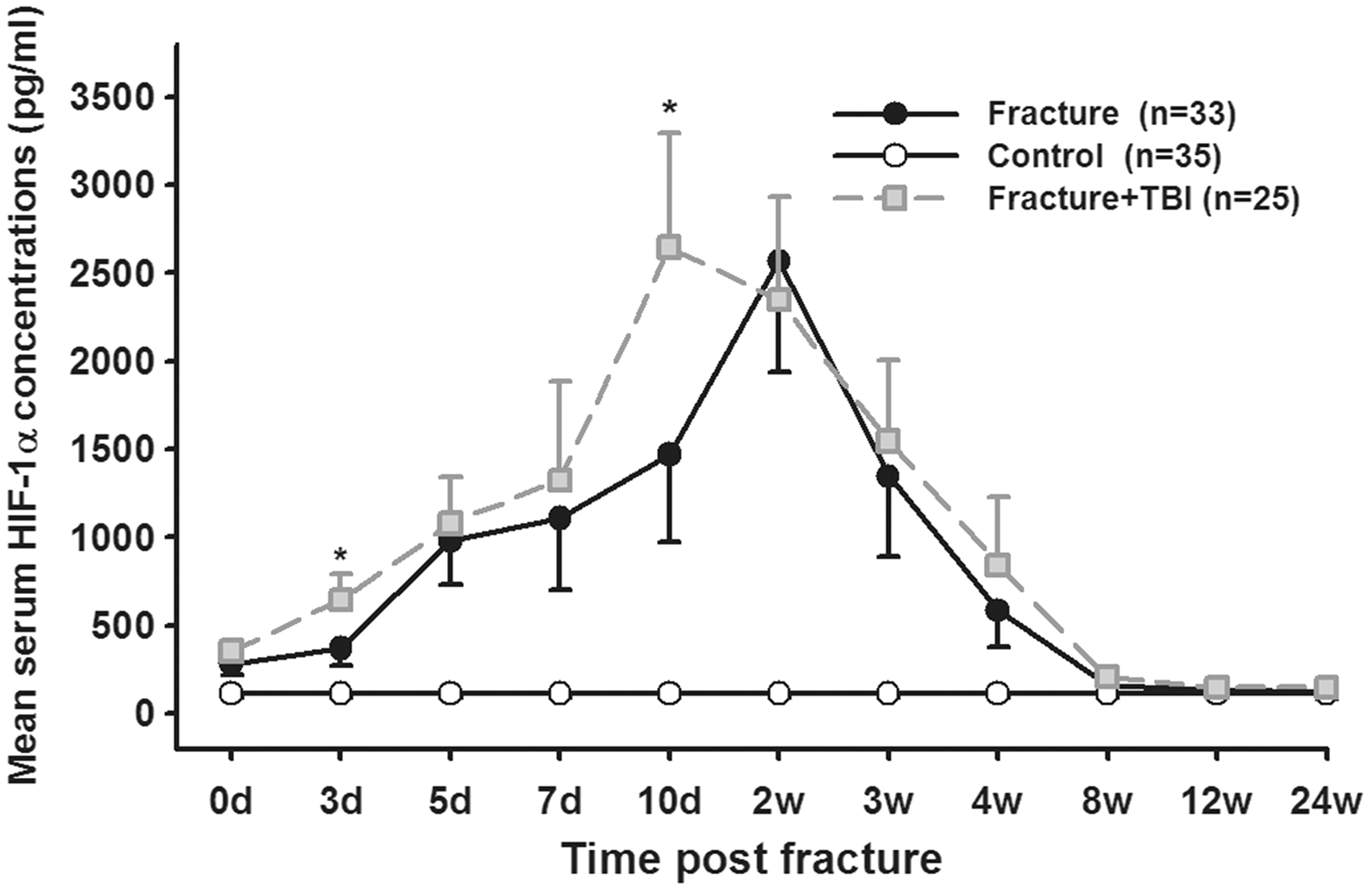
Serum HIF-1α concentrations in matched patients with fracture and concomitant TBI
To exclude the effects of fracture location or fixation pattern on serum HIF-1α concentration, we selected 10 pairs of patients with matched fracture type and location, extent of soft tissue damage and fixation pattern from both groups and their serum HIF-1α concentrations were analysed. As illustrated in Figure 2, serum HIF-1α concentrations in these 10 pairs of patients altered similar to the two groups aforementioned. In these 10 patients with fracture alone, mean HIF-1α concentration was 303.25 ± 45.87 pg/mL immediately after admission and increased to reach the highest concentration at week 2, followed by a continuous decrease and approach to the minimum concentration of 118.45 ± 28.63 pg/mL at the end of the observation period. In the matched groups of patients with fracture and concomitant TBI, initial HIF-1α concentration was 354.23 ± 85.45 pg/mL. Thereafter, HIF-1α concentrations were continuously increased and reached the maximum concentration of 2873.51 ± 672.85 pg/mL at day 10 after injury, followed by a continuous approach to the minimum concentration until the end of the observation period at week 24 (125.546 ± 26.31 pg/mL) after trauma. Comparison of serum HIF-1α concentrations in matched patients with fracture alone and concomitant TBI revealed significantly higher HIF-1α concentrations in patients with fracture and concomitant TBI at days 3, 5, 7, 10 and week 3 after trauma. Patients with fracture and concomitant TBI had a higher peak concentration of serum HIF-1α, and the higher concentration sustained longer than patients with fracture alone.
Time course of serum HIF-1α concentrations in matched patients with long-bone fractures alone (fracture) and patients with long-bone fracture and concomitant TBI (fracture + TBI) after surgery for fractures.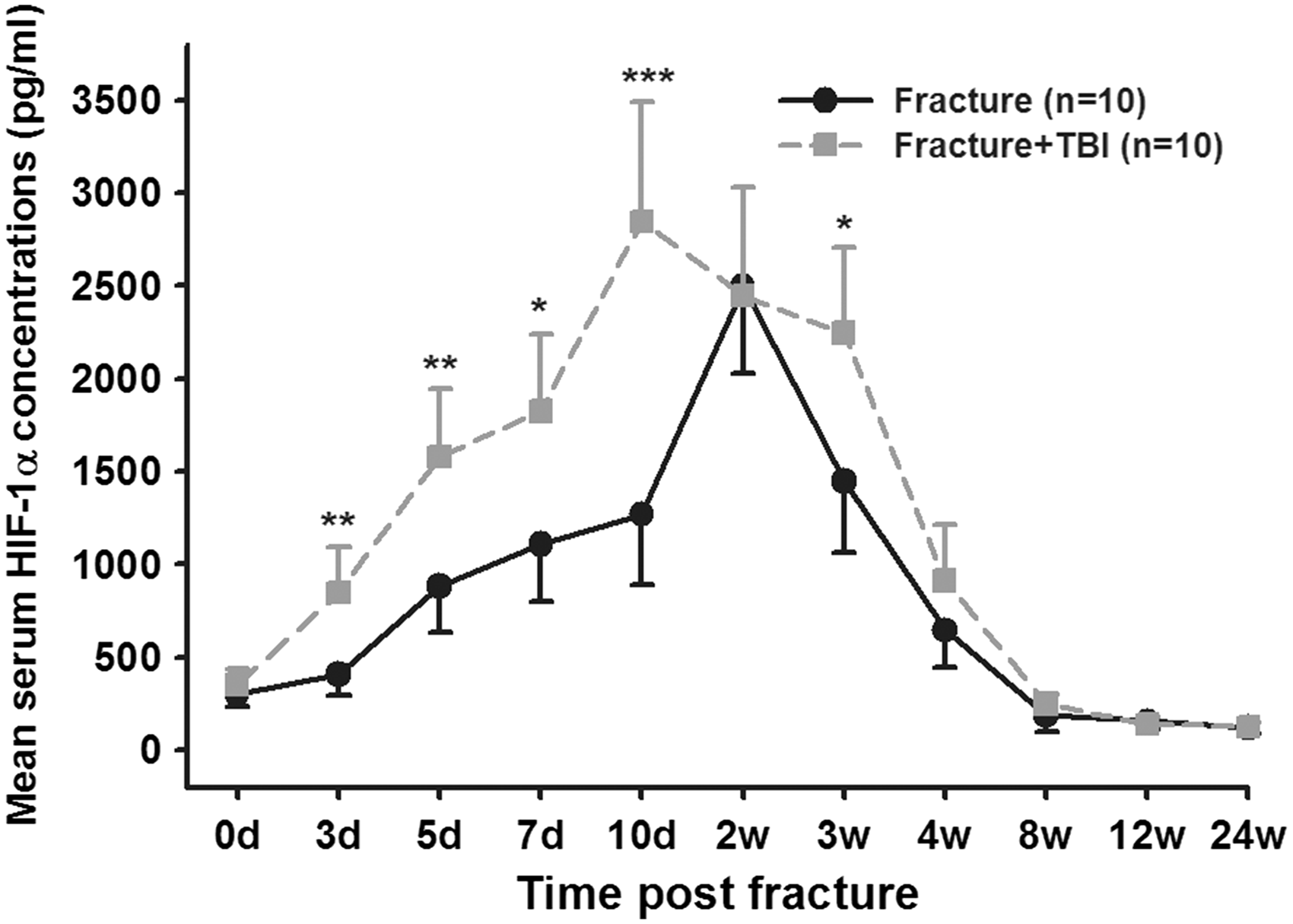
Comparison of HIF-1α concentration in fracture haematoma and serum
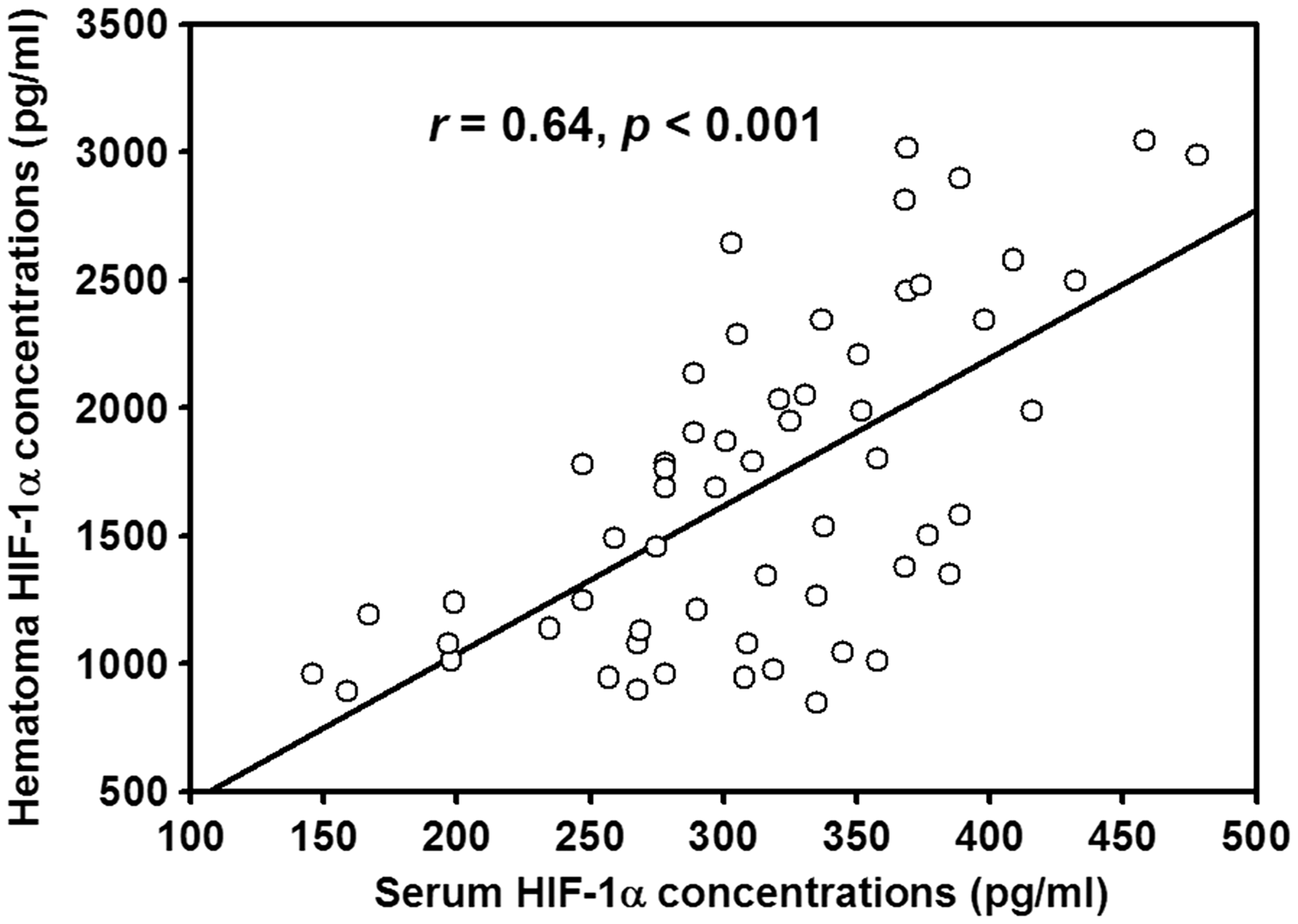
Concentrations of HIF-1α in fracture haematoma of patients in both groups are illustrated in Figure 3. Significantly higher mean HIF-1α concentrations measured in fracture haematoma were detected in FT group compared with patients in Fr group (2246.74 ± 487.28 pg/mL vs. 1265.35 ± 338.12 pg/mL, P < 0.05). Besides, HIF-1α concentrations in fracture haematoma were remarkably higher than that of paired serum samples in both groups (2246.74 ± 487.28 pg/mL vs. 354.23 ± 46.45 pg/mL, P < 0.001 and 1265.35 ± 338.12 pg/mL vs. 283.16 ± 58.69 pg/mL, P < 0.001, respectively). Additionally, we also analysed the association of HIF-1α concentrations in serum and fracture haematoma of patients in both groups; the results showed that serum HIF-1α concentrations had a positive correlation with HIF-1α concentrations measured in fracture haematoma (r = 0.64, P < 0.001) (Figure 4).
HIF-1α concentrations in fracture haematoma and serum of controls, patients with long-bone fractures (fracture) and patients with long-bone fracture and concomitant TBI (fracture + TBI). Correlation between HIF-1α concentrations in serum and fracture haematoma of patients with long-bone fractures (r = 0.64, P < 0.001).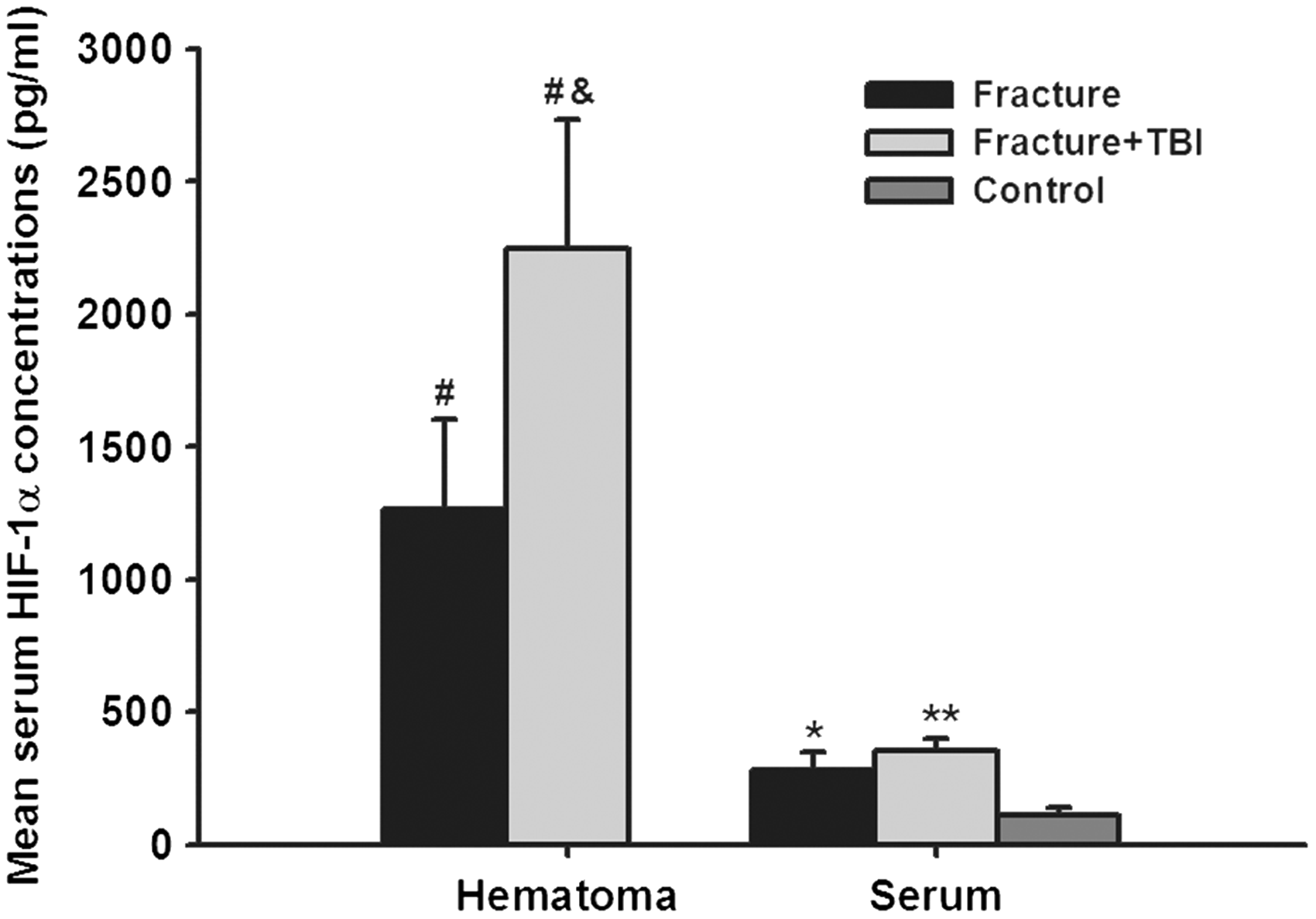
Discussion
A growing body of evidence has demonstrated that fracture healing is not a local process but is rather related to systemic reactions. Characteristic alterations in levels of the systemic and local factors have been previously described during fracture healing.21–23 Among various growth factors essential for the initiation and maintenance of the fracture healing, the angiogenic factors, such as VEGF, received more attention because the fracture repair process relies greatly on the formation of new blood vessels in the fracture site. 24 Blood vessels not only supply oxygen and nutrients to the fracture site but also play a critical role in bone regeneration and remodelling by mediating the interaction between osteoblasts, osteocytes, osteoclasts and vascular cells at a variety of levels. 25 Therefore, angiogenesis and osteogenesis are tightly coupled during bone development and regeneration.
HIF-1 family members been implicated in a diverse array of physiological and pathological processes such as tumourigenesis, inflammation, cell survival in ischaemic tissues. 26 The active subunit HIF-1α is responsible for oxygen responsiveness and is hydroxylated by oxygen-sensitive prolylhydroxylases under normoxic conditions, followed by targeting of the VHL tumour suppressor protein and degradation by the proteasome. 27 Recent studies have showed that HIF-1α is endogenously expressed in osteoblasts and functions as a critical regulator of angiogenic–osteogenic coupling.28,29 Constitutive activating HIF-1α through disruption Vhl in osteoblasts produces striking and progressive increase in bone volume that is secondary to an increase in osteoblast number and activity, whereas mice lacking HIF-1α in osteoblasts had impaired angiogenesis and bone healing; these processes appeared to be associated with involvement the action of VEGF.29–31 Considering the crucial role of HIF-1α in promoting fracture repair, here we analysed the local and systemic concentrations of HIF-1α in patients with fracture alone and concomitant TBI to test the hypothesis that BTI-caused acceleration of fracture healing might be associated with alterations in the local and/or systemic concentrations of HIF-1α.
In the present study, we confirmed the previous finding that TBI could shorten fracture healing time. Our data demonstrated for the first time that the local and the temporal systemic expression pattern of HIF-1α following fractures in patients with long bone fractures alone and concomitant TBI. Significantly higher concentrations of HIF-1α in fracture haematoma than serum within the first few hours after trauma detected in both groups elucidate active involvement of this molecule in the healing process, as early as from the inflammatory phase. It is well known that early bone formation is initiated by osteoblasts. Previous study demonstrated that HIF-1α could promote the differentiation of MSCs into osteoblasts. 32 So it appeared that early elevation of HIF-1α concentrations in the fracture site was necessary for the proliferation and differentiation of the osteoblasts during the inflammatory phase of fracture healing. In the association analysis, Spearman’s rank correlation revealed that HIF-1α concentrations in serum had a positive correlation with HIF-1α concentrations in fracture haematoma, which might imply the possibility that HIF-1α could be brought into the fracture site by blood vessels from the circulating system. However, it should be noted that HIF-1α concentrations in fracture haematoma was a great deal higher than that in serum measured immediately after trauma, which can be attributed to local secretion of HIF-1α by osteoprogenitor cells residing in fracture site.
Our results showed a fluctuation of the systemic HIF-1α concentration within 24 weeks after trauma in both groups. Serum HIF-1α concentrations in both patients with fractures alone and concomitant TBI were significantly higher than in controls. A continuous increase of serum HIF-1α concentration was observed immediately after fracture in both groups. The increase continued as highest HIF-1α concentrations were detected in the first two weeks after trauma. HIF-1α concentrations decreased at two weeks after surgery, followed by a continuous decline and approach to the minimum concentration until the end of the observation period at week 24 after surgery. These observations indicated that the oxygen tension in fracture site is extremely low in the early inflammatory phase; HIF-1α is triggered and required for the maintenance of anaerobic metabolism and matrix synthesis for cellular survival. The increase in serum HIF-1α concentrations suggests a systemic response to fracture and may partly be due to the absorption of HIF-1α from the fracture site into the circulating system; as evidence exists, not only local but also systemic release of growth factor is necessary to initiate and maintain fracture healing. 33 The serum concentrations of HIF-1α were sustainably elevated and maximum concentrations were detected during the intramembranous bone formation phase might suggest the chemotactic effect of HIF-1α on bone cells as an increasing number of osteoblasts, chondroblasts and immature progenitor cells invade the fracture area during the intramembranous bone formation phase. 34 Besides, as HIF-1α is potent to induce the differentiation of MSCs into osteoblasts, which express and release HIF-1α by themselves at the proliferations stage and exert stimulating effects on osteoblasts in turn. With the oxygen tension increased, HIF-1α is degenerated gradually and the serum concentrations decreased accordingly. But the concentrations remain higher than controls during the remodelling phase, which seems to be necessary to the sustained activation of osteoblasts during the remodelling phase. HIF-1α expression pattern observed in our study was very similar to the reports showing a high concentration of VEGF during the inflammatory phase of fracture healing in humans.35,36 This does not appear surprisingly because HIF-1α promotes angiogenesis and osteogenesis by elevating VEGF concentrations in osteoblasts. 30
Another important question addressed in our study was whether, and to what extent, HIF-1α concentrations in fracture haematoma and serum of patients with fracture alone would differ from patients with fracture and concomitant TBI. The results revealed significantly higher HIF-1α concentrations in fracture haematoma in FT group compared with Fr group and that serum HIF-1α concentration in FT group was significantly higher than that in Fr group at days 3 and 10 after injury. Besides, the maximum HIF-1α concentration was detected at week 2 after surgery in Fr group and at day 10 after surgery in FT group. Though without statistically significant differences, serum HIF-1α concentrations in Fr group were higher than that in FT group at days 0, 5 and 7 after trauma; systemic concentrations of HIF-1α in the two groups were very similar for the rest of the observation period after reaching the maximum concentration. These results indicated that TBI could significantly elevate concentrations of HIF-1α both in the local and circulating levels in the early inflammatory phase, which contributes to the accelerated fracture healing in TBI patients. Our results are in line with previous reports that in vitro incubating human stromal stem cells with serum from rats following TBI caused significant proliferation 37 and serum from TBI and fracture patients caused a increased proliferation of osteoblasts and accelerated differentiation of osteoprogenitor cells, 38 concluding that growth factors, hormones or peptides derived from brain may mediate a mitogenic effect on osteoprogenitor cells through circulating system. Previous studies have demonstrated that HIF-1α is endogenously expressed in the brain and will be upregulated in the context of TBI,39–41 which might be due to the effects of trauma-induced secondary cerebral ischaemia and hypoxia injury. Central nervous system-specific markers were detected in serum of TBI patient, indicating that the trauma in brain will cause disruption of the blood–brain barrier and allow the leakage of osteoinductive cerebrospinal fluid components into the systemic circulation. 42 Therefore, we propose here that elevated serum HIF-1α concentrations in FT patients may attribute, at least in part, to the flow of newly formed HIF-1α from the injured brain into the systemic circulation.
To clarify whether that the difference in serum HIF-1α concentrations between patients with fracture alone and concomitant TBI was associated with fracture location or fixation pattern, we performed an additional analysis in two fracture location and fixation pattern-matched subgroups. The results showed that HIF-1α concentrations of FT group compared with patients with Fr group were similar in the two matched subgroups of patients. Patients with fracture and concomitant TBI had more pronounced higher HIF-1α concentrations than patients with fracture alone, at days 3, 5, 7, 10 and week 3 after trauma and had a higher peak level and earlier peak time of serum HIF-1α. Besides, the higher serum HIF-1α concentration in FT subgroup also sustained longer and decreased more slowly than Fr subgroup. This analysis indicated no fracture location or fixation pattern-related difference in the HIF-1α serum concentrations between the two matched subgroups and confirmed the aforementioned results.
In summary, the present study demonstrated accelerated osteogenic effects of TBI in patients with fracture and concomitant TBI and revealed, for the first time, a significant elevation of HIF-1α concentrations in serum and fracture haematoma of both patients with fracture alone and concomitant TBI compared with healthy controls and that the HIF-1α concentrations in fracture haematoma and serum in fracture–TBI patients were higher compared with those with fracture alone. Our results indicates the involvement of HIF-1α in the human fracture healing and the accelerated fracture healing in fracture–TBI patients may be associated with the elevated systemic and local expression of HIF-1α, which at least in part are thought to result from the release from injured neural tissue or as part of a central nervous system response to brain injury. However, understanding the exact mechanism involved in accelerated fracture healing in TBI patients is still in its early stages; further studies with larger number of patients are needed to underlie the mechanism by which the central nervous system controls bone remodelling, which is of great interest and presents a challenge.
Footnotes
Acknowledgements
We are grateful to the patients for donating their samples to our research studies, staff within the research and clinical teams at Department of Emergency Surgery, Qilu Hospital of Shandong University for help in obtaining and processing samples for funding this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Department of Science and Technology of Shandong Province (No. 2008GG3002002).
Ethical approval
The ethics committee of Qilu Hospital of Shandong University approved this study (2010QHSU125).
Guarantor
XS.
Contributorship
XS researched literature and conceived the study. ZW, TQ and YL were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. XS and ZW wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
