Abstract
Background
Although the role of microparticles was recently implicated in stroke pathophysiology, the association between microparticles and inflammation is still not fully understood. The aim of this cohort study of 66 patients was to assess a relation between haemostatic factors, C-reactive protein and clinical outcome of ischaemic stroke.
Methods
Plasma microparticles procoagulant activity, concentrations of tissue factor-bearing microparticles, tissue factor and tissue factor pathway inhibitor in ischaemic stroke patients were determined with enzyme-linked immunosorbent assays at the time of initial diagnosis, along with serum C-reactive protein concentrations. Patients were divided into two groups depending on their C-reactive protein concentrations (C-reactive protein <3 mg/L; n = 28 vs. C-reactive protein ≥3 mg/L; n = 38). The analysed clinical outcome measures included the National Institutes of Health Stroke Scale and the Barthel Index.
Results
The two C-reactive protein groups did not differ significantly in terms of microparticles procoagulant activities, tissue factor-bearing microparticles, tissue factor and tissue factor pathway inhibitor concentrations. A significant correlation was observed between tissue factor pathway inhibitor and National Institutes of Health Stroke Scale score at admission (R = 0.3, P = 0.03). Patients with C-reactive protein ≥3 mg/L presented with significantly higher National Institutes of Health Stroke Scale scores (median, 9.00 vs. 5.50, P = 0.002) and lower Barthel Index scores (median, 20.00 vs. 65.00, P = 0.002) than individuals with C-reactive protein <3 mg/L. The C-reactive protein concentrations correlated positively with National Institutes of Health Stroke Scale scores (R = 0.3, P = 0.02) and inversely with Barthel Index scores (R = − 0.4, P = 0.002).
Conclusions
Altogether, these findings imply that haemostatic parameters (microparticles, tissue factor-bearing microparticles, tissue factor, tissue factor pathway inhibitor) do not account for elevated C-reactive protein concentrations in ischaemic stroke patients.
Introduction
Stroke represents the predominant cause of death and long-term disability of adult patients, thus imposing a large burden on public health. 1 According to the American Heart Association, up to 87% of all strokes result from ischaemia. 2 Although arterial hypertension, heart disease, diabetes mellitus, obesity and smoking are established risk factors for stroke, 3 the exact pathophysiology of this condition is still not fully understood. Nevertheless, haemostatic disturbances and thromboembolism are postulated to be pivotal events in ischaemic stroke pathogenesis.4,5
Circulating microparticles (MPs) are submicron-sized membrane structures involved in a number of biological processes. Different cell types, including blood and endothelial cells, can release MPs on activation, inflammation or during apoptosis. Importantly, while the expression of phosphatidylserine (PS) and tissue factor (TF) on the surface of MPs may enhance their procoagulant activity,6,7 TF is not a prerequisite of MPs-mediated coagulation. 8 TF is the central activator of the coagulation cascade, the activity of which is strictly regulated by the Kunitz-type proteinase inhibitor, tissue factor pathway inhibitor (TFPI).9,10 While the mechanisms of TF and TFPI action are well established, their role in ischaemic stroke remains unclear. We previously demonstrated that stroke patients present with elevated concentrations of tissue factor-bearing microparticles (MPs-TF) and TFPI, as well as with decreased concentrations of TF.4,5
Inflammation likely plays an important role in stroke pathophysiology. 11 Evidence from clinical studies points to elevated C-reactive protein (CRP) as an important risk factor for ischaemic stroke. 12 Notably, Idicula et al. 13 demonstrated that high CRP concentrations at admission are associated with greater severity of stroke and high mortality. Furthermore, elevated CRP concentrations were shown to be associated with an unfavourable functional outcome in patients with ischaemic stroke, as discussed by VanGilder et al. 14
The aim of this study was to investigate if haemostatic factors (MPs, MPs-TF, TF and TFPI) may account for the worse outcome of ischaemic stroke associated with elevated concentrations of CRP.
Materials and methods
Patients
A total of 66 patients with acute ischaemic stroke, aged 36 to 93 y, were included after obtaining a written informed consent, according to the protocol approved by the Local Ethics Committee at Collegium Medicum, Nicolaus Copernicus University (Bydgoszcz, Poland). The patients were recruited between 2011 and 2013, at the Neurology and Stroke Care Unit, Jan Biziel University Hospital No. 2, Bydgoszcz. All subjects were examined by the admitting neurologist, and their baseline data, including sex, age and stroke risk factors, were collected. The diagnosis of ischaemic stroke was established in line with World Health Organization criteria, 15 and further confirmed by non-contrast computed tomography scan of the head. The exclusion criteria of the study were: (1) haemorrhagic stroke, (2) transient ischaemic attack (symptom duration <24 h), (3) systemic malignancy, (4) renal and/or liver dysfunction, (5) sepsis and (6) pregnancy. The final exclusion criterion was the macroscopic evidence of haemolysis or lipaemia in obtained plasma samples. Stroke severity and functional status of the patients at admission were assessed using the National Institutes of Health Stroke Scale (NIHSS) and Barthel Index (BI), respectively. Furthermore, the type of ischaemic stroke was identified according to the Oxfordshire Community Stroke Project (OCSP) classification. A total of 18 patients (27%) received intravenous alteplase (alteplasum, recombinant tissue plasminogen activator, Actilyse 50®, Boehringer Ingelheim, Ingelheim, Germany) at 0.9 mg/kg (up to 90 mg) within 4.5 h of the stroke onset. 16
Definitions of the study variables
Arterial hypertension was defined according to the American Heart Association, as a systolic blood pressure level ≥140 mmHg and diastolic blood pressure ≥90 mmHg or as the need for antihypertensive therapy. Diabetes mellitus was diagnosed whenever at least one of the following criteria was satisfied: treatment with insulin or oral hypoglycaemic agents, or fasting blood glucose ≥7 mmol/L (≥126 mg/dL) on two occasions. Active cigarette smokers were defined as those reporting smoking prior to the stroke onset.
Blood
Blood samples for detection of MPs, MPs-TF, TF and TFPI were collected from each stroke patient at the time of initial diagnosis. The samples were collected into tubes (Becton Dickinson Vacutainer, Franklin Lakes, USA) containing 3.2% sodium citrate (final blood-to-anticoagulant ratio 9:1).
The samples for MPs and MPs-TF determination were centrifuged twice. First, blood was centrifuged at 1500 g for 15 min (room temperature). Then, plasma supernatant was pipetted into Eppendorf tubes and was rapidly centrifuged at 13,000 g for 2 min (room temperature). Obtained material was divided into 200 μL aliquots and stored at −80℃ until analysis. Plasma samples for MPs and MPs-TF determination were never centrifuged at 4℃. Blood for TF and TFPI determination was centrifuged at 1500 g for 20 min (+4℃), the plasma was divided into 200 µL aliquots and stored at −80℃ until analysis. No repeated freeze–thaw cycles were performed prior to analysis.
Determination of MPs, MPs-TF, TF and TFPI
Commercially available enzyme-linked immunosorbent assay (ELISA) kits for MPs, MPs-TF, TF and TFPI determination were used according to the manufacturers’ instructions. Absorbance was read on a plate reader (Multiskan EX, Thermo Fisher Scientific Inc., Waltham, USA). Optical density of the final reaction plate was detected at 405 nm or at 450 nm wavelength. The concentrations of MPs, MPs-TF, TF and TFPI were determined at the Department of Pathophysiology, Collegium Medicum of Nicolaus Copernicus University. To minimize the assay variance, plasma haemostatic parameters were measured on the same day in each individual.
MPs procoagulant activity was determined using a ZYMUPHEN MP-Activity kit (HYPHEN BioMed, SAS, Neuville-sur-Oise, France) and expressed as PS equivalents (nanomolar, nM PS). The kit is a functional assay based on the annexin V ability to bind PS. Plasma sample, supplemented with Ca2+, Xa factor and thrombin inhibitors, was introduced into the microplate well coated with streptavidin and biotinylated annexin V and incubated. After washing, the Xa-Va factor mixture containing Ca2+ was added, followed by prothrombin. If present in the sample, MPs bind to annexin V and expose their phospholipid surface, thus allowing Xa-Va factor mixture to activate prothrombin into thrombin in presence of Ca2+. Phospholipid concentration is directly proportional to the amount of generated thrombin determined on the basis of its specific activity on the thrombin substrate. The detection threshold of the test was ≤0.05 nM. The intra- and inter-assay coefficients of variation (CV) ranged from 3 to 8% and from 5 to 10%, respectively. MPs concentrations in normal human plasma are usually ≤5 nMPS.
The amount of MPs-TF in the plasma was determined with a ZYMUPHEN MP-TF kit (HYPHEN BioMed, SAS, Neuville-sur-Oise, France). The kit includes a micro-ELISA plate coated with a murine monoclonal antibody, specific for an extracellular domain of human TF and non-interfering with TF activity. Following an overnight incubation of the samples and washing thereof, factor VIIa and factor X were added to each wells. The TF–FVIIa complex activates factor X (X→Xa) in presence of Ca2+. Generation of factor Xa is dependent on TF and phospholipids present in the MPs. The kit has been optimized to avoid interferences associated with the presence of MPs that do not bear TF, and does not react with truncated TF or synthetic liposomes. The lower limit of detection was ≤ 1 pg/mL and the intra- and inter-assay CV were <8% and <10%, respectively. MPs-TF concentrations in normal human plasma are usually <2 pg/mL.
Plasma concentrations of TF were determined with a ZYMUPHEN Total Tissue Factor kit (HYPHEN BioMed, SAS, Neuville-sur-Oise, France), a two-site, enhanced, immunoassay for truncated and full-length human TF (total tissue factor). The kit has been optimized to minimize the interference associated with the presence of heterophilic antibodies, as this would result in the overestimation of TF concentration. The detection threshold for the assay was ≤10 pg/mL. The intra-assay CV was 8–9%, and inter-assay CV was 6–7%. The median TF in the healthy volunteers was 97.80 pg/mL (interquartile range (IQR): 89.09–132.96 pg/mL).
The concentrations of TFPI were determined with IMUBIND® TFPI ELISA kit (American Diagnostica GmbH, Pfungstadt, Germany). TFPI was detected using a biotinylated monoclonal antibody specific for the Kunitz domain 1 of native, complexed and truncated TFPI (total tissue factor pathway inhibitor). Binary complexes with factor Xa and quaternary complexes with TF, factor VIIa and factor Xa are also recognized by this test, but with a slightly lower affinity. According to the manufacturer, the intra-assay CV were between 6.2 and 7.1% and inter-assay CV were between 5.5 and 7.3%. The median TFPI in the healthy volunteers was 78.60 ng/mL (IQR: 67.68–105.00 ng/mL).
Determination of CRP
Serum concentrations of CRP (mg/L) at admission were extracted from patients’ medical records. CRP was measured with an immunoturbidimetric assay (laboratory analyzer AU680 Chemistry System, Beckmann Coulter, Brea, USA) at the central hospital laboratory (Jan Biziel University Hospital No. 2, Bydgoszcz, Poland). Based on the results of a previous study, 17 upper normal limit for CRP concentration was set at 3 mg/L.
Data analysis
Statistical analysis was conducted with STATISTICA® 10.0 for Windows (StatSoft, Cracow, Poland). Normal distribution of the analysed variables was verified with Shapiro–Wilk test. As none of the analysed parameters were distributed normally, their statistical characteristics were presented as medians (Me) and IQR. The groups were compared using the Mann–Whitney U test for the numerical data. The chi-square test and the chi-square test with Yates’s correction were used to compare the qualitative data. Furthermore, Spearman correlation coefficients (R) were determined and interpreted according to Zou et al., 18 i.e. R = 0.0 – no association, R = 0.2 – a weak correlation, R = 0.5 – moderate correlation, R = 0.8 – a strong correlation and R = 1.0 – perfect correlation.
The threshold of statistical significance for all the tests was set at P < 0.05.
Results
Demographic and clinical characteristics of the study subjects (Table 1)
Sixty-six patients met the inclusion criteria, including 56% of males (n = 37). Median age of the patients was 69 y (IQR: 62–79 years). As many as 76% (n = 50) of the patients presented with concomitant arterial hypertension, 27% (n = 18) with diabetes mellitus and 35% (n = 23) were smokers. Median NIHSS score at admission was 7 (IQR: 5–11) and median BI score at the time of diagnosis amounted to 35 (IQR: 5–75). According to the OCSP classification, the study subjects presented with partial anterior circulation infarct (58%, n = 38), lacunar infarct (21%, n = 14), posterior circulation infarct (15%, n = 10) and total anterior circulation infarct (6%, n = 4).
Demographic and clinical characteristics of patients with acute ischaemic stroke, stratified according to CRP concentrations.
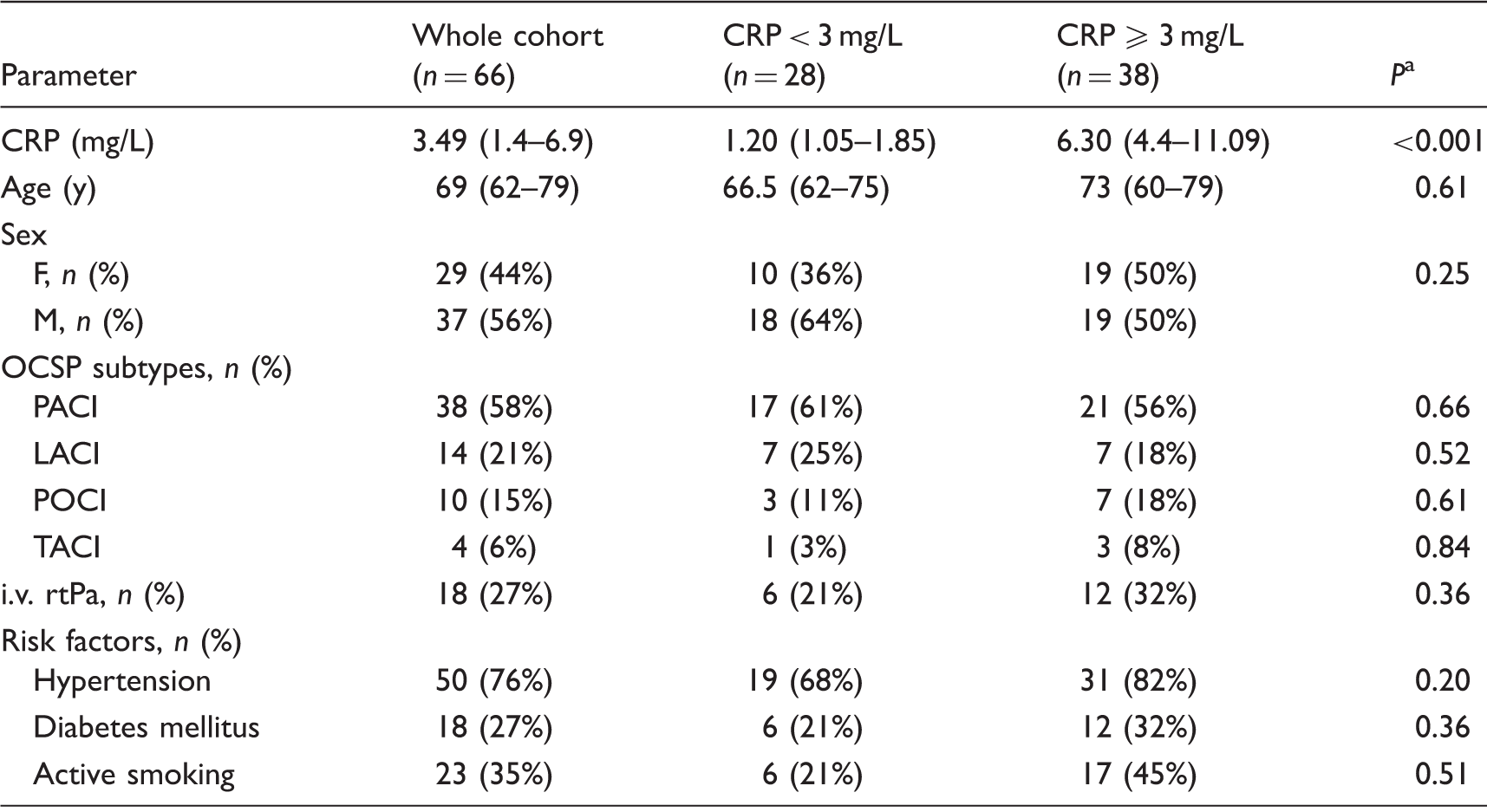
Note: values are median (interquartile range, IQR) or number (%) of patients in each group as appropriate.
CRP: C-reactive protein; F: female; M: male; n: number; OCSP: Oxfordshire Community Stroke Project classification; PACI: partial anterior circulation infarct; LACI: lacunar infarct; POCI: posterior circulation infarct; TACI: total anterior circulation infarct; i.v.: intravenous; rtPA: recombinant tissue plasminogen activator (alteplase).
P value obtained with the Mann–Whitney U test (numerical data) and with the chi-square test/the chi-square test with Yates’s correction (qualitative data).
Plasma MPs activity and plasma concentrations of MPs-TF, TF and TFPI in stroke patients at admission in relation to CRP concentrations
Comparison of MPs procoagulant activity and MPs-TF, TF, and TFPI concentrations in ischaemic stroke patients, stratified according to CRP concentrations.
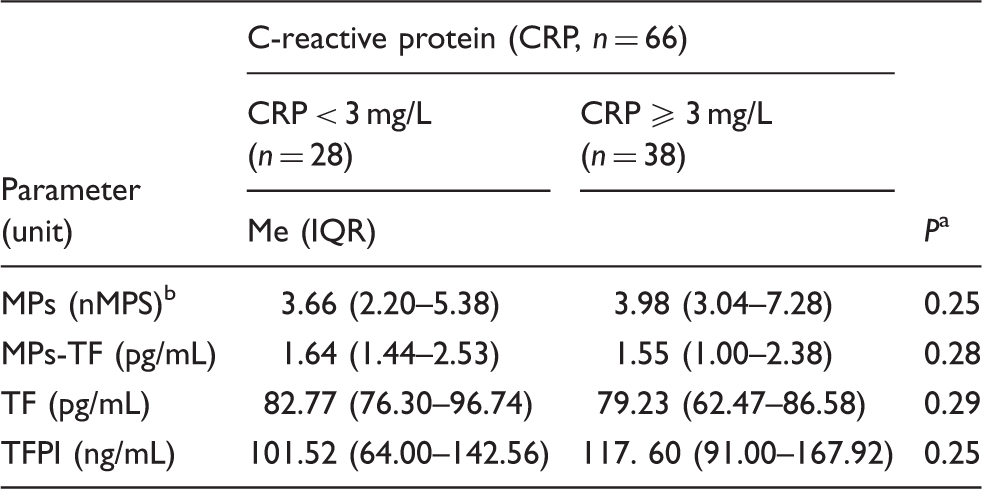
CRP: C-reactive protein; Me: median; IQR: interquartile range; n: number; MPs: microparticles; MPs-TF: tissue factor-bearing microparticles; TF: tissue factor; TFPI: tissue factor pathway inhibitor.
P value obtained with Mann–Whitney U test.
Nanomoles per litre relative to phosphatidylserine.
NIHSS and BI scores at admission of stroke patients in relation to CRP concentrations
Subsequently, we analysed an association between CRP concentrations, stroke severity (NIHSS score) and patients’ disability and handicap status (BI score) at admission. Median NIHSS score turned out to be significantly higher in patients with CRP ≥ 3 mg/L than in those with CRP < 3 mg/L (median: 9.00 vs. 5.50, P = 0.002; Figure 1(a)). In turn, individuals with CRP ≥ 3 mg/L presented with significantly lower BI scores at admission than patients with CRP < 3 mg/L (median: 20.00 vs. 65.00, P = 0.002; Figure 1(b)).
National Institutes of Health Stroke Scale (NIHSS) (a) and Barthel Index (BI) (b) scores at admission in relation to CRP concentrations (P-value obtained by using the Mann–Whitney U test).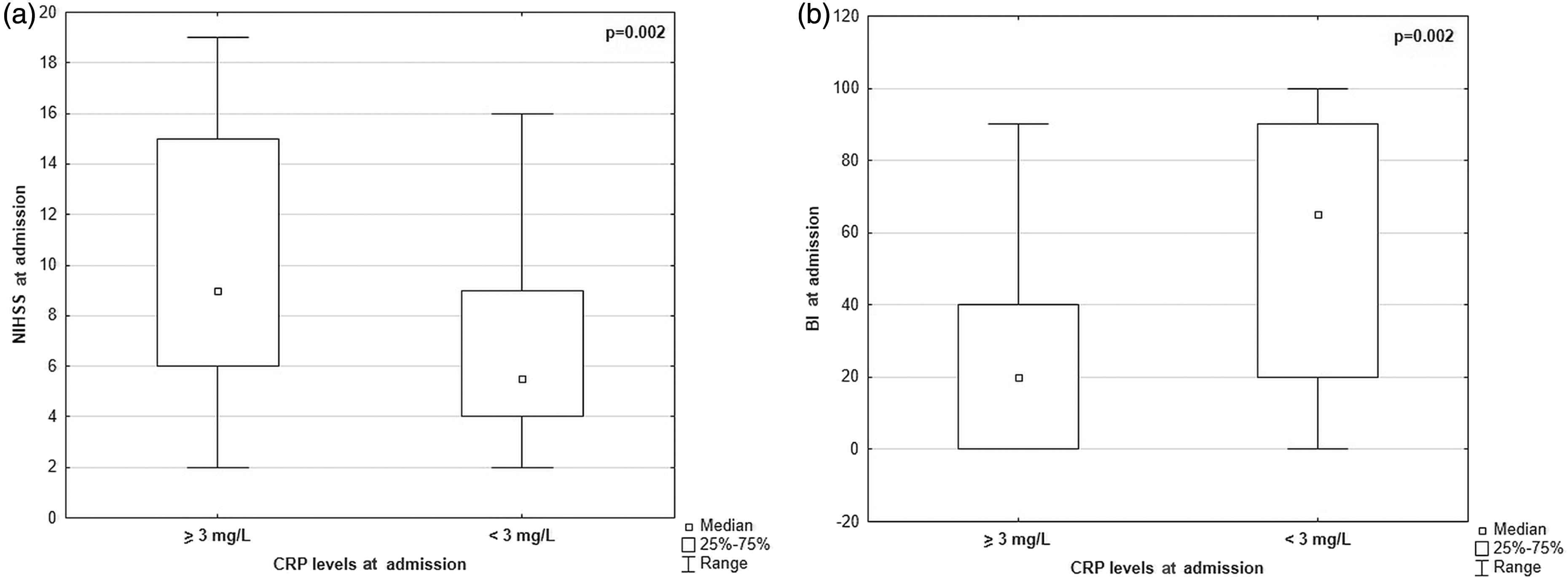
Correlation analysis
Finally, we analysed correlations between haemostatic parameters (MPs, MPs-TF, TF, TFPI), stroke scales (NIHSS, BI) and CRP concentrations.
Interestingly, a weak correlation was found between plasma concentrations of TFPI and NIHSS score at admission (R = 0.3, P = 0.03; Figure 2). Also CRP concentrations showed a weak correlation with NIHSS score at admission (R = 0.3, P = 0.02), as well as a moderate inverse correlation with BI score (R = −0.4, P = 0.002).
Correlation between plasma TFPI concentrations and NIHSS score at admission. TFPI showed significant correlation with NIHSS (Spearman’s R = 0.3, P = 0.03).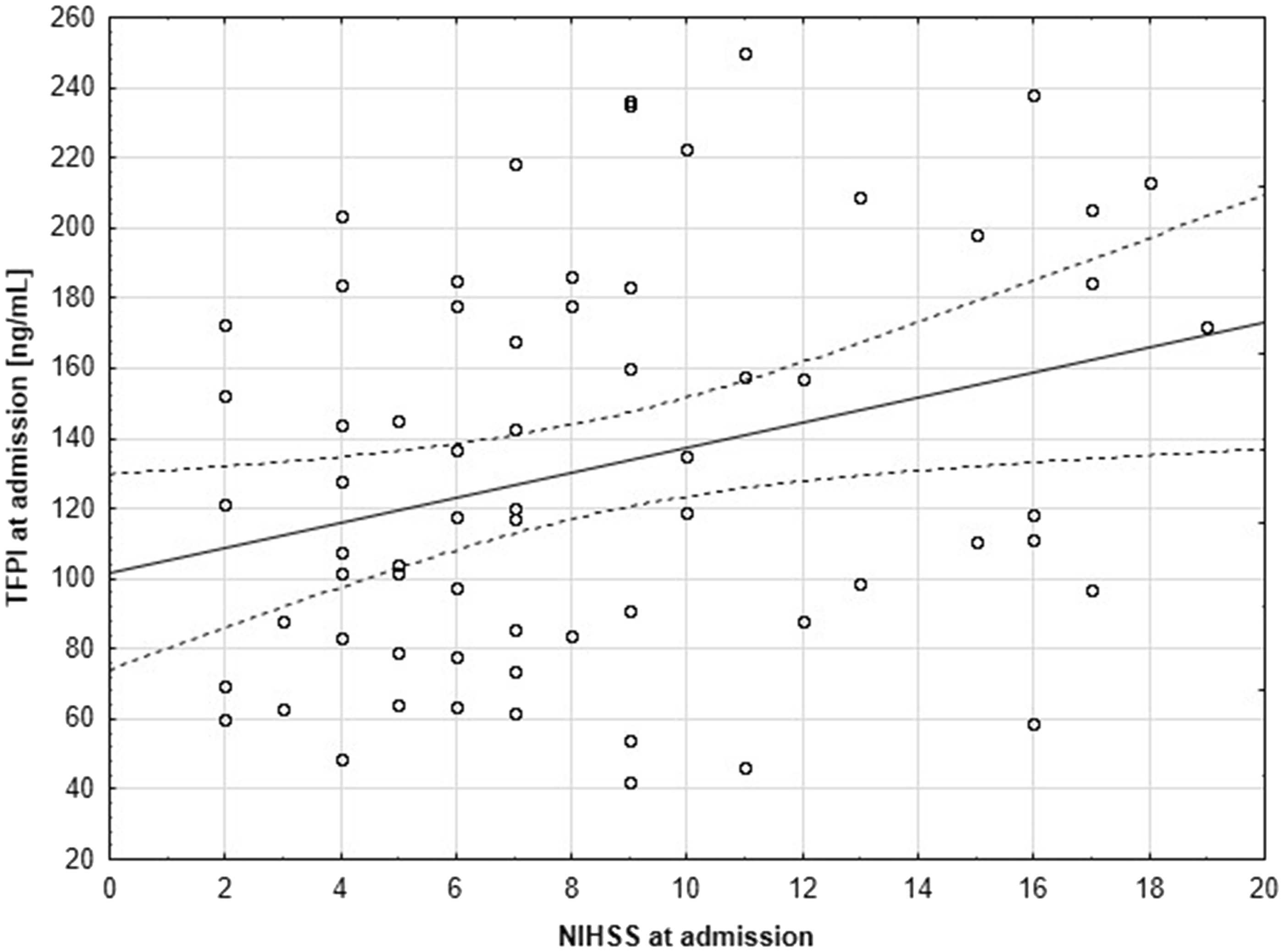
Discussion
The present study demonstrated that: (1) neither MPs procoagulant activity nor the concentrations of MPs-TF, TF and TFPI are associated to CRP concentrations, (2) patients with CRP ≥ 3 mg/L present with significantly higher NIHSS scores and significantly lower BI scores than individuals with CRP <3 mg/L and (3) TFPI concentrations in patients with ischaemic stroke correlate positively with their NIHSS scores at admission.
Despite the fact that elevated CRP is an established risk factor for stroke, we did not find an association between increased concentrations of CRP (≥3 mg/L) and haemostatic parameters. The results of previous studies conducted by Cherian et al. 19 and Simak et al. 20 pointed to involvement of MPs in stroke pathophysiology. Recently, we found increased MPs-TF concentrations in the blood of subjects with ischaemic stroke. 4 According to literature, the association between CRP and MPs may depend predominantly on MPs cellular origin.21–23 Cui et al. showed that endothelial and platelet MPs correlated significantly with CRP concentrations in patients with coronary heart disease, contrary to lymphocytic and monocytic MPs. 21 Chironi et al. 22 demonstrated that platelet and endothelial MPs concentrations in individuals without a cardiovascular disease, presenting with high and low/intermediate CRP concentrations are similar. However, the same study showed that the subjects with elevated CRP presented with higher concentrations of leukocytic MPs than those with normal CRP. Furthermore, Chirinos et al. 23 showed that CRP concentrations in healthy volunteers do not correlated with endothelial MPs expressing an apoptotic or activation phenotype. Unfortunately, the methods used in this study were not suitable to determine the cellular origin of MPs, which should be considered as a limitation of the current findings. Since the increased risk of stroke in individuals with elevated CRP concentrations is a well-established phenomenon, and CRP is an accurate measure of stroke severity, it is unclear why haemostatic factors did not account for elevated CRP in our study. CRP is an acute phase reactant, whose liver synthesis is predominantly mediated by interleukin-6 (IL-6), IL-1 and tumour necrosis factor alpha. Importantly, previous studies documented systemic proatherogenic properties of CRP.24,25 Also IL-6 was shown to be associated with poor outcome and mortality of stroke patients; 26 however, in one study, elevated concentrations of IL-6 did not correlate significantly with procoagulant markers in ischaemic stroke. 27
It is noteworthy that van der Zee et al. 28 identified CRP on the surface of MPs, and MPs from this pool were elevated in subjects with myocardial infarction. Moreover, two conformationally different CRP forms were identified recently: pentameric CRP (pCRP) and monomeric CRP (mCRP), a product of pCRP dissociation (reviewed in Thiele et al. 29 ). As mCRP shows a strong proinflammatory potential, 29 Habersberger et al. examined a link between MPs and mCRP. 30 The results of their study imply that circulating MPs are involved in conversion of pCRP to mCRP, and perhaps also the transfer of mCRP to the cell surface. 30 It would be advisable to evaluate a relationship between all cellular types of MPs and both CRP forms. Furthermore, our results do not exclude the possibility that MPs constitute the surface for mCRP generation in ischaemic stroke patients.
The results of this study suggest that CRP concentrations at admission may be associated with stroke severity and patient’s outcome, since individuals with elevated CRP presented with higher NIHSS scores and lower BI scores at admission. Furthermore, significant correlations were found between the values of both stroke scales results and CRP concentrations at admission. These observations are consistent with the results of a previous study. 13 Besides, CRP concentrations and NIHSS score were postulated to be equally accurate predictors of mortality. 31 While we did not use a high-sensitivity CRP assay in this study, also previous research conducted with the same method documented significantly worse outcomes in ischaemic stroke subjects with elevated CRP concentrations at discharge. 32
Interestingly, this study showed that total concentration of TFPI correlated positively with NIHSS score at admission. Previously, we demonstrated that individuals with ischaemic stroke present with higher TFPI concentrations than healthy controls. 4 TFPI is a physiological regulator of TF-induced blood coagulation, which can inhibit both activated factor X and activated factor VII–TF complex. TFPI is predominantly synthesized in endothelium, and approximately 80% of this compound is bound to endothelial cells. Out of the remaining 20%, 8% blood TFPI is bound to low-density lipoproteins, and 10% occurs as free full-length polypeptide.33–35 In general, increased TFPI concentrations are associated with atherothrombotic disease, reflecting the amount of endothelial perturbation and platelet activation. 36 Adams et al. found that the activity of TFPI, but not free TFPI concentrations, was higher in individuals in an acute phase of stroke than in the controls. 37 In contrast, Rossouw et al. 38 demonstrated that free TFPI concentrations, but not total TFPI or TFPI activity, were associated with stroke risk. One potential limitation of our study stems from the fact that we measured only total concentrations of TFPI. Furthermore, we did not find a correlation between TFPI and CRP concentrations, which implies that the former does not influence the acute phase response in humans. 39 With no doubt, more studies are needed to accurately clarify the results of this experiment and previous37,38 studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the Local Ethics Committee at Collegium Medicum, Nicolaus Copernicus University (Bydgoszcz, Poland).
Guarantor
E Ż .
Contributorship
AS and MŚ contributed equally to this work. AS and MŚ contributed to patient recruitment and compilation of data, analysed the plasma samples, performed statistics and wrote first manuscript. EŻ designed research and discussed the draft manuscript. WŚ discussed the draft manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
