Abstract
Background
Fibromyalgia (FM) is a syndrome characterized by widespread chronic pain. Its aetiology is still poorly understood, and there are no haematochemical or instrumental tests on which to base a diagnosis. Recent studies suggest that its pathogenesis may involve cytokines, in particular, chemokines – cytokines that regulate cell traffic under both physiological and pathological conditions. The aim of this study was to determine possible differences in the profile of systemic concentrations of chemokines between FM patients and healthy women (HW; controls).
Methods
The study participants were women diagnosed with FM (n = 17) and a control group of HW (n = 10). Serum concentrations of thymus and activation-regulated chemokine (TARC)/(CCL17), monokine induced by gamma-interferon (MIG)/(CXCL9), macrophage-derived chemokine (MDC)/(CCL22), interferon-inducible T-cell alpha chemoattractant (I-TAC)/(CXCL11), eotaxin (CCL11), pulmonary and activation-regulated chemokine (PARC)/(CCL18) and hemofiltrate CC-chemokine-4 (HCC-4)/(CCL16) were determined by enzyme-linked immunosorbent assay and compared between the FM and HW groups.
Results
FM patients had elevated serum levels of the following inflammatory chemokines: TARC (P < 0.001), MIG (P < 0.001), MDC (P < 0.01), I-TAC (P < 0.01) and eotaxin (P < 0.05). No differences were found in the circulating concentrations of PARC and HCC-4 (homoeostatic chemokines).
Conclusions
Since FM patients present higher serum concentrations of inflammatory chemokines than HW, the evaluation of these biomarkers could help in the diagnosis of this syndrome.
Introduction
CHEMOattractant cytoKINES or chemokines constitute a family of small soluble signalling molecules of approximately 70 amino acid residues with a molecular weight of 7–12 kDa. They play crucial roles in both homoeostasis and disease. Their homoeostatic roles include leukocyte maturation and trafficking, development, tissue repair and angiogenesis.1,2 As disease modulators, chemokines have roles in a wide variety of inflammatory and immune responses through the chemoattraction of innate and adaptive immune cells. 3 They selectively control, often with specificity, such phenomena as adhesion, chemotaxis and the activation of many types of populations and sub-populations of leukocytes, and hence are major regulators of leukocyte traffic. Besides their structural classification (CXC, CC, CX3C or XC, according to the arrangement of cysteine residues involved in the formation of disulphide bonds), another organization of chemokines has been proposed based on their expression and their functional activity. This classification groups chemokines into three ‘families’: pro-inflammatory, homoeostatic and mixed function. 4 Pro-inflammatory chemokines (e.g. monokine induced by gamma-interferon (MIG)/CXCL9, interferon-inducible T-cell alpha chemoattractant (I-TAC)/CXCL11 and eotaxin/CCL11) are up-regulated under inflammatory conditions and are involved in leukocyte recruitment to inflamed sites. Homoeostatic chemokines (e.g. hemofiltrate CC-chemokine-4 (HCC-4)/CCL16 and pulmonary and activation-regulated chemokine (PARC)/CCL18) are expressed constitutively at non-inflamed sites and are involved in homoeostatic migration and homing of cells in physiological conditions such as lymphocyte homing. Some chemokines have both properties, and are thus called mixed-function chemokines (e.g. thymus and activation-regulated chemokine (TARC)/CCL17 and macrophage-derived chemokine (MDC)/CCL22).2,5 In addition, chemokines appear to be molecules with a role in coordinating nociceptive events associated with injury. They enhance sensitivity to pain by direct action on chemokine receptors expressed in nociceptive neurons, 6 and also serve to regulate the inflammatory response by acting simultaneously on elements of the nervous system. 7
The diagnostic criteria of the American College of Rheumatology (ACR) currently define fibromyalgia (FM) as the presence of widespread pain (on both the left and the right sides of the body, both above and below the waist and obligatorily with axial skeletal pain being present) and tenderness at 11 or more of 18 specific tender point sites. 8 Thus, core symptoms of FM include pain, fatigue, and mood and sleep disturbances.9–11 Although there is much speculation about the aetiology of FM, one of the major theories is that cytokines may have a role in both the aetiology of the syndrome and in the intensity of core symptoms. 12 Based on the assumption that the origin of all pain is inflammation and the inflammatory response, 13 special attention has been paid to the inflammatory hypothesis of FM 14 with the implication of inflammatory cytokine involvement in the syndrome and in the modulation of pain. 12 Although there are conflicting results related to cytokine levels,15,16 FM might be caused by a mild inflammatory condition, since most FM patients show elevated concentrations of serum IL-8,15,17–19 of IL-6 in the supernatants of peripheral blood mononuclear cells (PBMC), 15 and of IL-1β, TNF (tumor necrosis factor)-α, IL-6 and IL-10 in the supernatants of isolated monocytes. 20 However, chemotactic cytokines, or chemokines, have been little studied in people with FM, with just a single study showing that MCP-1 (monocyte chemotactic protein-1) and eotaxin concentrations are elevated in the plasma of women with FM. 21
Valid and reliable biomarkers would help in standardizing the diagnosis of FM and in monitoring the response to treatment as well as improve the quality of outcome studies and clinical care. Thus, the aim of this study was to determine possible differences in the profile of systemic concentrations of chemokines between FM patients and healthy women (HW; controls), and to discuss their presumable diagnostic value.
Material and methods
Study design and FM patients
Anthropometric, demographic and clinical data.
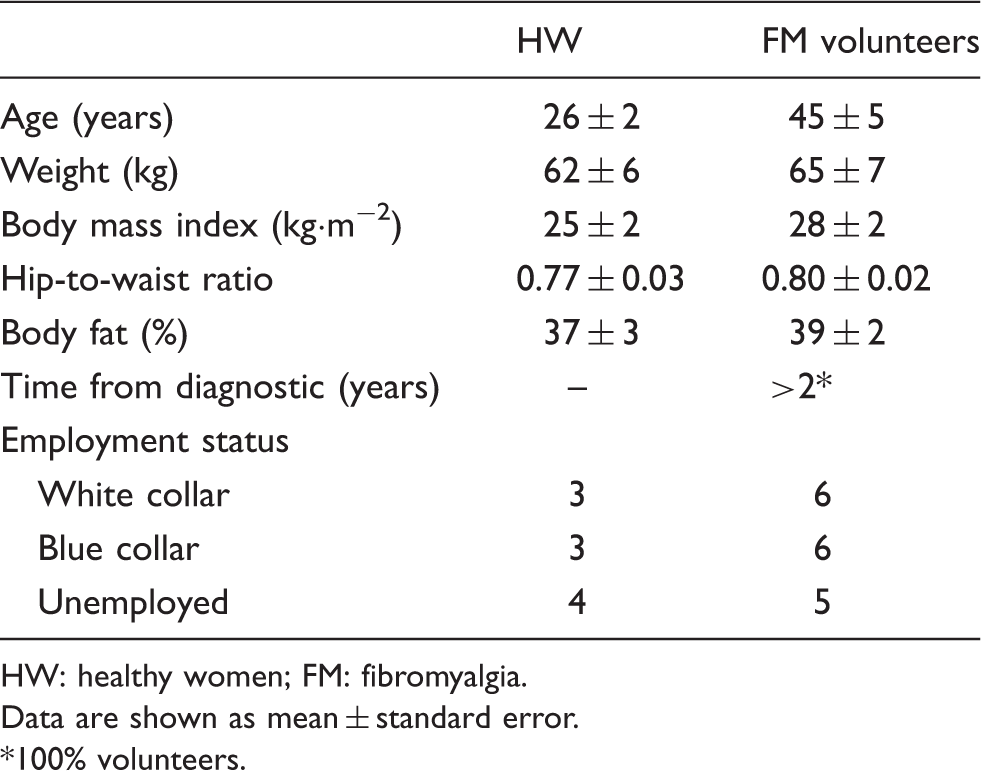
HW: healthy women; FM: fibromyalgia.
Data are shown as mean ± standard error.
100% volunteers.
Serum
Serum was obtained from the blood drawn by antecubital vein puncture. The blood extractions were performed by a qualified person. After extraction, blood samples for serum isolation were maintained for 15–20 min at room temperature. The blood was centrifuged at 700 g for 10 min. Finally, the serum samples were aliquoted and stored at −80℃ until assay.
Determination of systemic chemokine markers
Serum concentrations of TARC (CCL17) (RayBiotech, Norcross, GA, USA), MIG (CXCL9) (RayBiotech), MDC (CCL22) (CUSABIO), I-TAC (CXCL11) (RayBiotech), eotaxin (CCL11) (RayBiotech), PARC (CCL18) (RayBiotech) and HCC-4 (CCL16) (RayBiotech) were determined by enzyme-linked immunosorbent assay and compared between the FM and HW groups.
Statistical analysis
Values are given as the mean (± SEM). The variables were normally distributed (Kolmogorov–Smirnov test). Student’s t-test was used to compare the results between the HW and FM groups. The significance level was set at P < 0.05. The analyses were performed using the software GraphPad Prism 5.0.3.
Results
The FM patients had higher serum levels than HW in the following pro-inflammatory chemokines: TARC (56.83 ± 8.03 vs. 23.05 ± 2.79; P < 0.001, Figure 1(a)), MIG (8227 ± 1219 vs. 1792 ± 64.68; P < 0.001, Figure 1(b)), MDC (354.5 ± 17.17 vs. 190 ± 40.01; P < 0.01, Figure 1(c)), I-TAC (70.25 ± 10.97 vs. 24.83 ± 4.30; P < 0.01, Figure 1(d)) and eotaxin (6862 ± 545.2 vs. 5371 ± 564.4; P < 0.05, Figure 1(e)). We believe it is particularly interesting that 100% of the FM patients had higher serum MIG and MDC levels than the mean value obtained for the control group of HW, with the equivalent proportions for the other pro-inflammatory chemokines being 94.1% for TARC, 82.4% for I-TAC and 76.5% for eotaxin.
Serum concentrations of TARC (a), MIG (b), MDC (c), I-TAC (d), eotaxin (e), PARC (f) and HCC-4 (g) in healthy women (HW; n = 10) and in fibromyalgia patients (FM; n = 17). Each point represents the value of the concentration in one individual. The black line represents the means ± SEM. P < 0.05, P < 0.01 and P < 0.001 with respect to HW (Student's t-test).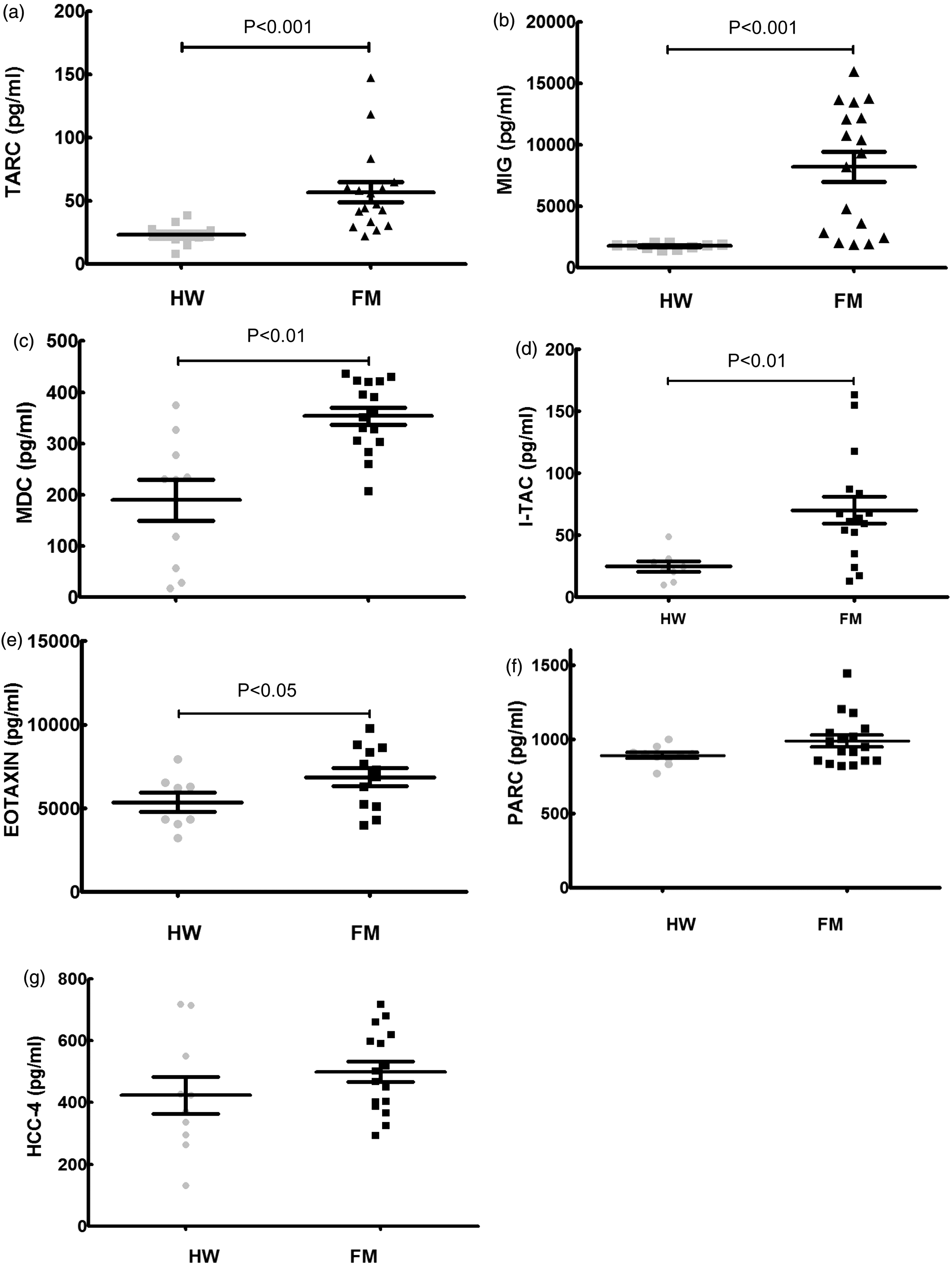
However, there were no statistically significant differences between the two groups in the serum levels of PARC (986.4 ± 40.36 vs. 890 ± 19.55; P > 0.05, Figure 1(f)) or HCC-4 (499.4 ± 33.17 vs. 423 ± 60.09; P > 0.05, Figure 1(g)), although there was a slight increase in the concentrations determined for both chemokines in the FM patients relative to the mean values of the controls (with 64.7% of the FM patients presenting values above the mean of the HW group).
Discussion
One of the major limitations in the FM research and the clinical care for FM patients is the lack of an objective marker to guide the diagnosis and to monitor the progression of treatment, or to predict the response to treatment. In previous studies by our research group, we found that FM patients have a high systemic concentration of IL-8 (CXCL8),18,19 but normal values of the pro-inflammatory cytokines TNF-α, IL-1β, IL-2, IL-6 and IL-18 and of the anti-inflammatory cytokines IL-10 and IL-4. 18 As well as being a pro-inflammatory cytokine, IL-8 is also considered to be an inflammatory chemokine since it is a potent chemoattractant for neutrophils 23 (cells essential for the development of the inflammatory response), besides being a potent promoter of sympathetic pain.19,24 With this in mind, we decided to study the systemic chemokine profile of FM patients to try to establish a characteristic profile that might be of potential use as an aid in the diagnosis of the disease. To the best of our knowledge, this study has been the first to jointly evaluate several chemokines, both inflammatory (MIG, MDC, I-TAC, TARC and eotaxin) and homoeostatic (PARC and HCC-4), in women diagnosed with primary FM. The results showed 100% of the FM patients to have higher serum levels of MIG and MDC than the mean values of these chemokines determined in the control group of healthy persons. There were also significantly higher values of I-TAC, TARC and eotaxin. Such raised levels of inflammatory chemokines in the FM patients are consistent with an underlying low grade inflammation. 19 The results are in agreement with the increased eotaxin and MCP-1 levels in FM patients reported by Zhang et al. 21 They may also, in part, explain the greater chemotactic capacity of neutrophils from these patients 19 since the CXCL-type chemokines (such as IL-8, MIG and I-TAC) are primarily strong chemoattractants for neutrophils.25,26
The observed increase in levels of MIG and TARC may be explained by the increased concentrations of IFN-γ that have been observed in some patients with primary FM, 18 since these two chemokines are produced in response to IFN-γ 27 while their release is blocked by IL-10 and IL-427 whose levels are unaltered in FM. 18 Similarly, IFN-γ has also been described as the inducer of I-TAC 28 and eotaxin, 29 so that an increase of this pro-inflammatory cytokine could be responsible for the observed increase of these two chemokines in FM.
Regarding the homoeostatic chemokines studied (PARC and HCC-4), the present results were also consistent with expectations, since the production of PARC30,31 and HCC-4 32 is stimulated by IL-10 and/or TNF-α whose levels are not elevated in the blood of FM patients, 18 and IFN-γ has an inhibitory effect on the production of PARC. 33
The present data confirm that in FM, there exists an inflammatory state which may contribute to the patients’ pain. This state is reflected in the elevated levels of inflammatory chemokines – MDC, MIG, TARC, I-TAC and eotaxin – observed in the present work, together with the previously described elevated level of IL-8.15,17–19 All these are molecules that mediate both inflammatory pain and neuropathic pain. 6 Since the main symptom of FM is chronic widespread pain, one is led to think that, as modulators of nociceptors, 6 these chemokines must be involved in the pathophysiology of the syndrome. Indeed, in the mechanisms of pain, chemokines increase sensitivity by directly acting on their receptors (which are present along the entire pathway of pain – in the peripheral nerves, the site of nerve damage, the dorsal root ganglia and the spinal cord). 6 Future research on the sites of action of these chemokines in FM would help clarify its pathophysiology.
Potential limitation of the investigation
Although the conclusions of the present pilot study are based on the statistical analysis between FM (n = 17) and HW (n = 10) volunteers, a limitation could be the limited size of sample. In any case, in our opinion, it can open further clinical investigations with a greater number of patients; in particular, on the elevated chemokines. Although, to the best of our knowledge, there are no published data reporting age-associated changes in concentration of the chemokines determined in the present investigation in a range of age between 20 and 50 years, and in our previous experience, we have not found variations in circulating inflammatory cytokines from women in the same range of age (‘adult women’); other limitation of the present study may be the difference in age between FM patients and HW.
In conclusion, FM patients present an altered profile of chemokines with higher serum concentrations of inflammatory chemokines (TARC/CCL17, MIG/CXCL9, MDC/CCL22, I-TAC/CXCL11 and eotaxin/CCL11) relative to HW. The evaluation of these chemokines could help in the diagnosis of this syndrome.
Footnotes
Acknowledgements
We thank the Fibromyalgia Associations of Don Benito and Badajoz, Spain, for their invaluable collaboration. We would like to thank Dr Robert Chatwin for language revision.
Declaration of conflicting interests
None declared.
Funding
This work was supported by grants PRIS10019 (FUNDESALUD), PRI09A003 and GR10020 (Junta de Extremadura-FEDER).
Ethical approval
The study was approved by the Ethical Committee of the University of Extremadura (Spain) according to the guidelines of the European Community Council Directives and the Declaration of Helsinki.
Guarantor
JJG.
Contributorship
JJG and EO researched literature and conceived the study. All authors were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. JJG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
