Abstract
Background
Inhaled steroids are widely used for the treatment of asthma. Concerns over adrenal suppression when used at high doses or in combination with drugs such as ritonavir exist, requiring the measurement of serum cortisol. Herein, we investigate the cross-reactivity of the inhaled steroids betamethasone, fluticasone and beclomethasone in the Roche cortisol immunoassay, in addition to five other steroids.
Methods
Five replicates were produced from a serum pool for each of the eight steroids at a final concentration of 0.1 and 1 µg/mL. Each steroid was dissolved in 50% methanol, with 50% methanol of the same volume added to the control sample. The cross-reactivity of each steroid in the cortisol assay was calculated.
Results
There was no statistically or clinically significant cross-reactivity in the measurement of cortisol when fluticasone, beclomethasone or betamethasone were spiked at 0.1 and 1.0 µg/mL, except for beclomethasone at a concentration of 1 µg/mL (1490 nmol/L) with a cross-reactivity of 1.6%, which is unlikely to be clinically significant. At both steroid concentrations investigated, prednisolone, 17-hydroxyprogesterone and 11-deoxycortisol exhibited statistically significant cross-reactivities that were greater than the least significant change of the assay (13.1%), whereas dexamethasone and metyrapone did not. Mean inter-assay precision was 1.5% (405–1586 nmol/L).
Conclusion
The cross-reactivity of the inhaled steroids; betamethasone, fluticasone and beclomethasone in the Roche cortisol immunoassay are unlikely to be clinically significant at the concentrations found in patients on therapeutic doses. This will enable confident assessment of adrenal status in patients at risk of adrenal suppression.
Introduction
Inhaled steroids are anti-inflammatory agents widely used for the treatment of asthma. Although these medications are thought to be safe at low-to-moderate doses, concerns exist over long-term use of high-dose inhaled corticosteroids, in particular, the risk of adrenal suppression. 1
This risk has become particularly important in patients being treated with the inhaled steroid fluticasone (Flixotide, A&H) in combination with the protease inhibitor ritonavir (Novir, Abbott). Ritonavir’s potent inhibition of hepatic cytochrome P450 3A4 causes an increase in serum fluticasone concentration, which may lead to iatrogenic Cushing’s syndrome and subsequent adrenal suppression. 2
When there is a high clinical suspicion of adrenal suppression following withdrawal of corticosteroid therapy, early diagnosis and prevention of an Addisonian crisis is vital. Measurement of serum cortisol at 9:00 h by immunoassay enables investigation of adequate adrenal reserve. A value of 500 nmol/L is widely quoted throughout literature across multiple immunoassay platforms as adequate, with below this requiring further investigation. 3 Automated cortisol immunoassays are convenient and rapid; however, they suffer from a lack of specificity due to interference from endogenous and prescribed steroids. As a result, some laboratories are using liquid chromatography-tandem mass spectrometry for measurement of serum cortisol due to its superior analytical specificity. To date, there have been no publications on whether inhaled steroids cross-react in automated cortisol immunoassays. Herein, we report on interference experiments carried out to assess whether eight steroids including the inhaled steroids fluticasone, beclomethasone and betamethasone cross-react in the Cobas 601 (Roche Diagnostics Inc., Lewes, East Sussex, UK) cortisol competitive immunoassay.
Methods
A serum pool was used to produce five separate replicates for each of the eight steroids investigated – betamethasone, fluticasone, beclomethasone, prednisolone, 17-hydroxyprogesterone, 11-deoxycortisol, dexamethasone and metyrapone. All steroids were purchased from Sigma Aldrich (Dorset, UK). Each steroid was dissolved in 50% methanol before being added to the serum replicates to produce a final concentration of 0.1 and 1 µg/mL with 50% methanol of the same volume added to the control samples (5% v/v methanol to serum). These concentrations were chosen as they were previously used in the cross-reactivity investigation by Roche, allowing comparison between results.
4
In addition, they are higher than those expected in patients being administered inhaled steroids and therefore show the highest simulated cortisol level that could be caused by these drugs.5,6 Each set of replicates were analysed within the same batch. The cross-reactivity of each steroid in the cortisol assay was then calculated using the following formula
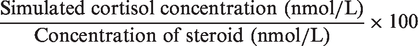
The cortisol concentrations produced by each steroid were compared with the control samples by analysis of variance with Bonferroni correction for multiple comparisons. All data were normally distributed, as evaluated by the Kolmogorov–Smirnov test. Values were considered significantly different at
Results
Cross-reactivity data for the measurement of cortisol when pooled serum samples were spiked with (a) 0.1 µg/mL and (b) 1.0 µg/mL of steroids.
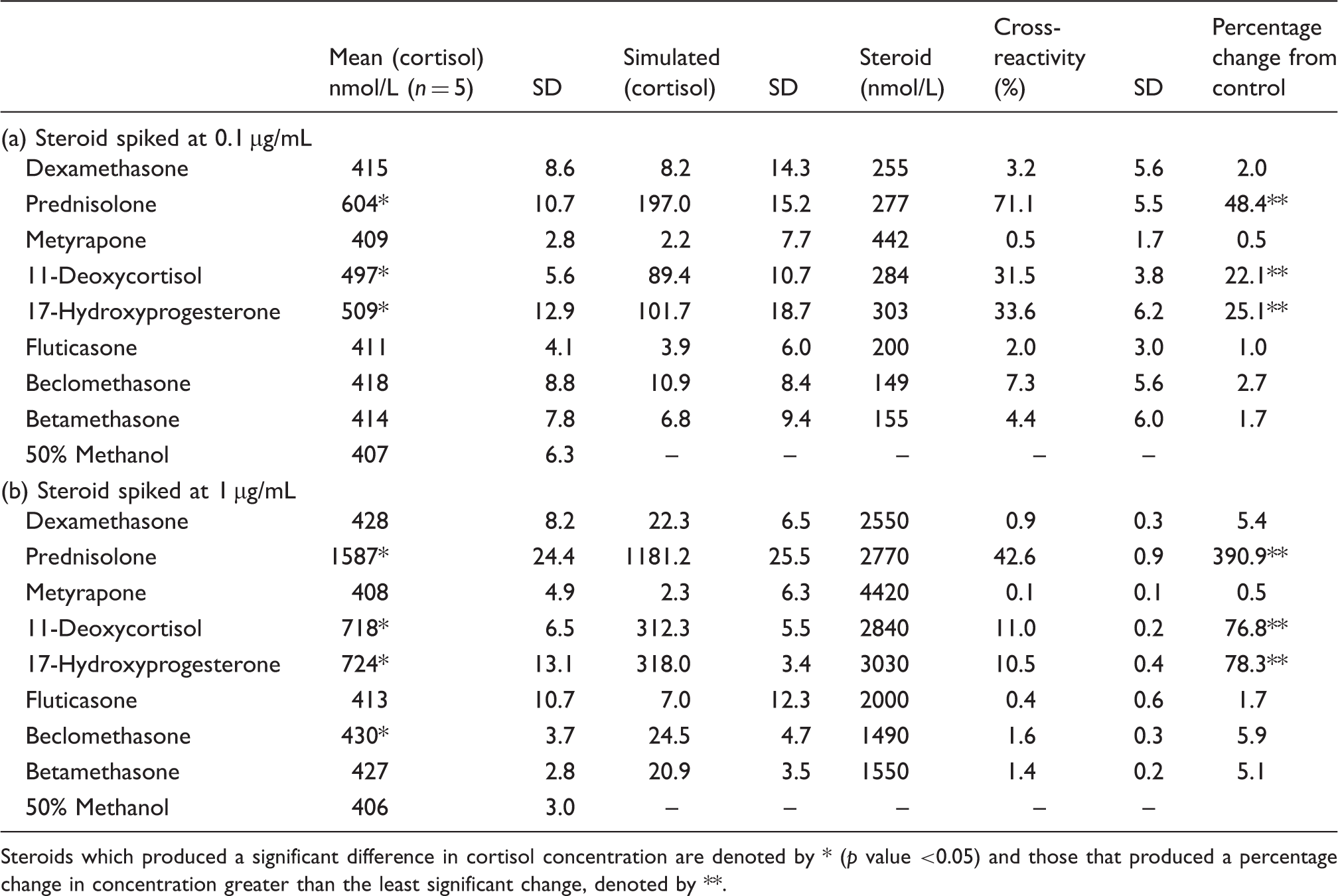
Steroids which produced a significant difference in cortisol concentration are denoted by * (
Discussion
Cross-reactivity data for serum cortisol measurement have previously been published by Roche (Roche Diagnostics Inc.) on prednisolone, dexamethasone, 11-deoxycortisol and 17-hydroxyprogesterone. 3 Our data are in agreement for the commonly prescribed drugs prednisolone and dexamethasone, showing minimal cross-reactivity in the cortisol assay by dexamethasone and prednisolone to significantly cross-react in the cortisol assay.
The percentage cross-reactivities for each steroid were found to be different at the two different concentrations investigated (0.1 and 1 µg/mL), with percentage cross-reactivity lower at the higher concentration. This could be because of the way the percentage cross-reactivity is calculated – dividing the simulated cortisol concentration by the concentration of steroid added to the sample. Therefore, although a higher concentration of steroid produces a higher simulated cortisol concentration (1 µg/L compared to 0.1 µg/L), this is may be out of proportion compared to difference in concentration. This is could be because the cross-reactivity of the steroids in the cortisol immunoassay does not have a linear relationship with the concentration of the steroid. This is likely to be because of the non-specificity of the antibodies to these steroids.
Our experiments showed statistically and clinically significant cross-reactivity from 11-deoxycortisol and 17-hydroxyprogesterone (Table 1) and to a greater extent than Roche. Although these metabolites are usually found in very low concentrations in the body, their cross-reactivity is important to be aware of during circumstances when they may be significantly elevated. For example, metyrapone therapy which may be used for treatment of Cushing’s syndrome can cause a raised 11-deoxycortisol, and patients with untreated congenital adrenal hyperplasia can have significantly elevated 17-hydroxyprogesterone concentrations.
Our studies found no statistically or clinically significant cross-reactivity in the measurement of cortisol from fluticasone, betamethasone, dexamethasone and metyrapone. Although beclomethasone showed statistically significant cross-reactivity at 1 µg/mL, this is unlikely to be clinically significant as it is below the LSC of the assay. In addition, the spiked concentrations investigated in this study are significantly higher than those observed in patients taking these medications. One study found the maximum fluticasone concentration following inhalation of 1000 µg was 0.12 ng/mL (0.02 nmol/L) (maximum recommended daily dose is 1760 µg).5,6 Therefore, a much lower simulated cortisol concentration would be produced at lower serum levels of beclomethsaone and the other steroids investigated.
Conclusion
These data confirm that the measurement of serum cortisol by immunoassay is suitable for the assessment of adrenal reserve in patients taking the inhaled steroids studied.
Cross-reactivity studies should be performed in-house to establish if oral or inhaled steroids cross-react with other cortisol automated immunoassays.
Footnotes
Declaration of conflicting interests
None.
Funding
None.
Ethical approval
None required.
Guarantor
FJS.
Contributorship
FJS performed laboratory experiments and wrote the first draft of the manuscript. ASD conceived and designed experiments and reviewed the manuscript. LMB and AG reviewed and edited final manuscript.
