Abstract
Background
Kisspeptin is a hypothalamic neuropeptide playing a physiological role in human reproduction. Genetic over-activation of kisspeptin causes precocious puberty in children. Concentrations of circulating kisspeptin are low in adults. The concentrations of plasma kisspeptin in boys and girls have not been studied previously.
Methods
Blood was obtained from 51 children and 63 adults. Plasma samples were analysed using radioimmunoassay. Children were aged 2–18 years, and attending hospital for a medically requested blood test unrelated to reproductive development. Data on pubertal status were not collected due to ethical reasons.
Results
Mean plasma kisspeptin was significantly higher in children when compared with adults (mean plasma kisspeptin in pmol/L: 12.3 ± 0.9, adults; 40.9 ± 3.3, children, P < 0.001 vs. adults). Overall mean concentrations of plasma kisspeptin were not significantly different between sexes (mean plasma kisspeptin in pmol/L: 39.5 ± 3.2, boys; 44.3 ± 6.3, girls, P = 0.48). In both sexes, concentrations of plasma kisspeptin increased with age to peak concentrations between 9 and 12 years of age, before decreasing beyond 12 years of age to adulthood. Plasma kisspeptin concentrations were highly significantly elevated in both girls and boys aged 9–12 when compared with adults (mean plasma kisspeptin in pmol/L: 59.5 ± 18.3, girls, P < 0.01 vs. adult women; 43.8 ± 6.2, boys, P < 0.001 vs. adult men).
Conclusions
We report that circulating kisspeptin is elevated in both boys and girls when compared with adults. Furthermore both boys and girls may have distinct, age-dependent concentrations of circulating kisspeptin. Further studies may determine if plasma kisspeptin could be used as a clinically useful biochemical marker of reproductive development in children.
Keywords
Introduction
Puberty is triggered by gonadotrophin releasing hormone (GnRH) secretion from the hypothalamus, which stimulates luteinising hormone (LH) and follicular stimulating hormone (FSH) secretion from the pituitary, which is needed for gonadal sex steroid synthesis and mature gamete release.
Kisspeptin is a neuropeptide encoded by the KISS1 gene, and released from specialized neurons which directly innervate hypothalamic GnRH neurons. 1 Hypothalamic expression of KISS1 and the kisspeptin receptor (KISS1R) increase during puberty in rats and monkeys. 1 Activating and inactivating mutations in KISS1 or KISS1R cause pubertal failure and precocious puberty in children, respectively.2,3 Intravenous administration of kisspeptin potently stimulates pituitary secretion of LH and FSH in humans; 4 however, this action is blocked by pre-treatment with a GnRH antagonist in other mammalian species. 5 Concentrations of hypothalamic kisspeptin are therefore causally associated with GnRH activation and reproductive maturation in humans.
A number of animal studies have examined concentrations of expression of KISS1 during reproductive development. However, there is a comparative paucity of data examining circulating concentrations of kisspeptin during human reproductive development. This is mainly attributable to the technical challenge of measuring plasma kisspeptin in blood samples; rapid centrifugation and separation is required in order to avoid substantial degradation of kisspeptin in blood samples. 6 It is therefore not possible to analyse stored serum or plasma samples retrospectively for concentrations of kisspeptin.
Kisspeptin circulates at high concentrations during pregnancy due to placental secretion (mean concentrations, 10,000 pmol/L), but at relatively low concentrations in other adults (mean concentrations, 1–10 pmol/L). 7
Concentrations of circulating kisspeptin are raised in girls with premature breast development. 8 A novel biochemical marker of pubertal activation might therefore help to manage children with pubertal disorders. This study aimed to measure for the first time, concentrations of plasma kisspeptin in boys and girls, and make comparison with circulating kisspeptin concentrations in adults.
Methods
Characteristics of subjects included in study. SDS, standard deviation score; M, male; F, female.

SDS: standard deviation score; M: male; F: female.
Blood was collected into lithium heparin Vacutainer tubes containing 5000 KIU aprotinin (0.2 mL Trasylol; Bayer, Newbury, UK). Samples were centrifuged immediately (3000 rotation per minute), separated and stored at −20℃ until measurement of kisspeptin immunoreactivity using a previously described, in-house radioimmunoassay based on a sheep GQ2 antibody. 6 Assay sensitivity was 2pmol/L, and the intra- and inter-assay coefficients of variation were 8.3% and 10.2%, respectively.
Data are presented as mean ± standard error of mean unless stated otherwise. Pairs of means were compared using the unpaired two-tailed t-test. Multiple means were compared using the one-way analysis of variance test with Tukey’s post hoc test. Linear regression was used to analyse associations between parameters. Standard deviation scores (SDS) were used to analyse height weight and body mass index (BMI) when corrected for age.
Results
The observed ranges of plasma kisspeptin concentrations were <2–32 pmol/L in adults, and <2–128 pmol/L in children (Figure 1(a)). Mean concentrations of plasma kisspeptin were significantly higher in children when compared with adults (mean concentrations of plasma kisspeptin in pmol/L: 12.3 ± 0.9, adults; 40.9 ± 3.3, children, P < 0.001 vs. adults). No significant associations were observed between concentrations of plasma kisspeptin and height SDS (r2 = 0.064; P = 0.08), weight SDS (r2 = 0.0091; P = 0.52) or BMI SDS (r2 = 0.0090; P = 0.52) of subjects (Figure 1(b)).
Concentrations of plasma kisspeptin in children and adults. (a) Concentrations of plasma kisspeptin immunoreactivity presented in children (n = 51) and adults (n = 63) attending outpatient clinic visits. Mean ± SD is presented; ***P < 0.001. (b) Scatterplot of plasma kisspeptin vs.SDS of height, weight and BMI. Linear regression (solid lines) revealed no significant associations.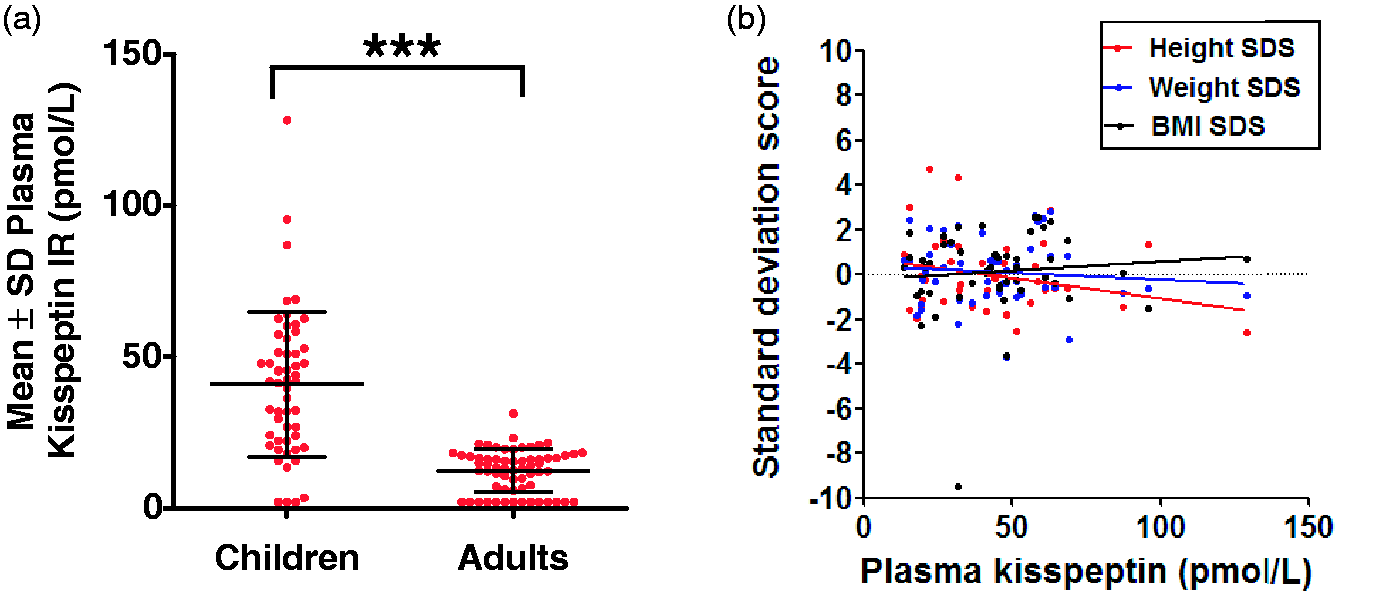
No clinical data on pubertal development were available for ethical reasons; therefore, we used subject age as a clinical surrogate of physical maturation in children. As expected, subject age was highly significantly and positively correlated with height (r2 = 0.71; P < 0.0001), weight (r2 = 0.60; P < 0.0001) and BMI (r2 = 0.38; P < 0.0001) of subjects (Figure 2(a)).
Age-dependent elevations in plasma kisspeptin in children when compared with adults. (a) Scatterplot of subject age vs. height, weight and BMI. Linear regression (solid lines) revealed highly significant associations between subject age and height (P < 0001), weight (P < 0.0001) and BMI (P < 0.0001). Mean plasma kisspeptin in males (b) and females (c) when classified by age. #P < 0.05 vs. adult concentrations. ##P < 0.01 vs. adult concentrations. ###P < 0.001 vs. adult concentrations. Data are mean ± SEM.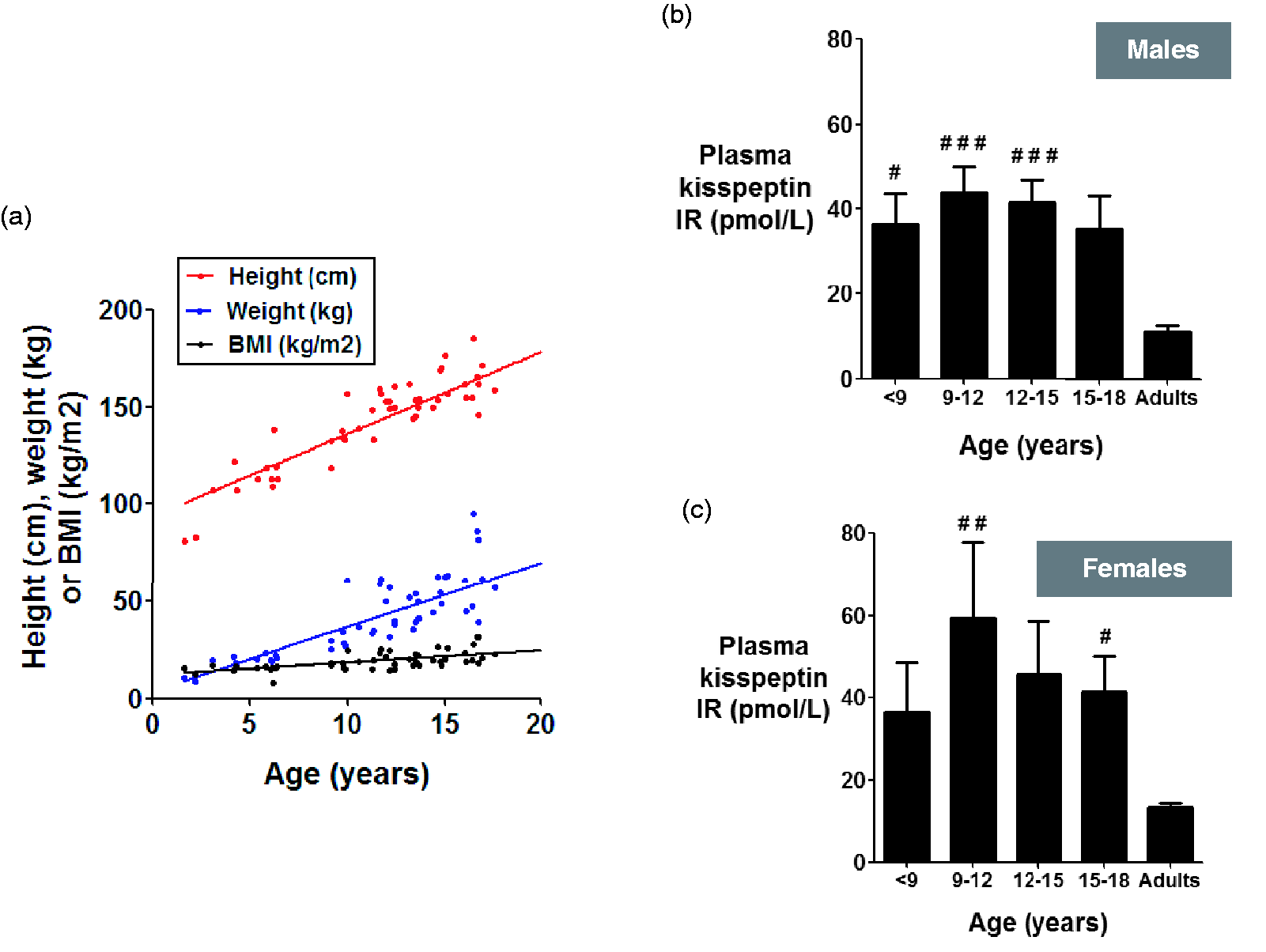
Overall mean concentrations of plasma kisspeptin were not significantly different between sexes (mean concentrations of plasma kisspeptin in pmol/L: 39.5 ± 3.2, boys; 44.3 ± 6.3, girls, P = 0.48) (data not shown); however, when subjects were classified into both sex and age, concentrations of plasma kisspeptin appeared slightly higher in girls when compared with boys (Figure 2(b) and (c)). In both sexes, concentrations of plasma kisspeptin appeared to increase with age to peak concentrations between 9 and 12 years of age, before decreasing beyond 12 years of age to adulthood. Plasma kisspeptin concentrations were highly significantly elevated in both girls and boys aged 9–12 when compared with adults (mean concentrations of plasma kisspeptin in pmol/L: 59.5 ± 18.3, girls, P < 0.01 vs. adult women; 43.8 ± 6.2, boys, P < 0.001 vs. adult men). Beyond 9–12 years of age, concentrations of plasma kisspeptin appeared progressively lower in children aged 12–15 and 15–18 years of age (Figure 2(b) and (c)).
Discussion
Animal and human genetic studies suggest that increased hypothalamic kisspeptin signalling is necessary for, and a trigger of pubertal maturation.1–3 We report that plasma kisspeptin is elevated in children when compared with adults, and that concentrations are most significantly elevated in children during ages commonly associated with puberty. Girls with premature breast development have been recently reported to have elevated plasma kisspeptin when compared with age and sex-matched controls. 9 These data collectively suggest that measurement of plasma kisspeptin could be a novel biochemical marker of reproductive development in children.
It is interesting to consider the underlying explanation for elevated concentrations of plasma kisspeptin during reproductive development. Kisspeptin is a potent activator of endogenous GnRH secretion, which is an orchestrator of pubertal development. Furthermore, it is worth noting that the observed elevation in plasma kisspeptin occurred during childhood ages commonly associated with the early stage of puberty. By contrast, established gonadal markers of puberty such as inhibin B are maximal during later stages of puberty. 9 Our findings therefore support the hypothesis that plasma kisspeptin may reflect a central drive to initiate pubertal maturation. However KISS1 is also expressed in the human pituitary gland 10 and ovaries 11 ; circulating kisspeptin may simply be a downstream marker representing concentrations of activity of these tissues during reproductive development.
We observed a non-significant elevation of plasma kisspeptin in girls when compared with boys. Furthermore, a recent report observed that plasma kisspeptin was marginally, but significantly, elevated in girls when compared with boys. Females have a higher number of hypothalamic kisspeptin neurons and higher concentrations of KISS1 expression when compared with males, in humans. 12 It is therefore possible that girls may have elevated concentrations of plasma kisspeptin owing to higher concentrations of hypothalamic kisspeptin signalling. These observations are consistent with a model proposing that higher concentrations of hypothalamic kisspeptin expression in girls may partially explain why they undergo earlier puberty and are more prone to precocious pubertal development when compared with boys. 13 Further work is required to investigate to what extent central kisspeptin signalling may influence sexual dimorphism in pubertal development.
It is important to consider the results of our study in the context of two limitations. For ethical reasons, we were unable to obtain blood from children who did not require a blood test for clinical reasons. Many children included in the study had a chronic gastrointestinal disorder, which might have affected their physical development. The current study was also limited by an absence of pubertal clinical data, due to ethical constraints. Nevertheless, our data suggest that circulating concentrations of kisspeptin are elevated during human juvenile development in an age-dependent manner, thus highlighting its potential importance as a novel clinical marker.
In summary, we report that concentrations of circulating kisspeptin are elevated in both boys and girls, and that children have higher plasma kisspeptin concentrations when compared with adults.
Footnotes
Acknowledgements
We are most grateful to the outpatient staff at Chelsea & Westminster NHS Foundation Trust, and Ms. Zohreh Farzad for assisting with sample collection.
Declaration of conflicting interests
None.
Funding
The research is funded by grants from the MRC, BBSRC, NIHR, an Integrative Mammalian Biology (IMB) Capacity Building Award, an FP7- HEALTH- 2009- 241592 EuroCHIP grant and is supported by the NIHR Imperial Biomedical Research Centre Funding Scheme. Other funding support: NIHR Clinical Lectureship (CNJ); AMS/Wellcome Starter Grant (CNJ); SFE Early Career Grant (CNJ); Wellcome Clinical Research Training Fellowship (GMKN); Wellcome/GSK Fellowship (ADS); NIHR Career Development Fellowship (WSD).
Ethical approval
The West London ethics committee approved this study (REC number 10/Q0707/4).
Guarantor
WSD
Contributorship
CNJ, SRB, NB and WSD obtained ethical approval; CNJ, GMKN and SN analysed the data; GMKN, AA, ADS and NB collected the samples; AA, ADS, CNJ and MAG performed the radioimmunoassay. All authors contributed to writing the manuscript.
