Abstract
Background
Fractalkine has been detected in synovial fluid (SF) from osteoarthritis (OA) patients. This study aims to examine the relation of fractalkine concentrations in serum and SF with the radiographic severity of OA.
Methods
Fractalkine concentrations of serum and SF were measured using an enzyme-linked immunosorbent assay method in 223 patients with knee OA and 165 healthy controls. The progression of OA was classified according to the Kellgren–Lawrence grading system.
Results
Elevated concentrations of fractalkine in serum were found in knee OA patients compared with healthy controls [all results median (interquartile range) 226.25 (183.19–259.91) vs. 127.42 (99.54–154.98) pg/mL, P < 0.001]. The case group included 71 knee OA patients with grade 2, 98 with grade 3, and 54 with grade 4. Knee OA patients with KL grade 4 had significantly higher fractalkine concentrations in serum and SF compared with those with KL grade 2 and 3 [serum: 247.68 (215.05–278.64) vs. 212.45 (169.19–247.96) pg/mL, P < 0.001, and 247.68 (215.05–278.64) vs. 222.00 (179.80–254.98) pg/mL, P = 0.005, respectively; SF: 94.95 (76.46–106.68) vs. 74.31 (63.64–84.79) pg/mL, P < 0.001, and 94.95 (76.46–106.68) vs. 80.34 (68.84–96.39) pg/mL, P = 0.001, respectively]. Knee OA patients with KL grade 3 showed significantly elevated concentrations of fractalkine in SF compared with those with KL grade 2 [80.34 (68.84–96.39) vs. 74.31 (63.64–84.79) pg/mL, P = 0.004]. Fractalkine concentrations in serum and SF of knee OA patients were both significantly associated with the disease severity evaluated by KL grading criteria (r = 0.261, P < 0.001 and r = 0.366, P < 0.001, respectively).
Conclusion
The fractalkine concentrations in serum and SF may serve as an effective biomarker for the severity of OA.
Introduction
Osteoarthritis (OA), the most common degenerative joint disease, is characterized by the degradation of cartilage, subchondral bone and inflammation of the synovial membrane. 1 Cartilage damage can lead to inflammation and ultimately pain. The balance between catabolic and anabolic functions of chondrocytes is very important in the maintenance of cartilage homeostasis. 2 Both biochemical factors and mechanical stress are considered to contribute to the disruption of this balance, and the destruction of cartilage. 3 In addition, inflammation is shown to be involved in the pathogenesis of OA. A series of inflammatory factors in the serum and synovial fluid (SF) have been indicated to be associated with the radiographic severity in patients with knee OA. 4
Chemokines are a family of small proteins that regulate cell migration into sites of inflammation. Based on the conserved cysteine motifs, chemokines are classified into C, CC, CXC and CX3C chemokines. 5 Fractalkine, also called CX3CL1, is a unique member of the CX3C subfamily in which the first two conserved cysteine residues are separated by three non-conserved amino acids. 6 Unlike other chemokines, fractalkine exists in two forms: a membrane-anchored and a soluble form, which enable it to function as both an adhesion molecule and a chemoattractant. 7 The receptor for fractalkine, CX3CR1, is capable of mediating both leukocyte migration and adhesion. CX3CR1 is expressed on several leukocytes such as monocytes, T cell subsets and natural killer (NK) cells. 8 In the process of OA, synovial fibroblasts proliferate excessively, eventually leading to destruction of bone and cartilage by secreting proinflammatory cytokines. 9 Fractalkine is found to function as a novel chemoattractant to induce OA fibroblast signaling. 10 Sawai et al. 11 found that fractalkine mediates T cell-dependent proliferation of synovial fibroblasts in rheumatoid arthritis. In addition, fractalkine was found to activate the production of matrix metalloproteases (MMPs) including MMP-2, 12 and MMP-9 13 which is involved in the degradation of many different components of extracellular matrix (ECM). 14 This indicates that fractalkine may play an important role in the pathogenesis of OA.
This study aims to examine the association of fractalkine concentrations in serum and SF with the disease severity of OA.
Materials and methods
Patients
This cross-sectional study was conducted in 223 patients with knee OA. All patients met clinical symptomatic criteria (American College of Rheumatology) and radiographic criteria for OA in at least one knee. Patients with infectious or inflammatory arthritis, knee injury, aseptic osteonecrosis and congenital abnormality, receiving various drugs including analgesics and non-steroidal anti-inflammatory drugs (NSAIDs) were excluded from this study. The patients did not receive any treatment for knee OA before enrolling this study. A total of 165 age- and gender-matched subjects with normal knee radiographs were also enrolled as controls.
Knee radiography was taken when each participant was standing on both legs with fully extended knee and the X-ray beam was centred at the level of the joint. The assessment of radiographic severity was performed using the Kellgren and Lawrence (KL) grading system. 15 Subjects having radiographic knee OA of KL grade ⩾2 in at least one knee were defined as OA patients. Subjects having KL grades of 0 for both knees were considered to be controls. The grading scale used for analysis was the one found higher upon comparison between both knees.
All tested subjects signed an informed consent form. The study was approved by the Ethics Committee of our university.
Laboratory methods
Venous blood samples collected from all participants were centrifuged and stored immediately at −80℃ until analysis. SF was taken from the most affected knee before the treatment of hyaluronic acid injection for the first time. The specimen was then centrifuged to remove cells and joint debris and stored at −80℃ for further measurement. The serum and SF samples were analysed for fractalkine using commercially available enzyme-linked immunosorbent assay (R&D Systems Inc., Minneapolis, MN, USA). Intra- and inter-assay coefficients of variations (CVs) were 2–3% and 5–9%, respectively. The detection limit ranged from 0.156 to 10 pg/mL.
Statistical analysis
Statistical analysis was carried out using the statistical package for social sciences (SPSS) software, version 16.0 for Windows. The data are presented as means ± SD or median (interquartile range). Data normality was analysed using Kolmogorov–Smirnov test. Demographic data between patients and controls were compared by Chi-square tests, unpaired Student's t-tests, or Mann–Whitney U test, where appropriate. Kruskal–Wallis test was utilized to compare fractalkine concentrations in serum and SF between knee patients with different KL grades. The correlation of fractalkine concentrations in serum and SF with disease severity was assessed by Spearman correlation analysis and multinomial logistical regression analysis. P-values < 0.05 were considered to be statistically significant for differences and correlations.
Results
Baseline clinical parameters
The characteristics between patients with knee OA and healthy controls.
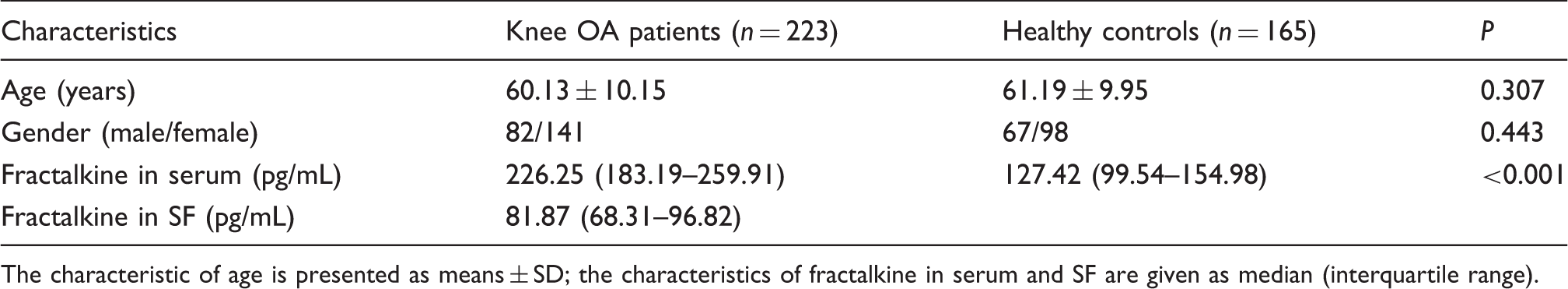
The characteristic of age is presented as means ± SD; the characteristics of fractalkine in serum and SF are given as median (interquartile range).
Fractalkine concentrations in serum and SF
Fractalkine concentrations in knee OA patients and healthy controls are shown in Table 1. Significantly elevated concentrations of serum fractalkine were found in patients with knee OA compared with healthy controls [226.25 (183.19–259.91) vs. 127.42 (99.54–154.98) pg/mL, P < 0.001].
Fractalkine concentrations in knee OA patients with different KL grades
The fractalkine concentrations of serum and SF in knee OA patients with different KL grades.
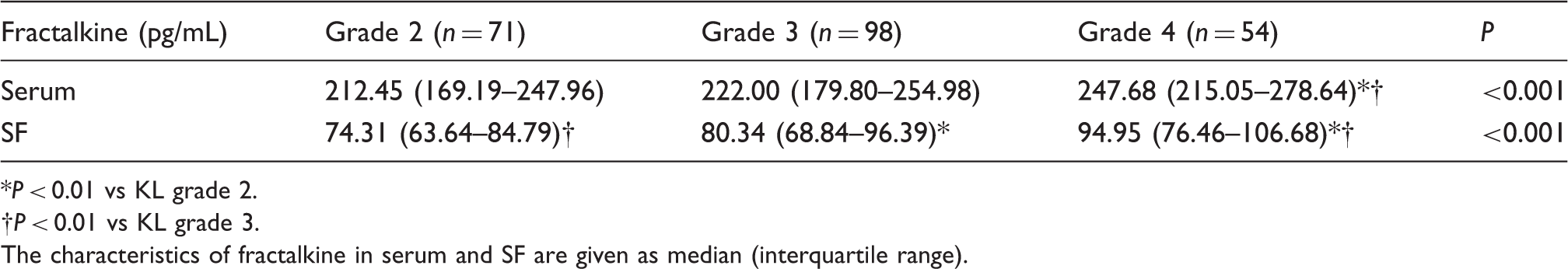
P < 0.01 vs KL grade 2.
†P < 0.01 vs KL grade 3.
The characteristics of fractalkine in serum and SF are given as median (interquartile range).
Association of clinical parameters with KL grades
The fractalkine concentrations in serum and SF were associated with KL grades (r = 0.261, P < 0.001 and r = 0.366, P < 0.001, respectively) by Spearman correlation analysis. Multinomial logistical regression analysis showed that fractalkine concentrations in serum and SF were both positively correlated with KL grades (P < 0.001 and P < 0.001, respectively).
Discussion
In this study, we examined the correlation of the fractalkine concentrations in serum and SF with the radiographic severity of OA. We observed that the fractalkine concentrations in serum and SF were closely related with the radiographic severity of OA, indicating that fractalkine concentrations in serum and SF may be a biomarker to evaluate the progression of OA. This is the first study to demonstrate that the fractalkine concentrations in serum and SF of knee OA patients are correlated with the radiographic severity of OA.
Previous studies suggested that OA patients had significantly higher concentrations of serum fractalkine compared with healthy controls. 16 This is consistent with the present study, which also demonstrated that serum concentrations of fractalkine were significantly higher in patients with OA compared with the control group. However, Ruth et al. 17 reported that serum concentrations of soluble fractalkine from patients with all rheumatic disease including OA, juvenile rheumatoid arthritis, psoriatic arthritis, polyarthritis and gout were lower than those in controls. In addition, they found that none of the rheumatoid arthritis (RA) serum samples contained detectable soluble fractalkine (>500 pg/ml). 17 This is inconsistent with another study which indicated that serum concentrations of soluble fractalkine were significantly higher in patients with RA compared with healthy controls, indicating that serum concentrations of fractalkine were detectable. 18 It seems that all of the results reported by Ruth et al. were inconsistent with most of other studies. This discrepancy may be due to ethnicity differences between the studied populations, differences of enrolled patients as well as different enzyme-linked immunosorbent assay kits used. Furthermore, our results indicated that fractalkine concentrations in serum and SF were closely related with the radiographic severity of OA. Our results point to a possible role of fractalkine in the pathophysiology of OA; there is an even stronger suggestion that fractalkine concentrations in serum and SF may be a useful biomarker to assess the progression of OA.
It is important to point out the limitations of our study. First, this is a cross-sectional study performed in a relatively small sample. Therefore, our results should be validated by further longitudinal studies in a larger population sample. Second, we did not measure fractalkine concentrations in SF from healthy controls due to ethical concerns. Last, we did not assess whether the treatment of hyaluronic acid injection had an effect on the serum and SF concentrations of fractalkine in knee OA patients.
In conclusion, fractalkine concentrations in serum and SF were positively correlated with the severity of knee OA. Fractalkine concentrations in serum and SF may serve as a new biomarker in addition of the traditional methods for assessing the risk and severity of knee OA.
Ethical approval
The ethics committee of Southern Medical University (2011SMU0045).
Guarantor
Yikai Li.
Contributorship
Yucong Zou, Yikai Li, Lu Lu, Liwei Huo and Yongliang Ye researched literature and conceived the study. You Lin, Zhiyong Su, Xusheng Wang, Hui Yang, Jianming Wang and Chengfu Yu were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. Yucong Zou and Weiming Liang wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
