Abstract
Background
Vitamin D deficiency is increasingly recognized in patients with primary hyperparathyroidism but some clinicians are reluctant to replace vitamin D due to concerns with aggravating hypercalcaemia. We investigated the impact of vitamin D repletion in asymptomatic patients with normocalcaemic and hypercalcaemic primary hyperparathyroidism.
Methods
This is a retrospective analysis of 111 patients with elevated parathyroid hormone concentrations (>6.4 pmol/L) referred to our endocrine clinic between January and December 2012; we identified 39 patients with primary hyperparathyroidism and vitamin D deficiency, i.e. 25 hydroxy vitamin D <20 µg/L. Patients were categorized into normocalcaemic (n = 23) and hypercalcaemic (n = 16) groups and the impact on biochemical parameters was recorded after at least six months treatment with either 1600 or 3200 units daily of cholecalciferol.
Results
Both normocalcaemic and hypercalcaemic groups showed a rise in 25 hydroxy vitamin D concentrations after replacement (p <0.0001). Parathyroid hormone concentrations fell in the normocalcaemic group (p = 0.08) but individually, five patients showed a rise (8–38% of baseline). In the hypercalcaemic group, parathyroid hormone remained static but the adjusted calcium concentration fell significantly (p = 0.006) except in two patients who showed mild rises (3 and 6%, respectively). There was no deterioration in renal function or calcium-related adverse events in any of the groups.
Conclusions
Our study supports the safety of vitamin D replacement in patients with mild asymptomatic primary hyperparathyroidism and coexistent vitamin D deficiency. Repletion does not aggravate hypercalcaemia and may limit disease progression. Patients with normocalcaemic primary hyperparathyroidism need further characterization from longitudinal studies.
Introduction
Vitamin D deficiency occurs more commonly in patients with primary hyperparathyroidism (PHPT) than in the general population. 1 Coexistent vitamin D deficiency may worsen the clinical expression of PHPT and individuals with both conditions tend to have higher parathyroid hormone (PTH) concentrations, larger parathyroid adenomas, and higher markers of bone turnover than those with PHPT alone.2,3 The mechanism of vitamin D deficiency in PHPT is not fully understood but is believed to be related to accelerated catabolism of 25 hydroxy-vitamin D (25OHD) by increased concentrations of PTH and 1,25-dihydroxyvitamin D.4,5 Although the serum 25OHD cut-off definition for vitamin D deficiency is still debated, a number of guidelines recommend initiating vitamin D therapy at a threshold of <20 micrograms/L. based on data in healthy subjects showing maximal PTH suppression when 25OHD concentrations are maintained above this concentration. 6 PHPT is traditionally characterized by raised PTH concentrations and hypercalcaemia but in recent years a new variant has been highlighted in which the serum calcium is normal and PTH concentrations are elevated in the absence of secondary causes of hyperparathyroidism. The natural history of this entity, termed normocalcaemic (NC) PHPT, is still uncertain but some authorities consider it to be a preclinical precursor of hypercalcaemic (HC) PHPT.7–11
The value of correcting vitamin D deficiency in PHPT is controversial. Early reports3,12–15 suggested that vitamin D replacement worsened hypercalcaemia in patients with PHPT but a few recent studies16–20 including a recent meta-analysis of 340 subjects in 10 observational studies 21 have shown that vitamin D reduces PTH concentrations without adverse effects on serum calcium. Furthermore preoperative vitamin D replacement has been reported to shrink adenoma size and reduce the risk of hungry bone syndrome following parathyroid surgery. 17 A trial of vitamin D may also allow diagnostic separation between PHPT and secondary hyperparathyroidism due to vitamin D deficiency. Persistently raised PTH concentrations after correction of vitamin D deficiency validate the diagnosis of PHPT whereas PTH returns to normal in patients with secondary hyperparathyroidism who are then spared further diagnostic studies or surgical intervention. Patients with true NC PHPT and coexistent vitamin D deficiency present a special quandary since such individuals can only be distinguished from vitamin D deficiency with secondary hyperparathyroidism after a trial of vitamin D. Lastly, there is a wealth of evidence to show that vitamin D has beneficial effects on aspects of human physiology including bone density, neuromuscular function, blood pressure, and other cardiovascular indices. 22
These considerations are reflected in current expert guidelines which recommend correcting vitamin D deficiency in asymptomatic patients with PHPT. 23 In practice, however, many clinicians remain reluctant to prescribe vitamin D to patients with PHPT due to concerns with aggravating hypercalcaemia coupled with the lack of pragmatic protocols to guide treatment. To date, only few studies have addressed the safety and efficacy of vitamin D replacement in asymptomatic individuals with PHPT and the optimal dose and duration of therapy is yet to be established. Our aim in this study was thus to evaluate the impact of vitamin D replacement in patients referred to our hospital endocrine clinic with asymptomatic PHPT. Specifically we sought to determine whether vitamin D induced or worsened hypercalcaemia in patients with NC and HC PHPT, respectively.
Patients and methods
Patients
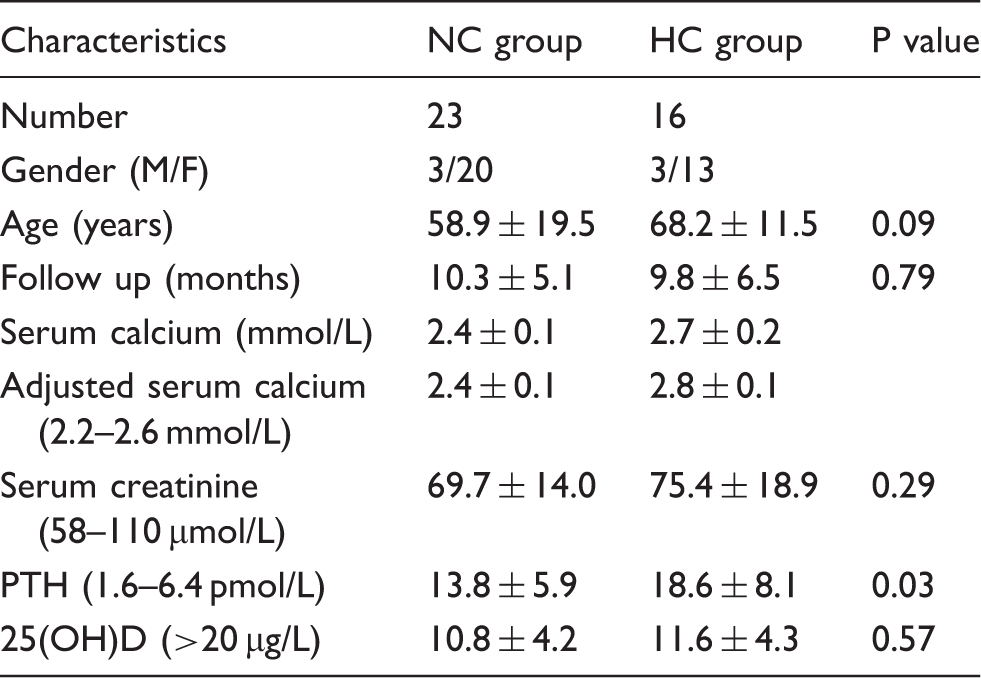
This is a retrospective analysis of 111 patients with raised PTH concentrations (>6.4 pmol/L) referred by general practitioners to our specialist endocrine clinic from January to December 2012. Patients were defined as vitamin D deficient if serum concentrations of 25OHD was <20 µg/L and severely deficient if 25OHD was <10 µg/L. Patients with 25OHD concentrations >20 µg/L at presentation were excluded from the study (n = 37, 25OHD − 32.10 ± 10.54, PTH − 13.6 ± 10.4, adjusted Ca − 2.5 ± 0.22, three patients had hypercalcaemia). We also excluded patients who did not receive vitamin D replacement in spite of being vitamin D deficient (n = 14) and patients with insufficient biochemical data (n = 11). None of the patients had symptoms or established complications of hyperparathyroidism. Patients with normal PTH concentrations after vitamin D replacement (n = 10) were considered to have secondary hyperparathyroidism and were also excluded. The analysis included 39 patients with persistently elevated PTH concentrations after vitamin D replacement. These patients were categorized according to their baseline serum calcium into a HC group (>2.6 mmol/L, n = 16) and a NC group (2.1–2.6 mmol/L, n = 23). The study was approved and registered in the clinical audit department of Cardiff and Vale University Health Board (approval reference number: 8154). All data were handled according to Caldicott principles.
Vitamin D replacement
Patients were treated with two different doses of oral cholecalciferol; 1600 (two tablets of 800 units each) units and 3200 (four tablets of 800 units each) units as available from the hospital formulary. Since there were no clear protocols to guide treatment, the choice of a particular fixed dose was at the discretion of the endocrinologist who initiated the primary treatment in the clinic. All patients completed at least six months of treatment with an intention to maintain a serum concentration of 25OHD of >20 µg/L.
Biochemical measurements
Serum calcium, albumin and creatinine were measured using the automated platform by Abbott Architect (Abbott Laboratories, North Chicago, Illinois, USA). The adjusted calcium was calculated by an in house derived equation applied to the calcium to correct it to account for albumin concentration. Serum 25OHD was measured by detecting parent to daughter ion transition method (multiple reaction monitoring) using UPLC-MS/MS (Waters, Manchester, UK). Serum PTH was measured by Abbott Architect Automated immunoassay (Abbott laboratories, North Chicago, Illinois, USA).
Data analysis
Data are presented as means ± standard deviation (SD) except where otherwise stated. We used a paired t-test to assess the difference in values before and after vitamin D therapy. The Student’s t-test was used to compare means between the two patient subgroups while differences in proportions between the groups were analysed with the Chi-squared test with Yates correction where appropriate. Differences were considered statistically significant at a P value of <0.05 using a two tailed test. All statistical analysis was undertaken using the statistical package SPSS for windows (version 16.0, SPSS Inc, Chicago, Illinois, USA).
Results
Baseline characteristics
Baseline characteristics of patients.
Impact of vitamin D replacement
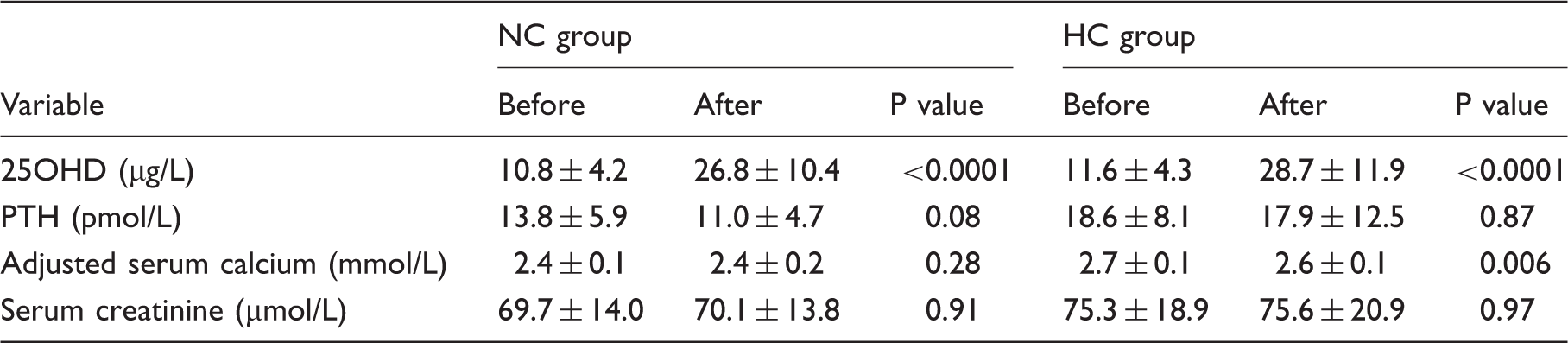
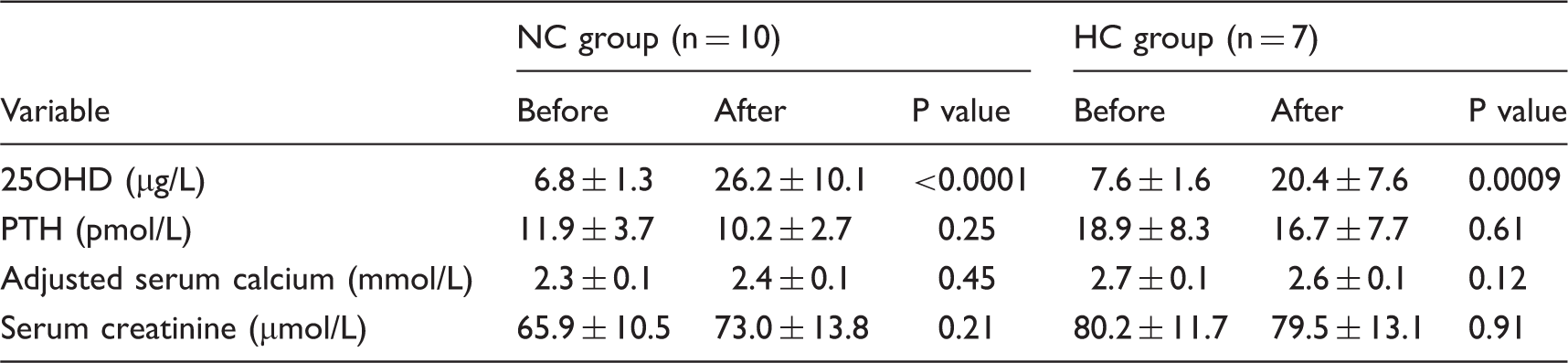
Table 2 shows the changes in biochemical parameters after treatment with vitamin D. In the NC group a significant rise in mean vitamin D concentration was seen after treatment while a drop in the mean PTH concentration was also observed although this fell short of statistical significance (P = 0.08). Similarly in the HC group a significant rise in mean vitamin D concentration was observed but PTH concentration remained the same after treatment. Mean serum calcium was unchanged in the NC group but fell significantly with treatment in the HC group (p = 0.006) The serum creatinine did not show any change from baseline in either group. In the subcategory of patients (Table 3) with severe vitamin D deficiency (25OHD <10 µg/L), replacement with cholecalciferol showed statistically significant improvement but there was no major impact on PTH or adjusted calcium concentrations. Figure 1(a) to (d) shows the individual level data for the change in serum PTH and adjusted calcium concentrations in both groups of patients. PTH concentrations rose mildly in five patients in the NC group (by 8–38% of baseline) and remained unchanged in one patient in this group. None of the patients with normal calcium concentrations at baseline developed hypercalcaemia. In the HC group, two patients developed a mild rise in adjusted calcium concentrations (by 6 and 3% of baseline, respectively). The creatinine concentration remained stable in all and there were no reported cases of renal impairment, nephrolithiasis, or other calcium-related adverse events.
(a) PTH concentrations after vitamin D replacement (NC group), (b) adjusted calcium concentrations after vitamin D replacement (NC group), (c) PTH concentrations after vitamin D replacement (HC group), (d) Adjusted calcium concentrations after vitamin D replacement (HC group). Biochemical variables before and after vitamin D replacement. Biochemical variables before and after replacement in severe vitamin D deficiency (25OHD <10 µg/L).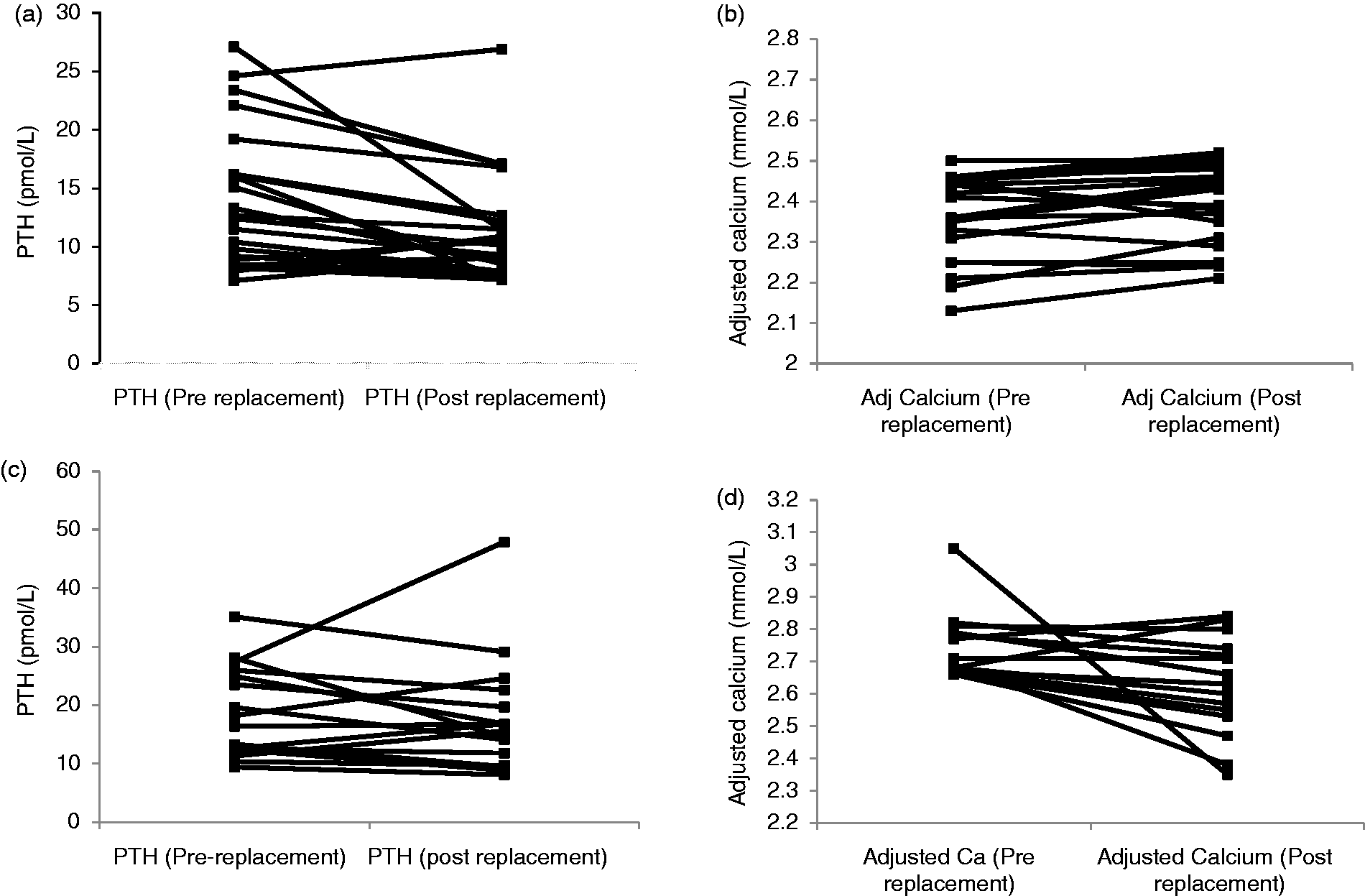
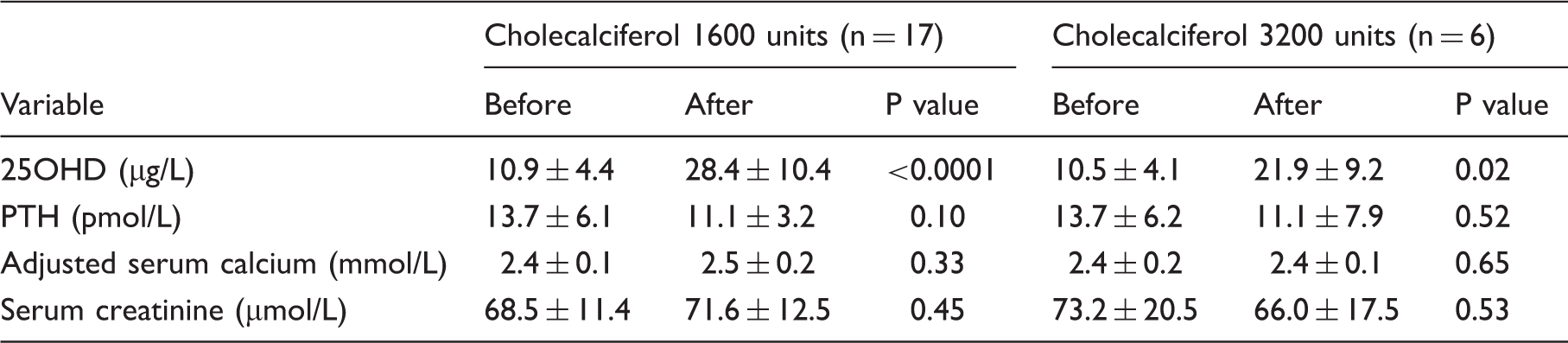
Impact of vitamin D dose
Biochemical variables before and after treatment with different doses of cholecalciferol in NC group.
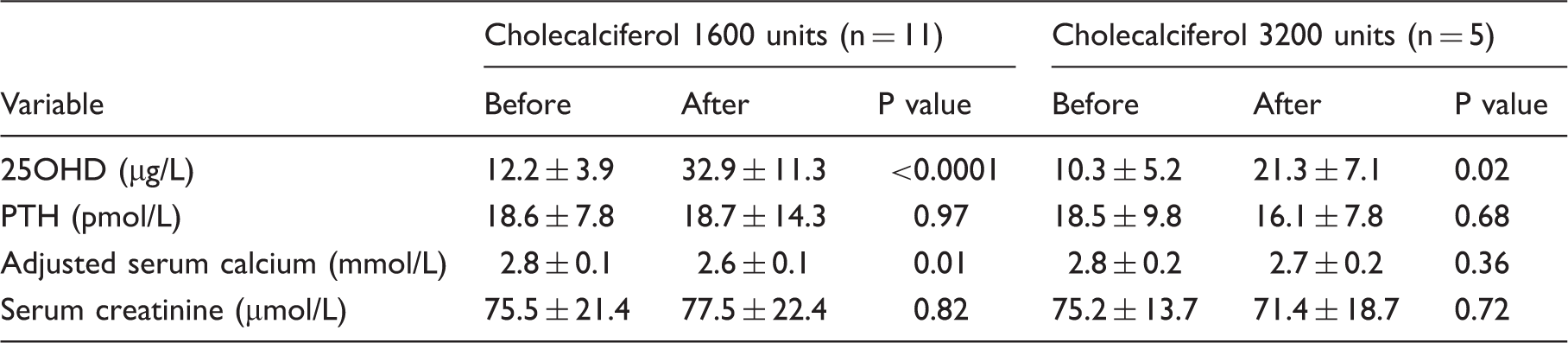
Biochemical variables before and after treatment with different doses of cholecalciferol in HC group.
Discussion
Clinicians nowadays encounter a growing number of asymptomatic individuals with combined vitamin D deficiency and PHPT, a trend driven by the high prevalence of these conditions coupled with the widespread use of automated multichannel laboratory analysers. Yet only a limited number of studies to date have specifically addressed the impact of vitamin D replacement in this group of patients and there is no guidance on the optimal dose, duration of treatment, and target 25OHD concentrations. Here we show that vitamin D supplementation is safe in patients with PHPT. After a minimum of six months of vitamin D replacement, none of our NC patients developed hypercalcaemia and only two HC patients showed mild increases in calcium. Renal function was stable and no cases of nephrolithiasis were recorded throughout the course of treatment.
Previous studies of vitamin D replacement in PHPT have focussed on patients with the classic HC presentation of the disease. In the present study, we have additionally explored the impact of replacement in patients with NC PHPT. There is scant data on the epidemiology, natural history, and clinical impact of NC PHPT. A prospective study of 41 patients by Lowe et al. showed a progression rate to hypercalcaemia of 20% over a three-year period. In addition, 40% of the cohort developed nephrolithiasis, bone demineralization, and fractures while remaining NC. These findings thus underpin the need for accurate diagnosis and monitoring in this group of patients. 24 In our study, vitamin D replacement considerably reduced PTH concentrations in NC patients with no major change in calcium concentrations or induction of hypercalcaemia. Out of 23 NC patients, PTH concentrations fell by a magnitude of 9–61% (mean 26%) in 16 patients and remained unchanged in one patient. In none of these patients did PTH return to normal after vitamin D repletion making it unlikely that these cases were due to vitamin D deficiency alone. Interestingly, five patients developed a rise in PTH with vitamin D treatment. These patients will require long-term monitoring for the development of hypercalcaemia or bone disease.
Our data also support the safety and efficacy of vitamin D repletion in patients with asymptomatic HC PHPT with coexisting vitamin D deficiency. In our patients we observed a mild decrease in PTH concentrations together with a significant decrease in the mean adjusted serum calcium concentrations. Most of our patients had mild hypercalcaemia (<3.0 mmol/L) with the exception of one patient whose calcium was 3.03 mmol/L at presentation and subsequently dropped to 2.55 mmol after vitamin D repletion. Our findings are broadly consistent with recent observations16–21,25 which have shown that vitamin D supplementation does not aggravate hypercalcaemia in patients with PHPT but actually causes a modest decline in serum calcium concentration which was suitably demonstrated in a recent meta-analysis. 21 The potential mechanism for this could be that vitamin D deficiency in PHPT triggers a concomitant elevation in calcium; hence after vitamin D replacement therapy there is reduction in PTH concentrations which attenuates the secondary or compensatory component to the primary PTH elevation. But the primary pathological process still remains in these patients as the serum calcium and PTH concentrations remain inappropriately elevated even after replacement. 17 Such a scenario is more pronounced when the reduction in serum calcium concentrations is from a higher baseline like in our HC group. Any direct comparisons of our findings with these other studies are limited by some degree of heterogeneity in dose, duration, and type of vitamin D formulation used for replacement.
In the 1970s, a number of small case series reported the development of clinically significant hypercalcaemia in patients with PHPT who were treated with vitamin D3. However, these patients had established bone disease and their vitamin D status were not routinely measured.26,27 A study published in the year 2000, involving five patients with PHPT and coexistent vitamin D deficiency, showed that vitamin D supplementation did not cause hypercalcaemia in any of the patients but led to hypercalciuria in three patients. 28 Subsequently, Grey et al. in their study of 21 patients with PHPT and vitamin D deficiency reported no change in serum calcium and a 26% decrease in PTH concentrations after administration of vitamin D3 at 50,000 units weekly for four weeks followed by 50,000 units monthly for 12 months. 16 Similarly, another study involving 56 patients with PHPT and vitamin D deficiency reported no change in serum calcium after treatment with 50,000 units of ergocalciferol (vitamin D2) weekly for eight weeks followed by varying maintenance doses. 18
Several other studies in comparable patient groups with PHPT and vitamin D deficiency have similarly reported stable calcium concentrations with or without an increase in urinary calcium in response to various doses of vitamin D supplements.17,25 More recently, Wagner et al. 19 assigned patients to treatment with either 1000–2000 IU daily or 50,000 IU weekly of vitamin D. PTH concentrations were unchanged in both groups and calcium concentrations remained stable in the high dose group but decreased in the low dose group although this became statistically insignificant after multiple adjustments for age, sex, vitamin D, and PTH concentrations. Lastly, Ranganathan et al. also showed that in postmenopausal female with PHPT and coexisting vitamin D deficiency prolonged treatment with vitamin D reduced serum PTH concentration with no adverse effects on adjusted serum calcium concentrations and renal function. 20
The cause of vitamin D deficiency in PHPT is usually multifactorial. Global geographic differences, seasonal variations, and duration of exposure to sunlight will have a significant bearing on 25OHD concentrations in patients with PHPT. Moosgaard et al. 29 have demonstrated a seasonal variation in plasma 25OHD concentrations in patients with PHPT which was higher in the late summer months in both the study (p <0.005) and control groups (p <0.05) but at all calendar months the average plasma 25OHD was numerically reduced in the PHPT patients compared with controls. This further emphasises that the proportion of vitamin D deficiency in PHPT patients will always be higher than that of the general population and needs replacing irrespective of seasonal variation.
Our study has limitations. First, our sample size was relatively small thus limiting a more detailed analytical breakdown of outcomes according to demographic characteristics, dose of vitamin D, and duration of treatment. Second, we did not have complete information on urinary calcium excretion in our patients and although we observed no cases of nephrolithiasis we are unable to demonstrate whether vitamin D repletion was associated with hypercalciuria in our cohort. Third, a small proportion of our patients failed to achieve a normal 25OHD concentration (>20 µg/L) despite vitamin D treatment of at least six months in each case. The reasons for treatment failure in these individuals are unclear but are likely to involve factors such as inadequate compliance, malabsorption, interacting medications, or undetected co-morbid conditions. The relationship between response and treatment dose did not reach statistical significance in our study and will require further exploration in larger adequately powered studies. Lastly, the retrospective design of our study meant that patients arbitrarily received different doses of vitamin D and were treated for varying lengths of time depending on clinician preference. Prospective controlled trials will be required to confirm the findings from our study.
In summary our data show that correction of vitamin D deficiency in patients with mild asymptomatic PHPT and coexisting vitamin D deficiency is safe and not associated with induction or aggravation of hypercalcaemia. Vitamin D repletion may limit disease severity in patients with PHPT and also allow the distinction between primary and secondary hyperparathyroidism. Patients with NC PHPT will benefit from characterization and monitoring for progression to hypercalcaemia or bone disease.
Footnotes
Acknowledgements
We would like to thank Dr Sarah Neale and Dr Carol Evans from the Medical Biochemistry department at Cardiff and Vale University health board.
Declaration of conflicting interests
JSD is a medical adviser to Internis; has received research and educational awards from Prostrakan (manufacturer of Adcal), Meda (manufacturer of Desunin), and Internis (manufacturer of Fultium); and has undertaken speaker meetings for Internis and Prostrakan. The remaining authors don’t have any conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
The study was approved and registered with audit department of Cardiff and Vale University health board (reference no: 8154/2013).
Guarantor
GD.
Contributorship
GD conceived and wrote the first draft of the article. GD, VE, LNRB, and JG collected and analysed the data and researched the literature. OO helped with statistical analysis. OO and SD supervised, modified, and edited the article at different stages. All authors reviewed and approved the final version of the manuscript.
