Abstract

We read with interest the article by Grasko et al. 1 on heterophilic antibody interference affecting adrenocorticotropic hormone (ACTH) immunoassay in a patient with Cushing’s syndrome. We describe a case of multiple immunoassay interferences complicating management of Cushing’s disease. A 54-year-old female was diagnosed with Cushing’s disease based on investigations on the Roche Cobas e602 and Siemens Centaur analyzers including an elevated plasma ACTH and high-dose dexamethasone suppression test that showed > 50% suppression of baseline serum cortisol. The patient underwent a transsphenoidal left hemi-hypophysectomy. Histopathology showed anterior pituitary gland tissue with moderate-to-strong staining for ACTH. Her blood pressure and glycaemic control improved with cessation of all antihypertensive agents and insulin. However, one week after the surgery, her serum cortisol concentrations on the Beckman Coulter DxI 800 analyzer were still within the normal range with lack of diurnal variation (cortisol results, 193–307 nmol/L). Given no biochemical cure, the patient underwent a completion hypophysectomy two weeks later. Histopathology showed anterior pituitary gland tissue with no abnormal cells. Plasma ACTH concentrations were unexpectedly high on the Siemens Immulite 1000 analyzer (ACTH, 20 pmol/L). Serum follicle-stimulating hormone (FSH) and luteinizing hormone (LH) were also detectable on the Beckman Coulter DxI 800 analyzer (FSH, 2.4 IU/L; LH, 2.8 IU/L). The remaining pituitary hormones were low, consistent with total hypophysectomy. An ectopic ACTH-secreting lesion was excluded with negative whole-body CT and Gallium DOTATATE positron emission tomography (GaTATE PET) scans.
Investigations for assay interferences.
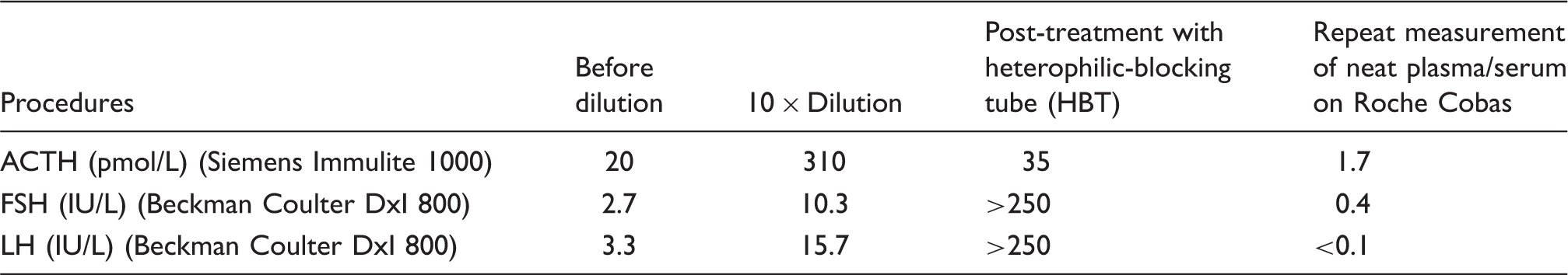
All laboratories should adopt a clear diagnostic algorithm to identify and investigate immunoassay interferences. 7 While the laboratory may hold the necessary expertise to investigate, the onus of identifying potential cases of interferences is on the astute clinician as well as the laboratory.8,9
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Patient consent obtained. No ethical approval required as all tests performed were part of routine investigation and management.
Guarantor
KWC.
Contributorship
JT and CYT were the attending physicians. KWC, NW and JG analysed the samples and data. KWC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
