Abstract
Background
Endocan (endothelial cell-specific molecule-1) is a proteoglycan and plays an important role in angiogenesis and inflammation. The aim of this study was to evaluate of serum and urinary concentrations of endothelial cell-specific molecule-1 in bladder cancer.
Methods
The study included 50 bladder cancer patients, 50 with urinary tract infection and 51 healthy volunteers. Serum and urinary endothelial cell-specific molecule-1 concentrations were measured with enzyme linked immunosorbent assay.
Results
In bladder cancer group, serum and urinary endothelial cell-specific molecule-1 concentrations were significantly higher than in the healthy subjects (P = 0.003 and P < 0.0001). Urinary endothelial cell-specific molecule-1 concentrations in cases with urinary tract infection were higher than in healthy volunteers (P = 0.002). There were no significant differences between bladder cancer and urinary tract infection groups in terms of serum and urinary endothelial cell-specific molecule-1 concentrations. Urinary endothelial cell-specific molecule-1 concentrations were higher than those of corresponding serum endothelial cell-specific molecule-1 concentrations (P < 0.0001 for bladder cancer and urinary tract infection groups, P = 0.002 for healthy subjects). In bladder cancer group, there was a positive correlation between serum endothelial cell-specific molecule-1 and urinary endothelial cell-specific molecule-1 concentrations (r = 0.32, P = 0.002). For serum endothelial cell-specific molecule-1, sensitivity and specificity were 50%, and 77%, and for urinary endothelial cell-specific molecule-1, 62%, and 71%, respectively.
Conclusion
Serum and urinary endothelial cell-specific molecule-1 concentrations increase in bladder cancer. This parameter also increases in serum and urine of cases with urinary tract infection. That urinary endothelial cell-specific molecule-1 values were higher than serum endothelial cell-specific molecule-1 values in all groups may be attributed to direct exfoliation of epithelial cells in bladder to urine.
Keywords
Introduction
Bladder cancer is the second most common cancer of the genitourinary system, the prognosis of which varies depending on the type. 1 The majority of patients are aged over 50, mean age being in the 70s. The incidence is four times higher in males than in females. 2 Interaction of various genetic and cancerogenic factors is thought to play a significant role in the aetiology. Genetic polymorphism has also been shown to play a significant role in cancer development and progression. 3
Bladder cancer can be classified as superficial (pTa, pT1 and CIS) and muscle-invasive (pT2-4) cancer based on whether tumour infiltration extends to the muscular bladder wall. 4 On the other hand, the revised World Health Organization classification proposes a distinction of non-invasive (pTa) and invasive (pT1-4) bladder cancers. 5
The gold standard for diagnosis of bladder cancer is cystoscopic transurethral resection, which establishes diagnosis and permits both pathological staging and treatment in superficial bladder cancers. Pathological staging of bladder cancer, determination of degree of disease and treatment strategy and estimation of prognosis are also possible with histopathological analysis of tissues. 6 However, cystoscopy is an expensive and invasive technique with high morbidity that results in patient discomfort. The identification of simple, low-cost, non-invasive methods for the diagnosis and monitoring of bladder cancer is therefore important.7–9
The heterogeneous character of the disease may make clinical and pathological parameters non-significant. New molecular markers are therefore needed in the diagnosis and prognosis of bladder cancer.10–12 Biochemical research is being conducted in order to identify novel tumour markers for use in predicting the morphological characteristics of tumours and ın predicting dispersion. This research has been intended to contribute to understanding the natural behaviour of bladder tumours. One of the molecules investigated for that purpose is endothelial cell-specific molecule-1 (ESM-1).
ESM-1 is 50 kDa proteoglycan constituted of 165 amino acid mature protein core (20 kDa), and a unique chondroitin/dermatan sulphate chain linked to serine residue at position 13713,14 and is primarily produced by kidney, liver, lung and gastrointestinal system endothelial cells.15–17 It plays a role in tumour growth and angiogenesis in cancer. Tumour angiogenesis is an important factor in recurrence and progression, and is an indicator of poor prognosis.18,19
Although ESM-1 parameter has been studied in some different tumours, there is only one study measuring blood (not urine) ESM-1 concentrations in bladder cancer. On the other hand, concordant results related to this parameter in inflammation have been published in the literature. Thus, the significance of the ESM-1 concentrations both in urine and serum of patients with bladder cancer is not clear yet.
The purpose of this study was to measure serum and urine ESM-1 concentrations in cases of bladder cancer and to compare those findings with serum and urine ESM-1 concentrations in patients with urinary tract infection (UTI) and healthy individuals. The study also investigated whether serum and urine ESM-1 concentrations are related to pathological staging in bladder cancer.
The fact that no previous study has investigated urine ESM-1 concentrations in bladder cancer means that this study is the first on the subject.
Materials and methods
Study population
The study was performed following approval from the Atatürk University Faculty of Medical Ethical Committee (No. B.30.2.ATA.0.01.00/22). Fifty patients presenting to the Urology Department with macroscopic haematuria and diagnosed with bladder cancer following cystoscopic tissue resection between June 2013 and February 2014 were enrolled as Group 1. Fifty patients with the same demographic characteristics as those in Group 1 and newly diagnosed with UTI comprised Group 2, while 50 healthy individuals with no previous diagnosis of cancer, with normal physical examination and routine test findings were enrolled as the control group, Group 3. All subjects in the study groups were informed about the study and were enrolled once they had given written consent. Inclusion criteria were new diagnosis of bladder cancer or UTI and age between 30–85 years. Healthy subjects aged 30–85 years were also enrolled. Criteria for presence of UTI were leukocytes exceeding 5 per high power field at microscopic analysis of urine, leukocyte esterase exceeding 25/µL at chemical analysis and urine culture positivity.
Exclusion criteria were presence of any malignity other than bladder cancer, hypertension, diabetes mellitus and cardiovascular system disorders being in receipt of active chemotherapy, immunotherapy or radiation, or a history of endoscopic invasion or surgery involving the urinary system in the previous three months.
Blood specimens
Blood samples were collected from the patient and control groups for routine biochemical tests, before any drug therapy or surgical procedure. These were first stored for coagulation and then centrifuged for 15 min at 4000 g at + 4℃. The serum samples obtained divided into aliquots and stored in a deep freeze at −80℃ until analysis.
Urine specimens
Urine samples were collected from the patient and control groups for routine urinary tests, before any drug therapy or surgical procedure. They were then centrifuged for 5 min at +4℃ at 2000 g. The supernatant obtained was divided into aliquots with the help of an automatic pipette. Specimens were stored in a deep freeze at −80℃ until analysis.
Analyte assay techniques
Serum and urine ESM-1 were measured on the same day in order to avoid any variation. ESM-1 concentrations in serum and urine samples were measured using enzyme linked immunosorbent assay (ELISA) with a ‘Human ESM-1 ELISA Kit’ (Lunginnov, Lot: LIK-1205-006, France). It uses a double-antibody ELISA. Samples like serum and urine and standards were pipetted into the 96-well microplate being coated on with a monoclonal antibody (also known as Capture Antibody that is specific for C-terminal of human ESM-1) and incubated for 1 h. ESM-1 present within a sample is bound by the capture antibody. After washing away of any unbound molecules, a secondary monoclonal antibody specific for N-terminal of ESM-1 that has been biotinylated, was added to the wells to incubate for another 1 h. After a washing step, streptavidin-horseradish peroxidase (HRP) (biotin-binding protein conjugated with polymers of HRP) was added and allowed to incubate for 30 min (protected from light). Unbound material was washed away. Chromogen solution was added and incubated for 10 min (protected from light) for the conversion of the colorless solution to a blue solution, the intensity of which was proportional to the amount of ESM-1 in the sample. As an effect of the acidic stop solution, the colour has become yellow. The coloured reaction product was measured using an automated ELISA reader at 450 nm. The results were expressed as picograms per milliliter (pg/mL).
The performance characteristics of this kit from the manufacturer’s sheat are given as: limit of quantification 300 pg/mL, detection 300 pg/mL to 10,000 pg/mL, intraassay precision 4.8%, interassay precision 7.6%. No interference was observed with haemolysed or hyperlipidaemic plasma or serum. We tested intra and interassay CVs for urine and found 5.8% and 9.3%. The values beneath the detection limit of 300 pg/mL were regarded as ‘0’.
Since urine samples were diluted two times for pH adjustment, and the results were multiplied by 2. The urinary ESM-1 concentrations obtained were divided by creatinine values measured from the same urine specimens in order to avoid dilution effects within the day, and the results were expressed as pg/mg creatinine. Urinary creatinine concentrations were measured using commercial kit by the colorimetric modified Jaffe method on a Beckman Coulter AU5811 device (Japan). The results were expressed as mg/dL.
Statistical analysis
Data recording and analysis were performed on ‘SPSS 20.0 for Windows’ (SPSS Inc., IL, USA) software. Descriptive data were expressed as number and percentage for categoric variables and median (minimum and maximum, 95% confidence interval [CI]) or mean ± standard deviation for numeric variables. Compatibility with normal distribution of age and serum and urine ESM-1 results was assessed using the Kolmogorov-Smirnov test. Since age results were normally distributed, comparisons of age among the groups were performed using parametric one-way ANOVA, while degree of significance of differences between groups was determined using the post hoc Tukey test. Since serum and urine ESM-1 results were not normally distributed, the Kruskal-Wallis test was used for comparisons between groups. The Mann Whitney-U test was used for two-way comparisons. Correlations between results were assessed using Spearman’s Rank correlation analysis. Difference in terms of gender distribution between groups was analysed using the chi square test. The Wilcoxon test was used to compare cases’ serum and urine ESM-1 concentrations. The ROC curve, an expression of a particular technique’s predictive power, was used to determine sensitivity, specificity and cut-off values for serum and urine ESM-1. A P < 0.05 was regarded as significant.
Results
Mean ages were 67.0 ± 8.9 years in the 50 patients with bladder cancer, 64.1 ± 10.9 in the 50 patients with UTI and 64.1 ± 11.1 in the 51 healthy controls. No significant difference was determined between the groups in terms of age (ANOVA, P = 0.26).
Four (8%) of the cases with bladder cancer were females, and 46 (92%) were males. Six (12%) of the cases with UTI were females and 44 (88%) were males, and 10 (20%) of the control group were females and 41 (80%) were males. Intergroup comparison revealed no significant differences in terms of gender distribution (χ2 test, P = 0.21).
Serum ESM-1 concentrations were beneath detection thresholds in four cases with bladder cancer, 11 with UTI and in 15 healthy individuals. Median (minimum–maximum) serum ESM-1 concentrations of all groups are shown in Figure 1. In the bladder cancer group, mean serum ESM-1 concentrations were 631 (0–2880, 95% CI 333–522) pg/mL, compared with 487 (0–2048, 95% CI 410–694) pg/mL in the cases with UTI and 472 (0–1379, 95% CI 351–551) pg/mL in the healthy individuals. A significant difference was determined among the groups in terms of serum ESM-1 concentrations (Kruskal-Wallis test P = 0.015). A two-group comparison revealed that serum ESM-1 concentrations in the bladder cancer group were statistically significantly higher compared with those in the healthy individuals (P = 0.003). There was no significant difference in serum ESM-1 concentrations between the bladder cancer and UTI groups (P = 0.18). There was also no significant difference in serum ESM-1 concentrations between the UTI and healthy control groups (P = 0.16). At a cut-off value of 630 pg/mL, serum ESM-1 concentrations exhibited 50% sensitivity and 77% specificity in differentiating cases of bladder cancer patients from healthy individuals.
Serum concentrations of ESM-1.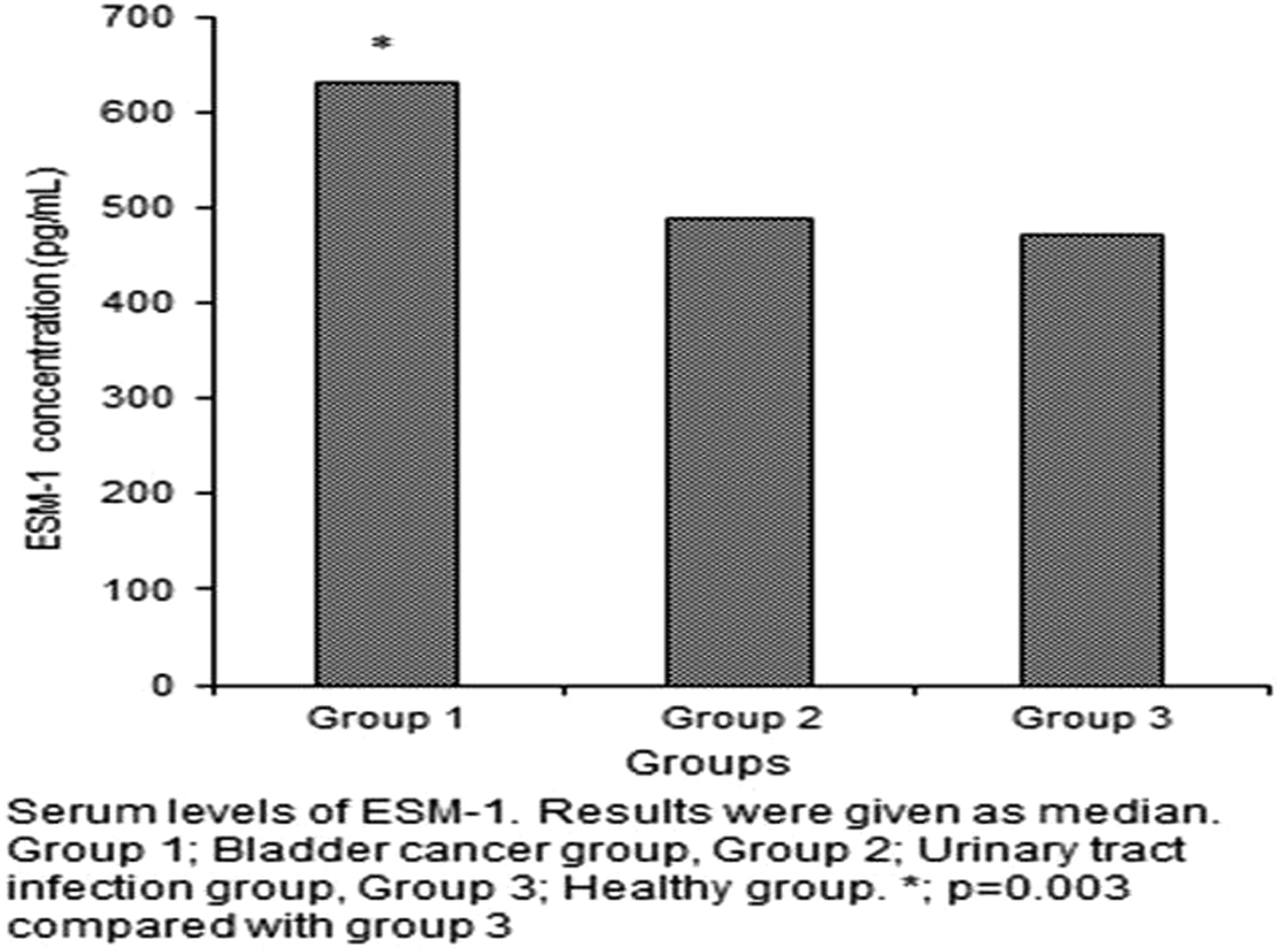
Urine ESM-1 concentrations were beneath detection thresholds only in nine members of the healthy control group. Urine ESM-1 values were expressed as pg/mg creatinine through division by urine creatinine values, and these results were used at statistical analysis. Median (minimum–maximum) urine ESM-1 concentrations for all groups are shown in Figure 2. Urine ESM-1 concentrations were 1514 (463–12840, 95% CI 704–999) pg/mg creatinine in the bladder cancer group, 1281 (211–4040, 95% CI 989–1408) pg/mg creatinine in the UTI group and 627 (0–6217, 95% CI 488–1052) pg/mg creatinine in the healthy control group. A significant difference was determined among the three groups in terms of urine ESM-1 results (Kruskal-Wallis test, P = 0.0001). A two-group comparison revealed no significant difference in urine ESM-1 concentrations between the bladder cancer and UTI groups (P = 0.10), but concentrations were significantly higher in the subjects with bladder cancer group than in the control group (P = 0.0001). Urine ESM-1 concentrations in the UTI group were also significantly higher than those in the control group (P = 0.002). Urine ESM-1 concentrations were 62% sensitive and 71% specific in differentiating cancer cases from healthy individuals. The cut-off value was set at 1100 pg/mL.
Urine concentrations of ESM-1.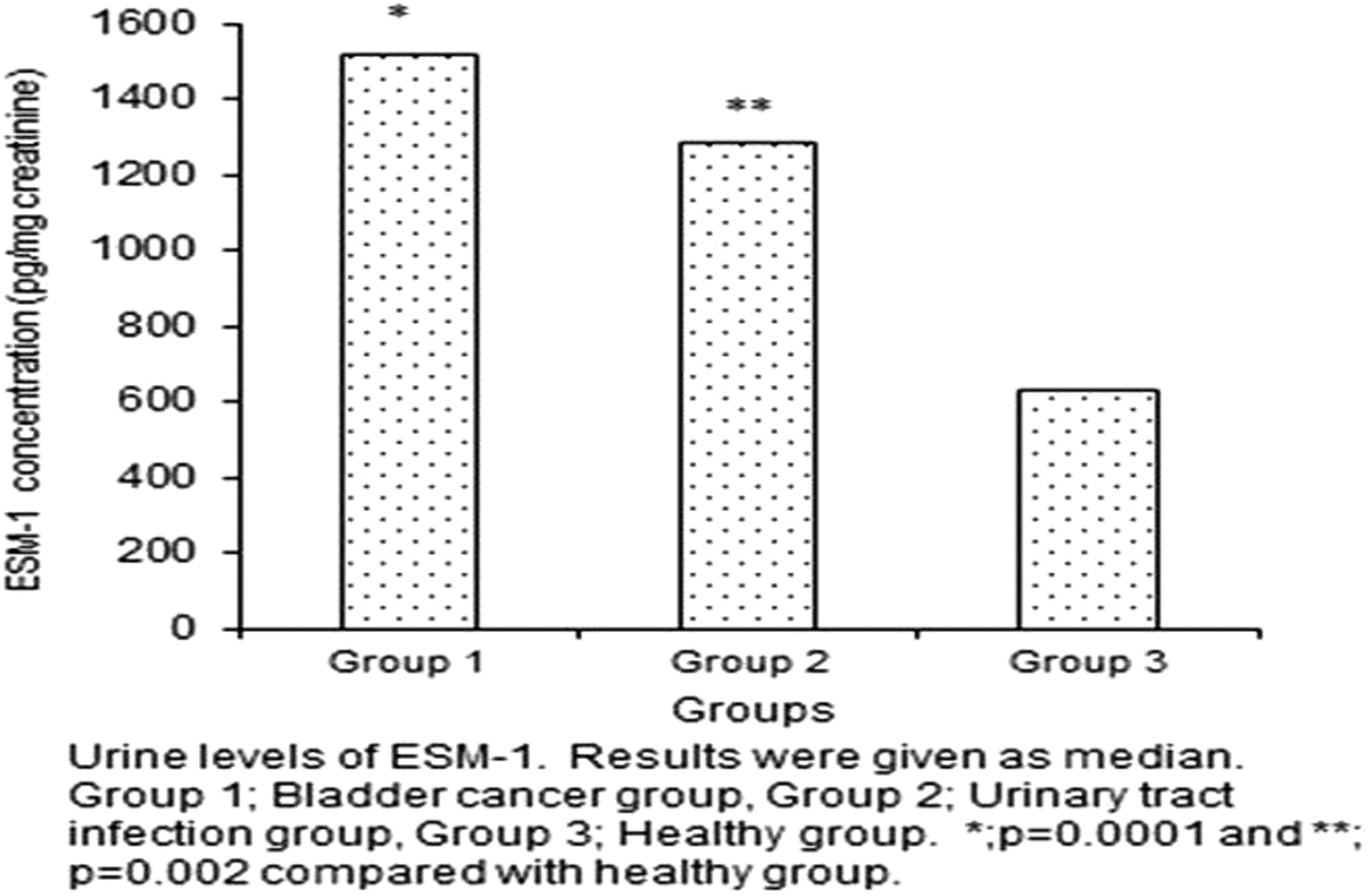
No correlation was determined between serum and urine ESM-1 concentrations when the patient and control groups were analysed together (P = 0.22). When the bladder cancer cases were analysed alone, a significant, positive moderate correlation was determined between serum and urine ESM-1 concentrations (r = 0.32, P = 0.002).
Patients with bladder cancer were staged on the basis of pathology results. Invasive bladder cancer was determined in 20 patients, while bladder cancer was superficial in 30 cases (histologic type of carcinoma was found to be 30 papillary non-invasive [pTa and papillary urothelial neoplasms of low malignant potential] and 20 invasive (pT1-4) tumours of the cases). Nine of the invasive bladder cancer cases were T2a, 5 T2b and 3 T3a, while one individual each was T3b, T4a or T4b. The difference between serum and urine ESM-1 concentrations in the two groups was not statistically significant (P > 0.05). Low-grade tumour was present in 20 of the patients with bladder cancer and high-grade tumour in 30. Patients were grouped on the basis of tumour grades, and statistical analysis was performed to determine any differences between them in terms of serum and ESM-1. Median (minimum–maximum) serum ESM-1 level in the low-grade group was 575 (0–2288) pg/mL and 676 (0–2880) pg/mL in the high-grade group. Serum ESM-1 concentrations exhibited no statistically significant difference between the low- and high-grade groups.
Urine ESM-1 concentrations were 1302 (469–12,840) pg/mg creatinine in the low-grade group and 1549 (463–7494) pg/mg creatinine in the high-grade group. No significant difference was determined between the two groups in terms of urine ESM-1 concentrations.
At individual investigation of cases of bladder cancer, tumour was low grade in all patients with serum ESM-1 concentrations of ‘0’.
Discussion
Bladder cancer is a common type of cancer with high morbidity and mortality. The high probability of bladder tumour recurrence and becoming invasive necessitates early diagnosis of the disease and close monitoring and changes of treatment modality when required. However, the diagnostic value of non-invasive tests such as cytology and tumour markers in early diagnosis and determining prognosis and invasion in bladder cancer is still not at the desired level. The fact that patients require monitoring at close intervals has led researchers to look for non-invasive biochemical markers instead of procedures with high morbidity and mortality. The promising nature of the use of markers such as nuclear matrix protein-22 in showing tumour and recurrence has led to increased interest in such researches.
Angiogenesis, defined as the emergence of new blood vessels, is an important factor in both tumour growth and in spread and metastasis. Studies have also investigated concentrations of endothelial markers involved in angiogenesis, tumour tissues, blood circulation and concentrations in urine.20–22
An increase is seen in concentrations in general circulation of molecules whose expressions increase for various reasons in the vascular endothelium. At the same time, increasingly expressed molecules may also infiltrate urine from the vascular endothelium on the bladder wall. In addition, entry into urine of bladder wall cells also causes an increase of these molecules in urine. Concentrations of potential markers in urine are therefore being investigated in addition to circulation in cases of bladder cancer.23,24
Lokeshwar et al. 25 measured urine hyaluronic acid (HA) from glycosaminoglycans (GAG) involved in tumour angiogenesis and metastasis and concentrations of hyaluronidase, the enzyme that breaks it down. They reported higher concentrations of HA and hyaluronidase in patients with bladder cancer compared with the control group. They determined that urine HA concentrations were 83.1% sensitive and 90% specific in identifying bladder cancer, while the hyaluronidase test exhibited 81.5% sensitivity and 83.8% specificity. In the light of those findings, they suggested that urine HA and hyaluronidase measurements represented tests with high sensitivity and specificity in the diagnosis and ‘grading’ of bladder cancer. 25 ESM-1, the serum and urine concentrations of which were measured in our study, is a proteoglycan containing dermatan sulphate, a member of the GAG family, like HA a marker in both tumour angiogenesis and in inflammatory response. We compared this marker in serum and urine specimens from patients with bladder cancer, patients with UTI and healthy individuals. Serum ESM-1 concentrations in the bladder cancer group were higher compared with healthy individuals but did not differ significantly from those in patients with UTI. There was also no statistically significant difference in terms of serum ESM-1 concentrations between the UTI and healthy control groups. In other words, serum ESM-1 concentrations increase in bladder cancer patients compared with healthy individuals. However, UTI may also cause a rise in ESM-1. In addition, while urine ESM-1 concentrations in cancer patients were not statistically different from those in patients with UTI, they were significantly higher than those of the control group. Urine ESM-1 concentrations in subjects with UTI were also higher than those of healthy subjects, and the difference between them was statistically significant.
Our review of the literature revealed no previous studies measuring urine ESM-1 concentrations. We identified one study investigating blood ESM-1 concentrations in cases of bladder cancer. In that study done, by Roudnicky et al., 26 bladder specimens were collected from normal and invasive bladder cancer, the purpose being to determine the presence and concentrations of ESM-1 in these tissues. Using real time PCR, ESM-1 expression was shown to be 1000–100,000 times higher in cancerous tissue. In addition, plasma ESM-1 concentrations in cases of invasive bladder cancer were also higher than those of healthy individuals. Due to the increase in plasma ESM-1 concentrations, they suggested that these could be used as a prognostic marker in cases of invasive bladder cancer. At a cut-off point of 0.63 ng/mL, plasma ESM-1 concentrations were calculated to exhibit 64% sensitivity and 80% specificity in differentiating invasive bladder cancer from healthy individuals. 26 Serum ESM-1 concentrations exhibited 50% sensitivity and 77% specificity in differentiating our cases of bladder cancer patients from healthy subjects. The sensitivity and specificity results from our study were lower than the values determined by Roudnicky et al. 26 Invasive bladder cancer represented 20 of the cases of cancer in our study, while bladder cancer was superficial in the other 30. In the study cited above, however, all cancer patients had invasive bladder cancer. We attribute this variation in the results to patients without invasion in our study group. In Roudnicky et al.’s study, immune staining of tumour specimens showed the presence of ESM-1 protein in vessels in the area of the tumour, while none was observed in vessels in areas of normal bladder tissue. Furthermore, ESM-1 staining revealed either no staining or diffuse staining in 53 out pf 70 cases of non-invasive bladder cancer. Staining was intense in cells in 23 out of 46 invasive bladder cancer tissues and diffuse in the others. A significant difference was determined between the groups in terms of staining. In conclusion, it was suggested that bladder cancer of an invasive nature increased ESM-1 expression. In that same study, it was shown that VEGF increase ESM-1 expression in vivo and in vitro. 26 Additionally, they determined that the interaction of the ESM-1 molecule with VEGF-A on the cell surface facilitates binding to VEGFR-2. On the basis of these findings, they concluded that increased synthesis of ESM-1, in association with VEGF-A rising due to tumour, might stimulate vascularization. In other words, ESM-1 molecules may be involved in tumour spread by increasing angiogenesis. 26 Cell culture studies have shown that ESM-1 increases the mitogenic activity of hepatocyte growth factor by binding to it through the same mechanism.13,14
Nault et al. 27 reported higher serum ESM-1 concentrations in the hepatocellular cancer cases compared with patients with non-cancerous alcoholic cirrhosis. They stated that serum ESM-1 may be a good marker of angiogenesis and may even be a potential target in the treatment of angiogenesis. Leroy et al. 28 found 3–10 times higher concentrations of ESM-1 in patients with renal papillary cancer compared with healthy individuals. They suggested that ESM-1 may represent a potential test for use in assessing tumour response to antiangiogenic treatments.
Matano et al. 29 investigated the relation between tumour invasion and ESM-1 expression in patients with pituitary adenoma and stated that increasing ESM-1 is involved in tumour angiogenesis and can be used to show pituitary adenoma invasion of the neighbouring cavernous sinus. Another study also reported increased ESM-1 expression in tissue samples from patients with ovarian cancer. 30
In terms of differentiation, cancer cells resemble normal cells both morphologically and functionally. In well differentiated tumours, cells resemble the tissue from which they derive, while in poorly differentiated tumours, cells do not resemble the tissues from which they derive. 31 In a study of patients with stomach cancer, Zhang et al. 32 not only reported elevated ESM-1 expression in cancerous tissue but also revealed the presence of a correlation between level of ESM-1 expression and degree of cancer differentiation. 32 Zuo et al. 33 determined a correlation between ESM-1 expression and cancer differentiation in patients with colorectal cancer.
Serum ESM-1 concentrations rise in patients with untreated acute myeloid leukemia. 34 ESM-1 increased again in individuals with developing complications associated with bacterial infections and antibiotic therapy caused a decrease in serum ESM-1 concentrations. In our study, too, urine ESM-1 concentrations were significantly higher in subjects with UTI than in the control group. This finding supports the idea that the presence of infection may also be responsible for ESM-1 elevation. In addition, urine ESM-1 concentrations were higher than serum ESM-1 concentrations in all groups (data not shown). This may indicate that direct passage of ESM-1 through the bladder wall is greater than that of the molecule into the general circulation.
Lassalle et al. 35 found that ESM-1 mRNA concentrations increased following the addition of TNF-α and interleukin-1β in a human vascular endothelial cell culture environment and they concluded that the ESM-1 expression was regulated through cytokines. 35
Studies have shown that serum ESM-1 concentrations increase in sepsis.17,36–38 Higher ESM-1 concentrations have also been determined in cases of severe sepsis compared with those with mild sepsis. It has therefore been suggested that ESM-1 can be used as a marker in detecting and determining the severity of sepsis. 36
Inflammation also plays an important role in the pathophysiology and complications of hypertension. 39 In a study of patient with essential hypertension, Balta et al. 40 determined higher serum ESM-1 concentrations in patients with essential hypertension than in healthy individuals. They also revealed a positive correlation between high sensitivity C-reactive protein and ESM-1 concentrations. They also stated that increased ESM-1 is a response to vascular injury, and the ESM-1 can provide more useful information in the development and progress of atherosclerosis than non-specific markers such as high-sensitivity C-reactive protein. Additionally, they suggested that increased ESM-1 concentrations in patients with essential hypertension can predict the risk of cardiovascular disease and can therefore be a useful marker in identifying at-risk patients. 40 This is because angiogenesis is a common characteristic of both atherosclerosis and hypertension, and synthesis of ESM-1 has been shown to be induced by proangiogenic factors such as VEGF-A and VEGF-C involved in the progression of cancer lymphoangiogenesis and angiogenesis. 41
Also in some inflammatory diseases related to skin such as Behcet 42 and Psoriasis, 43 increased serum ESM-1 concentrations were found.
In conclusion, serum and urinary ESM-1 concentrations increase in bladder cancer as well as UTI indicating that it may be regarded as both inflammatory and tumour marker. The marked increased in ESM-1 values in urine rather than serum may be attributed to direct exfoliation of epithelial cells in bladder into urine.
Footnotes
Acknowledgements
We would like to thank Dr Hulya Aksoy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Scientific Research Project of Ataturk University (No. 2013018).
Ethical approval
This study approved by the ethics committee of Medical School of Ataturk University (REC number: B.30.2.ATA.0.01.00/22).
Guarantor
LE.
Contributorship
LE designed research, analysed data and wrote the first draft of manuscript; AH performed statistics; AY and ÖF performed clinical monitoring and laboratory detection; AF helped to write English. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
