Abstract
Objectives
To explore microRNA-202 (miR-202) expression in serum of patients with multiple myeloma (MM), and investigate correlations between serum miR-202 expression and the development and prognosis of MM.
Design and methods
RNA was extracted from serum by QIAGEN miRNeasy Mini kit. Reverse transcription was performed with specific stem-loop primers. SYBR Green I QF-PCR was applied to detect the relative expression of miR-202 in 40 MM patients and 30 healthy controls. The linearity, specificity and reproducibility were evaluated. In addition, correlations between the relative expression of serum miR-202 and the concentrations of lactic acid dehydrogenase (LDH), β2M, λ light chain and κ light chain were assessed.
Results
The relative expression of miR-202 in MM patients 1.503 (0.161–9.831) was significantly higher than that in healthy controls 1.000 (0.105–3.046) (P < 0.01) and was significantly correlated with serum β2M and κ light chain concentrations (r = 0.366, P = 0.0305; r = 0.358, P = 0.0348).
Conclusions
The relative expression of serum miR-202 in MM patients was significantly higher than that in healthy controls, and therefore it may prove to be useful in the auxiliary diagnosis of MM.
Introduction
Multiple myeloma (MM) is a B-cell malignancy characterized by abnormal proliferation of plasma cells in the bone marrow, accounting for about 10% of all malignant haematological diseases. 1 Previous studies2,3 have demonstrated that microRNA (miRNA) expression concentrations are associated with MM occurrence. MiRNAs are a group of small noncoding RNAs with 21–24 nt that inhibit or degrade specific target gene mRNAs by specific binding with 3′-end non-translation region of mRNA and regulate the expression of target genes at the post-transcriptional concentration. They may participate in and regulate a series of biological activities including cell proliferation, differentiation, apoptosis and immune response. 4
It was found in our previous study 5 that the expressions of B-cell activating factor (BAFF) and receptor in the peripheral blood of MM patients were significantly higher than those in the normal controls. BAFF could promote survival and proliferation of MM cells, which was associated with the development and progression of MM, and microRNA-202 (miR-202) was predicted to regulate BAFF expression by means of biological software. Therefore, we postulate that miR-202 may participate in the development and progression of MM. Our previous study also revealed that the miR-202 concentration in peripheral mononuclear cells of MM patients was higher than that in healthy controls, giving preliminary evidence that miR-202 may participate in the pathogenesis of MM. Knowing that the pre-treatment of collecting peripheral mononuclear cells may be influenced by various factors, the present study was attempted to detect the miR-202 content in serum samples directly from MM patients to see its clinical significance.
Materials and methods
Study population
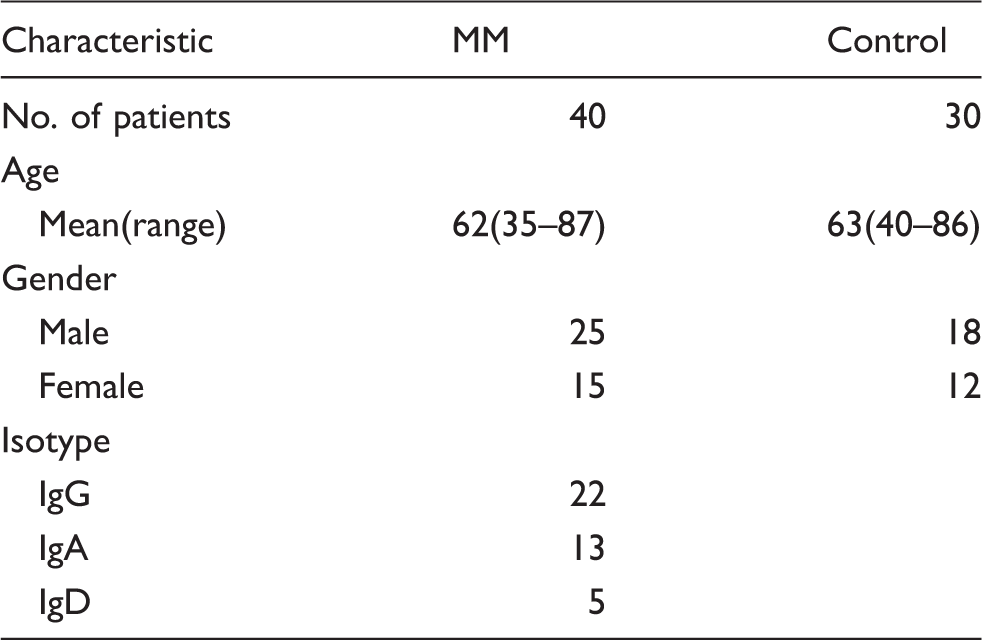
Characteristics of study subjects.
Main reagents and instruments
miR-202 and Arabidopsis thaliana (ath)-miR-156a looped RT primer, quantitative fluorescent polymerase chain reaction (QF-PCR) forward and reverse primers, ath-miR-156a standard (Guangzhou RiboBio Co., Ltd., Guangzhou, China); miRNeasy Mini kit (QIAGEN, Germany); DNA molecular marker (Shanghai Jie Rui Medical Reagents Co., Ltd., Shanghai, China); FastStart Universal SYBR GreenIMaster (Roche, Germany); high-speed refrigerated centrifuge (Hitachi, Japan); UV spectrophotometer (Limplen, Germany); 7500 Real Time PCR instrument (ABI, USA); ordinary PCR instrument (BIO-RAD, USA); reverse transcription reagent (Fermentas, Lithuania); special protein analyzer (Beckman Coulter, USA).
Specimen collection
Blood specimens were collected in separating gel vacuum collection tubes and centrifuged at 1000 × g for 10 min. The upper layer supernatant was stored in an RNase-free Eppendorf tube at −80℃ for use.
Serum RNA extraction and cDNA synthesis
RNA was extracted from 200 µL serum using miReasy Mini kit (Qiagen) according to the manufacturer’s protocol. To allow for the normalization of the samples variation in RNA isolation, 1 nM ath-miR-156a was added to serum before RNA isolation. RNA concentration and purity were measured by spectrophotometer. The 20 µL reaction volume included RNA 300 ng, 1.6 µL 62.5 nM looped RT primer, 4 µL 5 × Reaction Buffer, 2 µL dNTP (10 mM), 1 μL RNase inhibitor (20U/μl), 1 µL reverse transcriptase (200 U/µL) and nuclease-free H2O. The mix was incubated at 42℃ for 60 min and 70℃ for 5 min. The reverse transcription product was stored at −20℃.
Real-time QF-PCR
Real-time quantification was performed in triplicate with FastStart Universal SYBR Green Master (Rox) mix kit. Each reaction was performed in a final volume of 20 µL containing 9 µL SYBR Green I mix (Rox), 5 µL cDNA, 2 µL forward primer, 2 µL reverse primer and 2 µL RNase-free H2O. The mix was incubated at 95℃ for 10 min, followed by 42 cycles of 95℃ for 15 s and 60℃ for 60 min. Melting curve analysis (collected at 60–95℃) and 2% agarose gel electrophoresis were performed in order to validate the specificity of the PCR product. The relative expression of miR-202 was normalized to ath-miR-156a external reference, and was calculated by 2−ΔΔCt method, ΔCt = Ct(miR-202)-Ct(ath-miR-156a), ΔΔCt = ΔCt(patients)-ΔCt(control).
Statistical analysis
Statistical analysis was performed with SPSS 16.0 and graphs were generated with Graphpad Prism 5.0 software. The Ct value in each group was expressed as
Results
Linearity
After ten-fold serial dilutions (1:1, 1:10, 1:100, 1:1000 and 1:10,000) of miR-202 The standard curve of serum miR-202.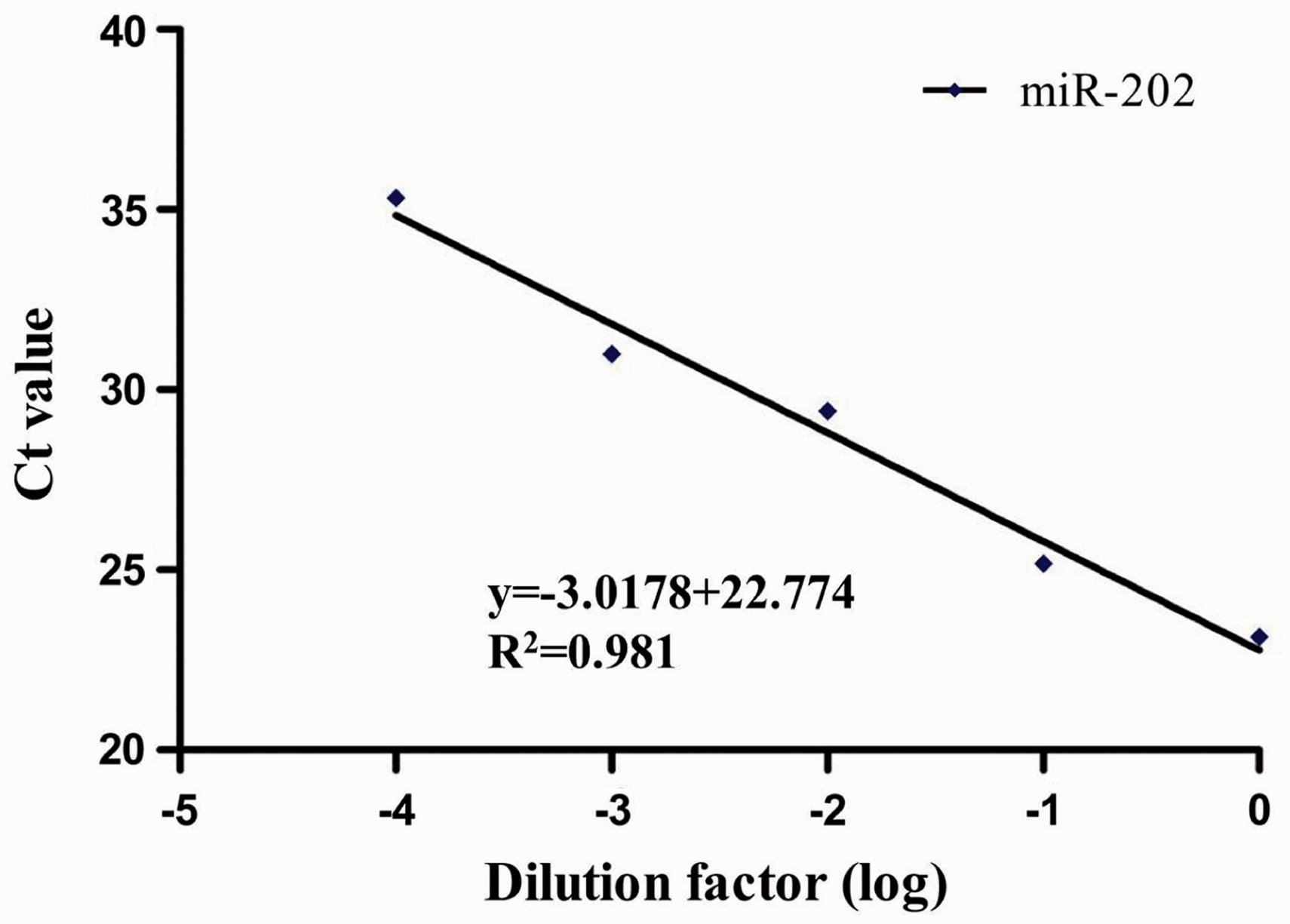
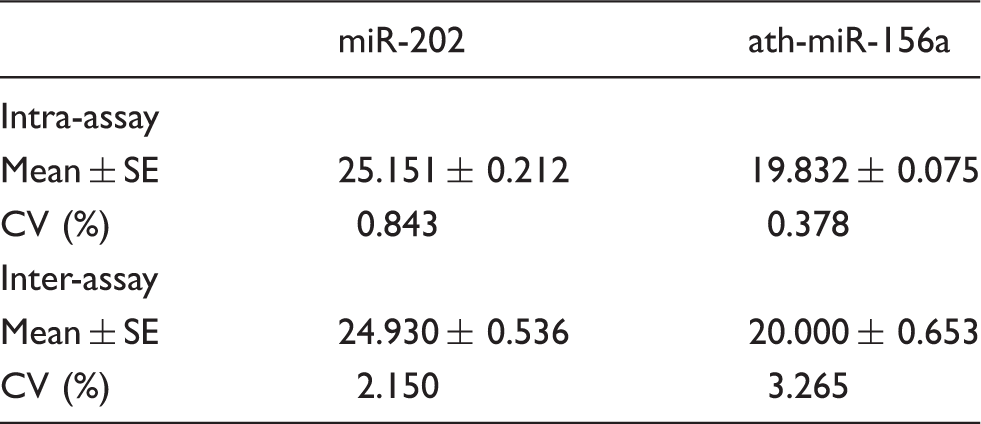
Reproducibility
Intra-assay and inter-assay repeatability.
Specificity
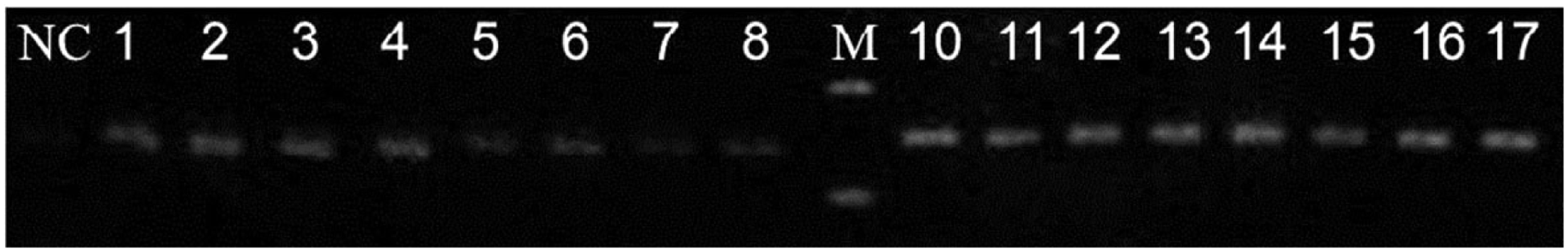
As shown in Figure 2, there was a specific single peak in QF-PCR melting curve. The melting temperature of miR-202 and ath-miR-156a was about 78℃ and 79℃, respectively. The specificity of QF-PCR product was validated by 2% agarose gel electrophoresis, showing that the miR-202 and ath-miR-156a products were 77 bp and 80 bp, respectively (Figure 3). The data aforementioned indicated our study had a high specificity.
QF-PCR melting curves of serum miR-202 and ath-miR-156a in MM patients and healthy controls. Agarose gel electrophoresis of serum miR-202 and ath-miR-156a QF-PCR products of MM patients and healthy controls.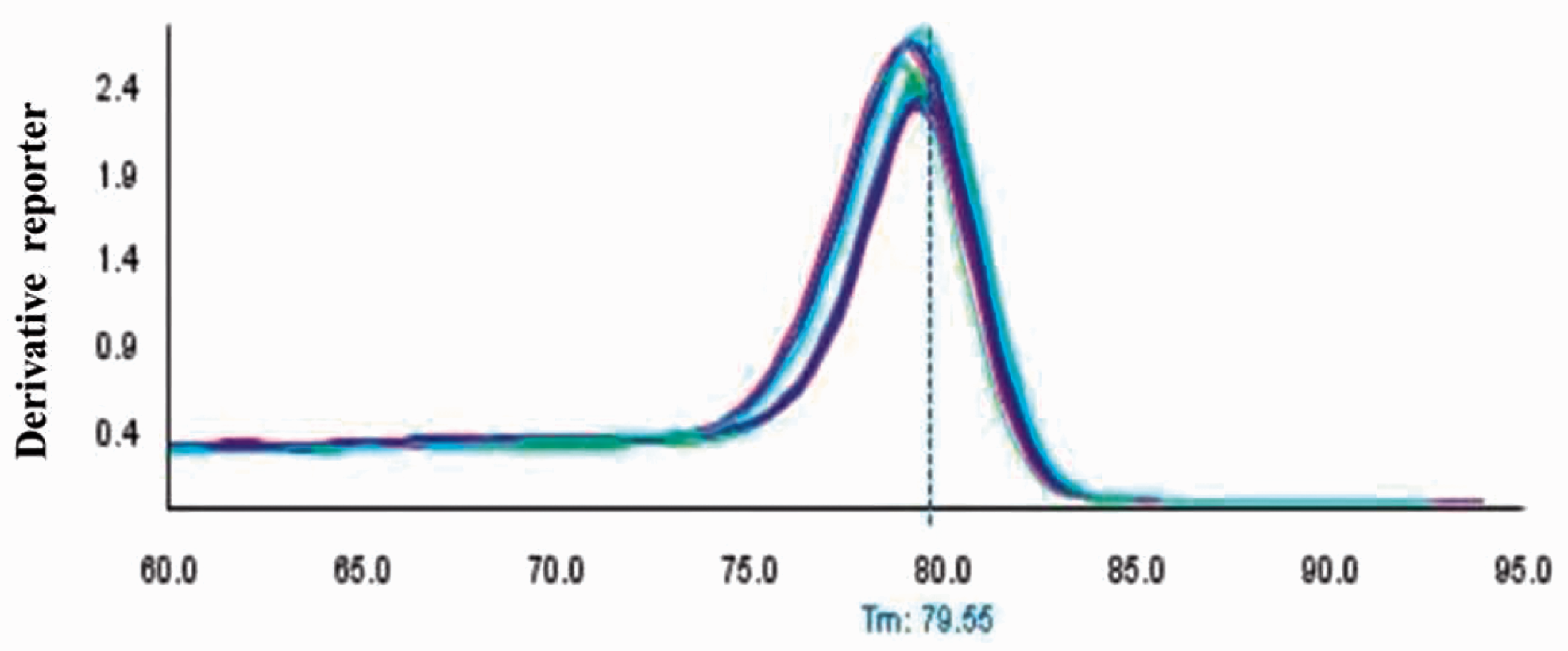
Serum miR-202 expression in MM patients and healthy controls
As normalized to ath-miR-156a, the relative expression of miR-202 in the 40 MM patients and 30 healthy controls was 1.503 (0.161–9.831) and 1.000 (0.105–3.046), respectively. Mann–Whitney test showed that there was significant difference between the two groups (U = 358.0, P < 0.01) (Figure 4), suggesting that the relative expression of miR-202 may be helpful in the auxiliary diagnosis of MM.
The relative expression of serum miR-202 in MM patients and healthy controls.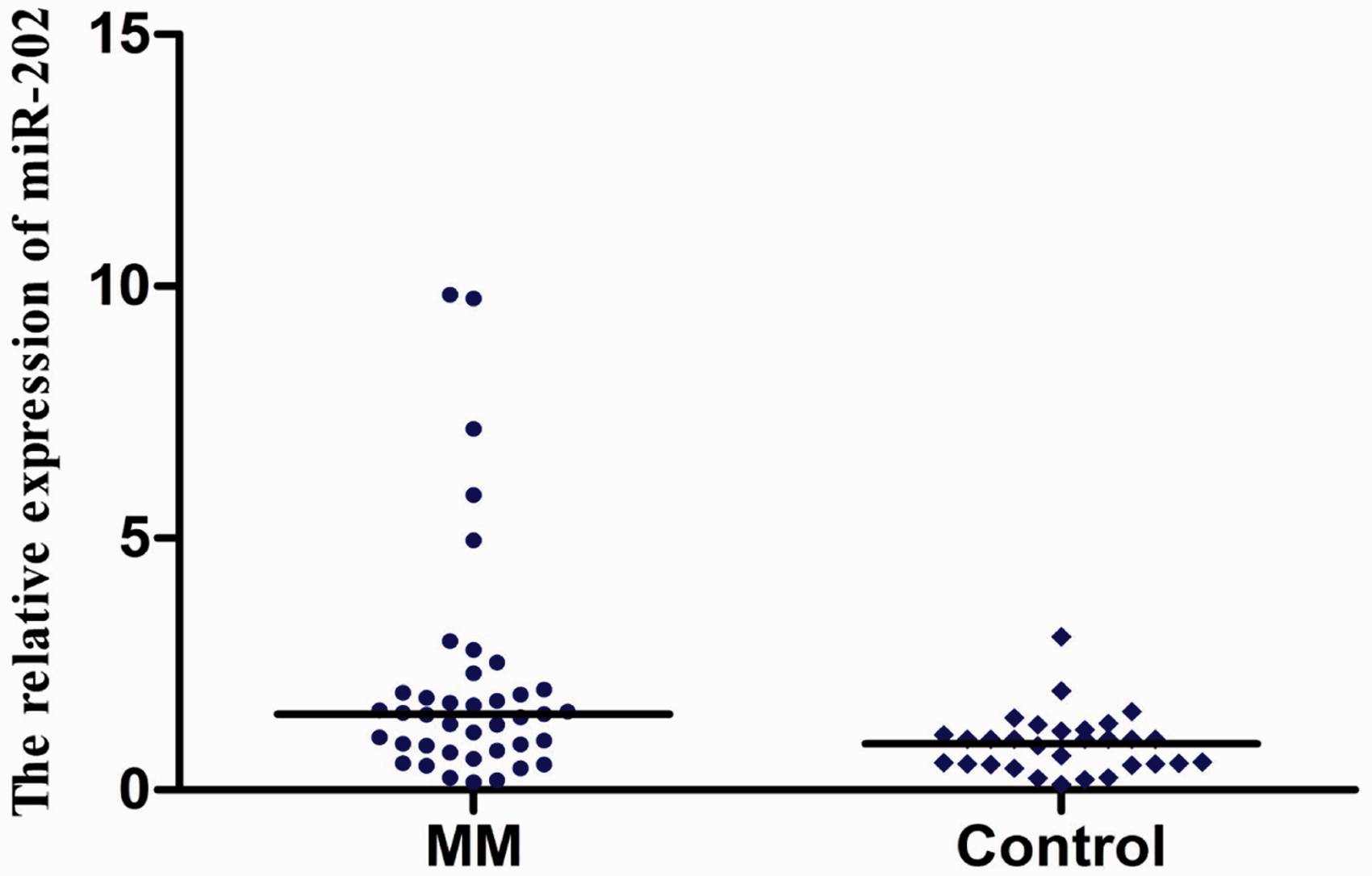
The clinical significance of expression concentration of miR-202 in the serum of MM patients
Receiver-operating characteristic (ROC) curve analysis was constructed to evaluate the diagnostic value of serum miR-202 for MM (Figure 5). The results showed moderate ability to differentiate the MM patients group from the healthy control group with an area under the curve of 0.711 (95% CI 0.579–0.825). At the cut-off values of 1.242, the sensitivity and the specificity for miR-202 in diagnosis of MM were 80.0% and 60.0%.
ROC curve of the miR-202 for differentiating the MM patients from healthy control group.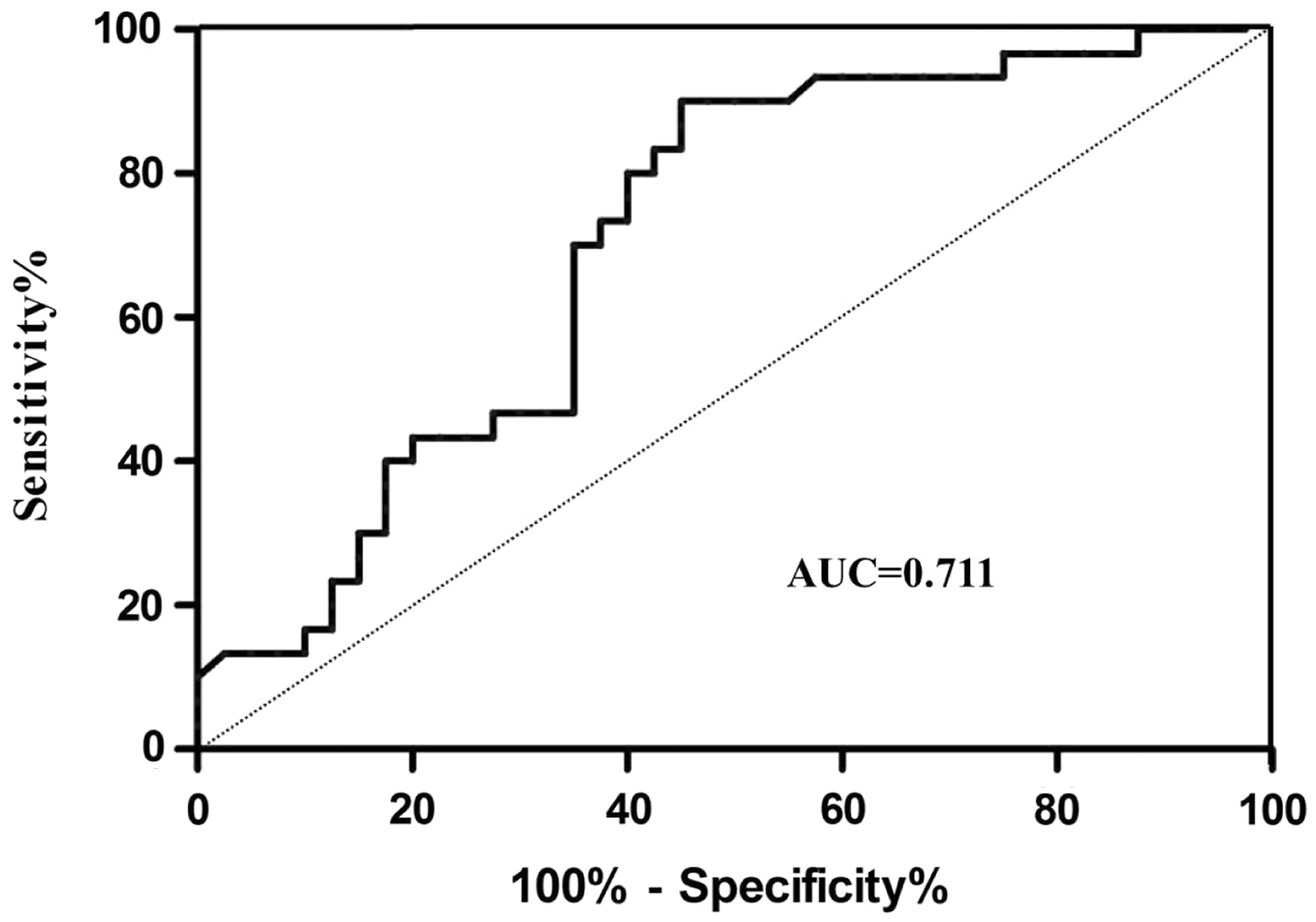
Correlations between the relative expression of serum miR-202 and clinical serum parameters in MM patients
Correlations between the relative expression of miR-202 and serum lactic acid dehydrogenase (LDH), β2M, λ light chain and κ light chain in the 35 MM patients were analysed. The results (Figure 6) showed that the relative expression of miR-202 was significantly correlated with β2M and κ light chain concentration (r = 0.366, P = 0.0305; r = 0.358, P = 0.0348), but not correlated with the LDH and λ light chain concentration (r = 0.330, P = 0.0528; r = 0.304, P = 0.0756).
Correlations between the relative expression of serum miR-202 and LDH, β2M, λ light chain and κ light chain in MM patients.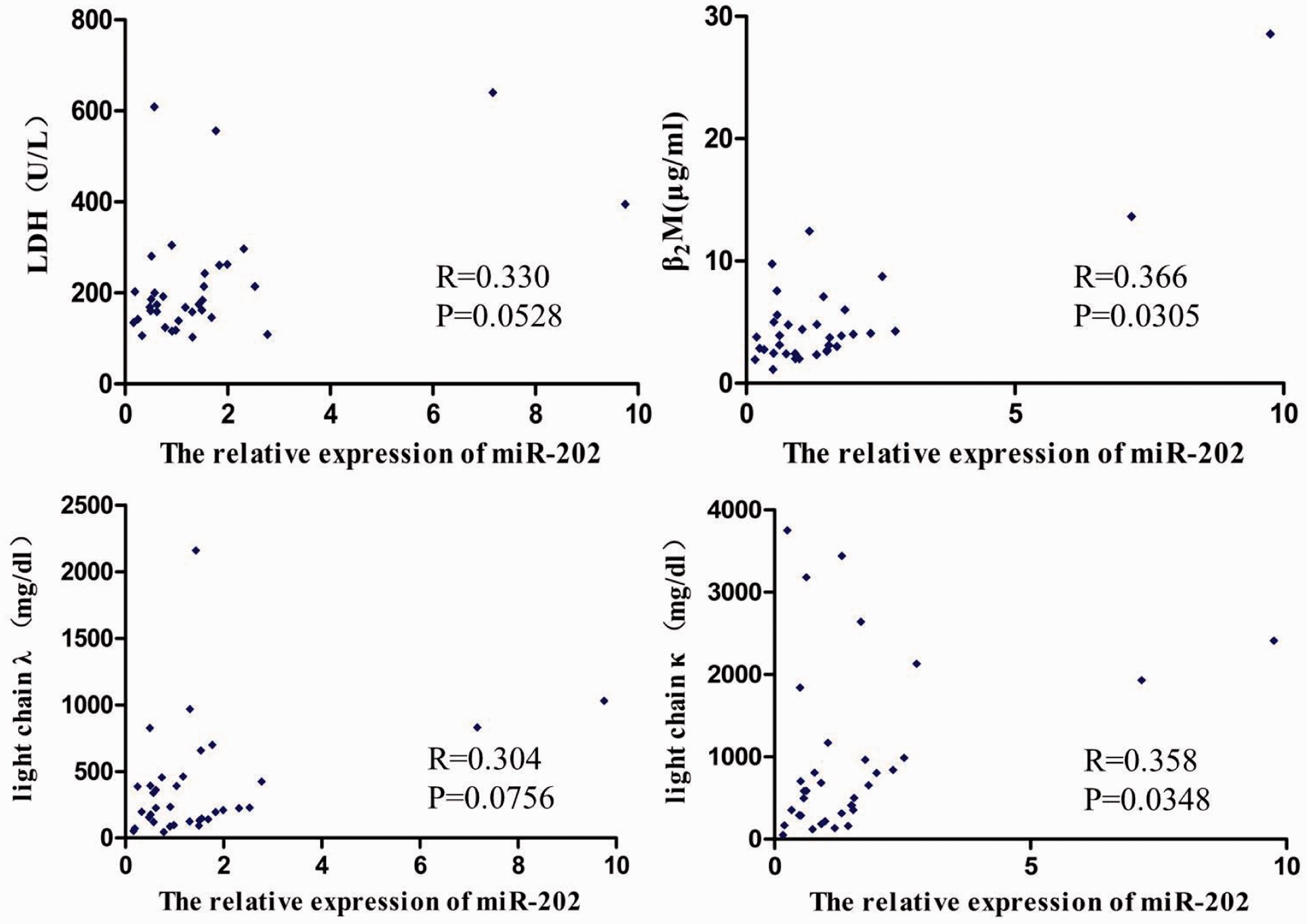
Discussion
After Lee et al. 6 discovered the first miRNA molecule (lin-4) in their study on the growth of Caenorhabditis elegance in 1993, Reinhart et al. 7 discovered the second miRNA molecule (let-7) in the same species. MiRNAs are a group of small noncoding RNAs with 21–24 nt that inhibit or degrade specific target gene mRNAs by specifically binding with 3′-end non-translation region of mRNA, and regulate the expression of target genes at the post-transcriptional concentration. MiRNAs may act as oncogenes or tumour suppressors and participate in a series of biological activities including cell proliferation, differentiation, apoptosis and immune response. 4 MiRNAs have recently elicited a growing interest as new therapeutical targets due to their ability to target multiple genes involved in cancer promotion or repression. 8 Duchaine and Slacks 9 have recently summarized the ‘double strategies’ concept: miRNAs as anticancer agents or miRNA as cancer targets, thus the treatment protocols should rely on the reconstitution of underexpressed miRNAs or on the antagonism of aberrantly expressed miRNAs. An important issue for this therapeutic approach is the efficient delivery in tumour tissues and the uptake by tumour cells. Di Martino et al. 10 have reported that synthetic miR-34a exerts a powerful antitumour activity in clinically relevant xenograft models of human MM via a novel lipid-based delivery vehicle. Tagliaferri et al. 11 have summarized the promises and challenges of miRNA-based treatment of MM. In addition, miRNA could be used in treatment of other solid tumours.12–15 Although current available data do not predict toxicity of this therapeutic approaches, it is necessary to conduct further identification with more specimens to confirm the conclusion.
Chen et al. 16 first found that miRNAs were present in the serum and plasma of humans and other animals, and could be detected directly in serum, thereby greatly facilitating clinical use of such tests. They also demonstrated that serum miRNAs were stable, reproducible and consistent among individuals of the same animal species. They identified specific expression patterns of serum miRNAs for specific diseases, providing evidence that serum miRNAs contain fingerprints for various diseases. Then Lawrie et al. 17 demonstrated that serum miRNA could be used as a potential tumour biomarker. Their study showed that the expression of miR-155, miR-210 and miR-21 in the serum of patients with diffuse large B-cell lymphoma was significantly higher than that in normal controls, and that the miR-21 expression concentration was closely correlated with patient survival. Large numbers of subsequent studies also confirmed that miRNA could be used for auxiliary diagnosis and differential diagnosis of tumours, prognosis prediction and monitoring of the disease course.16,18–20 Combination of serum miR-720 and miR-1308 together provides a powerful diagnostic tool for distinguishing normal healthy controls from pre-cancerous myeloma and myeloma patients. In addition, the combination of serum miR-1246 and miR-1308 can distinguish monoclonal gammopathy of undetermined significance (MGUS) from myeloma patients. 21 The plasma miR-92a concentration in symptomatic MM patients was significantly downregulated compared with smoldering MM patients, MGUS patients and normal subjects. The plasma miR-92a concentration in the complete remission group became normalized, whereas the partial response group did not reach the reference range. Therefore, the plasma miR-92a concentration may serve as a biomarker for monitoring therapeutic response in MM patients and disease progression in asymptomatic MM patients. 22
Previous studies 23 have demonstrated that compared with miRNA in normal tissues and cells, circulating miRNAs have a better RNase-resistance ability and can exist stably in the serum for a long time, or even remain stable under boiling, repeated freeze-thaw cycles, acidic and alkaline conditions. QF PCR was the main method for detecting serum miRNA due to quickness, high sensitivity and specificity. However, there was a low concentration of miRNA in the serum. To control the difference between specimens, it was often necessary to select appropriate internal or external references. Internal references used in different laboratories were often different, such as miR-16, 17 miR-142-3p, 24 miR-24 25 and RNU6B. 26 Some laboratories even added artificially made exogenous miRNA 18 and total RNA 16 to the circulation. Some studies in the literature reported that addition of exogenous miRNA control (cel-miR-45 or cel-miR-238) would not reduce the precision of the experiment, while internal reference miRNA-16 was likely to be affected by haemolysis and other factors, and its expression in the serum was not stable enough. Therefore they concluded that miR-16 was not a suitable internal reference. 27 In addition, some other studies 28 had reported that the expression of RNU6B in the plasma was relatively low and unstable, they thought that RNU6B was not a suitable internal reference, either. Recently, ath-miR-156a and cel-miR-39 were usually used as external reference of circulating miRNA because of the absence of homologous sequences in humans and their stability.22,29,30
In the present study, we added artificially synthetic ath-miR-156a to the serum and used QIAGEN miRNeasy Mini kit to extract serum RNA, which was reversely transcribed into cDNA using specific Bulge-Loop™ primer. Then, we detected the relative expression of miR-202 by SYBR Green I QF-PCR. The result of 2% agarose gel electrophoresis on the product obtained from this method exhibited a single bright band with a specific single peak without non-specific bands, showing our study had good specificity. After ten-fold serial dilution of miR-202 cDNA in the serum of the same patient, the R2 of the standard curve was 0.981, indicating a good linearity and the amplification efficiency of target gene was 1.1. The intra-assay and inter-assay CV of miR-202 were 0.843% and 2.150%, respectively, showing our study had a good repeatability. The result showed that serum miR-202 was differentially expressed in MM patients versus healthy controls. Serum λ light chain, κ light chain and β2M were closely correlated with the tumour-bearing status of MM patients. LDH can reflect tumour-proliferating activity, and therefore was of important value in assessing the therapeutic effect and prognosis. 31 It was found in this study that the relative expression of miR-202 was significantly correlated with the concentration of β2 M, and κ light chain ((r = 0.366, P = 0.0305; r = 0.358, P = 0.0348), indicating that circulating miR-202 may indirectly reflect the tumour-bearing status, and further investigation was needed to estimate if these serum miRNA were associated with changes occurring in MM. Therefore, the relative expression of serum miR-202 may prove to be useful in auxiliary diagnosis of MM. Unfortunately, we failed to find a significant correlation between the relative expression of miR-202 and the LDH, λ light chain concentrations (r = 0.330, P = 0.0528; r = 0.304, P = 0.0756), probably because we did not conduct stage classification of the patients.
The present study has suggested that miR-202 may be useful in auxiliary diagnosis of MM. As the sample size of the present study is relatively small, it is necessary to conduct further identification with clinical specimens to confirm our conclusion, and we need further fundamental research to confirm whether miR-202 concentrations correlated with the development and progression of MM.
Footnotes
Acknowledgements
We would like to thank Surgical Comprehensive Laboratory of Affiliated Hospital of Nantong University.
Declaration of conflicting interests
None.
Funding
This research was funded by the National Natural Science Foundation of China (81271920), the Natural Science Foundation of Jiangsu Province (BK2010284) and the Six Major Human Resources Project of Jiangsu Province (2008095).
Ethical approval
The study was approved by the Ethics Committee of Nantong University affiliated Hospital with the permit number: 2012(005).
Guarantor
SJ.
Contributorship
JY and XQ researched the literature and conceived the study. JY, XS, Ws and XW were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. JY wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
