Abstract
Background
As plasma is the recommended sample type for Roche adrenocorticotropic hormone (ACTH) assay, we evaluated the effect of EDTA concentration on Cobas ACTH assay.
Methods
Samples containing twofold and fourfold higher concentrations of EDTA were prepared by adding plasma to empty K2EDTA tubes and by making under-filled EDTA tubes. All measurements were performed with four replicates.
Results
Increased EDTA concentration resulted in a significant decrease in ACTH concentration. Fifty-per cent-filled EDTA tube showed 19% decrease in ACTH concentration and 25% filled EDTA tube showed 50% decrease in ACTH concentration.
Conclusion
We recommend that inadequately filled EDTA specimens should be rejected when using Cobas ACTH assay.
Introduction
Adrenocorticotropic hormone (ACTH) is known to be subject to proteolytic degradation and previous studies confirmed the instability of ACTH.
1
Rapid processing of specimens is recommended, also usage of aprotinin tubes and addition of maleimide was reported to improve
Siliconized K2EDTA and K3EDTA tubes are the recommended specimen type according to Roche ACTH assay sheet and many other manufacturers. EDTA interference should be evaluated when plasma is the specimen of choice as insufficient sample volume may increase EDTA concentration in the sample. In the present work, we evaluated the effect of EDTA on Cobas ACTH assay.
Material and methods
Samples containing twofold and fourfold higher concentrations of EDTA were prepared by adding plasma to empty K2EDTA tubes. Pooled plasma was prepared from anonymous patient samples. Three millilitres of plasma were added to empty K2EDTA tubes and mixed by inverting the tube to dissolve EDTA. The process was repeated for two times and four times to obtain the intended EDTA concentration.
The same process was repeated with whole blood by adding pooled whole blood to empty K2EDTA tubes to increase EDTA concentration.
Furthermore a completely filled, a 50% filled and a 25% filled EDTA tube were collected from a subject to confirm the effect.
We used 3 mL K2EDTA tubes (BD Diagnostics, Franklin Lakes, NJ, USA) for sample collection. Collection tubes contain 5.4 mg spray-coated K2EDTA. EDTA concentration in completely filled, 50% filled and 25% filled tubes are 1.8 mg/mL, 3.6 mg/mL and 7.2 mg/mL whole blood respectively.
Plasma pools were prepared at two different ACTH concentrations (21.3 ng/L vs 378.4 ng/L). ACTH was measured in four replicates on Cobas E 411 analyzer (Roche Diagnostics). Measurements were conducted in 1 h after preparation. The limit of detection of the ACTH assay is 1 ng/L and the within-run coefficient of variation (CV) of the ACTH assay was 2.9% at a concentration of 4.9 ng/L and 2.0% at a concentration of 74.2 ng/L per manufacturer data sheet. Results were compared with one-way analysis of variance test.
Results
ACTH values in different EDTA concentrations.
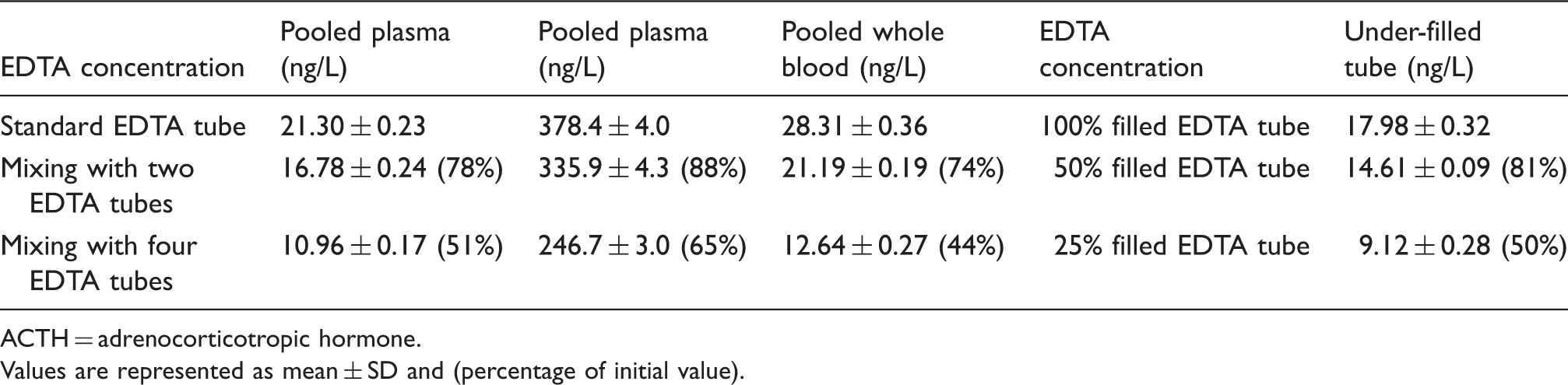
ACTH = adrenocorticotropic hormone.
Values are represented as mean ± SD and (percentage of initial value).
Discussion
Cobas ACTH assay is composed of three reagent components: Streptavidin-coated microparticles, Biotinylated monoclonal ACTH-specific antibody and Ruthenylated monoclonal ACTH-specific antibody. EDTA possibly affects the binding kinetics of the antibodies used in reaction. EDTA is a chelating agent, binding divalent cations. Binding of cations present in serum, can change antigen conformation and the measurable analyte concentration.2,3
İncreasing EDTA concentrations resulted in a considerable decline in ACTH concentrations. Given the <3% within-run CV of Cobas ACTH assay, >10% decline which can be caused by a 50% filled EDTA tube is a highly remarkable analytical effect. Our results suggest that appropriate volume of blood draw is an important factor for correct measurement of ACTH. Under-filled EDTA tubes should be considered unsuitable for ACTH measurement. There is not a widely accepted definition for total allowable of ACTH however, College of American Pathologists (CAP) reported a maximum acceptable intra-assay CV of 15.2% and Royal College of Pathologists of Australasia (RCPA) reported a maximum error limit of ±9.0 ng/L for <90 ng/L and ±10% for >90 ng/L.4,5 Fifty-per cent-filled EDTA tube demonstrated a 19% decline in ACTH concentration which is beyond the CAP allowable limit but falls within the RCPA allowable error limit. We think that this interference has the potential to affect clinical decision making especially in low concentrations. Low ACTH concentrations are used for distinction of ACTH-independent and ACTH-dependent Cushing’s syndrome. In addition, low plasma ACTH is considered a useful parameter for the diagnosis of subclinical Cushing’s syndrome in patients with an adrenal incidentaloma. 4 It was previously discussed that even best performing assays yielded 15–20% misclassifications at low ACTH concentration 6 and EDTA interference in Cobas ACTH assay may increase the number of misclassified patients.
The limitation of the study is that we did not directly measure EDTA concentration in samples; however, the under-filled tube confirms the effect. The distribution of EDTA in plasma water and blood cells is not clear and the accurate EDTA concentration in plasma is unknown.
ACTH is a parameter that is affected by many preanalytical factors such as temperature, proteolysis and hemolysis. In addition to these factors, we report that Cobas ACTH assay is affected by EDTA concentration in sample. We recommend that inadequately filled EDTA specimens should be rejected when using Cobas ACTH assay.
Footnotes
Declarations of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
None.
Contributorship
BT and AC designed the study and wrote the paper; HY and EA contributed to data analysis and interpretation. All authors reviewed and edited the paper. All authors approved the final version of the manuscript.
