Abstract
Background
Inflammation and hypercholesterolaemia contribute to atherosclerotic changes which can start in childhood. Children with hyperlipidaemias are at high risk for early coronary atherosclerosis. This study evaluates the relationship between lipoprotein-associated phospholipase A2 (Lp-PLA2), carotid intima-media thickness (CIMT) and flow-mediated dilatation in hypercholesterolaemic dyslipidaemic children.
Methods
We performed a case–control study consisting of 43 cases, aged 2 to 17 years, and 24 age-matched controls. Fasting blood samples were obtained from both groups for the measurement of a lipid profile (total cholesterol, LDL-C, HDL-C and triglycerides) and Lp-PLA2 in mass units. The latter was determined with a turbidimetric immunoassay method (PlacTest, DiaDexus Inc.) applied to an automated analyser. CIMT and flow-mediated dilatation measurements were undertaken by a paediatric cardiologist, using high-resolution B-mode ultrasonography.
Results
Total cholesterol, LDL-C and Lp-PLA2 concentrations were significantly higher in the cases than in the controls (p < 0.001 for all three parameters). While CIMT values were also significantly higher in the patients compared to the controls (P = 0.001), flow-mediated dilatation values were significantly lower (P = 0.001). We found positive correlations between Lp-PLA2 and total cholesterol (r = 0.41, P = 0.001), Lp-PLA2 and LDL-C (r = 0.36, P = 0.004), Lp-PLA2 and CIMT (r = 0.44, P = 0.019) and LDL-C and CIMT (r = 0.41, P = 0.032); there were negative correlations between Lp-PLA2 and flow-mediated dilatation (r = −0.15, P = 0.045), total cholesterol and flow-mediated dilatation (r = −0.45, P = 0.017), LDL-C and flow-mediated dilatation (r = −0.51, P = 0.006) and CIMT and flow-mediated dilatation (r = −0.45, P = 0.016).
Conclusion
Lp-PLA2 concentrations are significantly elevated in hypercholesterolaemic dyslipidaemic children. Given the association of Lp-PLA2 with markers of atherosclerosis (total cholesterol, LDL-C, CIMT and flow-mediated dilatation), the finding of increased concentrations of Lp-PLA2 could be used to identify early atherosclerotic changes in hypercholesterolaemic dyslipidaemic children and may inform their clinical management.
Keywords
Introduction
Hypercholesterolaemia is a multifactorial metabolic disorder. Many factors, especially genetic disorders, have important roles in its development. In hypercholesterolaemic patients, elevated circulating concentrations of total (TC) and LDL-cholesterol (LDL-C) are among the major constituents responsible for the development of atherosclerosis. The process of atherogenesis begins with high concentrations of cholesterol triggering an increase in the expression of adhesion molecules, pro-inflammatory genes and cytokines, thus triggering a systemic inflammatory response. 1 Consequently, circulating leukocytes adhere to and penetrate the vascular endothelial wall, initiating the atherosclerotic process within the vessel. 2 Atherosclerosis is among the most common causes of cardiovascular morbidity and mortality, which include myocardial infarction (MI), and cerebrovascular and peripheral vascular diseases. Studies indicate that atherosclerosis begins in early childhood and continues as a life-long process. 3
Lipoprotein-associated phospholipase A2 (Lp-PLA2) is an enzyme associated with HDL and LDL lipoproteins and plays a role in the hydrolysis of phospholipids. It hydrolyzes platelet-activating factor (PAF), which is an important inflammatory mediator originating from the cell membrane, and PAF-like phospholipids that have been subject to oxidative damage; for these reasons, it is also called PAF acetylhydrolase (PAF-AH).
4
The role of Lp-PLA2 in atherosclerosis is mainly related to its ability to hydrolyze oxidized phospholipids and oxidized LDL (oxLDL).
5
This reaction yields the pro-inflammatory products lyso-phosphatidylcholine (lysoPC) and oxidized fatty acids (oxFA). LysoPC disturbs endothelial function and acts as a chemo attractant on monocytes, causing them to migrate into the intima, becoming first macrophages and then foam cells (Figure 1).
6
In atherosclerotic plaques, activated macrophages and foam cells within the fibrous cap close to rupture have been shown to cause the increased production of Lp-PLA2.
6
When released into the circulation, 85% of Lp-PLA2 is carried with LDL-C and 15% with HDL-C.
7
Schematic representation of the role played by Lp-PLA2 in plaque formation. Lp-PLA2 hydrolyzes oxidized LDL-cholesterol (LDL-C) yielding to the proinflammatory lysophosphatidylcholine (LysoPC) and oxidized fatty acids (FA). LysoPC acts as chemoattractant on monocytes, causing them to migrate into the intima, becoming first macrophages and subsequently foamy cells.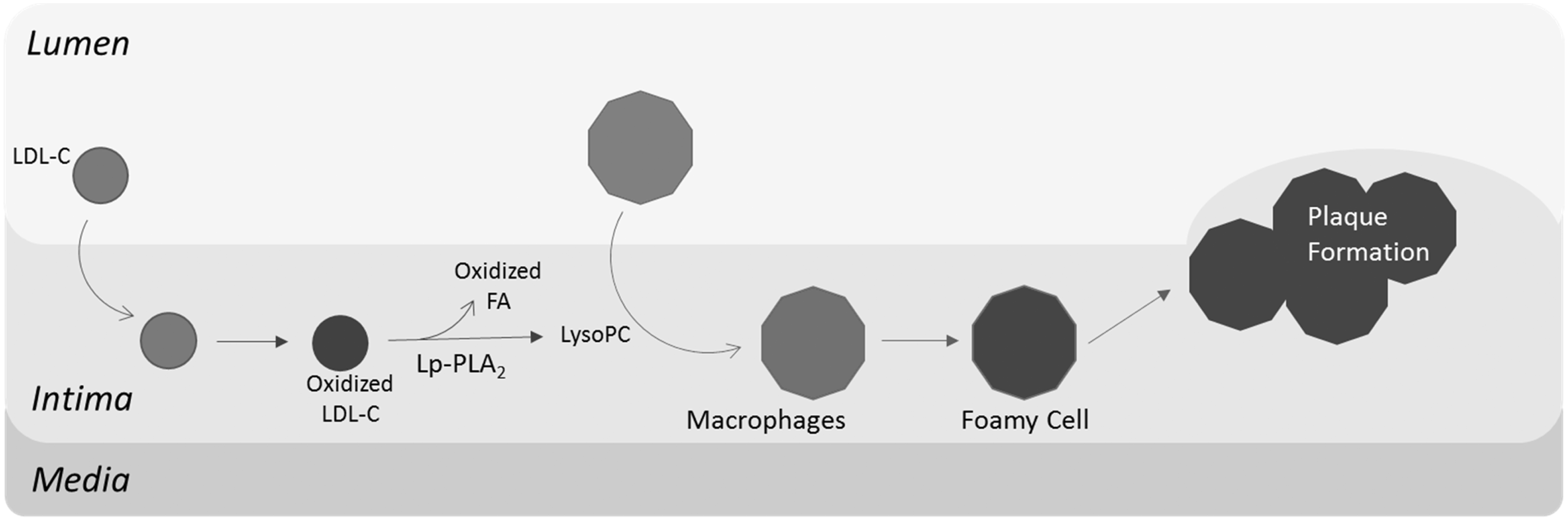
Since Lp-PLA2 is produced within the atherosclerotic plaque and its concentrations do not rise during the systemic inflammatory processes, this enzyme is regarded as a specific marker for vascular endothelium, rather than being considered a systemic inflammatory marker. 8 Compared with patients who do not have atherosclerosis, patients with coronary atherosclerosis have increased circulating concentrations of Lp-PLA2 and as a result, increased concentrations of lysoPC and inflammation. Previous studies have shown that Lp-PLA2 is a marker of inflammation that is primarily produced by macrophages and lymphocytes; thus with its pro-atherogenic role, it plays a critical role in the development of atherosclerosis. Moreover, inhibition of Lp-PLA2 has been shown to have an anti-atherogenic effect. 9 Taken together, these findings suggest that Lp-PLA2 also plays a role during the early stages of the atherosclerotic process. 10
LysoPC, generated as a result of Lp-PLA2 action, can trigger endothelial dysfunction by inhibiting endothelial-derived nitric oxide production, increasing oxidative stress, stimulating apoptosis in endothelial cells, or preventing endothelial cell migration to the site of endothelial damage. 6
Endothelial function is generally evaluated by changes in the endothelial vascular tone. Evaluation of flow-mediated dilatation (FMD) in brachial artery with Doppler ultrasonography is commonly used for the interpretation of endothelial function. 11
Many recent studies have described the measurement of carotid intima-media thickness (CIMT), as an early marker for atherosclerosis. 12 CIMT has been reported to correlate with the presence of coronary artery disease, and largely, to provide further information for the better stratification of non-fatal cerebral and cardiovascular risk. 12
In adults, many studies have been undertaken of both FMD and CIMT as measures of atherosclerosis, and there is some limited evidence that FMD’s and considerable evidence that CIMT’s add value to risk stratification. 13 Despite the fact that non-standardized measurement methodologies in these imaging techniques result with inconsistent findings, 14 these markers are generally recommended as tools to improve the definition of cardiovascular risk in individuals who need early intervention. 15 However, the use of these techniques for clinical evaluation of atherosclerosis in paediatric patients at high risk for cardiovascular events is limited. Thus, precise and reliable markers are required that will facilitate the estimation of the cardiovascular hazards especially in children with high risk. Such improved risk profiling will then allow close monitoring of the highest risk group for the highly aggressive interventions to prevent hard cardiovascular end-points. 3
Based on these findings, the aims of this study were to measure Lp-PLA2 in children with and without hypercholesterolaemia and to examine the relationship between Lp-PLA2 and CIMT, an early marker for atherosclerosis, and between Lp-PLA2 and FMD, an indicator of early endothelial dysfunction, in hypercholesterolaemic dyslipidaemic (HD) children.
Method and materials
Identification of patient and control groups
The patient group consisted of cases who were referred to the Outpatient Clinic in the Department of Metabolism (this is a specialized, second step health care facility at Ege University Faculty of Medicine, Children’s Hospital), between April 2011 and December 2012, either from Outpatient Clinic of Healthy Children or from primary care. All cases had either a history of familial hypercholesterolaemia and/or randomly established hyperlipidaemia (discovered following laboratory investigations undertaken for other reasons) and/or had some subtle clinical signs (chest pain, visual-skin related symptoms, etc.) that inferred a preliminary diagnosis of familial hypercholesterolaemia. In all cases referred, no meaningfully persistent pathological physical or clinical signs (chest pain, decrease in effort capacity, skin colour changes, xanthomatous plaques, xanthelesma, arcus cornea, etc.) were detected by physical examinations and/or patient history. Disorders of lipoprotein metabolism were identified by measurement of the following serum lipid parameters (TC, HDL-cholesterol, LDL-cholesterol, triglyceride, Lp(a), Apo B, Apo A1) and by undertaking lipid electrophoresis. Individuals with systemic and chronic disease (e.g. renal or liver failure) and secondary hyperlipidaemia (e.g. nephrotic syndrome or obesity) were excluded. Out of the 52 individuals referred, 43 cases, aged between 2 and 17 years, were subsequently diagnosed as having heterozygous familial hypercholesterolaemia and met all of the criteria of the study. All of the cases had at least one parent with hypercholesterolaemia. As none of these patients had been diagnosed with hyperlipidaemia before, they were not taking any lipid lowering drugs.
The control group (n = 24) consisted of age-matched children who presented to Ege University, Faculty of Medicine, Department of Paediatrics, Division of General Paediatrics, for various reasons. Exclusion criteria for the selection of the control group were as follows: (1) the presence of a metabolic and systemic disease, (2) the presence of a chronic inflammatory disease, (3)taking lipid-lowering drugs and (4) the presence of cardiovascular disease and/or hypercholesterolaemia among first-degree relatives. All individuals who met inclusion criteria were informed verbally about the purpose and scope of the study, and a signed informed voluntary consent form was obtained from the legal guardians of the children included in the study. The study was approved by the Local Ethics Committee on 17 May 2010 with the item no: 10-4.1/30.
Analytical methods
Venous blood samples were obtained from all individuals in the patient and control groups following an 8-h fasting period. The serum obtained was stored at −80℃ until analyses were performed. Serum Lp-PLA2 mass levels were measured using a commercially available Lp-PLA2 enzyme-linked immunosorbent assay (ELISA) kit (PLAC test) on a Syncron LX20 analyser (Beckman Coulter, USA) in the Biochemistry Laboratory of Ege University, Faculty of Medicine, Department of Paediatrics. A diagnosis of dyslipidaemia was made based on serum concentrations of TC, triglyceride (TG), LDL- and HDL-cholesterol, according to the age- and sex-specific percentiles as described in the Nelson Textbook of Paediatrics. 16 Plasma glucose concentrations were measured using the glucose oxidase/p-aminophenazone (GOD/PAP) method. Serum lipids were determined using the following methods: TC, cholesterol oxidase/p-aminophenazone (CHOD/PAP); TG, glycerol-3-phosphate oxidase; HDL-C, precipitation. All measurements were undertaken on an I Lab 300 Plus analyser (Clinical Chem. Systems, USA). LDL-C concentrations were determined using the Friedewald equation; this equation was not used when the serum TG level was above 4.5 mmol/L.
CIMT and FMD measurements
All individuals were referred to Ege University, Faculty of Medicine, Department of Paediatrics, Division of Cardiology, where a paediatric cardiologist conducted CIMT and FMD measurements in a blind setting. CIMT was measured using high-resolution B-mode ultrasonography (GE Vingmed, Vivid 7.0, Ultrasound AS, Horten, Norway), equipped with a 12-MHz linear probe, on the right carotid artery, at end-diastole; three measurements were recorded for each patient, and the average determined to be the CIMT value. 17 Endothelial FMD measurement was performed while the patient was lying supine with the right arm extended after a brief resting period. The site where the brachial artery was best visualized, 2–5 cm above the cubital area, was marked using a 12-MHz probe, and the probe was fixed on this position. All the measurements were recorded by measuring the internal diameter of the brachial artery at end-diastole. Following the optimal basal measurement, the arm was subjected to ischaemia for 5 min by applying pressure (50 mmHg above the systolic pressure) with a cuff on the upper arm; the pressure was then relieved, and the measurements at the brachial artery were repeated at 60 s intervals. Dilatation at the brachial artery following basal measurement and ischaemia was calculated as the percentage (%) of the FMD value. 18
Statistical analysis
Statistical analyses were performed using SPSS (Version 11.0) software. Student’s t test was used to determine the significance in the difference between groups. Pearson’s correlation analysis was used for multiple comparisons and p < 0.05 was accepted as being statistically significant.
Results
Demographical, clinical and laboratory data in control and patient groups.
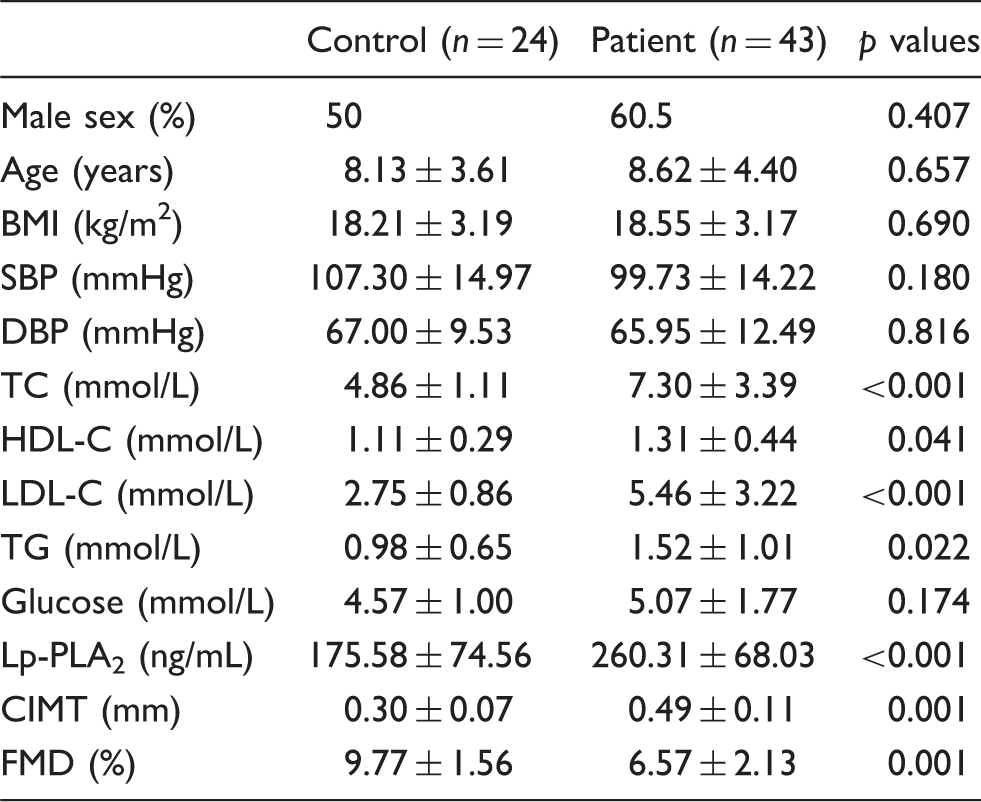
When demographical and clinical data were compared between the control and the hypercholesterolaemic patient group, there was no statistically significant difference in sex, age, BMI, SBP, DBP and glucose concentrations. When laboratory findings were compared, concentrations of TC (p < 0.001), HDL-C (p = 0.041), <LDL-C (p < 0.001), TG (p = 0.022) and Lp-PLA2 (p < 0.001) were significantly higher in the patients compared to the controls.
When ultrasonography findings were compared in the two groups, CIMT values were significantly higher in the patients than in the controls (p = 0.001); FMD values were significantly lower in the patients than the controls (p = 0.001).
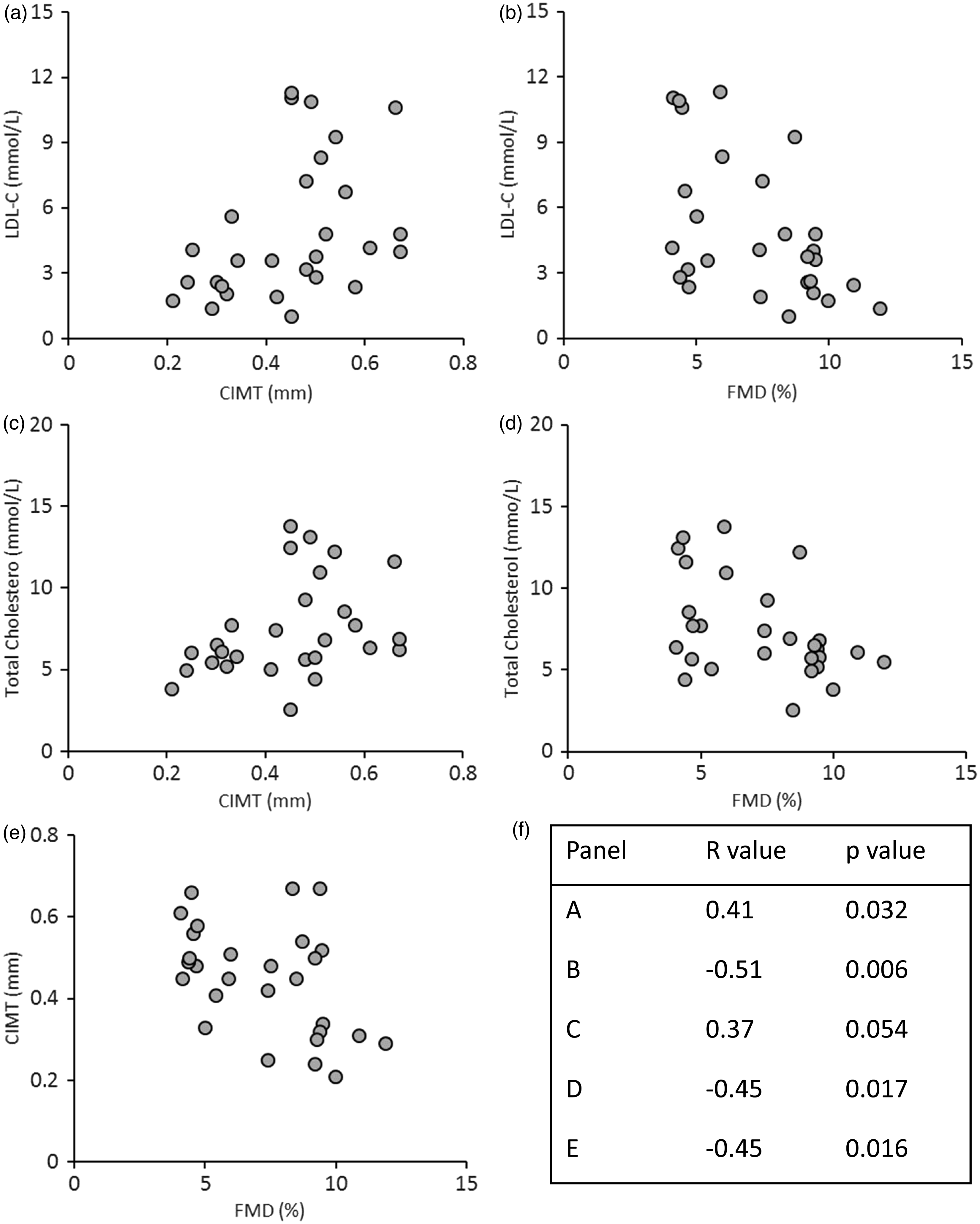
According to the correlation analysis between the parameters of the study, Lp-PLA2 showed a positive correlation with TC (r = 0.41, P = 0.001, Figure 2(a)), LDL-C (r = 0.36, P = 0.004, Figure 2(b)), HDL-C (r = 0.28, P = 0.024, Figure 2(c)) and CIMT (r = 0.44, P = 0.019, Figure 2(d)), and a negative correlation with FMD (r = −0.15, p = 0.045, Figure 2(e)). Both LDL-C and TC showed a positive correlation with CIMT (r = 0.41, p = 0.032, Figure 3(a) and r = 0.37, p = 0.054, Figure 3(c), respectively), but a negative correlation with FMD (r = −0.51, p = 0.006, r = −0.45, p = 0.017, Figure 3(b) and (d), respectively). Moreover, we observed a negative correlation between CIMT and FMD (r = −0.45, p = 0.016, Figure 3(e)). There were no significant correlations between Lp-PLA2 and TG, CIMT and TG, as well as FMD and TG. Similarly, HDL-C concentrations showed no significant correlation with either CIMT or with FMD (data not shown).
Correlation analysis of Lp-PLA2 levels with total cholesterol (a), LDL-C (b), HDL-C (c), CIMT (d) and FMD (e). The related r and P values of each correlation are also listed (f). Correlation analysis of LDL-C with CIMT (a) and FMD (b), total cholesterol with CIMT (c) and FMD (d) and CIMT and FMD (e). The related r and P values of each correlation are also listed (f).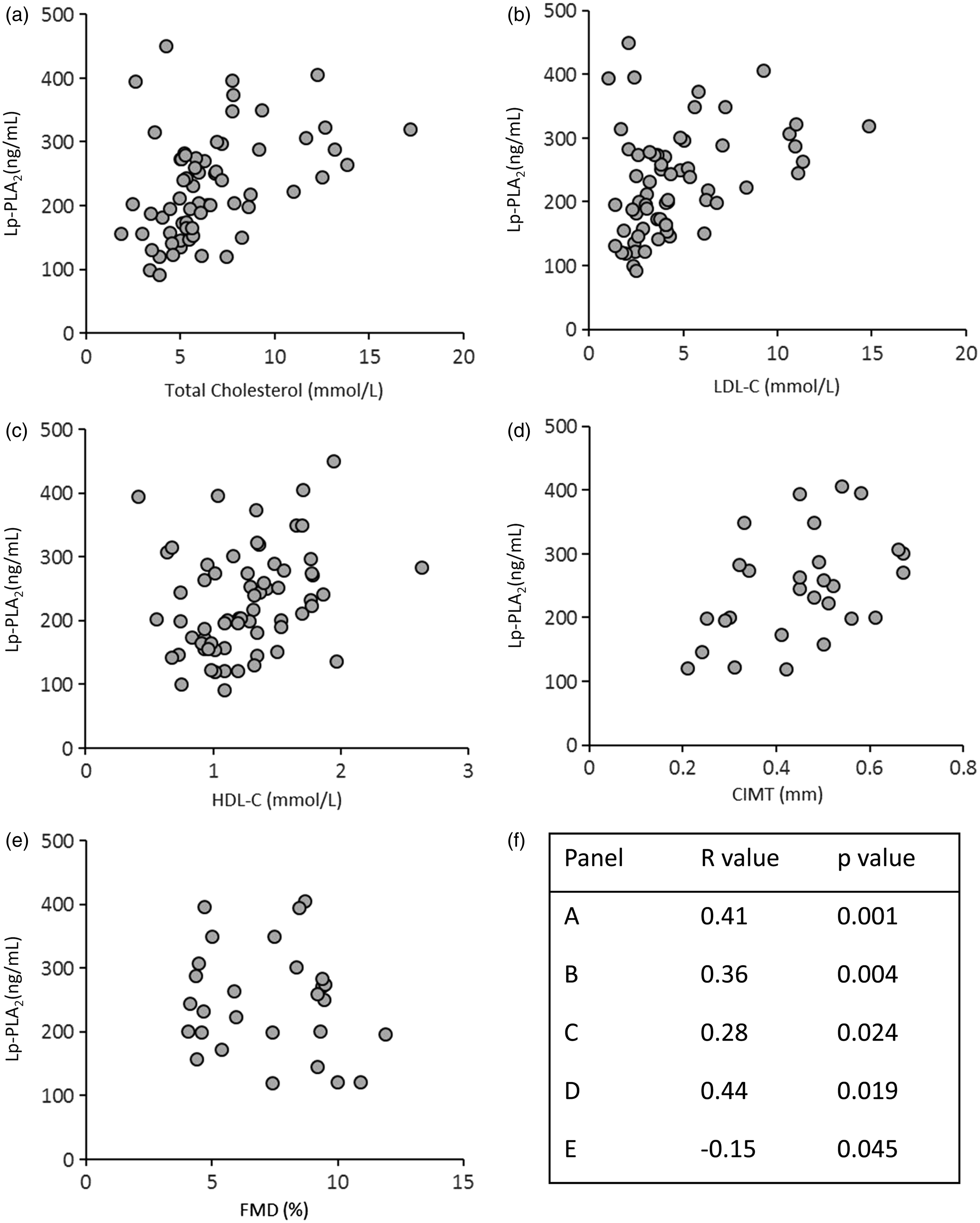
Discussion
The present study demonstrates that Lp-PLA2 concentrations are significantly elevated in HD children and that Lp-PLA2 could indeed be used as a marker for the detection of early atherosclerotic changes in these patients.
Atherosclerosis is a multifactorial pathology that can start in childhood especially in cases with risk factors that include obesity, hypertension and dyslipidaemia and continues as a life-long process. 19 Hypercholesterolaemia is an independent risk factor for this pathology in which high concentrations of serum TC and LDL-C are known to be among the most important factors in the development of atherosclerosis, especially in children. 20 Patients known to have dyslipidaemic disorders, such as high concentrations of serum TC and LDL-C during childhood and adolescence, carry a greater risk for early onset of atherosclerosis, subclinical progression and associated complications. 21 Therefore, close monitoring of dyslipidaemic children (and particularly those with familial hypercholesterolaemia), together with early diagnosis, and appropriate treatment are crucial steps for the prevention of further health issues. 22
Many studies have investigated Lp-PLA2 levels in adults with atherosclerotic diseases and although most of them have found an association between Lp-PLA2 and atherosclerosis as well as coronary heart disease,23–26 some have not.27,28
Other studies have investigated the concentrations of Lp-PLA2 in childhood. Ryu et al. 29 explored Lp-PLA2 mass and activity levels in children with heterozygous familial hypercholesterolaemia and found that they were significantly higher compared to their unaffected siblings. While some found a correlation between BMI and Lp-PLA2 levels in overweight and obese children,30–32 others did not. 33 Castro et al. 34 reported elevated Lp-PLA2 activity in young diabetic adults and adolescents compared with activities found in healthy controls. In the EARLY study involving 50 hyperlipidaemic children between 7 and 19 years of age, Lp-PLA2 levels in hyperlipidaemic cases were found to be close to the high-risk values determined for adults. 35
In our study, we found that Lp-PLA2 concentrations were significantly higher in HD children than in controls (P = 0.001).
The interpretation of measured Lp-PLA2 concentrations is dependent on the establishment of a reference interval for serum Lp-PLA2. Reference intervals for Lp-PLA2 have been reported in the range of 120–342 ng/mL for women and 131–376 ng/mL for men. Previous studies indicated 235 ng/mL as a clinical cut-off value for adults; however, after a review by an expert committee, this cut-off value was lowered to 200 ng/mL. 36
As yet however, there are no data to determine the cut-off value in children. In our study, measured values of Lp-PLA2 in the patient group were 260.31 ± 68.03 ng/mL, much higher than the cut-off value defined as ‘high risk’ in adults (Lp-PLA2 > 235 ng/mL). On the other hand, the control group in our study showed Lp-PLA2 values within normal limits (175.58 ± 74.56 ng/mL).
Overall, these results indicate that HD children have an increased risk of developing atherosclerosis and plaque rupture. Moreover, our findings suggest that Lp-PLA2 levels are moderately correlated with TC (r = 0.41, P = 0.001) and LDL-C (r = 0.36, p = 0.004) and weakly correlated with HDL-C (Figure 2). These findings are consistent with the fact that 85% of Lp-PLA2 is associated with LDL-C and 15% with HDL-C. 7
In our study, we found a positive correlation between Lp-PLA2 and CIMT (r = 0.44, p = 0.019) and a negative correlation between Lp-PLA2 and FMD (r = −0.15, p = 0.045) and between CIMT and FMD (r = −0.45, p = 0.016) (Figures 2 and 3). Moreover, while CIMT values were higher in HD children, FMD values were lower compared to controls (P = 0.001 for both parameters).
Despite some studies stating the opposite, 37 the general perception is that CIMT is a predictor of cardiovascular diseases.38,39 In the Rotterdam study, Van Der Meer et al. 23 showed that CIMT measurement is an independent predictor of MI (Hazard Ratio: 1.95 (1.19–3.19)). In the ARIC study, the risk of cardiovascular events was found to be significantly higher in individuals who had a CIMT above 1 mm, compared with those who had a CIMT below 1 mm. 40
In our study, CIMT values were significantly higher in the hypercholesterolaemic patient group compared with the controls. This result indicates that hypercholesterolaemic children have morphological atherosclerotic changes that start at an early age. There is one published study that shows a borderline to no significant association between CIMT and Lp-PLA2 in familial hypercholesterolaemic children. 29 There are, however, several studies investigating this relationship in adults. Campo et al. 41 and Kardys et al. 42 found no correlation between Lp-PLA2 levels and CIMT >1 mm while Li 43 found that CIMT and Lp-PLA2 levels were closely correlated with the severity of coronary heart diseases.
Many studies have investigated the association between atherosclerosis and FMD.44,45 In a study aimed at characterizing the 5-year-prognostic value of FMD in patients with chest pain, Neunteufl et al. 46 determined that cardiovascular events occurred more frequently in patients who had impaired FMD. Moreover, FMD was found to be significantly lower in children with familial hypercholesterolaemia and adults with cardiovascular disease 47 and was a useful screening tool for cardiovascular risk stratification. 48 In healthy individuals, FMD is between 7% and 10% of the basal brachial artery diameter, whereas it varies between 0% and 5% in patients with cardiovascular diseases. In our studies, FMD values are consistent with these reports, as the FMD percentage in the patient group (6.57%) is significantly lower than in the control group (9.77%, p = 0.001). These results suggest that endothelial dysfunction has already started in the hypercholesterolaemic patients in our study.
In our research, an inverse relationship was observed between CIMT and FMD (r = −0.45, p = 0.016, Figure 3(e)). This result is important as it indicates that early morphological atherosclerotic changes accompany physiological changes at the vascular wall even in childhood. Increased CIMT 49 and lower FMD 50 were previously reported in paediatric patients with familial hypercholesterolaemia. However, to our knowledge, our study is the first that shows the inverse correlation between these parameters in these children.
The main limitation of our study is the relatively small sample size; moreover, the case–control study described here was unable to determine whether patients with elevated Lp-PLA2 had indeed an increased risk of developing cardiovascular diseases. Future studies should aim to determine the cut-off value for Lp-PLA2 in children; furthermore, cardiovascular events should be serially monitored in dyslipidaemic children to assess whether Lp-PLA2 is indeed a bona fide marker for predicting serious cardiovascular risk.
In conclusion, in this study, we measured Lp-PLA2 concentrations in HD children and compared these measurements with those of CIMT and FMD. Our results show that Lp-PLA2 concentrations are significantly elevated in HD children. Given its association with other markers of atherosclerosis such as TC, LDL-C, CIMT and FMD, and the fact that increased levels of Lp-PLA2 are associated with atherosclerotic changes, we believe that this parameter should be used to determine atherosclerotic changes in children as well as in adults. Not only is the measurement of Lp-PLA2 straightforward and able to be undertaken on automated equipment alongside lipid measurements, but its measurement would also offer the advantage of specificity. Indeed, while lipid parameters such as LDL-cholesterol might be elevated because of confounding factors (e.g. hypothyroidism), Lp-PLA2 is site-specific (being released from the actual plaque) and elevated concentrations would therefore indicate the susceptibility of the plaque to rupture. The determination of cardiovascular risk is extremely important, especially during childhood and in asymptomatic individuals. Even though most of the times, a stenosis is absent, these individuals are prone to cardiac attacks; therefore, the identification of people at risk is paramount and could be achieved by early detection of the change in Lp-PLA2 levels.
The increased incidence of childhood metabolic syndrome, insulin resistance and diabetes mellitus, which are associated with childhood obesity, inevitably increases the risks of developing premature atherosclerosis and cardiovascular disease. Novel markers, like Lp-PLA2, along with the already known markers TC, LDL-C, TG and HDL-C might help physicians in improving the risk analysis of paediatric patients and play a significant role in the detection and monitoring of early atherogenic events in children.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by Ege University Research Fund (2010-TIP-075).
Ethical approval
The study was approved by the Local Ethics Committee on 17 May 2010 with the item no: 10-4.1/30.
Guarantor
FGS.
Contributorship
BO, FGS, TK, EA, and ES conceived the study, performed measurement of Lp-PLA2 and other tests and researched literature. SKU and MC were involved patient recruitment and gaining ethical approval. BO and TK were involved obtaining venous blood samples from all individuals in the patient and control groups. EL conducted CIMT and FMD measurements in a blind setting. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
