Abstract
Aim
To investigate the diurnal variability and pre-analytical stability of soluble α-klotho in serum.
Background
Recent evidence suggests that the cleaved extracellular domain of the α-klotho receptor, soluble α-klotho, affects phosphate homeostasis, ion channel regulation and antifibrotic/antioxidant pathways. However, soluble α-klotho measurements have yielded inconsistent results when related to renal function, markers of mineral metabolism and patient outcomes. Pre-analytical factors such as biological variation and analyte stability may affect the interpretation of soluble α-klotho results but are yet to be formally assessed.
Methods
Clotted blood samples were collected from 10 healthy adult volunteers at three time-points during the day to assess diurnal change. Additional samples were collected and allowed to stand at room temperature for 30, 60 and 120 min, prior to centrifugation and analysis to evaluate analyte stability. Serum soluble α-klotho was measured using a validated commercial enzyme-linked immunosorbent assay.
Results
Delayed processing of samples had no significant effect on serum soluble α-klotho concentrations over a 2-h period. Serum soluble α-klotho concentrations remained stable over morning, midday and afternoon time-points in this pilot study.
Conclusion
Serum soluble α-klotho demonstrates short-term pre-analytical stability and minimal diurnal variability in this pilot study. Larger studies are warranted to confirm these findings.
Background
The α-klotho gene encodes a single-pass transmembrane protein that acts as an obligate co-receptor for fibroblast growth factor 23 signaling. First described as an anti-ageing gene, we now know that α-klotho has multiple key functions. Although most attention has focused on its regulation of phosphate homeostasis in the kidney, α-klotho also has a range of endocrine and paracrine functions, including growth factor signalling, ion channel regulation, and anti-insulin, antifibrotic and antioxidant activities. 1
α-klotho exists in two forms–membrane-bound (mKl) and soluble entities (sKl). mKl is variably expressed in different tissues, whereas sKl can be produced by both ectodomain cleavage of mKl (∼130 kDa) and from an alternative splice variant leading to a shorter form of sKl (∼70 kDa). Proteomic analysis of various extracellular fluids suggests that the major circulating form of sKl in humans is that resulting from mKl ectodomain cleavage.2,3
Measurement of serum sKl concentrations in health and disease, however, has yielded inconsistent results when related to renal function, markers of mineral metabolism and patient outcome. 4 In general, assays for such proteins are often impacted by a number of pre-analytical variables, including, amongst others, sample collection and storage. These may affect the interpretation of sKl concentrations and consequently our understanding of the α-klotho protein, its metabolism and effects. As these factors have not been formally assessed, the present study was designed to examine both analyte stability and diurnal variation in a group of healthy volunteers.
Methods
Study population
The study was approved by the Melbourne Health Human Research Ethics Committee, Melbourne, Australia and was performed in accordance with the Declaration of Helsinki. A total of 10 healthy volunteers, six male and four females, all over 18 years of age and with normal renal function (eGFR>90 ml/min/1.73 m2) were recruited to this study. Mean (± SD) age was 40.5 ( ±9.8) y. Two participants had mild hypercholesterolaemia and were on treatment with a lipid-lowering agent. No significant medical histories were reported from any other volunteers.
Blood collection
A total of five blood specimens were collected in serum separator tubes (BD Biosciences, Franklin Lakes, NJ, USA) on the same day for all individuals; three tubes at 08:00–09:00, one at 12:00–13:00 and one at 15:00–16:00. These times were chosen to reflect routine outpatient pathology sampling. Morning samples were allowed to stand at room temperature for 30, 60 and 120 min, prior to centrifugation (10 min, 4℃, 3000 g) and processing, while midday and afternoon samples stood for 30 min. All samples were stored at −80℃ until batched analysis.
Freeze–thaw analysis
To assess the effect of freeze-thawing on the stability of serum sKl, an extra blood sample was taken from three individuals at 09:00 on the day of analysis. These samples were allowed to clot for 30 min at room temperature before centrifugation (as described above). From these samples, one aliquot of serum was taken for immediate analysis without freezing (<45 min); a second aliquot was subjected to a single freeze–thaw (frozen to −80℃ and thawed at room temperature) and a third aliquot was subjected to three freeze–thaw cycles, prior to measurement.
Soluble klotho measurements
Unless otherwise indicated, samples were thawed to room temperature (once only) prior to analysis. Serum sKl concentrations were measured using the IBL soluble klotho enzyme-linked immunosorbent assay (ELISA) kit (Immuno-Biological Laboratories Co., Ltd., Gunma, Japan) according to the manufacturer’s protocol. The manufacturer’s reported intra-assay and inter-assay analytical coefficient of variation (CVA) are 3.1% and 6.9%, respectively. All samples were measured in duplicate.
Statistical analysis
Data are represented with median values and interquartile ranges (IQR) and analysed using non-parametric statistics. The effect of freeze-thawing and temporal changes in sKl concentration was analysed using the Friedman test with Dunn’s test for multiple comparisons. Intra-individual biological variation was determined according to Fraser and Harris. 5 All analyses were performed using GraphPad Prism 6 for Windows (La Jolla, CA, USA).
Results
The intra-assay and inter-assay analytical coefficients of variation (CVA) for this study were 4.9% and 8.8%, respectively.
Pre-analytical stability
Delayed separation of morning blood samples for 30, 60 and 120 min post-venipuncture yielded median (IQR) serum sKl concentrations of 499 (409–739) pg/mL, 514 (402–675) pg/mL and 541 (409–674) pg/mL, respectively (Figure 1(a)). There was no evidence of significant analyte instability over this period (Friedman test, p = 0.366). Additionally, single and repeated (thrice) freeze–thaw processing of serum samples was found to have no significant effect on sKl measurements (Friedman test, p = 0.361).
Pre-analytical stability (a) and daytime diurnal variability (b) of serum sKl concentration in 10 healthy adult participants.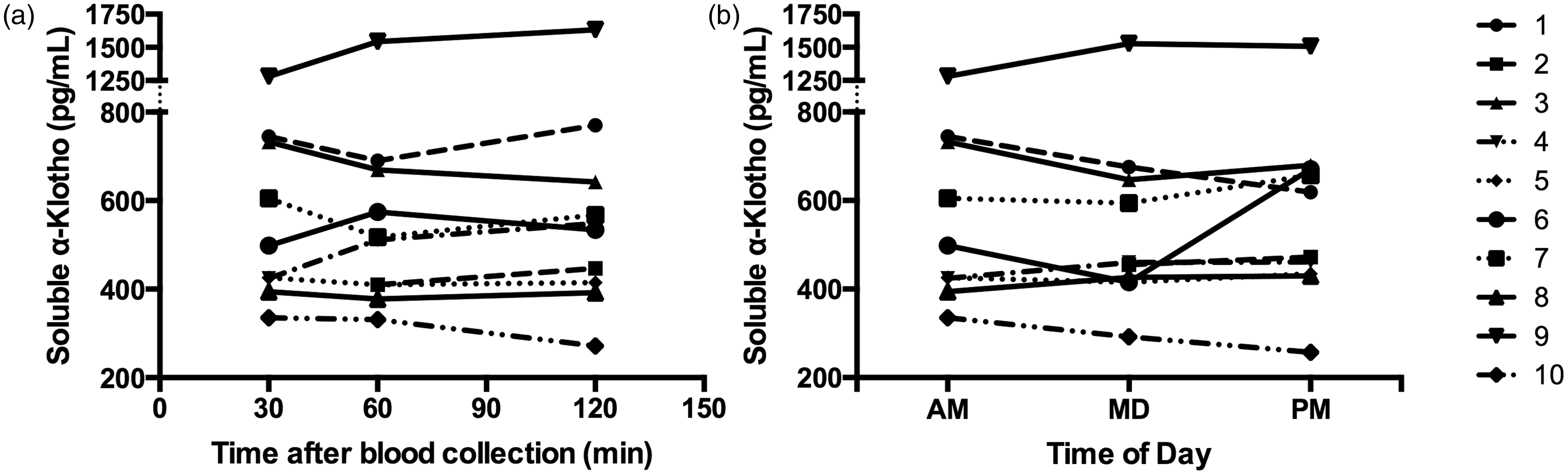
Diurnal variation
Median (IQR) sKl concentrations were 499 (409–739) pg/mL, 498 (424–654) pg/mL and 545 (434–673) pg/mL at morning (08:00–09:00), midday (12:00–13:00) and afternoon (15:00–16:00) time-points, respectively (Figure 1(b)), showing no significant daytime diurnal variability (Friedman test, p = 0.366). Based on these measurements, the intra-individual coefficient of variation (CVI) for serum sKl was 2.5%. Accordingly, assuming a CVA of 8.8% this yields a reference change value for sKl of ∼25% (at a 95% confidence level).
Discussion
Our findings from healthy volunteers suggest minimal diurnal variation in sKl concentrations, minimal analyte degradation over 2 h after blood sampling as well as stability to repeated freeze–thaw cycling.
Although this pilot study is not powered to detect small magnitude intra-individual changes in sKL concentration (<40%), the apparent lack of variability in all participants throughout the day suggests that the timing of blood collection is unlikely to impact upon sKl measurements substantially. Reassuringly, the finding of minimal daytime variation presented here is also consistent with a larger study of patients with X-linked hypophosphataemia (n = 44), where levels remained within 20% of 09:00 values at 12:00 and 16:00 time-points. 6 The low short-term intra-individual biological variation for sKl (2.5%) implies that relatively small changes in sKl concentration may be clinically significant if such low levels of variability are maintained in disease states. Furthermore, these results highlight the need for stringent control over other sources of variation in the pre-analytical and analytical phases of sKl assessment.
Since most clinical studies prescribe rapid sample processing protocols, the apparent pre-analytical stability of serum sKl demonstrated here suggests that analyte instability is unlikely to contribute significantly to the marked inconsistencies in results noted previously. 4 Differences in analyte specificity and other assay characteristics are therefore more likely to account for such findings. 7 Further assessment of pre-analytical stability may be needed if greater delays in sample processing (>2 h) are expected (e.g. collection from remote sites).
Limitations in our study include a small sample size and the assessment of only short-term biological variation. Estimates of day-to-day biological variation and assessment of pre-analytical factors in diseased individuals are needed to appraise the applicability of these findings to other populations.
In conclusion, serum sKl displays minimal daytime variation in healthy individuals. Although prompt processing of samples for sKl measurement is advisable, minimal degradation is observed in clotted blood over 2 h at room temperature and with repeated freeze–thaw cycles.
Footnotes
Disclaimer
The contents of this article are solely the views of the individual authors and do not reflect the views of NHMRC or the Jacquot Foundation.
Declaration of conflicting interests
ERS has received research funding from Amgen and Baxter, honoraria from Shire, and served as a consultant for Vifor Pharma. SGH has received research funding or honoraria from Amgen, Baxter, Gilead, Novartis and Shire. NDT has received consultancy fees, honoraria and research funding from Amgen and Shire Pharmaceuticals.
Funding
The research was funded by a Jacquot Foundation Research Establishment Award and Australian National Health and Medical Research Council (NHMRC) Postgraduate Research Scholarship.
Ethical approval
The Melbourne Health Human Research Ethics Committee approved this study.
Guarantor
SJT.
Contributorship
SJT and ERS designed the study. SJT conducted the study, performed the analysis and wrote the first draft of the manuscript. All other authors contributed to critically revising the manuscript and approved the final draft.
