Abstract
Background
High sensitivity C-reactive protein (hsCRP) is an acute phase reactant and a sensitive marker of inflammation. Age and adiposity can potentially promote the production of C-reactive protein (CRP). However, only a few studies have investigated any interaction between age and adiposity on hsCRP concentrations.
Methods
We recruited 822 men (mean age, 61 ± 14 years) and 1097 women (63 ± 12 years) during their annual health examination from a single community. We cross-sectionally examined whether age, body mass index (BMI), and the interaction are significantly associated with hsCRP concentrations, independent of confounding factors.
Results
Multiple linear regression analyses for hsCRP showed that in men, age (β = 0.169, P < 0.001) and BMI (β = 0.114, P = 0.004) as well as smoking status, triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), uric acid, and gamma-glutamyl transferase (GGT) were significantly associated with hsCRP; and in women, age (β = 0.203, P < 0.001) and BMI (β = 0.225, P < 0.001) as well as HDL-C, fasting plasma glucose (FPG), uric acid, GGT and high-molecular weight adiponectin were significantly associated with hsCRP. In addition to their direct associations, the interaction between age and BMI was also a significant and independent determinant for hsCRP in both men (F = 4.407, P = 0.036) and women (F = 5.487, P = 0.019). In participants aged < 75 years, overweight (BMI ≥25.0 kg/m2) persons were more likely to have a prevalence of elevated hsCRP concentrations (≥1.00 mg/L) compared with the normal-weight (BMI <22.0 kg/m2), but such an association was not observed in those aged ≥75 years.
Conclusions
Adiposity promotes low-grade systemic inflammation in community-dwelling persons aged <75 years.
Background
C-reactive protein (CRP), an acute-phase reactant, is an exquisitely sensitive systemic marker of inflammation. Accumulating evidence supports the concept that high-sensitivity C-reactive protein (hsCRP), even when within the clinical normal reference range, is an important precursor of the metabolic syndrome (MetS) and type 2 diabetes,1–3 and a strong predictor that reflects early stage cardiovascular disease (CVD).4,5
There is growing evidence that adipose tissue plays a critical role in the induction of chronic low-grade inflammation (i.e. hsCRP production) in the liver largely by synthesizing cytokines, including interleukin-6 (IL-6). 6 Indeed, measures for overweight are among the strongest correlates of CRP concentrations, and the close relationship between inflammation and overweight may help to explain the greater susceptibility to CVD among these individuals.7,8 Furthermore, blood concentrations of acute-phase reactants such as IL-1 receptor antagonists (IL-1 ra) and IL-6 have been known to increase with age.9,10 Thus, hsCRP values also increase with age. 11 However, only a few studies have investigated any interaction between age and adiposity for CRP.
The aim of this study was to determine whether there is an interaction between age and body mass index (BMI) that modifies hsCRP in both genders. We examined the cross-sectional data from Japanese community-dwelling persons.
Methods
Subjects
The present study is designed as a part of the Nomura study. 12 Subjects were selected through a community-based annual check-up process in a rural town located in Ehime prefecture, Japan. Information on medical history, present conditions and drugs were obtained by interview. Other characteristics, such as smoking and alcohol habits, and medication, were investigated by individual interviews using a structured questionnaire. The sample population included 822 men and 1097 women. The Ethics Committee of Ehime University School of Medicine approved all the procedures and each subject gave his/her informed consent to participate.
Evaluation of risk factors
Information on the demographic characteristics and risk factors was collected using clinical files. Body mass index was calculated by dividing weight (in kilograms) by the square of the height (in metres). We measured blood pressure with an appropriate-sized cuff on the right upper arm of the subjects in a sedentary position using an automatic oscillometric blood pressure recorder (BP-103i; Colin, Aichi, Japan) while they were seated after having rested for at least 5 min. Smoking status was classified into never smokers, past smokers and current smokers. Daily alcohol consumption was measured using the Japanese liquor unit in which a unit corresponds to 22.9 g of ethanol, and the participants were classified into never drinkers, occasional drinkers (<1 unit/day) and daily drinkers (≥1 unit/day). Total cholesterol (T-C), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), fasting plasma glucose (FPG), creatinine (enzymatic method), uric acid, high-molecular weight (HMW) adiponectin (Fujirebio, Tokyo, Japan), γ-glutamyl transferase (GGT) and hsCRP concentration (Dade Behring Inc., Marburg, Germany) were measured during fasting. Plasma hsCRP concentration was measured using a Behring BN II nephelometer (Dade Behring Inc., Marburg, Germany) and the inter- and intra-assay coefficient variations were 3.2 and 6.7%, respectively. We eliminated individuals with hsCRP ≥10.0 mg/L in order to exclude those with underlying inflammatory conditions. Low-density lipoprotein cholesterol (LDL-C) concentration was calculated by the Friedewald formula. Participants with TG concentrations ≥4.52 mmol/L were excluded. Glomerular filtration rate was estimated using the following equation: eGFR = 194 × Age−0.287×Cr−1.094 × 0.739 (if female). 13
Statistical analysis
All the statistical analyses were performed according to gender using IBM SPSS Statistics Version 20 (Statistical Package for Social Science Japan, Inc., Tokyo, Japan). Data are presented as the mean ± standard deviation (SD) unless otherwise specified, and for parameters with non-normal distributions (TG, FPG, GGT, HMW adiponectin, and hsCRP) the data are presented as median (interquartile range) values. In all the analyses, parameters with non-normal distributions were used after log-transformation. Differences among two groups divided according to gender were analyzed analysed by Student’'s t-test or chi-square (χ2) test. Correlations between various characteristics and hsCRP were determined using Pearson's correlation coefficient. Multiple linear regression analysis was used to evaluate the contribution of each explanatory variable for hsCRP. Moreover, additive effect of age (category, <50, 50 –64, 65–74, ≥75 years) and BMI (category, <22.0, 22.0–24.9, ≥25.0 kg/m2) on hsCRP was evaluated using a general linear model. The Models with hsCRP as the objective variables were also constructed to evaluate for interactions of age *BMI, with further adjustment for the significantly confounding factors above. In addition, elevated hsCRP concentrations was defined as the fourth quartile of hsCRP concentrations, and the relationship between BMI category and prevalence of elevated hsCRP concetrations according to age category was examined by stepwise multiple logistic regression analysis. Adjustments were made for potential confounders which were significant in the multiple linear regression analysis. A P- value <0.05 was considered significant.
Results
Characteristics of subjects categorized by gender
Characteristics of various risk factors of subjects categorized by gender.
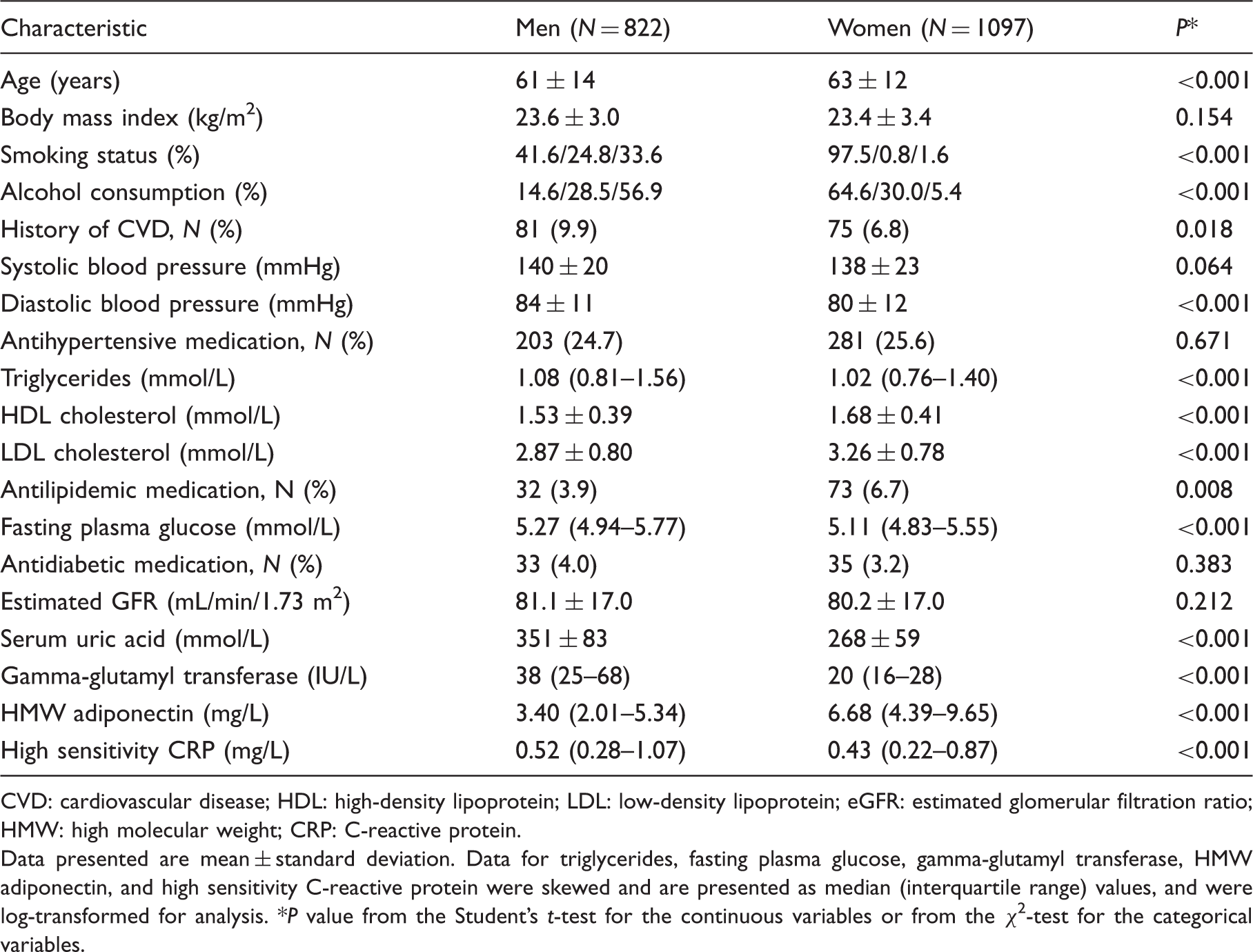
CVD: cardiovascular disease; HDL: high-density lipoprotein; LDL: low-density lipoprotein; eGFR: estimated glomerular filtration ratio; HMW: high molecular weight; CRP: C-reactive protein.
Data presented are mean ± standard deviation. Data for triglycerides, fasting plasma glucose, gamma-glutamyl transferase, HMW adiponectin, and high sensitivity C-reactive protein were skewed and are presented as median (interquartile range) values, and were log-transformed for analysis. *P value from the Student’s t-test for the continuous variables or from the χ2-test for the categorical variables.
Relationship between various confounding factors and hsCRP of subjects categorized by gender
Relationships between various confounding factors and high sensitivity C-reactive protein of subjects categorized by gender.
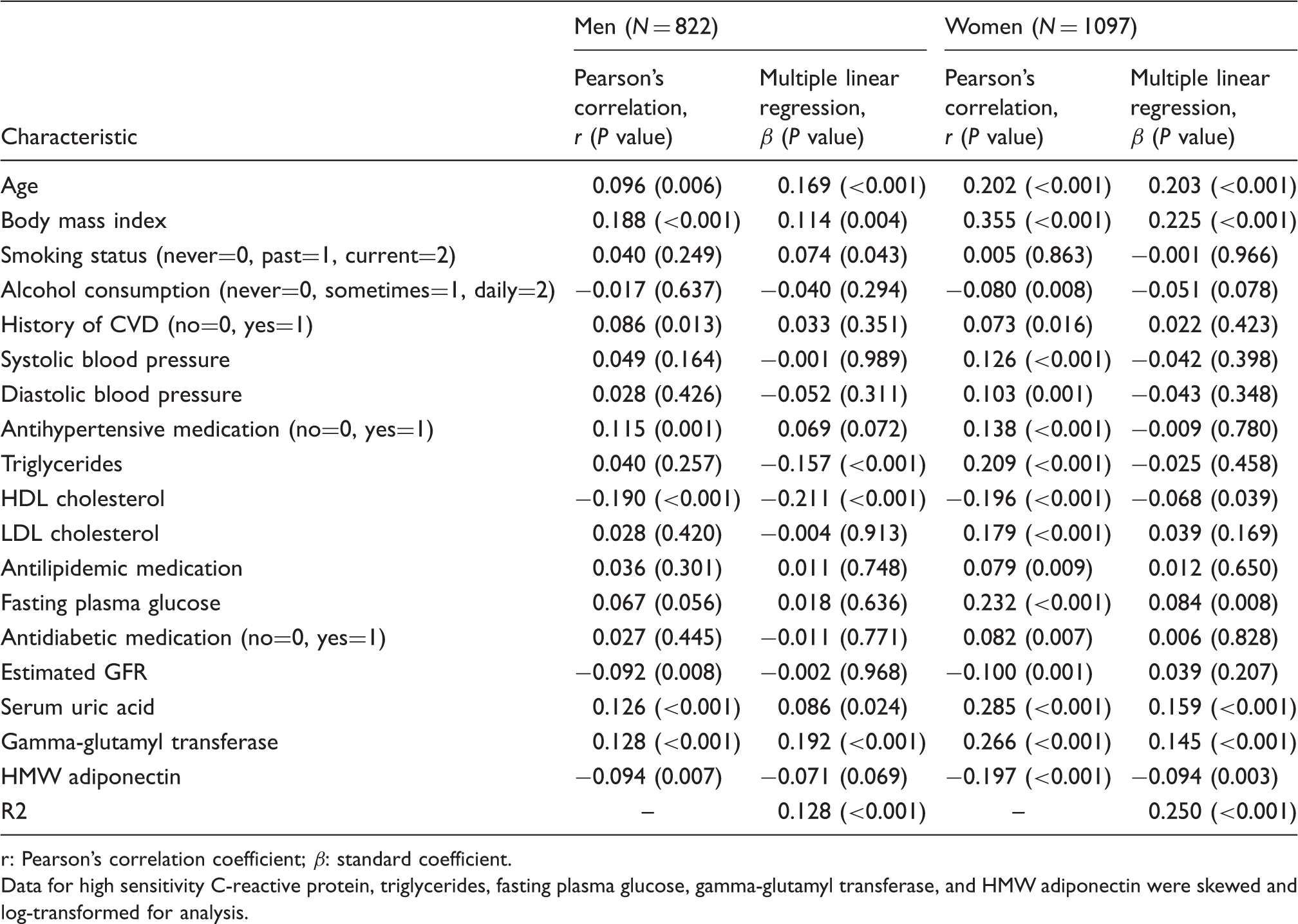
r: Pearson’s correlation coefficient; β: standard coefficient.
Data for high sensitivity C-reactive protein, triglycerides, fasting plasma glucose, gamma-glutamyl transferase, and HMW adiponectin were skewed and log-transformed for analysis.
Interaction between age and BMI for hsCRP
Interaction between age and body mass index for high sensitivity C-reactive protein of subjects categorized by gender.
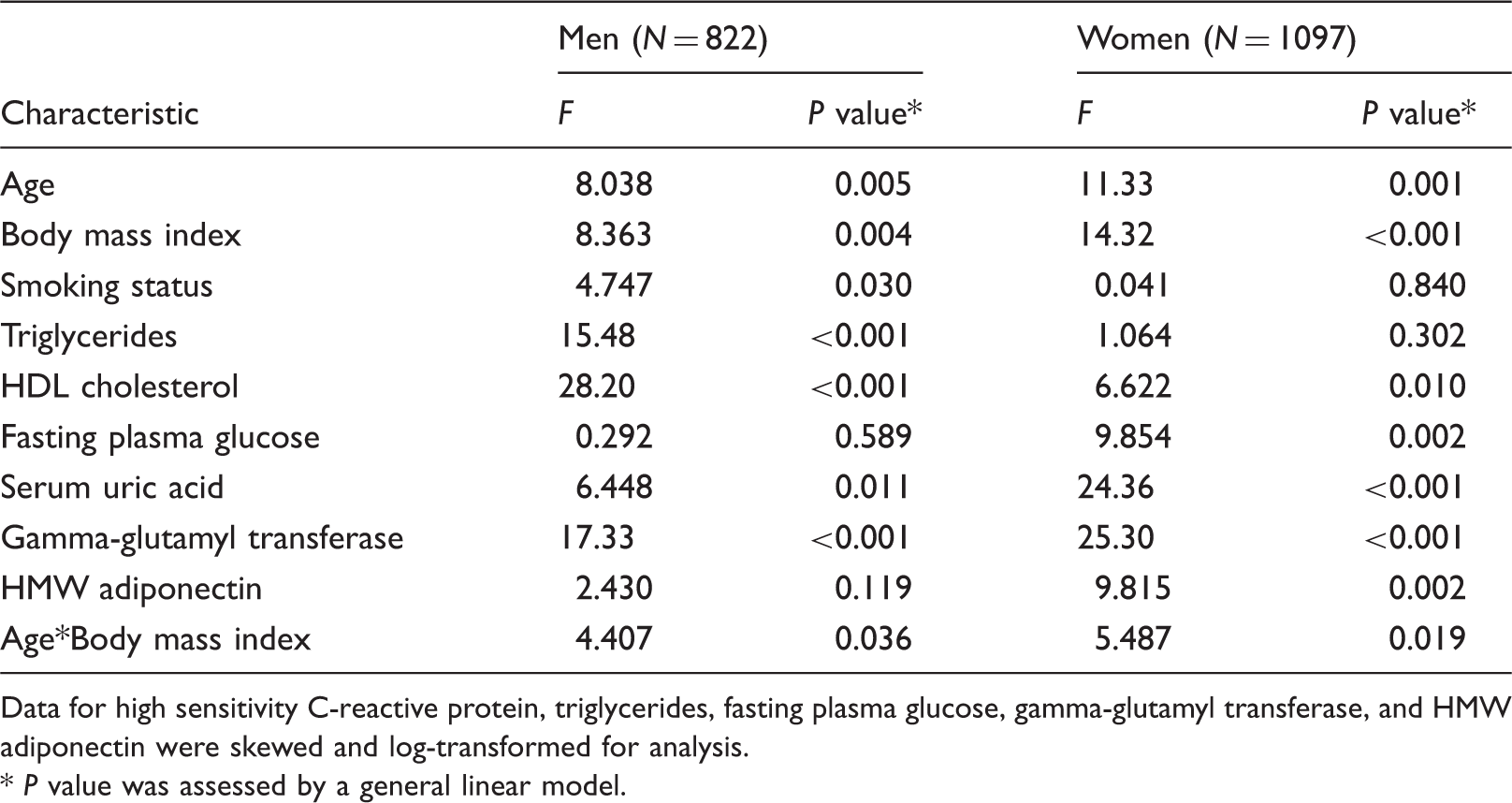
Data for high sensitivity C-reactive protein, triglycerides, fasting plasma glucose, gamma-glutamyl transferase, and HMW adiponectin were skewed and log-transformed for analysis.
P value was assessed by a general linear model.
Correlation between age category and BMI category with prevalence of elevated hsCRP concentrations (≥0.100 mg/dL) of subjects categorized by gender
Elevated hsCRP concentrations (≥1.00 mg/L) were present in 27.9% of men and 22.5% of women. As shown in Figure 1, the study subjects were divided into twelve 12 groups according to the categories of age and BMI. In both genders, the BMI category exerted a gradual additive effect with prevalence of elevated hsCRP concentrations in either group aged <75 years, however, in the group aged ≥75 years such an association was not observed. The increase in CRP concentrations with BMI is more different for group aged <75 years than for the group aged ≥75 years in both men and women. In brief, we observed an interaction between age and BMI on hsCRP concentrations in men (F = 6.469, P = 0.011) and women (F = 7.345, P = 0.007).
Correlation between age category and body mass index (BMI) category with prevalence of elevated hsCRP (≥1.00 mg/L) of subjects categorized by gender. Study subjects were divided into twelve groups according to categories of age and BMI. Statistical significance was assessed by analysis of covariance.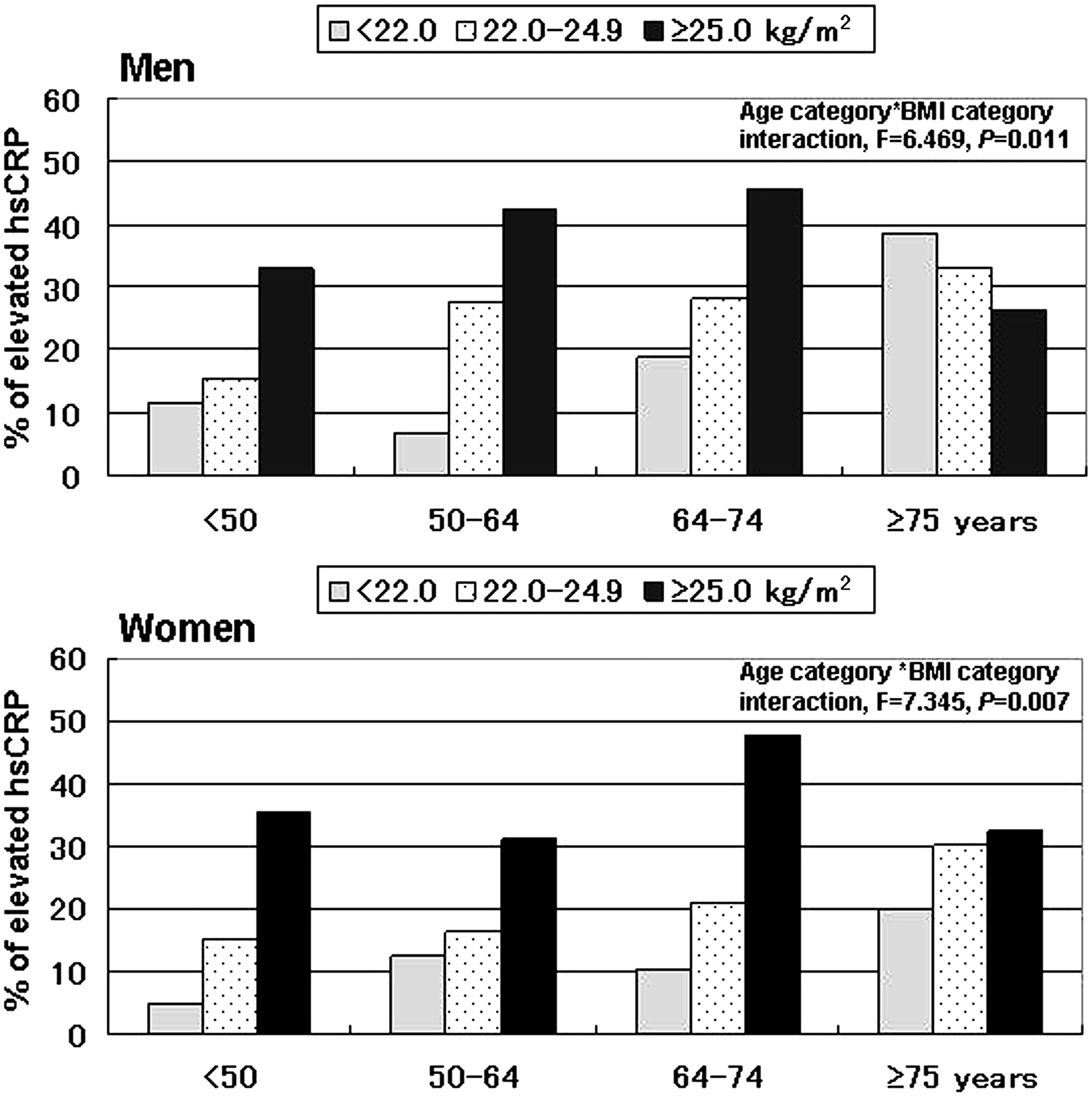
Adjusted odds ratios (95% confidence intervals) for prevalence of elevated hsCRP concentrations (≥1.00 mg/L) of subjects categorized by gender
Association of body mass index category with elevated high sensitivity C-reactive protein concentrations (≥1.00 mg/L) of subjects categorized by gender.
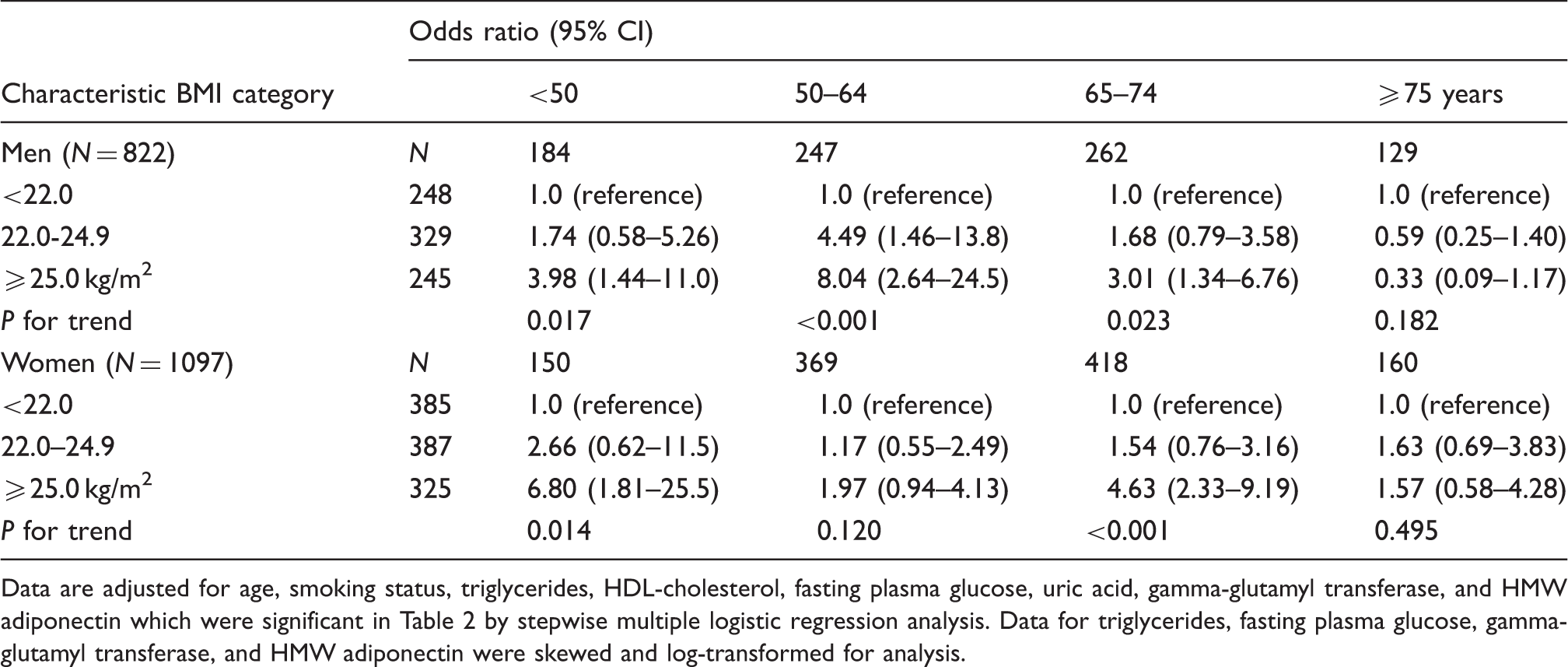
Data are adjusted for age, smoking status, triglycerides, HDL-cholesterol, fasting plasma glucose, uric acid, gamma-glutamyl transferase, and HMW adiponectin which were significant in Table 2 by stepwise multiple logistic regression analysis. Data for triglycerides, fasting plasma glucose, gamma-glutamyl transferase, and HMW adiponectin were skewed and log-transformed for analysis.
Discussion
To examine any possible contribution of age and BMI to hsCRP in the general population, we studied the relationship between the explanatory variable and hsCRP. This study showed that age and BMI as well as various factors were independently and significantly associated with hsCRP in both genders, and the association between BMI and hsCRP concentrations was influenced by age. Obese (BMI ≥25.0 kg/m2) participants (i.e. men and women) aged <74 years have a higher prevalence of elevated hsCRP concentrations than those with normal-weight (BMI <22.0 kg/m2), but such an association was not observed among those aged ≥75 years. Thus, we found that an interaction between age and BMI was also a significant and independent determinant for hsCRP as well as confounding factors in Japanese community-dwelling persons.
In this study, the relationship between various confounding factors and hsCRP was consistent with previous studies in that sex, age, BMI, smoking status, HDL-C, and TG were significantly associated with hsCRP.14–17 From a randomly sampled population of 303 English men aged 50–69 years, Mendall et al. demonstrated that BMI was an independent confounder, followed by age and smoking status. In a total of 16616 US men and nonpregnant women aged ≥17 years, higher BMI was independently associated with higher CRP concentrations, even among young adults aged 17–39 years and precipitants excluding smokers, persons with inflammatory disease, CVD, or diabetes mellitus and estrogen users. 16 In a total of 637 Japanese men aged 40–49 years, the ORs of elevated CRP (≥0.60 mg/L) were 1.83 (95% CI: 1.25–2.69), 2.63 (1.69–4.11), and 10.31 (2.17–48.98) for upper normal-weight (BMI: 22.0–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (≥30.0 kg/m2) persons, respectively, compared with lower normal-weight persons (18.5–22.0 kg/m2). 18 In our study, hsCRP concentrations increased with higher BMI among participants aged <75 years.
Age is also an important determinant factor, and there are many studies about an association between age and CRP concentrations. Plasma CRP values in general adult populations in Germany (2291 males and 2203 females; ages, 25–74 years) and Scotland (604 males and 650 females; ages, 25–64 years) were very similar, and the median CRP approximately doubled with age, from approximately 1 mg/L in the 25–34 years to approximately 2 mg/L in 65–74 years. 19 In the US population, CRP concentrations are higher among women than among men (median, 2.7 mg/L vs. 1.6 mg/L) and increase with age (median, 1.4 mg/L among those aged 20–29 years vs. 2.7 mg/L among those aged ≥80 years). 20 In a cross-sectional study of 2675 healthy Japanese adults aged ≥55 years, Log CRP showed a sigmoid curve against BMI with the base at BMI 21.0–22.9 kg/m2, and after adjustments for age, sex, smoking status, and weight change over the past two years, the adjusted means of Log CRP still had a similar sigmoid curve against BMI in the elderly. 21 Also in our study, elderly persons aged <74 years with high BMI values may have subtle low-grade inflammation irrespective of a favourable cardiovascular risk.
CRP is the most widely measured marker of systemic inflammation, and adiposity is the other major determinant of hepatic CRP synthesis via the production of proinflammatory cytokines {(IL-6, IL-1, and tumor necrosis factor-α}).22,23 Especially, IL-6 plays an important role in the regulation of acute phase protein synthesis in human hepatocytes, and is secreted in the endocrine system in proportion to the accumulation of fat mass, particularly in the visceral region. 22 Visceral fat has been shown to be a promoter of low low-grade CRP-inflammation24,16 and can produce higher concentrations of IL-6 than subcutaneous fat. 25 Production of CRP as well as IL-6 and IL-1Ra wereas higher in 711 elderly participants in the Framingham Heart Study (mean age, 79 years) than in 21 young healthy volunteers (mean age, 39 years). 9 BMI thought to be a surrogate for visceral fat, was shown in our study to be positively associated with elevated hsCRP concentrations in both men and women aged <75 years. Although, such an association was attenuated in participants aged ≥75 years. As IL-6 concentrations, which are responsible for altered acute-phase protein, increases with aging, 26 the influence of aging on hsCRP may be greater than the influence of BMI.
Some limitations of this study must be considered. First, the cross-sectional study design is limited in the ability to eliminate causal relationships between age and BMI, and hsCRP. Second, our analyses of hsCRP concentrations are based on a single assessment of blood sample, which may introduce a misclassification bias. Third, we could not eliminate possible effects of the underlying diseases, and medication, especially diuretic use and antilipidemic drug use, on the results. These points need to be addressed again in a large population-based sample in a prospective manner. Therefore, the demographics and referral source may limit generalizability.
In conclusion, the present study showed that low-grade systemic inflammation was more likely to present in overweight persons aged <75 years compared with the normal-weight persons aged <75 years, but such an association was not observed in those aged ≥75 years. The underlying mechanism behind this relationship is unclear, but seems to be independent of traditional confounding factors such as smoking status, TG, HDL-C, FPG, uric acid, GGT, and HMW adiponectin. For community-dwelling healthy persons, prospective population-based studies are needed to investigate the mechanisms underlying this association to determine whether interventions, such as effective lifestyle modifications that decrease BMI will decrease hsCRP. 27
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This work was supported in part by a grant-in-aid for Scientific Research from the Foundation for Development of Community (2012).
Ethical approval
This study was approved by the ethics committee of Ehime University School of Medicine (17-3, The longitudinal genetic epidemiological study for metabolic syndrome, atherosclerotic disease, and anti-aging) and each subject gave their informed consent to participate.
Guarantor
RK
Contributorship
RK participated in the design of the study, performed the statistical analysis and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
