Abstract
Background
Microbiological culture of cerebrospinal fluid is the gold standard to differentiate between aseptic and bacterial meningitis, but this method has low sensitivity. A fast and reliable new marker would be of interest in clinical practice.
Objective
Interleukin-6, secreted by T cells in response to meningeal pathogens and quickly delivered into cerebrospinal fluid, was evaluated as a marker of acute meningitis.
Design and Methods
A total of 150 cerebrospinal fluid samples were analysed by an electrochemiluminescence method, selected according to patient diagnosis: (a) bacterial meningitis confirmed by positive culture (
Results
Cerebrospinal fluid interleukin-6 concentration showed significant differences between all pathologic groups and the control group (
Conclusion
Interleukin-6 measured by electrochemiluminescence method is a promising marker for early differentiation between aseptic and bacterial meningitis. More studies are needed to validate clinical implications for future practice in an emergency laboratory.
Introduction
Meningitis is the inflammatory response of leptomeningeal cells and the subarachnoid space to injury that can be of infectious, chemical, tumoral or autoimmune aetiology. Acute infectious meningitis represents an important cause of emergency consultation, especially in paediatrics. Viral meningitis has a higher incidence than bacterial meningitis, but its clinical course is usually more benign. 1
In Spain, 3000–4000 new cases of meningitis are declared each year, with a mortality rate of up to 20%. It is estimated that approximately 70% of bacterial meningitis occur in children under five years of age, mainly due to three pathogens:
During the initial phase of either bacterial or viral meningitis, the clinical signs and symptoms are usually non-specific (headache, nuchal rigidity, fever, nausea and convulsions). Whenever meningitis is suspected, cerebrospinal fluid (CSF) is extracted by lumbar puncture to enable an aetiologic diagnosis by means of cell count, as well as biochemical and microbiological studies.
The gold standard for the diagnosis of septic meningitis is microbiological culture. However, this method shows low sensitivity 3 and the diagnosis may be delayed. In untreated patients, Gram staining of CSF reveals the presence of bacteria in 50–80% of cases, and microbiological culture is positive in 85% of cases of bacterial meningitis. 4 However, this percentage can decrease by up to 50% when the patient has been previously treated with antibiotics. Moreover, in microbiological culture, bacterial growth requires at least two days; for viral meningitis, culture may take even longer, although the most common pathogens (enterovirus and herpes simplex virus) can be detected with high sensitivity, by the polymerase chain reaction (PCR), reducing the laboratory turnaround time. Therefore, although microbiologic results are needed to confirm the bacterial infection and optimize the treatment, the initial aetiologic diagnosis of meningitis usually relies on the cytological and biochemical characteristics of CSF.
A cell count higher than 5 or 7 leukocytes/μL is considered pathological in adults and children, respectively, with a differential count being indicated after sample staining. Bacterial meningitis usually develops with pleocytosis (more than 1000 leukocytes/μL) and a clear predominance of polymorphonuclear cells (PMN) over lymphocytes. Conversely, viral meningitis produces a moderate pleocytosis, rarely over 1000 leukocytes/μL, showing clear lymphocyte predominance after the initial stages.5,6 Thus, a leukocyte count does not allow early diagnosis of viral meningitis. Biochemical tests such as glucose and total protein provide complementary information. Viruses are the most common cause of aseptic meningitis (i.e. negative bacterial culture), although it is not always possible to demonstrate the viral origin of meningitis. 5 Therefore, a faster and highly sensitive new method would be of interest in clinical practice.
Interleukin-6 (IL-6) is secreted by T cells, macrophages and fibroblasts. These cells mediate the immune response, and are involved in the regulation of the acute-phase response to injury and infection. 7 Serum and umbilical cord blood IL-6 measurements have been described as a useful tool in the diagnosis of neonatal sepsis. 8
In this study, we hypothesized that IL-6 may be delivered into CSF during septic meningitis. Subsequently, IL-6 may be detected after lumbar puncture and be useful as a biomarker for diagnosis and monitoring of these patients.
Materials and methods
During two periods (January 2008–June 2009 and February 2013–November 2013), 1326 CSF samples were received at the Department of Clinical Biochemistry, Hospital Universitario Central de Asturias (HUCA), Spain. A cohort of 377 patients who were admitted to the emergency department (ED) or intensive care unit (ICU) was retrospectively selected. Patients with a clinical diagnosis of meningitis (fever, headache and meningeal signs) and sufficient sample volume after performing routine tests were included as cases (
The CSF cell count was carried out immediately after specimen collection into sterile tubes following European Federation of Neurological Societies (EFNS) clinical guidelines.
9
One CSF sample was analysed per patient, as a part of their diagnostic process, and a differential leukocyte count was performed on the stained sample, when more than 6 or 20 leukocytes/μL were observed, in adults and children, respectively. After differential count, when needed, all the CSF samples were centrifuged at 1500
Another CSF sample was routinely sent to microbiology laboratory for specific tests according to individual clinical suspicion. Bacterial tests panel included Gram staining, culture and antibiogram. Viral PCR tests were applied for enterovirus, herpes virus-1, herpes virus-2, Epstein-Barr virus, cytomegalovirus and parvovirus. Immunoglobulin G (IgG) test was used for varicella zoster virus.
Consideration of bacterial meningitis depended on clinical criteria and presence of more than 60% PMN in differential white cell count, with total >100 leukocytes/
Samples were classified into five groups, according to the final diagnosis, before being processed for this study: (a) bacterial meningitis confirmed by positive culture (
IL-6 was measured in a Cobas e-601 analyzer (Roche Diagnostics) at the state laboratory of the HUCA (Oviedo, Spain), by an electrochemiluminescence (ECLIA) method able to provide a result within 19 min. This method has been standardized against the National Institute for Biological Standards and Control’s (NIBSC) first international standard (89/548). The limit of detection was 1.5 pg/mL, and linearity up to 5000 pg/mL was observed. When higher IL-6 concentrations were obtained, (>5000 pg/mL) samples were diluted 1:10. However, some samples (
The imprecision of the method had already been estimated by using a pool of plasma samples for low concentrations (experimental mean 3.20 pg/mL), showing 7.6% and 8.0% of intra- and inter-assay variability. 11 Quality control material for medium and high concentrations (PreciControl MultiMaker, 33.3 pg/mL and 210 pg/mL, respectively) showed coefficient of variation (CV) of 3.0% and 1.0%, respectively, for intra-assay, with the inter-assay CV being 4.5% and 3.7%, respectively.
No significant cross-reactions were described with other interleukins: IL-1, IL-2, IL-3, IL-4, IL-8, γ-interferon and TNF-α.
Statistical analysis was performed with SPSS Statistics software (IBM version 19) and MedCalc (version 12.5). A
Results
Population characteristics and classic markers from all patients included in the study.
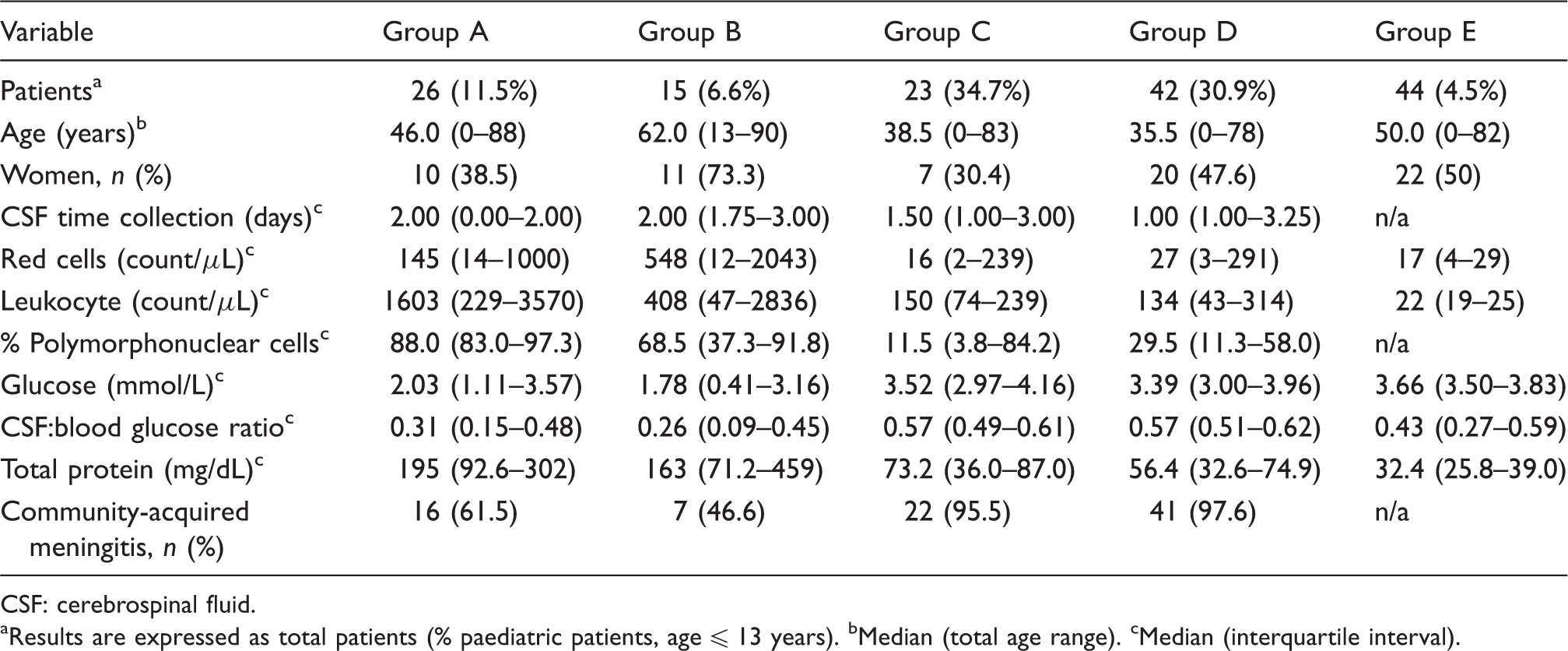
CSF: cerebrospinal fluid.
Results are expressed as total patients (% paediatric patients, age ≤ 13 years). bMedian (total age range). cMedian (interquartile interval).
Diverse pathogens were found in the 49 CSF samples from groups A and C (46.2% of cases). Among those from group A, the most common were
Outcomes of all the followed case-patients stratified according to pathologic groups.
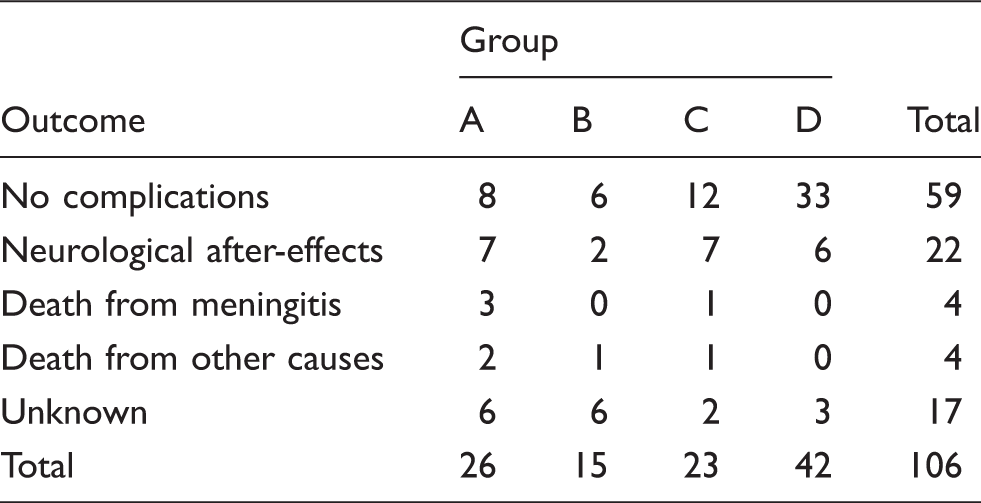
Six patients (40%) in group B (bacterial meningitis not confirmed) had antibiotic therapy administered 72 h prior to admission, but in group A (bacterial meningitis confirmed) this percentage decreased by up to 23.1%. In group B (
The IL-6 concentration was determined for all the groups, as well as the median and 95% confidence intervals (CI) calculated for each group. CSF IL-6 concentration showed significant differences between the control group and all other groups ( Distribution of IL-6 concentrations and % PMN on CSF from different study groups: (a) bacterial meningitis confirmed by positive culture, (b) clinical suspicion of bacterial meningitis with negative or not performed culture, (c) viral meningitis confirmed by polymerase chain reaction (PCR) or IgG determination, (d) clinical suspicion of viral meningitis with negative or not performed PCR and (e) control group.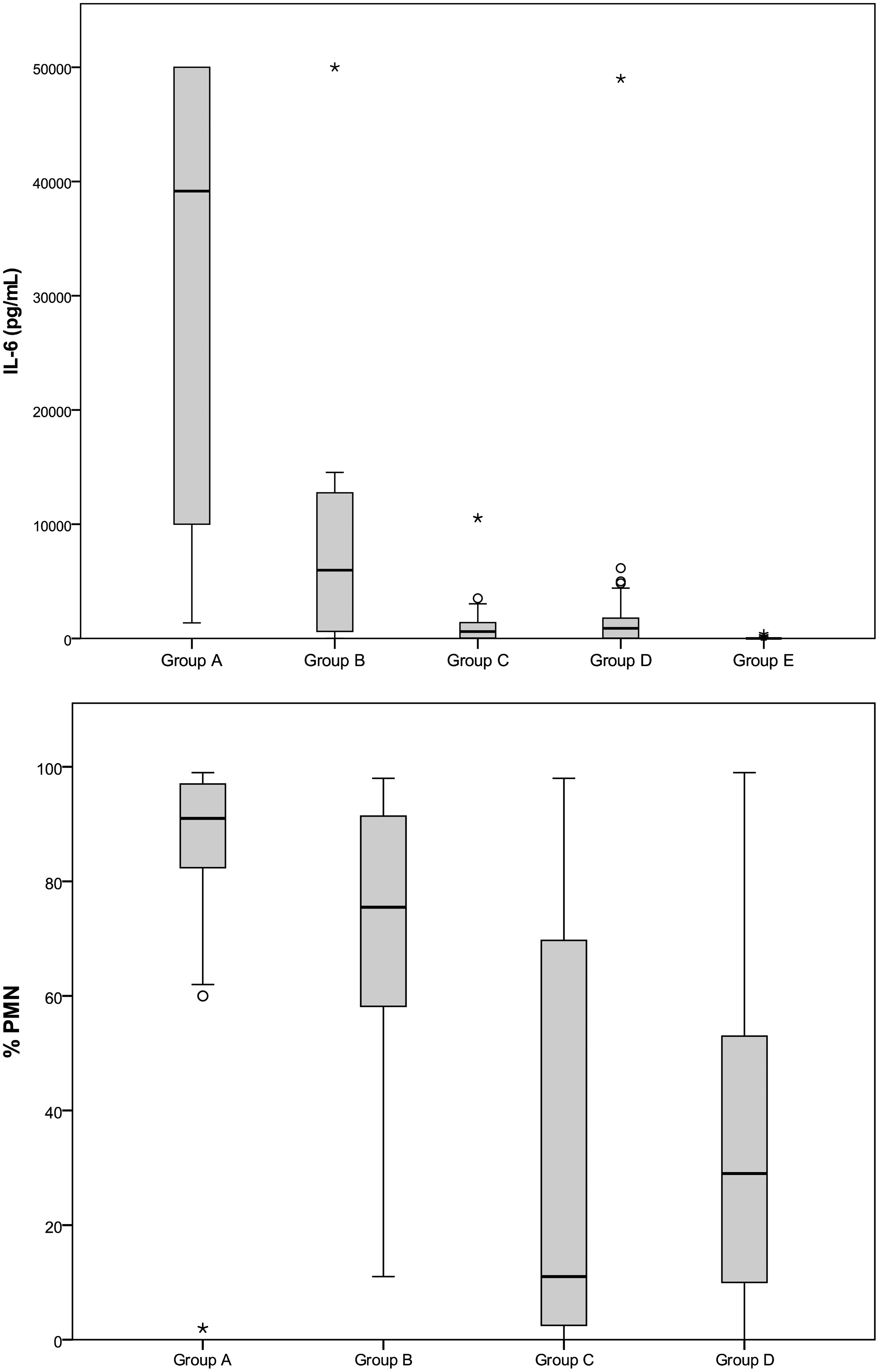
Outlier values in group B (bacterial meningitis not confirmed by microbiological tests).
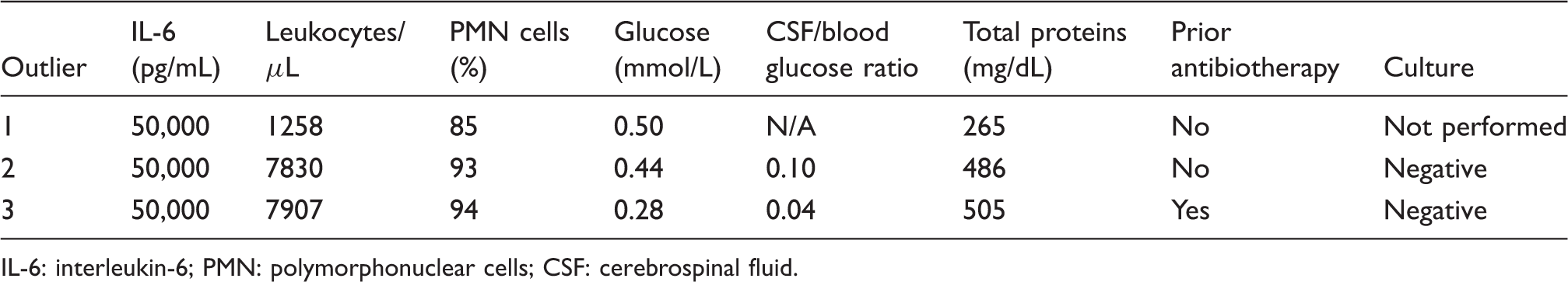
IL-6: interleukin-6; PMN: polymorphonuclear cells; CSF: cerebrospinal fluid.
There was also another outlier value in group D (IL-6: 49,000 pg/mL). Routine tests results were as follows: 304 leukocytes/μL (54% PMN cells), CSF glucose: 4.22 mmol/L, CSF/blood glucose ratio: 0.38; total proteins: 173 mg/dL. No recent prior antibiotic therapy, negative culture and viral studies were performed. This sample could not be classified into group A or C because there was no confirmation by microbiological techniques. Moreover, classical biochemical parameters were discordant: low CSF/blood glucose ratio with low percentage of PMN cells.
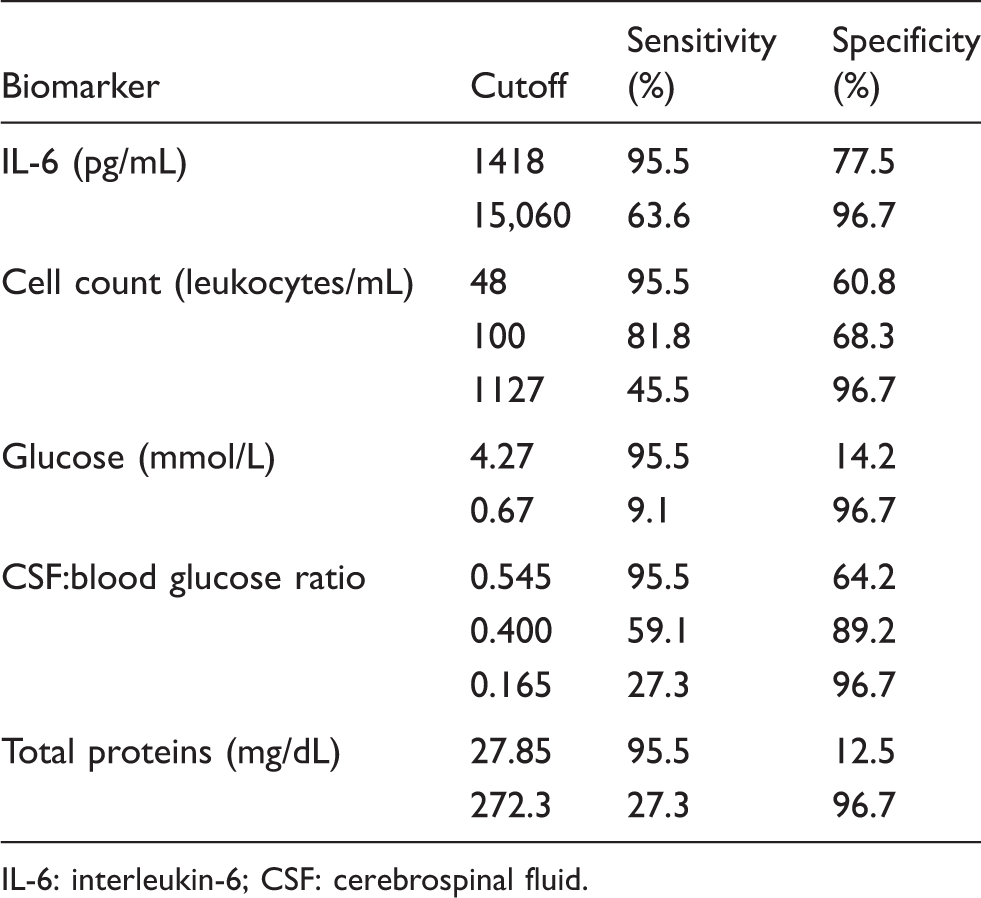
When CSF IL-6 was tested as a diagnostic tool for bacterial meningitis (group A), an area under the curve (AUC) of 0.937 (95% CI: 0.895–0.978) was obtained. Figure 2 shows the comparison between the AUC of CSF IL-6 and classical markers, such as CSF leukocyte count (AUC 0.872; 95% CI: 0.802–0.943), total protein concentration (AUC 0.795; 95% CI: 0.680–0.910) and decrease of CSF/blood glucose ratio (AUC 0.865; 95% CI: 0.791–0.939). Statistically significant differences were found for all three comparisons, demonstrating the higher diagnostic power of IL-6 despite having found some degree of overlap between the respective ROC curves. Selecting a CSF IL-6 cut-off value of 1418 pg/mL, 95.5% sensitivity and 77.5% specificity were obtained for the diagnosis of bacterial meningitis, whereas concentrations higher than 15,060 pg/mL showed 63.6% sensitivity and 96.7% specificity. On the other hand, the best classical biomarker was the CSF/blood glucose ratio, showing 95.5% sensitivity and 60.8% specificity at a cut-off value of 0.545 (Table 4). It is important to point out, however, that in six patients from group A the ratio could not be calculated: two of them because no blood sample had been obtained at admission and the other four because the value of CSF glucose was below the detection limit of our method (<0.11 mmol/L, enzymatic hexokinase reference method); therefore, these patients had to be excluded from the numerical analysis. However, the usual cut-off value of 0.4 for CSF/blood glucose ratio only showed 59.1% sensitivity and 89.2% specificity. Additionally, a conventional cutoff of 100 cells/μL for the leukocyte count only achieved 81.8% sensitivity and 68.3% specificity.
Comparison of the diagnostic power of IL-6 and classic biomarkers for bacterial meningitis confirmed by positive culture (group A). Cut-off points showing the best sensitivity or specificity for each marker on diagnosis of bacterial meningitis. IL-6: interleukin-6; CSF: cerebrospinal fluid.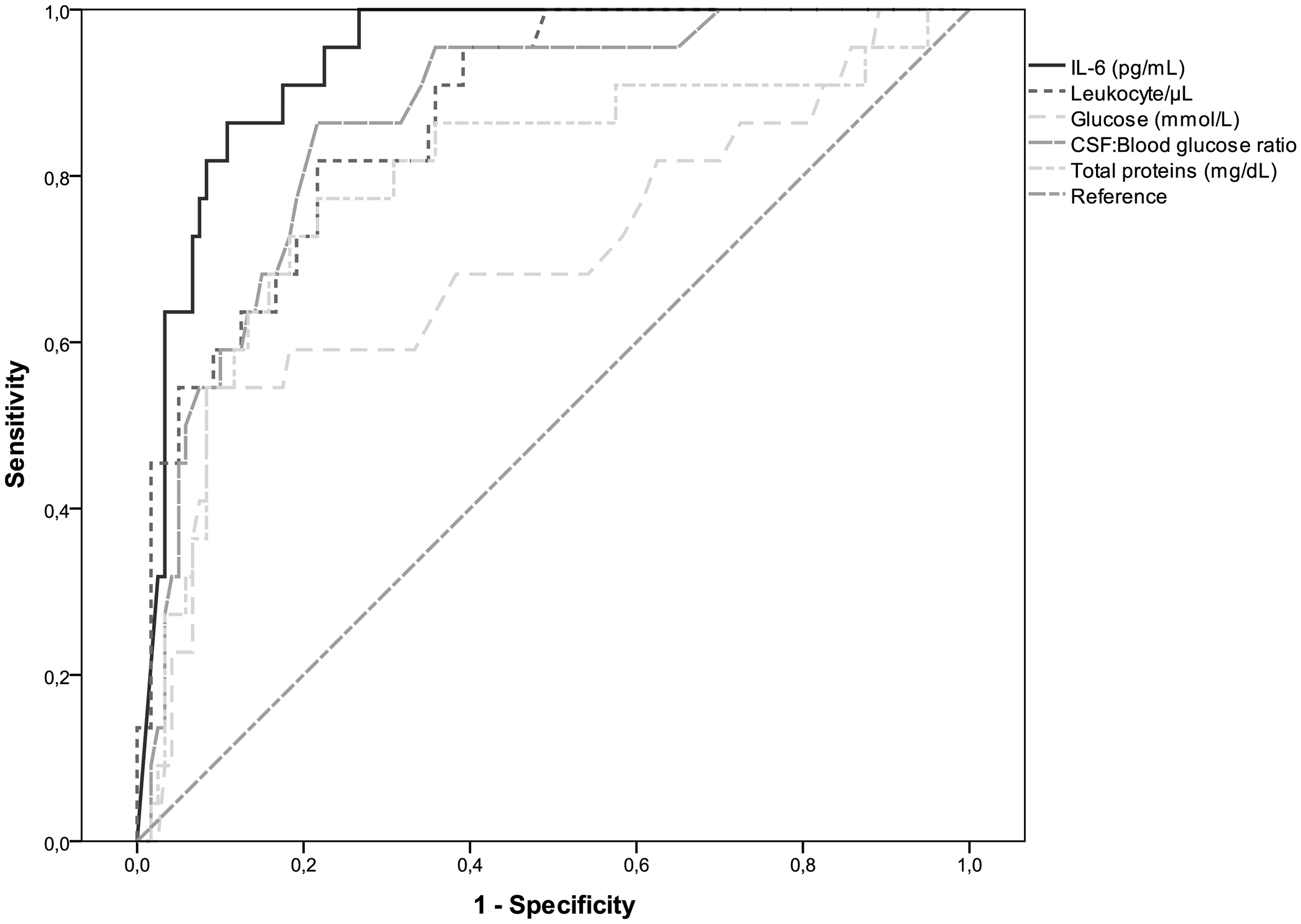
Discussion
The diagnosis of meningitis is based on the clinical presentation of the patient and the cytological and biochemical characteristics of CSF obtained by lumbar puncture.1,12 Establishing the aetiology of meningitis is essential, if appropriate treatment is to be administered promptly. Unlike viral or aseptic meningitis, bacterial meningitis may be fatal, especially if diagnosis and antimicrobial therapy are delayed.13,14
Low CSF glucose concentrations (<2.22 mmol/L) are present in approximately 50–60% of cases of bacterial meningitis; often CSF protein is increased. However, the CSF/blood glucose ratio provides better diagnostic performance (a ratio ≤ 0.4 theoretically showing up to 80% sensitivity and 98% specificity). On the other hand, Gram staining of CSF allows rapid diagnosis in 60–90% of cases of bacterial meningitis, but the false negative rate is high. Microbiological culture of CSF is positive in 70–85% of cases of bacterial meningitis 1 but at least 48 h are required to obtain a result. Thus, microbiological tests are more useful to confirm the infection and optimize the antibiotic treatment than for diagnosis.
For all these reasons, finding new biochemical markers that allow fast and reliable diagnosis of meningitis is of great value. The potential usefulness of measuring inflammatory biomarkers, such as C-reactive protein (CRP), in bacterial meningitis has been previously evaluated.15,16,17 However, controversial results were described with sensitivities ranging from 18 to 100% and specificities between 75 and 100% according to different cut-off points. Procalcitonin (PCT) has also been postulated as an efficient marker for the differential diagnosis of meningitis,18,19,20 but Viallon et al. 21 found undetectable concentrations of PCT in most of the 105 CSF samples analysed from different patients, including both viral and bacterial meningitis; on the other hand, Shimetani et al. 22 showed that PCT concentrations in patients with bacterial meningitis were higher in serum than in CSF samples.
IL-6 has been regarded as a useful marker for the diagnosis of neonatal sepsis in either umbilical cord blood or neonatal serum,23,24 especially during the first 48 h of life due to better kinetic profile than PCT in those patients. Interestingly, several studies have demonstrated that high CSF IL-6 levels can be found in bacterial and viral meningitis.25,26,27 However, slight elevations of CSF IL-6 levels have also been found in patients with schizophrenia, major depressive disorder, trauma and after exhaustive exercise.25,28,29
Fast, highly sensitive tests that can help in the differential diagnosis of bacterial from aseptic meningitis would be of great interest. In the present study, IL-6 was able to discriminate between both aetiologies at an IL-6 cutoff of 1418 pg/mL showing 95.5% sensitivity and 77.5% specificity for bacterial meningitis. Classical tests reached the same sensitivity only at a cost of lower specificities: 60.8% and 64.2% for a cut-off point of 48 leukocytes/mL and 0.545 CSF/blood glucose ratio, respectively, that differ considerably from the classical cutpoints well established in clinical practice (Table 4). In fact, IL-6 showed significantly higher diagnostic power (Figure 2) when compared to classic biomarkers (
However, our proposed IL-6 cutoffs disagree with those previously published. Taskin et al. (Accucyte ELISA) showed that IL-6 in CSF could be an early marker in differential diagnosis of bacterial and viral meningitis as well as other diseases causing meningeal irritation, showing a mean IL-6 of 349.3 (± 169.2) pg/mL for the diagnosis of bacterial meningitis.
4
Prasad et al. (RayBio ELISA)
27
proposed an IL-6 cut-off level of 300 pg/mL for bacterial meningitis with 87.5% sensitivity and 53% specificity. Other authors described even much lower CSF IL-6 concentrations than ours in bacterial meningitis. For instance, Mukai et al. (Quantikine HS ELISA)
30
and Hsieh et al. (R&D Systems ELISA)
31
reported a median of 70.37 pg/mL and 45.2 pg/mL, respectively, whereas Vázquez et al. (Immulite ELISA)
26
showed 92.3% sensitivity and 100% specificity with a cutoff of 90 pg/dL. On the other hand, Pinto-Junior et al. (R&D Systems ELISA)
32
found a mean of 536 pg/mL for bacterial meningitis, but a mean of 415 pg/mL for aseptic meningitis, with no significant differences. Nevertheless, all these studies have several limitations. First, the numbers of cases of bacterial meningitis (
In our study, outlier values (IL-6 ≥ 50.000 pg/mL) were not only found in group A (
Finally, the retrospective design of the present study allows for the possibility of bias. Randomized selection of controls aimed to minimise this effect. However, our results provide a basis for further prospective studies to establish the diagnostic role of CSF-IL-6 in clinical practice.
Conclusion
In the present study, measurement of CSF IL-6 by automated ECLIA has been demonstrated to be a potentially useful tool to aid the management of patients under suspicion of meningitis. Prospective studies are needed to validate these findings.
Footnotes
Acknowledgements
The authors thank Roche diagnostics for providing reagent kits.
Conflict of interest
None.
Funding
IL-6 reagent was supplied by Roche Diagnostics.
Ethical approval
The ethical committee of clinical research of the Principality of Asturias, Ref 002/14.
Guarantor
FVA.
Contributorship
PG, BP and FVA researched the literature and conceived the study. PG and VR analysed the samples. PG and BP performed the statistical analysis. PG wrote the first draft of the manuscript. BP and EM reviewed and modified the paper. All authors reviewed and approved the final version of the manuscript.
