Abstract
Background
As lifespan in HIV infection increases, cardiovascular disease has emerged as a cause of morbidity and mortality. Asymmetric dimethylarginine is an established marker of endothelial dysfunction and predicts cardiovascular events. The role of asymmetric dimethylarginine in HIV-related cardiovascular disease has not been established. Our aim was to determine whether asymmetric dimethylarginine concentrations were elevated in treatment naïve, HIV-infected subjects and to correlate these with markers of immune activation and disease progression.
Methods
Serum samples were collected from HIV-positive and -negative subjects attending a primary health care clinic over a 12-month period. Asymmetric dimethylarginine concentrations were measured and correlated with CD4 count, viral load, hsCRP, IL-6, IgG, adenosine deaminase and CD8/38 T lymphocytes.
Results
Sixty HIV-positive participants (mean age 32.0 years) and 20 HIV-negative controls (mean age 32.4 years) were studied. All were of black ethnicity. The mean asymmetric dimethylarginine concentration in the infected group measured 0.67 µmol/L (95% CI 0.62–0.72 µmol/L) which was significantly higher than in the control group of 0.48 µmol/L (95% CI 0.40–0.56 µmol/L). Asymmetric dimethylarginine correlated inversely with CD4 counts and positively with IgG, adenosine deaminase and CD8/38 T lymphocytes. No significant correlation was found with hsCRP, IL-6, or viral load.
Conclusion
We demonstrated that asymmetric dimethylarginine is elevated in HIV infection, in patients with relatively well-preserved CD4 counts not yet on anti-retroviral treatment. We showed significant correlations of asymmetric dimethylarginine with CD8/38 T lymphocytes, IgG and adenosine deaminase, suggesting that T-cell activation and the adaptive immune response underlie asymmetric dimethylarginine elevation in this population.
Introduction
The lifespan of HIV-infected patients is increasing as treatment regimens improve. Cardiovascular disease has now emerged as an important cause of morbidity and mortality in this population.1,2 The increased cardiovascular risk is likely multi-factorial. Lipid disorders and insulin resistance associated with the use of anti-retroviral therapy (ART) were initially widely implicated.3–5 However, atherosclerosis is now recognized as an inflammatory process 6 and there is growing evidence that the disordered systemic inflammatory response associated with HIV is likely to contribute to endothelial dysfunction and atherosclerosis in this patient group.
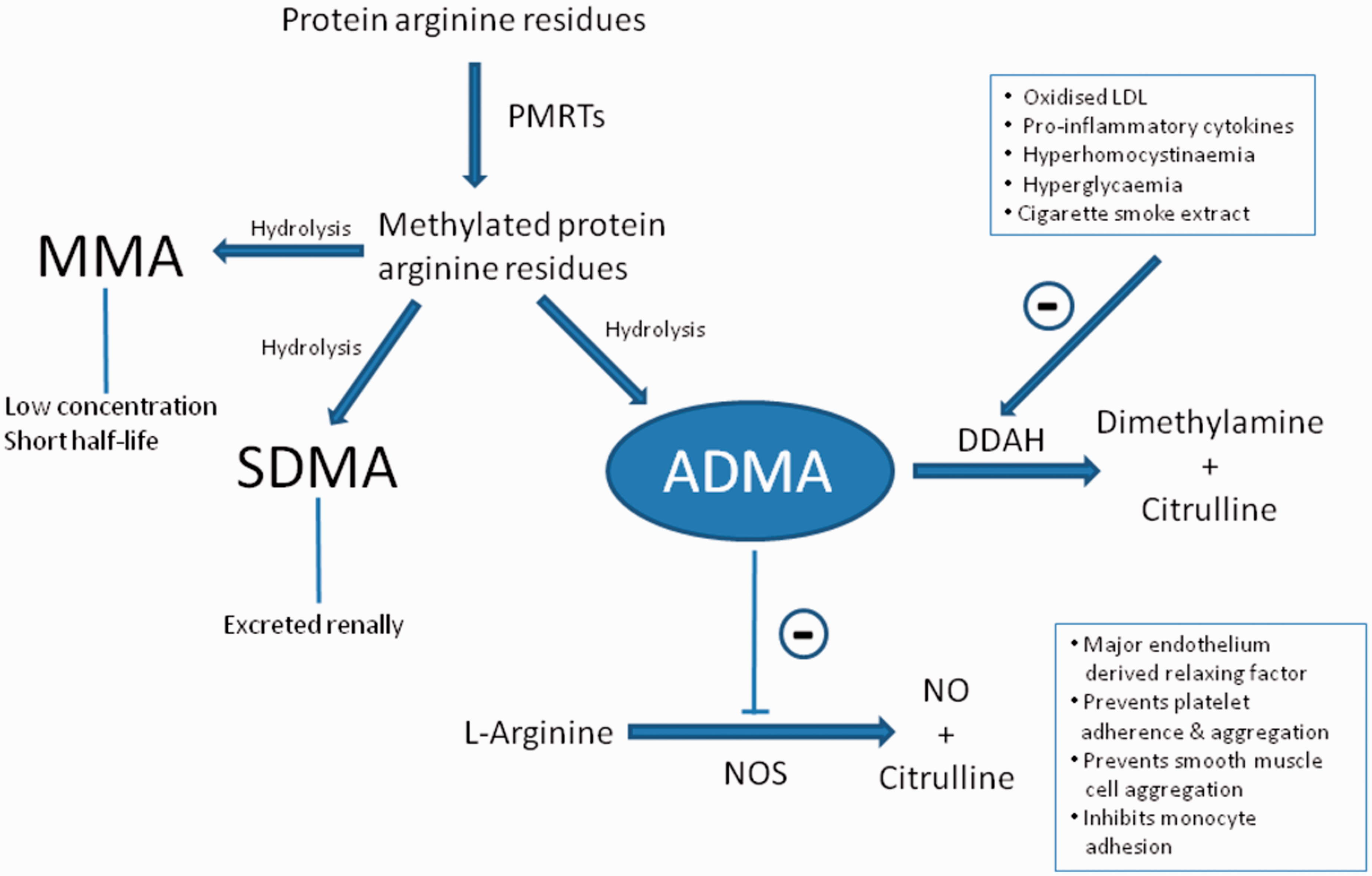
Asymmetric dimethylarginine (ADMA) is an established marker of endothelial dysfunction in a number of pathological states.7–13 It functions as an endogenous inhibitor of all nitric oxide synthase (NOS) isoforms. Nitric oxide (NO) is an anti-atherogenic molecule functioning as the major endothelium-derived relaxing factor. Apart from its vasodilatatory effects, NO has also been shown to prevent platelet adherence and aggregation, suppress smooth muscle cell proliferation and inhibit monocyte adhesion.
14
Post-translational arginine residue methylation is a ubiquitous, enzymatic process that contributes to the function of various proteins and has an important role in gene transcription.
15
Degradation of ADMA is mediated principally by dimethylarginine dimethylaminohydrolase (DDAH). It has been proposed that the elevation in plasma ADMA that occurs with vascular disease is largely due to impaired activity of DDAH.
16
This may result from a number of oxidative stress-inducing factors such as oxidized LDL and pro-inflammatory cytokines,
16
hyperhomocystinaemia,
17
hyperglycaemia
18
and cigarette smoke extract.
19
The reduction in DDAH activity allows ADMA to accumulate, resulting in a decrease in NOS activity and a loss of its vaso-protective effects (Figure 1). ADMA may thus represent the summative effect of numerous risk factors on endothelial health.
20
ADMA generation, elimination and inhibition of nitric oxide synthase (NOS). PRMTs, protein arginine methyltransferases; DDAH, dimethylarginine dimethylaminohydrolase; NO, nitric oxide; LDL, low density lipoprotein (adapted from Blackwell
15
).
ADMA concentrations have been shown to reliably predict cardiovascular events and mortality in patients with chronic kidney disease,7,8 cardiac disease,9,10 diabetes11,12 and in critical illness. 13 The role of ADMA in the premature development of cardiovascular disease in HIV remains to be established. A few studies have demonstrated elevated concentrations of ADMA in established HIV infection.21,22 However, concentrations have not been studied previously in early disease and have not been correlated with a more extensive panel of markers of immune activation and inflammation. Therefore, the aim of our study was to establish whether ADMA concentration is increased in untreated, asymptomatic HIV infection and whether these concentrations correlated with markers of disease progression and immune activation. The marker of immune activation, CD38 expression on CD8+ T lymphocytes has been identified as an important prognostic marker independent of CD4 counts and viral load. 23 Our hypothesis is that the persistent and aberrant inflammation associated with HIV contributes to endothelial dysfunction and would correlate with ADMA concentration in our study participants. We therefore included several markers of inflammation such as high-sensitivity CRP (hsCRP) and IL-6 as well as non-specific markers of chronic inflammation such as adenosine deaminase (ADA) and IgG. ADA is well described as a catabolic enzyme that is up-regulated in chronic inflammatory conditions. 24 Early work suggested that ADA activity increases in the serum and erythrocytes of HIV-positive subjects and correlates with disease stage. 25 B lymphocyte activation and polyclonal hypergammaglobulinaemia are characteristic of HIV infection 26 and serum IgG concentrations are a useful indicator of activation of the humoral arm of the adaptive immune response.
Methods
Patient selection
This was a cross-sectional cohort study. Sixty HIV infected, ART naïve participants and 20 HIV-negative controls were recruited from a single HIV counselling and testing (HCT) prevention clinic in Crossroads, Cape Town, South Africa. The prevention clinic employs the national HCT testing algorithm with accredited rapid tests. Clinical information was recorded on a standard proforma by the investigator from information obtained during patient interview. Data collected included: (i) Demographic details: age, gender, ethnic group. (ii) Medical history: HIV status, date of HIV diagnosis, WHO stage and most recent CD4 count if available. (iii) Medication use: including vitamins and herbal preparations. Patients taking ART, anti-tuberculous medications or therapy for any other infections were excluded from the study.
Determination of ADMA
Samples collected in serum separator tubes were spun at 1000 g for 15 min and the separated serum was stored at –70℃ before analysis in a single run. ADMA concentrations were measured in duplicate using an enzyme-linked immunosorbent assay (ELISA) (DLD Diagnostika, Hamburg, Germany). The reported reference interval of this assay is 0.40–0.75 µmol/L. The kit has an intra-assay CV of 5.7% at 0.66 µmol/L and an inter-assay CV of 10.3% at 0.66 µmol/L. The manufacturer quoted 1.2% cross-reactivity with symmetric dimethylarginine. The assay had a limit of detection (LOD) of 0.05 µmol/L and an average recovery of 104%.
Determination of markers of disease progression
HIV-1 viral load testing and CD4 counts were determined within 6 h of sampling. HIV-1 RNA quantifications were performed with a Nuclisens Easy Q HIV-1 v.1.2 kit (BioMerieux Inc., Boxtel, Netherlands), an assay with a lower detection limit of 40–50 copies/mL. CD4 T-cell counts were determined by flow cytometry on whole blood. The BD MultiTEST CD3-FITC/CD8-PE/CD45-PerCP/CD4-APC reagent was utilized together with BD TruCOUNT tubes (BD Biosciences, San Jose, CA).
Determination of markers of inflammation
A nephelometric rate method utilizing anti-CRP antibody-coated latex particles was used for hsCRP measurement (Immage® hsCRP, Beckman Coulter Inc. Brea, CA, USA). The assay had a reported total CV of 3.8% at 1.34 mg/dL and a reference value of <0.74 mg/dL. Serum IL-6 was determined using a Quantikine® ELISA assay (R&D Systems Inc, Minneapolis, MN). The assay had a reported inter-assay CV of 7.2% at 2.78 pg/mL and a LOD of 0.039 pg/mL.
Determination of markers of immune activation
CD38 expression on CD8+ T lymphocytes (CD8/38) was determined by flow cytometry. Whole blood samples were incubated with the monoclonal antibodies; CD8 Per-CP; CD38 APC; CD3 FITC (BD Biosciences, San Jose, CA) and analysed on a BD FACSCalibur instrument using BD Cell Quest Pro (Version 2) software. Lymphocytes were gated on forward vs. side scatter, CD3 and CD8 expression. Serum IgG concentrations were determined using a PEG-enhanced automated immunoturbidometric method on the ADVIA™ 1800 (Siemens Medical Solutions Diagnostics, Tarrytown, NY, USA). An enzymatic method generating a coloured quinine dye monitored in a kinetic manner was utilized for determining serum ADA. The quoted reference range was 0–15 U/L (Diazyme laboratories, Paway, CA, USA).
Statistical analysis
Data were analysed using STATISTICA® version 9.0 software (StatSoft Inc., Tulsa, OK, USA) and Excel® (Microsoft, Seattle, WA, USA). Descriptive statistics were used to analyse each parameter in terms of distribution, mean, median, quartiles, maximum and minimum values and standard deviation. Non-parametric tests were applied as not all the data fitted a Gaussian distribution. The Mann-Whitney U test was used to compare mean ADMA concentrations between the HIV-positive and -negative populations. Spearman’s rank correlations were utilized in order to compare the continuous variables.
Results
The 60 HIV-positive participants had a mean age of 32.0 years and 80% were females. They had a relatively well preserved CD4 count of 416.1 cells/mm3 (95% CI 369–463).
Parameter means and 95% confidence intervals (CI).

The mean ADMA concentration in the HIV-infected group measured 0.67 µmol/L (95% CI 0.62–0.72 µmol/L) was significantly higher (P < 0.00018) than the mean in the control group of 0.48 µmol/L (95% CI 0.40–0.56 µmol/L), (Table 1). The mean CD4 count, CD8/38, ADA and IgG concentrations were significantly different in the HIV-positive versus the HIV-negative control group while the concentrations of the acute inflammatory markers, hsCRP and IL-6 were not significantly different between the two groups.
ADMA concentration correlated negatively with CD4 counts (r = –0.24, P = 0.03) but not with viral load. However, significant associations were found between ADMA and markers of cellular immune activation, including CD38 on CD8 T-cells (r = 0.29, P = 0.01), IgG (r = 0.25, P = 0.02) and ADA (r = 0.36, P < 0.01). There was no significant correlation between ADMA and the acute inflammatory markers hsCRP and IL-6.
Discussion
In agreement with current literature, we found ADMA concentration to be significantly raised in HIV-infected subjects. Kurz et al. 21 demonstrated that ADMA concentration was elevated in HIV-1 infected patients versus HIV-negative controls. However, in contrast to our study of relatively young (mean age 32.0 years) and asymptomatic patients, their study population of 112 patients, had an average age of 40.3 years and relatively advanced disease, with a mean CD4 count of 181 and 55% of the patients met criteria for AIDS. A number of patients had previously received nucleoside monotherapy. In the Kurz et al. 21 study, ADMA concentration correlated with plasma neopterin leading the authors to conclude that ADMA accumulation is due to a Th-1 type immune response. Notably, ADMA concentration did not correlate with viral load, CD4 counts or CRP concentrations. A smaller study of 37 patients confirmed elevated ADMA concentration in HIV and found them to be associated with higher coronary artery calcium scores. 22 CD4 counts and viral loads were not determined and patients on ART were not excluded from this study. The average age of this cohort was 45 years. In concordance with our study, they also found no significant differences in CRP concentration between the positive and negative groups.
Notably, the difference in mean ADMA concentration between HIV-positive and -negative subjects was considerably greater in our study population (0.19 µmol/L) than the mean differences of 0.03 µmol/L 21 and 0.05 µmol/L 22 found in the two previous studies. ADMA concentrations are known to increase with age 27 and therefore it was conceivable that our relatively young cohort would have relatively low baseline ADMA concentrations. This was reflected in the average ADMA values obtained in our study which were lower than those previously reported in a reference population of 19–75 year olds. 27 Older subjects, as were recruited in previous studies, may already have elevated ADMA concentrations due to age-related disease and lifestyle factors. Thus, there may already be a degree of subclinical inflammation that blunts the DDAH response to the immune activation associated with HIV. Hence the difference in ADMA concentrations between HIV-positive and -negative subjects may be more distinct in a younger cohort. We also considered that previous studies measured ADMA with an HPLC method whilst we used a commercial ELISA. The HPLC methods tend to offer greater precision and produce lower results which may explain some of the differences in the study results.
Consistent with the view that immune perturbations underlie cardiovascular disease in HIV, ADMA concentration demonstrated a significant negative correlation with CD4 counts. Recent clinical outcome studies have demonstrated the link between low CD4 counts and cardiovascular disease.28,29 These studies were supported by others that used surrogate markers of cardiovascular disease such as carotid plaque 30 and arterial stiffness. 31 However, as reviewed by Hsue et al. 32 and Ipp and Zemlin, 33 others have argued that a lowered CD4 count may merely be a surrogate marker of a more generalized form of chronic inflammation and cardiovascular disease, as mediated by a composite of factors such as a leaky gut and/or other co-infections such as cytomegalovirus (CMV). The finding that ADMA concentration did not correlate with viral load in our cohort would attest to this, implying that factors other than the virus itself contribute to the ongoing inflammatory process in these patients.
T-cell activation predicts HIV disease progression independently of viral load in treatment naïve patients.34,35 The immune activation marker CD38 on CD8+ T lymphocytes has proven to be a stronger marker for disease progression and death than either CD4 count or viral load.23,36 The role of T-cell activation in HIV-related cardiovascular disease remains undetermined. There are currently conflicting data on T-cell activation and evidence of vascular dysfunction in HIV. In a cross-sectional study there was no association between CD38 expression and either carotid intima medial thickness (IMT) or endothelial function. 37 A case-control study from the National Institute of Allergy and Infectious Disease cohort added weight to this finding by demonstrating no evidence for an association between T-cell activation and subsequent cardiovascular events in chronic HIV infection. 38 In contrast, Kaplan and colleagues found that markers of T-cell activation, including CD38, were associated with carotid artery stiffness 39 and increased risk of carotid plaques. 40 Our study is the first to positively correlate CD38 on CD8 T lymphocytes with a plasma marker of endothelial dysfunction, supporting the hypothesis that immune activation, and specifically T-cell activation, are central to the pathogenesis of cardiovascular disease in this population.
Our finding that CRP did not correlate with ADMA concentrations is in keeping with the the finding of Kurz et al. 21 Intuitively, however, we expected that hsCRP, a marker of ongoing inflammation and an established predictor of future coronary events in the general population, would be increased in those patients with an elevated ADMA. In the Multicentre Aids Cohort Study (MACS) CRP was found to be a marker for disease progression in the enrolled HIV-infected males. 41 However, its value as a marker of cardiovascular risk in HIV, above that of traditional markers, such as those included in the Framingham risk score, remains to be determined. Current data are limited and contradictory. 42 Our treatment-naïve cohort results indicate that ADMA correlates better with markers of disease progression and immune activation than with CRP. Our finding that ADMA concentrations did not correlate positively with IL-6 heightens our suspicion that ADMA is increased in response to factors other than those involved in the acute inflammatory response.
The strong correlation of ADMA with CD38 on CD8 T lymphocytes indicates that ADMA may increase more in response to factors involved in the adaptive immune response in those living with HIV. ADA catalyses the conversion of adenosine to inosine and is up-regulated in chronic inflammatory conditions. Results from our study suggest that it may be a useful marker of non-specific immune activation and disease progression as it correlated positively with ADMA and negatively with CD4 count. Polyclonal hypergammaglobulinemia is well described in HIV infection and concentrations of IgG particularly, have been shown to be increased in untreated individuals. 43 The use of total IgG as a marker of non-specific immune activation has not been well explored until recently. 44 However, here we show the value of this marker by demonstrating its inverse correlation with CD4 counts and CD38 on CD8 T lymphocytes. In addition, IgG, along with ADA, correlated positively with ADMA demonstrating that ADMA concentrations may be increased in response to the adaptive immune response.
We recognize that our cross-sectional study design allows for only a ‘snapshot’ view of these markers in our cohort. Longitudinal studies are necessary to determine whether an elevated ADMA concentration has clinical relevance, translating into an increased risk of cardiovascular events in the HIV-infected population. The link between T-cell activation, chronic inflammation and elevated ADMA concentration, remains to be determined. The mechanism may involve increased propensity to protein arginine methylation or reduced DDAH enzyme activity possibly as a result of oxidative stressors. Given that protein methylation is a highly regulated process 14 we speculate that it is likely to be DDAH that is affected by factors associated with ongoing inflammation in HIV. Factors to be considered are those that were not measured in our study, including pro-inflammatory cytokines other than IL-6, oxidized LDL 16 and homocysteine 17 which are all known to inhibit DDAH activity. Recent work has already demonstrated that ADMA concentrations fall with ART 45 but whether treatment intervention and the use of specific classes of ART will improve endothelial dysfunction and reduce vascular risk, remains to be determined. We did not include smoking status as part of our questionnaire which may have influenced ADMA concentrations if smoking rates differed significantly between the HIV-positive and -negative groups. However, as both groups were recruited from the same primary health care clinic, the socioeconomic factors were likely to be similar for the whole cohort. We also did not include measurements of arginine and symmetric dimethylarginine which may have provided some insight into the mechanism underlying the ADMA increase in HIV. This was a drawback of the ELISA method we chose to employ over HPLC which has the ability to simultaneously measure these related metabolites. However, we wanted to utilize a relatively simple, affordable assay that could ultimately be developed into a point of care test. Along with other basic tests such as IgG and ADA, we envisage that ADMA could enable the early identification of patients at increased risk of disease progression or inflammatory associated complications, including cardiovascular complications, before CD4 counts drop below 350 cells/mm3.
Conclusion
In summary, our study is the first to demonstrate that the cardiovascular risk marker, ADMA, is elevated early in the course of HIV infection, in patients with well-preserved CD4 counts who have not yet received ART. Further, we showed that ADMA concentrations correlated inversely with CD4 lymphocyte counts but not with viral load, suggesting that factors other than the virus itself were contributing to ongoing inflammation in our cohort. ADMA concentrations correlated with the immune activation marker CD38 on CD8 T lymphocytes, ADA and IgG but not with hsCRP or IL-6. This introduces the concept that it is T-cell activation and the chronic inflammatory response which underlie the elevation of ADMA in HIV and that this mechanism should be considered when developing new treatment strategies to reduce cardiovascular morbidity and mortality in this population.
Footnotes
Acknowledgements
We wish to thank the patients and staff of the Emavundleni Prevention Centre of the Desmond Tutu HIV Centre in Crossroads Cape Town for their participation in this study and particularly Dr Surita Roux for her involvement and co-ordination. In addition, we would like to thank Muiruri Macharia for his help with running the ADMA ELISAs and Martin Kidd for his aid in the statistical analysis of the results.
Declaration of conflicting interests
None declared.
Funding
This research was supported by the National Health Laboratory Services Research Trust, South Africa, the Poliomyelitis Research Foundation (PRF), South Africa, the Department of Science and Technology of South Africa (through the SHARP initiative) and the Harry Crossley Foundation.
Ethical approval
The study was approved by the ethical committees of the University of Stellenbosch (HREC N07/09/197) and the University of Cape Town (REC 417/2006).
Guarantor
HI.
Contributorship
HI and AEZ conceived and designed the study. HI was involved in gaining ethical approval and patient recruitment. CLH and AEZ researched the literature and CLH wrote the first draft of the manuscript. All authors reviewed and edited the paper and approved the final version.
