Abstract
Background
Monitoring of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) in remission usually includes indirect immunofluorescence (IIF), proteinase 3 (PR3)-ANCA and myeloperoxidase (MPO)-ANCA. Typically, PR3 and MPO-ANCA are both performed because patients sometimes switch specificity during follow up. Published data are limited to case reports and incidence of change is not reported. The aim of this study was to quantify the incidence of antibody switching.
Methods
Hull and East Yorkshire Hospitals National Health Service Trust serves a population of 720,000 for ANCA testing. We reviewed all ANCA results from January 2000 to August 2012 to quantify incidence of antibody switching. A total of 22,002 IIF screens (14,518 patients) were performed. A total of 9838 (45%) also had PR3- and MPO-ANCA (6439, 44% of patients). Patients that changed specificity from PR3- to MPO-ANCA and vice versa were identified and case notes reviewed.
Results
A total of 218 patients with confirmed AAV positive for PR3/MPO-ANCA were followed for a mean of 2.6 years (range <0.1 to 12.4 years; with 113 (52%) patients followed for >1 year). Five patients (2%) changed specificity during follow up (3 GPA, 1 MPA & 1 EGPA). In two patients this was associated with relapse. Incidence of specificity change was 1 per 82 years (including two reversions to presenting specificity) and one per 286 years for changes associated with relapse. Monitoring using only the initial antibody specificity would have resulted in missed relapse in one patient.
Conclusion
Antibody specificity changes in AAV are rare. Monitoring only the initial antibody specificity would have missed clinical events but rising C-reactive protein presaged relapse in these cases.
Keywords
Introduction
The anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitides (AAV) are a group of related inflammatory small vessel disorders, comprising granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA) and eosinophilic GPA (EGPA). The international consensus statement on ANCA methodology recommends screening by indirect immunofluorescence (IIF) using ethanol-fixed neutrophils with identification of the IIF pattern and further testing of positive samples for autoantibodies to proteinase 3 (PR3-ANCA) and myeloperoxidase (MPO-ANCA).1,2 The presence of PR3-ANCA typically correlates with IIF C-ANCA pattern and MPO-ANCA with IIF P-ANCA pattern but these are occasionally reversed and not all patients display these associations. 1
These laboratory tests have an important role in facilitating rapid diagnosis and early warning of relapse to enable early intervention to preserve organ function. The role of ANCA in predicting disease relapse has been controversial as a rise in ANCA titre during remission (PR3- or MPO-ANCA) is only weakly associated with disease relapse (diagnostic odds ratio 2.7). 3 Monitoring of AAV patients in remission therefore usually includes IIF and quantitation of PR3/MPO-ANCA by enzyme immunoassay (EIA). It is normal practice in follow up of patients during treatment and remission to perform both PR3-ANCA and MPO-ANCA, regardless of the previous results observed or the current IIF pattern, as there have been a number of cases described in which autoantibody specificity has switched between PR3-ANCA and MPO-ANCA. 4 Though considered unusual, the incidence of antibody specificity switching has not been reported in a large case series, published data being limited to a few case reports summarized in Saleem et al. 4 Here we present data from a case series of 218 patients with AAV spanning nearly 13 years of practice.
Our aim was to establish the incidence of antibody specificity switching between PR3-ANCA and MPO-ANCA in monitored patients with AAV and to consider if follow up ANCA testing could be limited to the single specificity identified at presentation.
Method
Hull and East Yorkshire Hospitals serve a population of about 720,000 for ANCA testing. This service covers primary and secondary care. Tertiary renal and immunology referral services are provided to a wider population.
During the period reviewed (January 2000 to August 2012) we performed IIF as our primary screen.
Specific EIA for PR3-ANCA and MPO-ANCA were performed if:
IIF was positive for any pattern including atypical patterns; An antinuclear antibody was present (which may obscure an underlying ANCA pattern); PR3-ANCA or MPO-ANCA were previously positive; Clinical presentation suggested a high probability of AAV despite a negative IIF screen. These included destructive upper airways disease, rapidly progressive glomerulonephritis, pulmonary-renal syndrome, cutaneous vasculitis with systemic symptoms, mononeuritis multiplex and pulmonary inflammatory disease.
5
Ethanol-fixed neutrophils for ANCA IIF were either prepared in-house from a normal pool (until end of August 2000) or from a commercial source (Euroimmun UK Ltd, London, UK). A number of different immunometric methods were used during this period – Biodiagnostics (Worcester, UK) EIA was used to April 2005; Euroimmun (Lubeck, Germany) EIA to May 2009; Grifols Chorus (Cambridge, UK) EIA to January 2011; Biorad Bioplex (Hercules, California, USA) Luminex subsequently. The manufacturer’s recommended reference interval was used for each method after local validation.
The Pathology Service Laboratory Information Management System contains a database of all pathology patient results generated since December 1999. We utilized this database to identify all ANCA results and, for those patients with multiple requests, identify those in whom ANCA specificity had switched from PR3-ANCA to MPO-ANCA and vice versa.
We reviewed results from all ANCA performed between January 2000 and August 2012. All patients identified as having a positive PR3-ANCA or MPO-ANCA at anytime during this period had all of their PR3- and MPO-ANCA results reviewed for evidence of switching of specificity. Results were considered positive if they exceeded the upper limit of the reference interval. Clinical diagnosis of AAV was confirmed from laboratory and/or case note records. Of the 218 cases 147 (67%) were confirmed by biopsy and an additional 15 (7%) had inadequate or inconclusive biopsy results. A total of 56 patients (26%) did not have biopsy performed prior to initiation of therapy. All patients had received treatment for a presumptive diagnosis of AAV.
Results
During this period (January 2000 to August 2012) 22,002 ANCA screens were performed in 14,518 patients and 9838 (45%) of these serum samples had PR3- and MPO-ANCA tested (6439 patients; 44% of all patients). A total of 1523 (6.9%) samples were positive for PR3-ANCA and/or MPO-ANCA. Of these, 250 patients had multiple PR3/MPO-ANCA measurements and 218 (87%) of these had a diagnosis of AAV.
These 218 patients were followed for a mean of 2.6 years (range <0.1 to 12.4 years). A total of 113 (52%) were followed for at least one year and 156 (72%) for at least three months. Total patient follow up years was 571.
Antibody specificity changed in five (2%) patients during follow up (3 GPA, 1 MPA and 1 EGPA). The incidence of specificity change was 1 per 82 follow up years (including two reversions to original specificity) and 1 per 286 follow-up years if only those associated with relapse are considered.
Figure 1 summarizes the outcomes and details for these five patients. Interpretation of these changes is complicated by the changes in method over the study period: Patient 1 had a specificity switch without a method change, associated with relapse and then reverted to the original specificity on change of method while in remission. Patient 2 had a specificity switch without a method change, and no evidence of relapse. Given the poor predictive value of ANCA for relapse and the association of low level ANCA with other inflammatory disease such as infection, the cause of this change is unclear. Patient 3 had a specificity switch associated with a method change while in remission. Patient 4 had a specificity switch without a method change during a chest infection and probable relapse. Patient 5 had a specificity switch without a method change, and no evidence of relapse. As for Patient 2 the cause of this change is unclear. Specificity switch was therefore associated with clinical relapse (and increased serum C-reactive protein (CRP)) in two patients.
Summary of patient outcomes. Superscript letters refer to the method in use at the time: aBioidiagnostics; bEuroimmun; cGrifols; dBioRad; CRP, C-reactive protein; GPA, granulomatous polyangiitis; MPA, microscopic polyangiitis; EGPA, eosinophilic granulomatous polyangiitis; ANCA, anti-neutrophil cytoplasmic antibody; AAV, ANCA-associated vasculitis; PR3, proteinase 3; MPO, myeloperoxidase.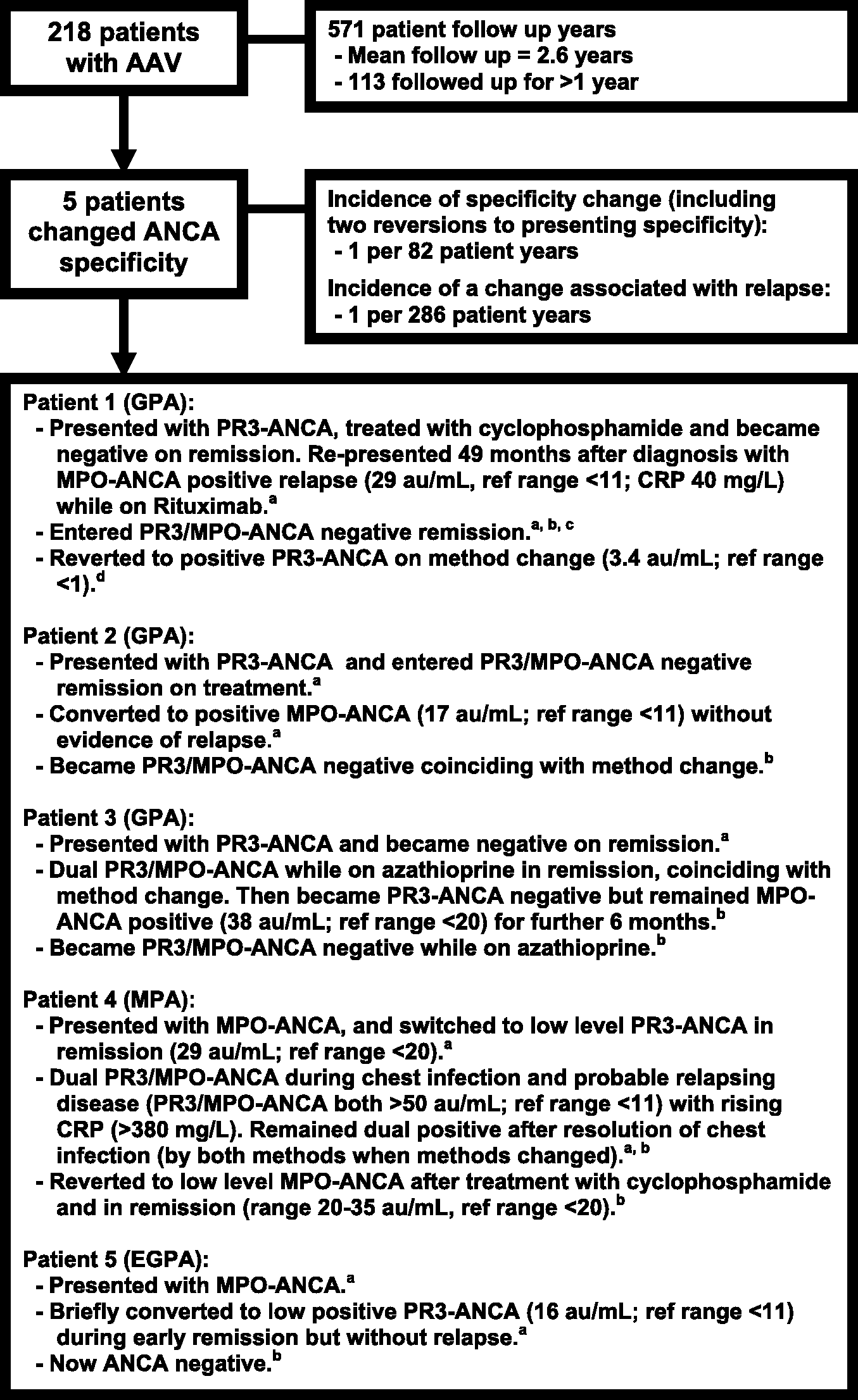
Discussion
There are two issues related to these findings.
Incidence of PR3/MPO-ANCA switching: We observed specificity switches in 2% (5/218) of patients. For four of these, change from the original specificity occurred without a change in method (Patients 1, 2, 4 and 5) and in two of these patients this was associated with relapse. In two patients there was a return to the presenting specificity, while the other three became PR3/MPO-ANCA negative, giving seven specificity switches in total. These seven changes equates to 1 per 82 patient years, 1 per 114 patient years if only the change from presenting specificity is counted, and 1 per 143 years if only the four changes from presenting specificity not associated with a method change are counted. The incidence therefore lies somewhere between 1 in 82 and 1 in 143 patient years. Significance of the changes in specificity: In two patients (Patients 1 and 4) the changes potentially had clinical significance as they were associated with relapse. If reliance had been placed on the presenting abnormal ANCA assay for monitoring, one relapse would have been missed (Patient 1). However, the poor predictive value of ANCA assays for relapse means that other markers are used in parallel with the ANCA assays and in this case the raised CRP should have alerted the clinician to a potential relapse.
6
The relapse in Patient 4 was associated with development of dual PR3/MPO-ANCA positivity and so would not have been missed if only the presenting specificity had been checked. In two patients (Patients 2 and 5) switching constituted only a low level transient positivity and was not considered clinically significant (Figure 1). We therefore observed only one clinically significant event (relapse in Patient 1) that may potentially have been missed if only the presenting ANCA specificity was monitored. This equates to a missing clinically significant event once every 571 patient years.
The cause of specificity switching is unknown. In some patients it has coincided with drug regime changes but this was not the case in any of our patients, who were all on standard maintenance or induction therapy. 4
There are a number of limitations to this study. Due to its long term (13 years) a number of different methodologies were used and may have accounted for some of the specificity switches as variability between ANCA assays is well recognised. 7 However, this was a relatively minor influence as four of the five changes from presenting specificity occurred without a change in method. Additionally, only 52% of patients were monitored using ANCA for more than a year (113 of the 218 patients) and all of the patients that switched fell into this group. It may be that longer term monitoring would show more switches. Finally, only 67% of patients had their diagnosis confirmed by biopsy. In the other patients biopsy was unsatisfactory, refused or contraindicated. However, all had a presumptive clinical diagnosis of AAV and were treated on this basis.
Even given these limitations, it is clear that specificity switching was rare in this cohort, with an incidence between 1 in 82 and 1 in 143 patient years. This and the practice of using other markers of relapse (clinical examination, CRP, renal function, proteinuria and selective imaging) indicate that the probability of missing a relapse if only the presenting ANCA is monitored is small. Decisions on the optimum monitoring strategy for AAV in remission require balancing the clinical benefits of additional testing against the costs involved.
Footnotes
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Ethical approval was not required as tests were routine care/audit and anonymised by the care team before analysis.
Guarantor
SH.
Contributorship
SH conceived and designed the study, researched the literature, collected data, performed analysis and wrote the report. VF collected data, performed analysis, researched the literature, contributed to the report writing and reviewed the article for intellectual content. MA reviewed the article for intellectual content.
