Abstract
Aim
To evaluate the influence of age of onset and duration of diabetes on the positivity of glutamic acid decarboxylase antibody (GADA) in South-Brazilian type 1 diabetes mellitus (DM) patients.
Methods
GADA was evaluated in 92 patients with type 1 diabetes, in 147 gestational DM patients, and in 59 subjects with normal glucose tolerance.
Results
Type 1 patients with positive GADA (N = 44, 48%) were older at the onset of diabetes (22 ± 9 versus 18 ± 10 y, P = 0.043) and had a shorter DM duration (12 ± 8 versus 19 ± 9 y, P < 0.001), as compared with negative GADA patients. A logistic regression with antibody positivity as the dependent variable and diabetes duration as the independent variable, showed that the shorter diabetes duration was related to the presence of the antibody with an odds ratio (OR) = 5.6; (CI 95% = 2.1–14.6); P < 0.001. Another model, with age at diagnosis as the independent variable, did not show any association with antibody positivity. However, analysing only men, a shorter DM duration (OR = 6.5; CI = 1.7–24.0; P = 0.006), and also a higher age at diagnosis (OR = 5.5; CI = 1.5–21.0; P = 0.01) were significantly related to the antibody positivity. The performance of GADA was similar in up to 15 y of duration of DM (P = 0.78), but significantly diminished with higher duration (P = 0.001).
Conclusion
GADA testing is a helpful tool in the diagnosis of type 1 DM starting in young adults and older individuals. Even though the positivity rate declines along the course of disease, it still provides useful information up to 15 y after the diabetes diagnosis.
Introduction
Diabetes mellitus (DM) classification is currently divided into four categories: type 1 DM, type 2 DM, other types (which includes several subgroups with different pathogenesis) and the category of gestational DM.1,2 However, sometimes it is difficult to distinguish one type of diabetes from another based only on clinical information. Overlapping clinical features frequently occur in early onset type 2 diabetes, maturity onset diabetes of the young (MODY) and recently diagnosed type 1 DM with preserved pancreas function. In these situations, the presence of autoimmunity against the pancreatic β-cell is decisive information to establish the diagnosis of type 1A diabetes.2–5
Autoantibodies to a number of islet β-cell constituents, including glutamic acid decarboxylase (GADA), islet antigen-2, insulin and zinc transporter-8 are characteristic of patients with type 1 DM. Only 2–4% of these patients are autoantibody negative, fewer than 10% have only one marker, and around 70% have three or four markers. 4 GADA has a sensitivity of 75–84%,6–9 and specificity of 95–98.8% for the diagnosis of type 1 DM.10–12 A harmonization programme was recently established by international consortia studies to increase comparability of the antibody results between laboratories.4,13
GADA persist for longer in patients than other islet autoantibodies after the onset of type 1 diabetes. 14 However, its accuracy can differ among different ethnicities, age of onset of diabetes and diabetes duration.9,15,16 Therefore, the purpose of this study was to evaluate the performance of GADA in the diagnosis of type 1 diabetes in a South-Brazilian population, taking into account the duration and the age of onset of diabetes.
Patients and methods
Patients
This study included 298 individuals: 92 patients with an established diagnosis of type 1 DM, 59 healthy individuals and 147 women with gestational diabetes, evaluated between January 2010 and July 2011. The patients with DM were consecutively recruited from the Endocrinology Outpatient Clinic at Hospital de Clínicas de Porto Alegre (HCPA).
The clinical diagnosis of type 1 DM was based on a history of documented diabetic ketoacidosis, insulin use within up to three years after diagnosis and fasting baseline levels of C-peptide lower than 0.3 nmol/L. 17 Fifty-nine healthy individuals matched by age, with no history of DM in family members and a normal oral glucose tolerance test (OGTT) according to ADA criteria,1,3 were included to assess antibody quality control and provide cut-off values of normality. Gestational DM was diagnosed based on the Guidelines of the Second Meeting of The Diabetes and Pregnancy Task Force in Brazil, with diagnostic cut-off points of a fasting plasma glucose ≥6.1 mmol/L or a 2 h plasma glucose ≥7.77 mmol/L. 18
The study was approved by local ethical committee.
Methods
A cross-sectional study design was employed, and a complete medical history and physical examination were performed. Patients’ weight was assessed in light clothes without shoes. Body mass index (BMI) was calculated from weight in kilograms (kg) divided by height in square meters (kg/m2). The waist was measured as recommended by World Health Organization. Blood pressure was measured after a five-min rest, using a calibrated mercury sphygmomanometer.
OGTT was performed by measurement of fasting and two hours glucose after the intake of 75 g of glucose, with an enzymatic method. A fasting glucose below 5.55 mmol/L and a two hours value below 7.77 mmol/L were considered normal.1,3 GADA was evaluated by radioimmunoassay, RSR LIMITED® Cardiff UK (inter-assay variation coefficient of 4.9% and 7.0% for values of 6.1 and 42.9 U/mL, and intra-assay of 3.6% and 3.7% for values of 6.4 and 42.7 U/mL, respectively). 19 GADA values above the 95th percentile value of controls were considered positive. C-peptide was measured by chemoluminescence using an IMMULITE ® kit (inter-assay variation coefficient of 6.3% and 4.4% for values of 0.30 and 1.47 nmol/L, respectively, and limit of detection of 0.10 nmol/L). 20
Variables without Gaussian distribution were log transformed before analyses. Logistic regressions were performed taking the positivity of the GADA as the dependent variable, and gender, age at diagnosis and duration of DM as independent variables.
Sensitivity, specificity, positive predictive value, negative predictive value and accuracy of GADA were calculated. The significance level was set at 5%. The analyses were performed using statistical package SPSS 16 for Windows.
Results
Clinical and laboratory characteristics of patients with type 1 diabetes, healthy individuals and gestational diabetes mellitus
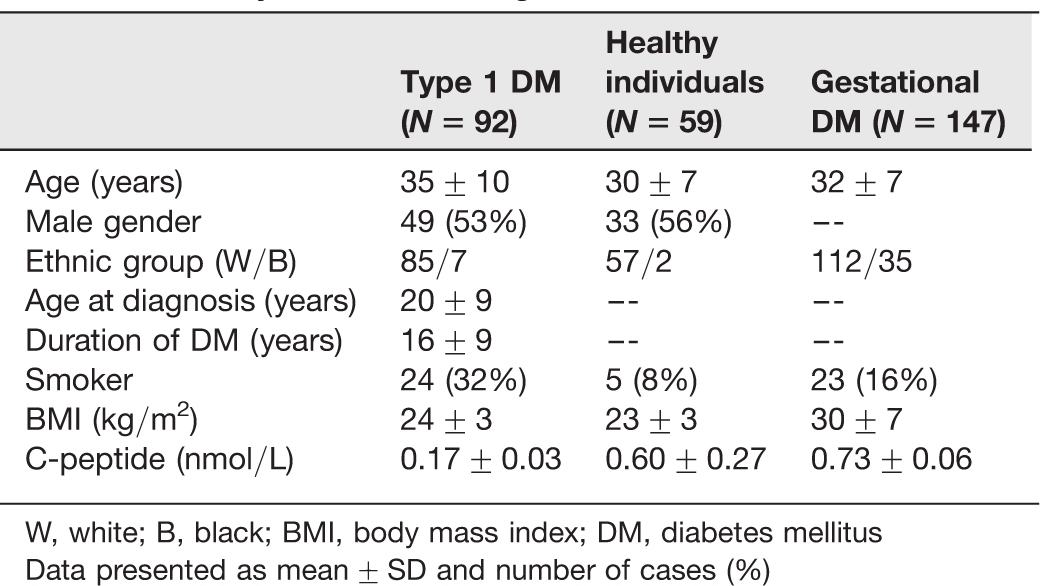
W, white; B, black; BMI, body mass index; DM, diabetes mellitus
Data presented as mean ± SD and number of cases (%)
Clinical and laboratory characteristics of positive versus negative GADA type 1 diabetes patients
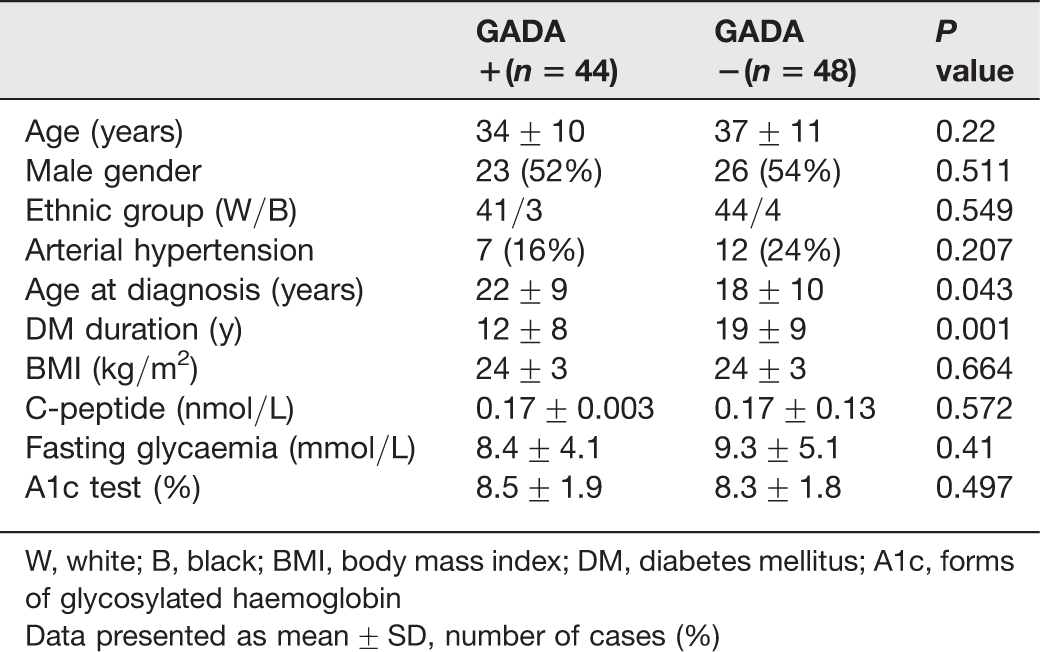
W, white; B, black; BMI, body mass index; DM, diabetes mellitus; A1c, forms of glycosylated haemoglobin
Data presented as mean ± SD, number of cases (%)
Diabetes duration was stratified into five-year periods to evaluate the diagnostic performance of GADA in each range of duration. The performance was similar in up to 15 y of duration of DM (P = 0.78), but significantly diminishing with higher duration (P = 0.001).
In order to evaluate the specific influence of age at onset of DM on the accuracy of test, the different sensitivities and specificities were calculated according to age of diagnosis. The diagnostic performance of GADA in patients with DM onset at age ≥16 years (33 positive tests in 56 patients) was significantly superior to <16 years, and showed sensitivity of 58.9% (CI: 45–71.6), specificity of 98.3% (CI: 89.7–99.9), PPV of 97.1% (CI: 82.9–99.8), NPV of 71.6% (CI: 60.3–80.8) and accuracy of 79.1% (Figure 1). On the other hand, in patients in whom type 1 DM began before the age of 16 years (11 positive tests in 36 patients), GADA had a sensitivity of 30.6% (CI:16.9–48.3), specificity of 98.3% (CI:89.7–99.9), PPV of 91.7% (CI:59.8–99.8), NPV of 69.9% (CI:58.7–79.1) and accuracy of 72.6%.
Distribution of GADA titres according to age at diagnosis of DM. GAD, glutamic acid decarboxylase; DM, diabetes mellitus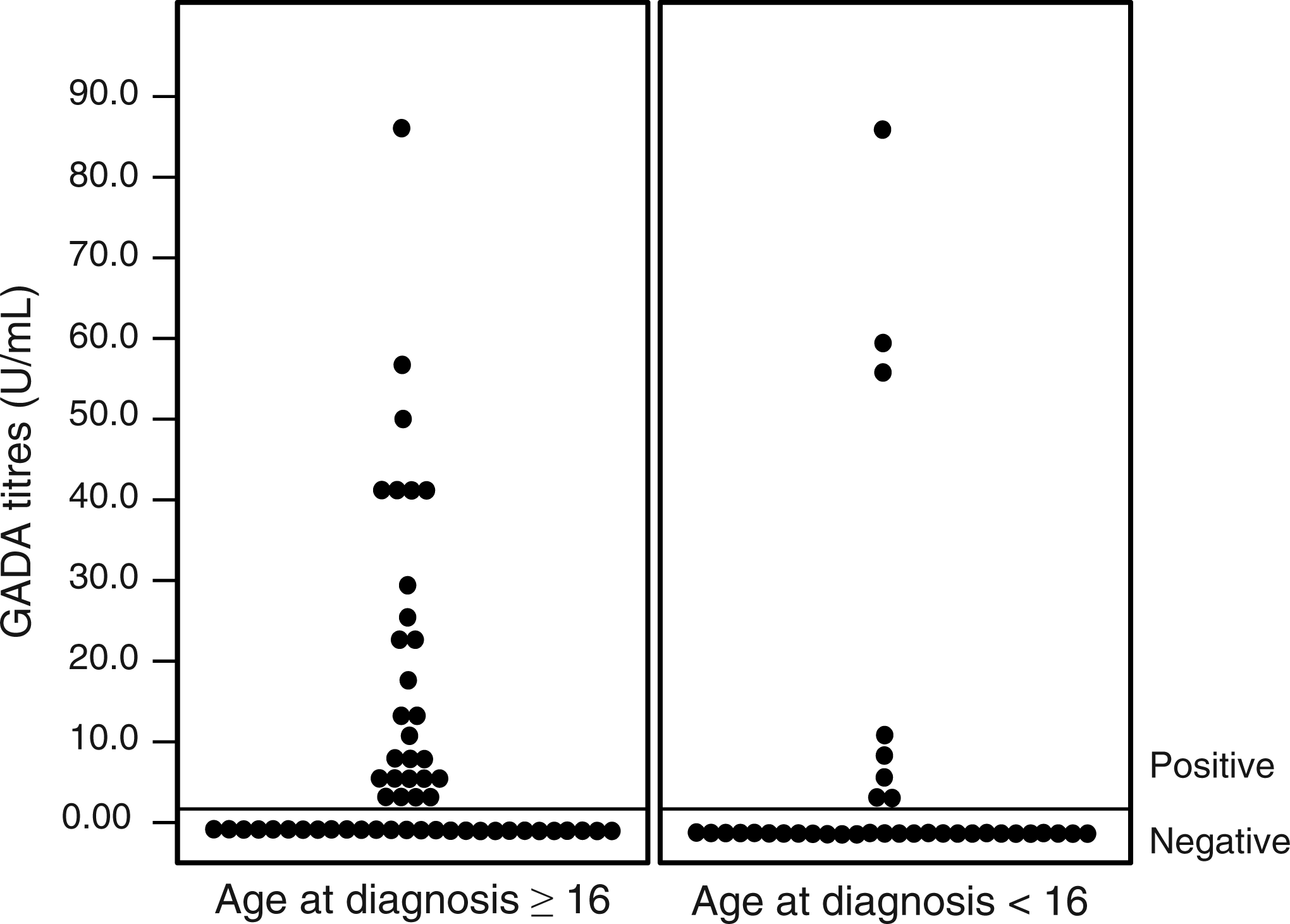
Univariate logistic regressions performed with GADA positivity as the dependent variable and diabetes duration and age at diagnosis of diabetes as the independent variable in each model
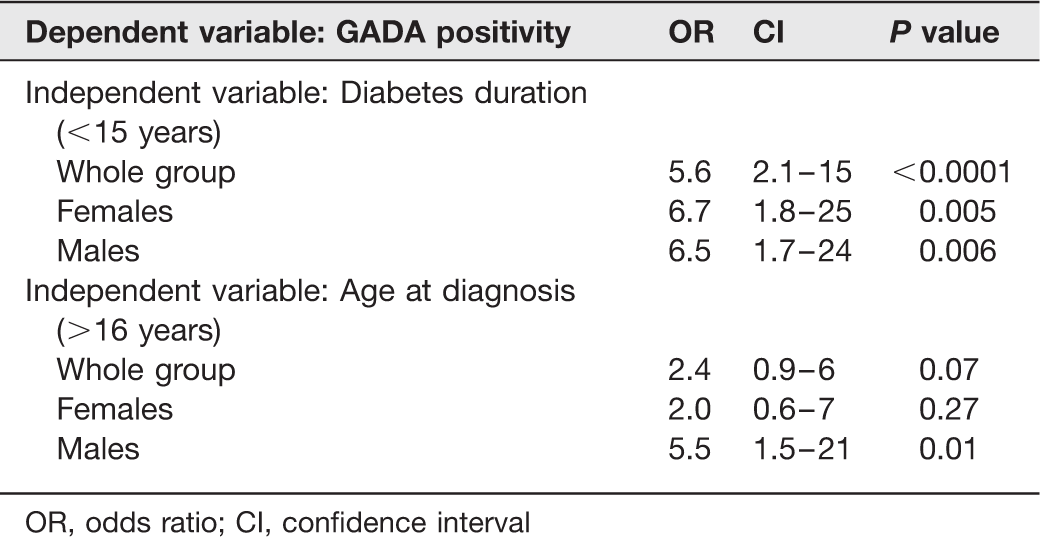
OR, odds ratio; CI, confidence interval
Evaluating the diagnostic performance of GADA in the subsets of age at diagnosis and DM duration simultaneously, the best performance was in patients with a diagnosis of DM over 16 years of age, and duration of the disease less than 15 years simultaneously (28 positives tests out of 37) with a sensitivity of 75.7% (CI: 58.4–87.6), specificity of 98.3% (CI:89.7–99.9), PPV of 96.6% (CI:80.4–99.8), NPV of 86.6% (CI: 75.5–93.3) and accuracy of 89.5%.
Discussion
In our study, the best diagnostic performance of GADA test was in patients with the duration of the disease of less than 15 years, and in the subset of men, also with the onset of DM after 16 years of age.
Diagnosis of the correct type of diabetes is usually made based on clinical information, but the use of autoimmunity markers is sometimes necessary. Therefore, the accuracy and the limitations of these antibodies must be studied in detail. Taking the whole group of patients, the specificity of GADA found in our study is practically identical to that of previous studies in other populations – around 98% – although our sensitivity was lower.10,11 This is probably due to the fact that about half of the patients had a DM duration over 15 years in our sample. When analysing the patients with diabetes duration of less than 15 years, a best diagnostic performance of GADA was demonstrated, with a sensitivity similar to that reported by other studies, that ranged from 75 to 84%.6–9,21
Age at diagnosis of type 1 diabetes is an important characteristic for the interpretation of the GADA result. Our data is in agreement with international studies that show that sensitivity of the GADA is greater in older individuals at diagnosis.8,9,15,21–25 GADA is considered the most prevalent antibody in type 1 DM with age of onset above 20 years, 9 and its rate of positivity is constant with ages in the range of 10–40 years in some series.9,26 GADA sensitivity is also greater than the anti-islet and the anti-insulin antibodies in adults, and it is, therefore, the most sensitive autoimmunity marker in this age range.9,14,21,22
We have considered the period of less than three years to start insulin as a criteria for type 1 diabetes, and though it is quite a long period for patients with classical type 1 diabetes, it has been described that patients with LADA (latent autoimmune diabetes in adults) patients may present a highly variable median period to insulin initiation, sometimes up to 30 months after diagnosis or longer. 27
The longer the duration of DM, the lower the positivity of GADA. In the recent study of McDonald et al, 5 patients with type 1 diabetes duration of less than six months had GADA and/or islet antigen-2 antibody in 82% of cases. The positivity is reported to decrease with time, but more than 50% of the patients with type 1 DM have positive GADA after 10-year duration of the disease, and it is the antibody that remains positive the longest after diabetes onset.9,15,23,26 Our study reproduced this finding in the Brazilian population, but expanded the range of satisfactory test performance up to 15 years of diabetes.
GADA sensitivity also varies with gender.8,21,24 In women, the sensitivity is reported to be around 80%, without variation according to age at diagnosis; and, in men, it is 50–60% below the age of 10 years and 75–90% above this age.8,9,21,24 However, no gender difference has been shown above the age of 20 years. 9 In our study, both age at diagnosis and duration of DM determined GADA positivity in men, while in women only the duration of diabetes influenced the positivity. This finding suggests that the stimuli to autoimmunity may operate differently at various ages, and they may also be sex-related. Any time gender is an issue, it raises the differences between men and women regarding lifestyle, and occupation, as well as influence of sex hormones and sex chromosomes. This highlights the complexity of the mechanisms. 28 Environmental factors predisposing to diabetes might also be different for men and women before and after puberty. For example, there is an increased risk of diabetes in boys with any neonatal infections and a decreased risk in girls, indicating a gender difference in the impact of neonatal infections on the risk of type 1 diabetes in children. 29
In the Brazilian population, the study of Pardini et al. 30 also reported a negative correlation of type 1 DM duration with GADA positivity: GADA positivity of 80% in recent-onset (<12 months) and 54% in longer duration diabetes (>12 months). A recent study in Southern Brazil found 63% of GADA positivity in patients with type 1 DM duration between three and five years. 31 On the other hand, the study of Rodacki et al. 32 described GADA positivity prevalence of 45.8% of type 1 diabetes patients with a mean duration of disease of 10 y, not affected by disease duration or age at diagnosis. The authors speculate that this low positivity may be due to the fact that only patients with more than one-year of diabetes duration were included in the analysis. So, the differences in GADA prevalence in our country appear to be influenced mainly by the subjects characteristics, such as diabetes duration or by the use of different assays.
Racial and ethnical differences have also been shown to influence the prevalence of GADA. 16 Prevalence of GADA positivity in other countries is variable, reported to be around 45.8% in Chinese population, 25 23% in Mexicans 33 and, in European patients, it varies from 39% in longstanding patients with age at diabetes diagnosis below 15 years, to 97% in patients recently diagnosed and with age at diagnosis older than 15 years. 15 Therefore, the ethnic background should be taken into account to predict the performance of the test. Furthermore, the exact prevalence of type 1 diabetes (insulin dependent not caused by autoimmunity) is not well described in these populations, and is probably responsible for a subset of cases, a phenomenon that remains to be further investigated.
The clinical importance of antibodies evaluation for the classification of DM was recently shown in the so called Aβ classification. This classification evaluates the presence of autoimmunity (A = antibodies) and the function of the β cell (β = C-peptide level) and so far presented the higher diagnostic accuracy in comparison with the traditional clinical classifications.2,34,35 Therefore, it is important to take into account the effect of age of onset and diabetes duration upon the GADA test interpretation in order to more properly use this tool.
Our study presents some limitations. The most important is the cross-sectional design employed. Obviously, a longitudinal design would allow the observation of the evolution and influence of several factors on the antibody course, and not merely an association between putative variables. Moreover, the sample size was relatively small, limiting conclusions to a similar population.
In conclusion, GADA testing is a helpful tool in the diagnosis of type 1 DM starting in young adults and older individuals. Even though the positivity rate declines along the course of disease, it still provides useful information up to 15 years after the diabetes diagnosis. The interpretation of the GADA results in children and in patients with long-lasting diabetes should be made with caution.
