Abstract
Objectives
Urinary hormone concentrations are often adjusted to correct for hydration status. We aimed to determine whether first morning void urine hormones in growing adolescents require adjustments and, if so, whether urinary creatinine or specific gravity are better adjustments.
Design and methods
The study population was adolescents aged 10.1 to 14.3 years initially who provided fasting morning blood samples at 0 and 12 months (
Results
Fasting first morning void hormone concentrations correlated well and were unbiased between unadjusted or adjusted by either creatinine or specific gravity. Urine creatinine concentration increases with Tanner stages, age and male gender whereas urine specific gravity was not influenced by Tanner stage, age or gender. Adjustment by creatinine or specific gravity of urinary luteinizing hormone, estradiol, testosterone, dihydrotestosterone and dehydroepiandrosterone concentrations did not improve correlation with paired serum concentrations.
Conclusions
Urine steroid and luteinizing hormone concentrations in first morning void samples of adolescents are not significantly influenced by hydration status and may not require adjustments; however, if desired, both creatinine and specific gravity adjustments are equally suitable.
Introduction
Measurements of urinary gonadotropins and steroids in children and adolescents emerged as methods to estimate pubertal development and gonadal function early in the immunoassay era.1–3 Urine sampling provides an integrated measurement especially for hormones such as luteinizing hormone (LH) 4 secreted in pulsatile manner or diurnally like sex steroids in early puberty and is more acceptable to children and adolescents than venipuncture. The relatively high hormone concentrations in urine compared with blood or saliva, together with the ability to concentrate urine, is advantageous for assays with low sensitivity or analytes at low concentrations. However, an inherent problem of using urine is the wide and unregulated variation reflecting the individual’s fluid status.
Urine dilution or concentration creates corresponding changes in urine solute concentrations so that adjustment of urine concentration may be required to avoid misinterpreting hormone excretion due to variation in hydration. 5 Osmolality, specific gravity (SG) and creatinine measurements are used to adjust hydration. 6 Although measurement of osmolality by freezing point depression is considered the reference method, 7 it is laborious, time consuming and expensive and so is usually replaced by SG and creatinine measurements particularly for large-scale field studies. Urinary SG is measured using a refractometer to compare light refraction of a urine sample against pure water standard or by reagent strips which measure the ionic strength of urine by colour changes. Urine SG of sample is normalized to a population reference value. While SG measurement has been largely superseded by urine creatinine adjustment in clinical laboratories, SG adjustment for urine dilution remains standard in antidoping laboratories and is used in some toxicology studies. 8 Creatinine adjustment is based on the assumption that (a) this end-product formed endogenously from muscle creatine is released into the bloodstream and excreted in urine at a constant rate depending only on total muscle mass 9 and (b) endogenous hormones and creatinine undergo renal excretion at the same rate. 6 Yet, creatinine excretion rate may be influenced by the growing muscle mass during puberty leading to potential systemic errors in using creatinine adjustments. 10
Some11,12 but not other13–17 studies suggest creatinine or SG adjustment for measurement of urinary substances although such adjustments may be either unnecessary or even introduce additional measurement errors. Furthermore, none has focused on situations where creatinine is changing systematically due to somatic growth. Thus, the present study aimed to determine whether the first morning void hormonal assessments carried out in growing young adolescents at various stages of pubertal progression require adjustments and, if so, to determine whether creatinine or SG adjustment was better.
Materials and methods
Samples
Adolescents aged 10.1 to 14.3 years initially were recruited from local secondary schools in the state of New South Wales (NSW), Australia. Ethical approval was obtained from the Human Research Ethics Committee, University of Sydney (HREC 13094). Fasting morning blood samples were collected at 0 and 12 months (
Assays
Urine SG was measured by immersing a reagent strip (ChoiceLine 10, Roche Diagnostics) in freshly voided urine samples. Dipstick colour changes were compared visually with the colour chart to estimate the SG. Urine samples were subsequently stored at −80℃ and subjected to three freeze–thaw cycles for LH, creatinine and steroid analysis. The urine samples were first thawed and assayed for LH measurements with the Immulite 1000 LH (Siemens) as described previously. 18 The within-assay coefficients of variation were <10%. The samples underwent second freeze-thaw cycle for the creatinine measurements. Urine creatinine concentrations were determined by the colorimetric alkaline-picrate (Jaffé) method (CREJ2, Roche Diagnostics, Cat. No. 04810716 190) on a Cobas C501 analyzer (Roche Diagnostics GmbH, Indianapolis, IN). Calibrators (Roche Diagnostics, Cat. No. 10759350 190) were used for this automated system to generate a linear curve ranging between 375 and 55,000 µmol/L and the limit of detection of 375 µmol/L. The final urine thaw was for urine steroid measurement. Urinary and serum estradiol (E2), testosterone (T), dihydrotestosterone (DHT) and dehydroepiandrosterone (DHEA) were measured by liquid chromatography tandem mass spectrometry (LC–MS/MS) as modified from a previously described method for serum 19 and adapted for urine specimens following deconjugation, with details described in the Supplementary Materials.
The LH and steroid concentrations were adjusted to standard SG of 1.020 according to the formula [hormone concentrationsample × (1.020−1)/(SGsample−1)]
20
and to standard CR measurement of the present study adolescent population (12.40 mmol/L [
Data analysis
The steroid and LH concentrations unadjusted and adjusted for SG or creatinine were compared by Deming (orthogonal) regression and deviance (Bland–Altman) analysis using MedCalc software. Based on using the same analyte with different adjustments, the variance ratio in the Deming regression was assumed to be unity. Non-independence according to variations in the number of samples provided by each individual was ignored in this analysis. Descriptive statistics including mean, standard deviation (SD) and standard error of mean (SEM) were performed by SPSS version 21. The
Results
In first morning urine void samples ( Plot of urinary creatinine measurements of adolescents groups according to age (left panel) and Tanner stage (right panel). Data represent the creatinine mean and SEM. For the age plot, samples were from 3-monthly intervals (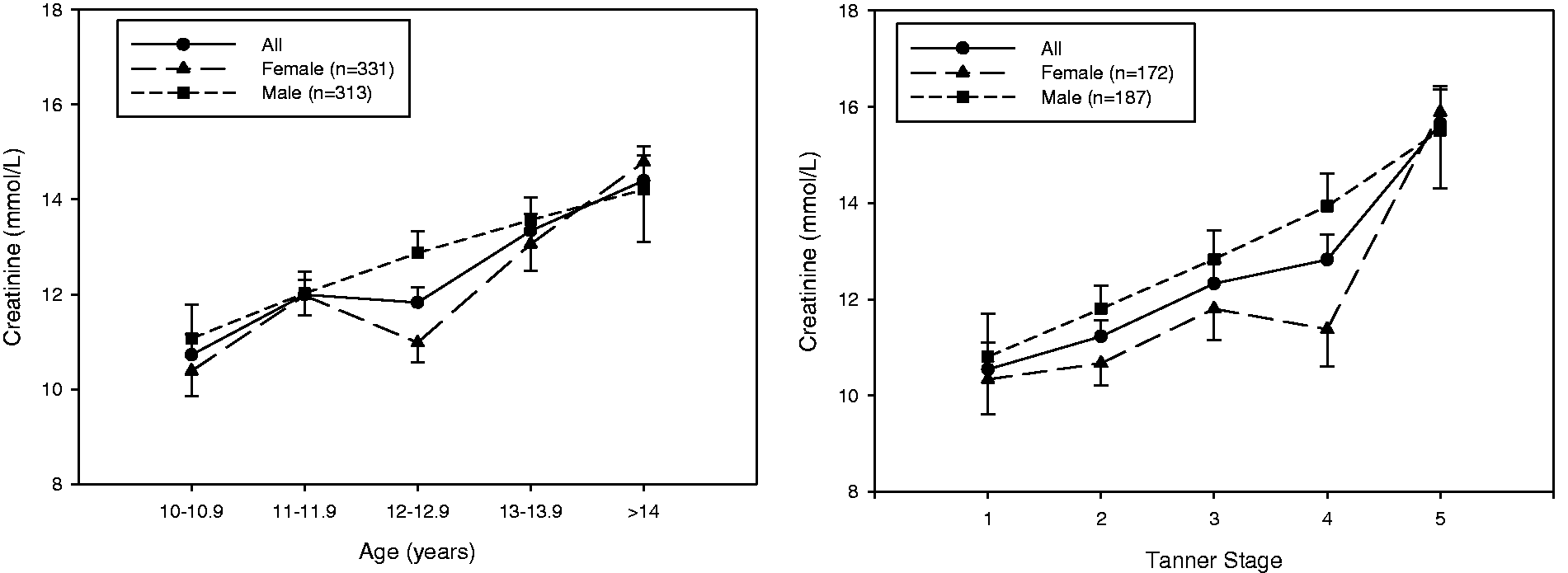
Urinary LH, E2, T, DHT and DHEA concentrations, adjusted for either SG or creatinine, are compared according to Deming regression line, and the deviance plots are shown in Figure 2. For each urinary hormone concentration, there was a good correlation between the SG and creatinine-adjusted concentrations ( Comparison of urinary LH, E2, T, DHT and DHEA concentrations adjusted by SG and creatinine. Comparison was made according to Deming regression analysis (left panels) and Bland–Altman plots (right panels). For the Deming plots, the slope is shown as a solid line and line of identity in fine dotted line. Insets are the regression formula and 95% confidence limits on the intercept and slope. The Bland–Altman plots represent the differences between creatinine and SG-adjusted hormone concentrations against the averages of the hormone concentrations adjusted with the two correction method. The solid line and the dashed lines represent the observed average and the 95% limit of confidence (±1.96 SD), respectively.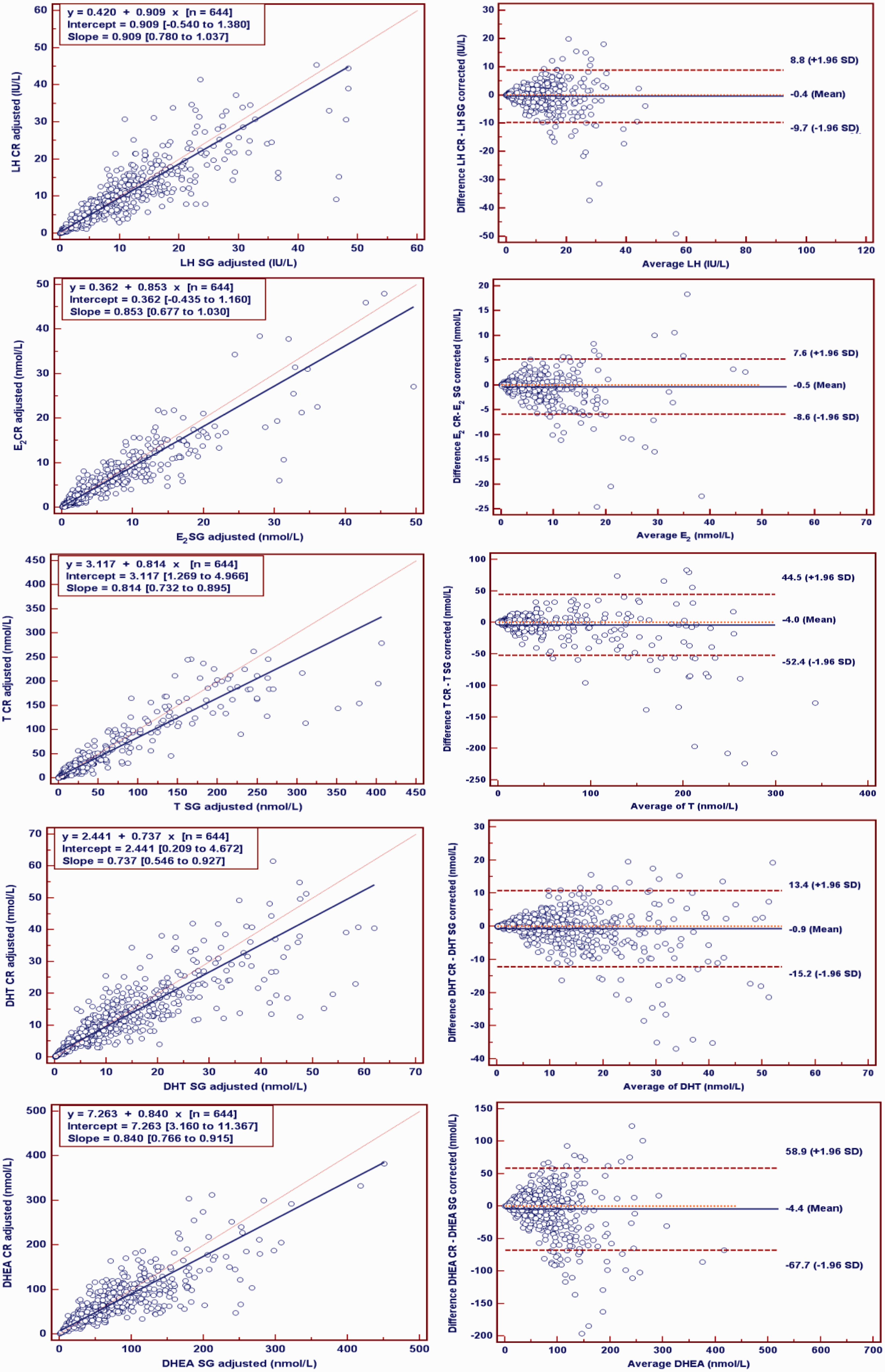
Comparison of unadjusted against SG- and creatinine-adjusted urinary hormone measurements.
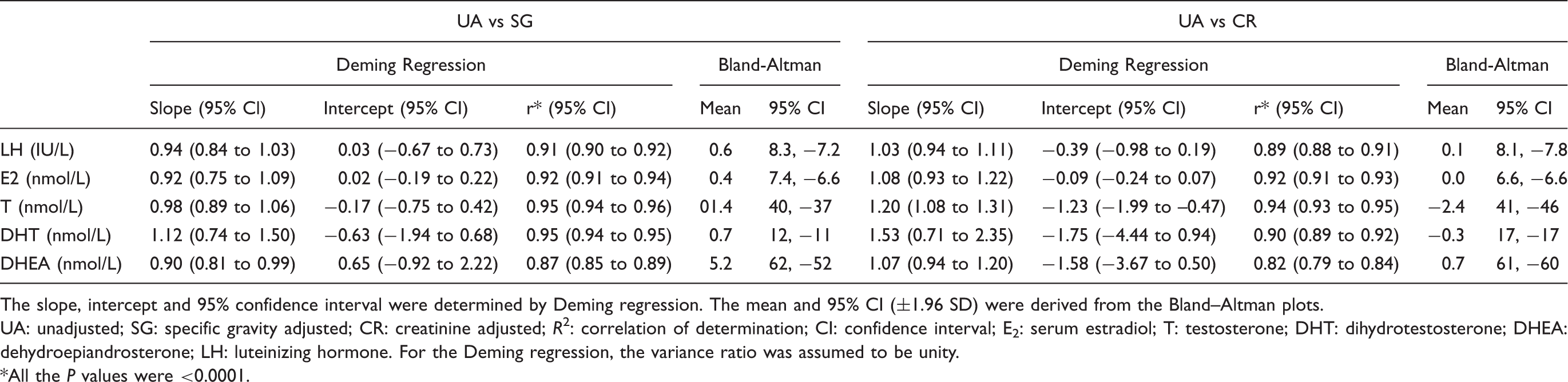
The slope, intercept and 95% confidence interval were determined by Deming regression. The mean and 95% CI (±1.96 SD) were derived from the Bland–Altman plots.
UA: unadjusted; SG: specific gravity adjusted; CR: creatinine adjusted;
*All the
The mean, SD and
Pearson’s correlation coefficient and confidence intervals (in parentheses) of paired urinary and serum LH, E2, T, DHT and DHEA (
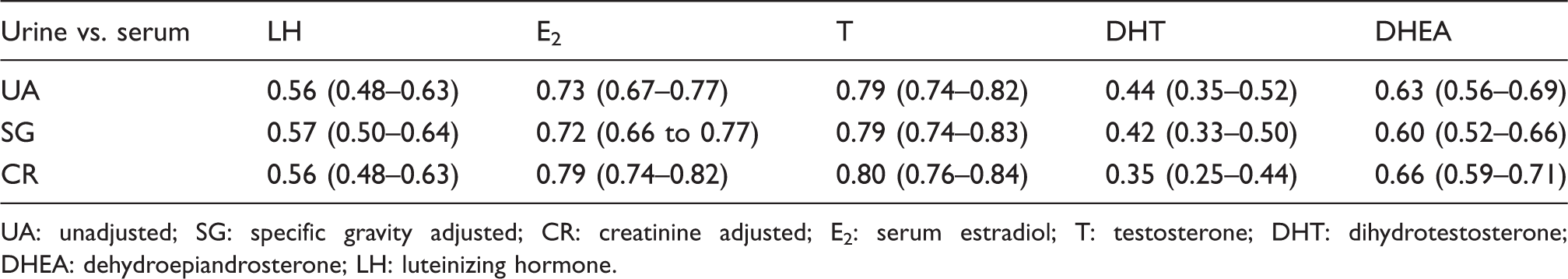
UA: unadjusted; SG: specific gravity adjusted; CR: creatinine adjusted; E2: serum estradiol; T: testosterone; DHT: dihydrotestosterone; DHEA: dehydroepiandrosterone; LH: luteinizing hormone.
Discussion
Urinary measurement of reproductive hormones is a convenient means to evaluate pubertal status and gonadal function for field population studies. In clinical settings, adjustment based on the assumption of stable urine creatinine excretion is commonly used to adjust for variations in hydration although other techniques such as regression normalization or log transformation are proposed.10,21 As an end metabolite of muscle creatine, urine creatinine is determined by total muscle mass in addition to other factors such as age, gender, diet (meat consumption), physical activity and body mass index, some of which exert their effects via changes in muscle mass.6,17,22,23 Hence, one aim of the present study was to determine for the first time whether creatinine adjustment was valid or required for longitudinal studies of growing adolescents.
Our findings confirm that the first morning urine creatinine concentration increases with age and Tanner stages and was higher among men. However, adjustment for urine creatinine was no better or worse than adjustment for SG or even no adjustment. This may reflect the fact that we studied first morning void urine samples which control hydration, whereas similar interpretation may not apply to urine sampled at random when hydration state may vary more. Our findings are consistent with previous studies showing prominent intra- and interindividual variability in creatinine excretion of second morning and 24 h urine samples in adults due to variable fluid intake. 17 Significantly higher creatinine concentrations in morning versus afternoon, 24 in evening spot samples 6 and creatinine loss due to multiple freeze–thaw cycles have also been reported25,26 all of which introduce systematic errors in use of urine creatinine for dilution adjustments. Thus, although studies have suggested alternative adjustment based on SG in adult humans and primates,27,28 none has focused on the need for SG adjustments in first morning voids of growing adolescents.
SG is readily measured by reagent strip for field studies without needing a laboratory. Previous studies demonstrate good agreement between SG measurements by reagent strip versus refractometer29,30 or osmolality.30–32 SG measurement by reagent strip is widely used in clinical applications.32,33 Although refractometer urine SG may be influenced by disease states leading to high serum protein or glycosuria,7,34 reagent strip SG is not affected by glucose, only minimally by urea and albumin, but may be affected by the rare instances of alkaline urine. 31 Urine SG reading may also be influenced by diet, environment and the renal reabsorption capacity. 35 Among adolescents, we find that urine SG measured with reagent strips is systematically not influenced by age or gender consistent with previous reports.22,36
Limits of acceptable creatinine and SG measurements vary between studies. Generally, urine is considered too dilute when the SG and creatinine concentrations are lower than 1.010 and 0.5 g/L (4.4 mmol/L), respectively, and too concentrated where SG and creatinine concentrations higher than 1.030 (or 1.035) and 3 g/L (26.5 mmol/L), respectively.17,35 However, due to the standardized method of collection and hydration (first morning void), the present study did not discard any samples as too dilute or too concentrated.
The present study demonstrated that the fasting first morning void urine hormone concentrations adjusted by creatinine correlated well with those adjusted by SG in this adolescent population. This is consistent with previous reports that used randomly collected or timed urine collection from children and adults showing good correlations when creatinine and SG adjustments were compared directly5,12,21,23,35,37 or with adjustment according to both5,27,28 including a reduced variation using these adjustments in some studies.10,12 However, the present study shows that neither of the adjustment methods for first morning void urine sample of adolescents were significantly improved compared with unadjusted hormone concentrations. These observations are consistent with previous reports for creatinine adjustment of urine steroid measurements in adult women.38,39
In studies where the urinary hormone concentrations were correlated with paired circulating serum concentrations, the urinary unadjusted concentration or concentration expressed by volume of urine correlates better than the adjustment based on analyte to creatinine ratios,11,13,40 although some studies have shown improved correlation with creatinine adjustments.41,42 The present study demonstrated that the urinary hormone concentrations adjusted with creatinine, and SG did not improve the correlation with paired serum concentrations. These samples were also grouped into three creatinine and SG percentile ranges (25th, 25–75th and 75th) to replicate non-fasting conditions with wider variation in hydration status. However, no improvement was observed in terms of correlation between the unadjusted or adjusted urine hormone and paired serum concentrations. These findings further support that the adjustments may not be necessary for first morning void urine samples.
In conclusion, the present study shows that adjustment of urinary steroid and LH concentration for hydration state may not be required for first morning void specimens of even growing adolescents. If adjustments are required, then either creatinine or SG is equally suitable and provides comparable results. Reagent strip SG measurements are simple and sufficiently reliable, economical and time-saving for large numbers of urine sampling in long-term field studies.
Footnotes
Acknowledgments
None.
Declarations of competing interests
None declared.
Funding
This research was funded by Australian Government National Health and Medical Research Council (NHMRC) (grant number 1003312).
Ethical approval
Ethical approval was obtained from the Human Research Ethics Committee, University of Sydney (HREC 13094).
Guarantor
DJH.
Contributorship
GKSS was responsible for assay development and conducting the assays, as well as statistical analyses, writing the first draft and editing the manuscript. BWRB, RD and MJ were involved in the writing and editing of the manuscript. RD was also involved in the assay development. KSS was involved in study design, study oversight and writing and editing this manuscript. DJH was responsible for study design, assay development, statistical analyses, writing and editing the manuscript. All authors approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
