Abstract
A compromised vitamin B12 status is common in older people despite dietary intakes that typically far exceed current recommendations. The maintenance of an optimal status of vitamin B12 is not only dependent on adequate dietary intake but more critically on effective absorption which diminishes with age. The measurement of vitamin B12 is complicated by the lack of a gold standard assay. There are a number of direct and functional indicators of vitamin B12 status; however, none of these are without limitations and should be used in combination. Vitamin B12 is of public health importance, not only because deficiency leads to megaloblastic anaemia and irreversible nerve damage, but also because emerging evidence links low B12 to an increased risk of a number of age-related diseases, including cardiovascular disease, cognitive dysfunction, dementia and osteoporosis. Furthermore, there are concerns relating to potential adverse effects for older adults with low vitamin B12 status of over-exposure to folic acid in countries where there is mandatory fortification of food with folic acid. The aim of this review is to examine the known and emerging issues related to vitamin B12 in ageing, its assessment and inter-relationship with folate.
Keywords
Introduction
Major advances in medicine over the last century have resulted in a dramatic increase in life-expectancy. This, however, has been accompanied by a subsequent increase in the prevalence of age-related chronic diseases; with morbidity, disability and poor quality of life frequently present in older age. Poor nutritional status is recognized as a contributing factor and, recently, the role of vitamin B12 in ageing has attracted considerable attention. Apart from the clinical features of B12 deficiency (i.e. megaloblastic anaemia and irreversible neuropathy), emerging evidence indicates that subclinical deficiency (i.e. low biomarker status) of B12 may be implicated in the development of several chronic age-related diseases. The aim of this paper is to consider this evidence and also review its inter-relationship with folate in ageing. The stages of vitamin B12 depletion and the analytical methods used to assess biomarker status will be considered. The scientific evidence linking low/deficient B12 status to adverse health outcomes will be reviewed, and consequences for relevant public health policies will be considered.
Structure and physiological functions of vitamin B12
Vitamin B12 is one of a group of cobalt containing compounds known as cobalamin. There are a number of different forms of the vitamin including the two metabolically active forms methylcobalamin and deoxyadenosylcobalamin and cyanocobalamin, the synthetic form of the vitamin typically used in supplements and fortified foods. Vitamin B12 is required as a co-factor for only two mammalian enzymes, methionine synthase and methylmalonyl CoA mutase. Adenosylcobalamin acts as a co-factor for the latter enzyme which converts methylmalonyl CoA to succinyl CoA, a metabolite in the tricarboxylic acid cycle (Figure 1). This is an important reaction in the metabolism of branched-chain amino acids and odd-chain length fatty acids.
1
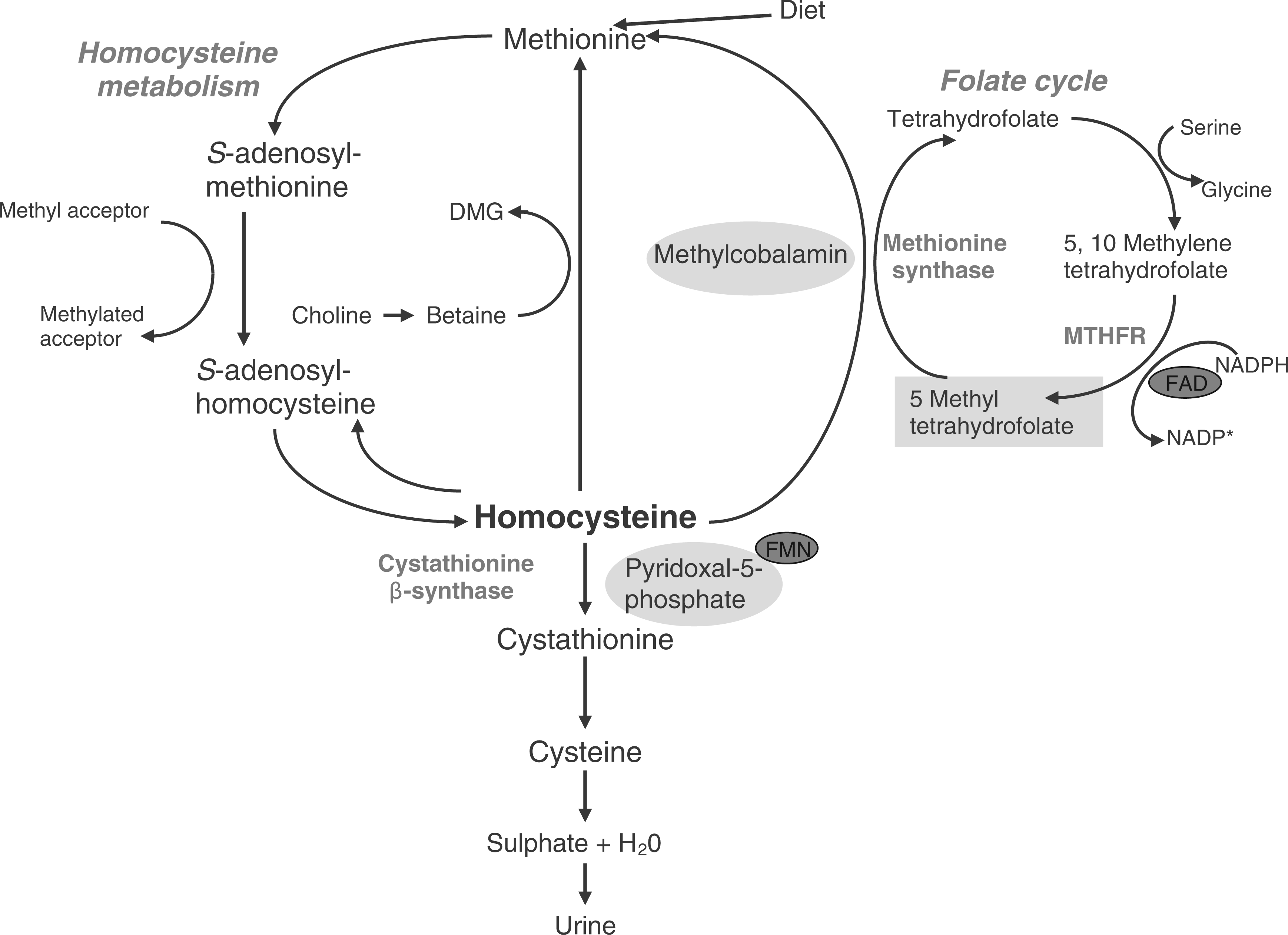
In vitamin B12 depletion, the reduced activity of methylmalonyl-CoA mutase causes the concentrations of its substrate methylmalonyl CoA to build up and form the by-product methylmalonic acid (MMA), which is then excreted into the circulation. Methylcobalamin acts as a co-factor for the enzyme methionine synthase, which catalyses the re-methylation of homocysteine to methionine, the precursor of S-adenosylmethionine (Figure 2). S-adenosylmethionine is a universal methyl donor essential for the methylation of phospholipids, neurotransmitters, amines, DNA, RNA and myelin basic protein. In vitamin B12 depletion, inactivity of the methionine synthase enzyme leads to a reduction in important methylation reactions and elevated homocysteine concentrations in the circulation.
2
The vitamin B12 (i.e. adenosylcobalamin)-dependent conversion of methylmalonyl-CoA to succinyl-CoA. Metabolic pathway involving vitamin B12 of the remethylation of homocysteine to methionine and the recycling of folate. Shows co-factor forms for vitamin B12 (methylcobalamin), vitamin B6 (pyridoxal-5-phosphate) and riboflavin (vitamin B2; flavin mononucleotide [FMN]; flavin adenine dinucleotide [FAD]). Also shows the metabolism of betaine and dimethylglycine (DMG).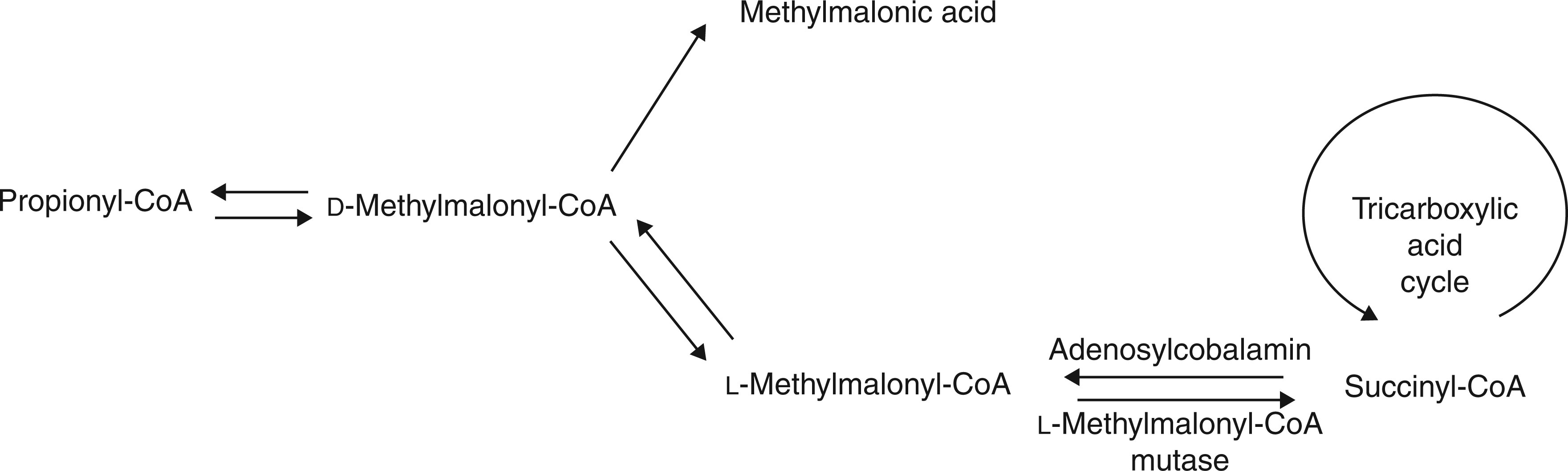
Vitamin B12 and folate interrelationship
The haematological sign of vitamin B12 deficiency is megaloblastic anaemia characterized by megaloblastic (immature, enlarged) blood cells that occur as a result of impaired DNA synthesis. An identical anaemia arises with folate deficiency because these two vitamins are intrinsically linked via the enzyme methionine synthase. 2 Methionine synthase sits at the junction between two major biosynthetic pathways: the remethylation cycle and the folate cycle (Figure 2). Methionine synthase is the only known enzyme to use 5-methyltetrahydrofolate, which in turn is converted to the metabolically active form, tetrahydrofolate, needed in one-carbon metabolism. 2 In vitamin B12 depletion, methionine synthase activity is reduced and the formation of tetrahydrofolate is blocked, with folate essentially becoming trapped as 5-methyltetrahydrofolate (i.e. the methyl-trap hypothesis). This is an unusable form of the vitamin as it cannot be converted back to its precursor 5,10-methylenetetrahydrofolate because the reduction of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate (by the enzyme methylenetetrahydrofolate reductase [MTHFR]) is irreversible. S-adenosylmethionine is an allosteric inhibitor of MTHFR, thus reduced S-adenosylmethionine concentration (as seen in vitamin B12 depletion) removes the inhibition of MTHFR, promoting the reduction of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate so that it can be used to re-synthesise methionine (the precursor of S-adenosylmethionine) thus further trapping folate as 5-methyltetrahydrofolate. In this situation, cells suffer from a folate pseudo-deficiency; folate is available but it is not in an active form that can be used by the cell, and consequently, the production of purines and pyrimidines needed for DNA synthesis is disrupted. 3 This affects the rapidly dividing cells of the bone marrow, leading to the production of megaloblastic blood cells and subsequent megaloblastic anaemia. In addition, 5-methyltetrahydrofolate is a poor substrate for folylpolyglutamate synthase, the enzyme responsible for cellular retention of folate. In order for folate to be retained by the cell 5-methyltetrahydrofolate must first be converted to tetrahydrofolate via methionine synthase and thus, intracellular erythrocyte folate concentration has been reported to be lower in individuals with low vitamin B12. 3–5
Absorption, transport and storage
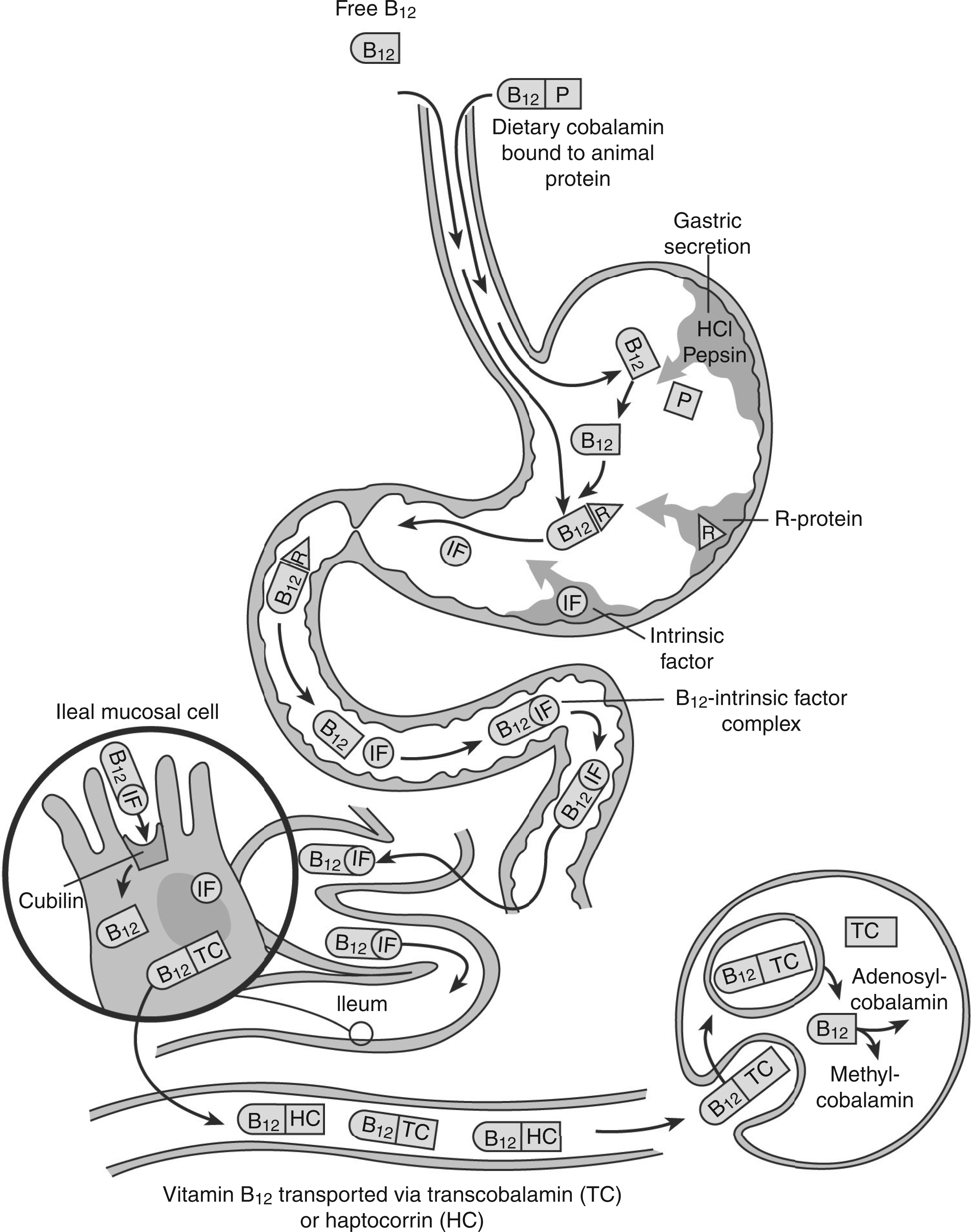
The absorption of vitamin B12 is a highly complex process that is dependent on normal functioning of the gastrointestinal tract (Figure 3), consequently, it often becomes less efficient with age. Vitamin B12 is tightly bound to protein in food and must be released by hydrochloric acid and pepsin in the stomach in order for B12 to be absorbed. The synthetic form of vitamin B12, found in fortified foods and supplements, is however already free and thus has no gastric acid requirement. Free vitamin B12 immediately binds with R-binding proteins and then with intrinsic factor (IF) in the duodenum forming the IF-B12 complex which travels to the terminal ileum where it is absorbed via specific cubilin receptors by calcium-dependent endocytosis. There is a limit to the amount of vitamin B12 that can be absorbed at any given time via the IF-related pathway; receptors are reported to become saturated with B12 intakes greater than 1.5–2.0 µg of vitamin B12.
6
In addition, an alternative, albeit inefficient pathway exists whereby small amounts (1%) of vitamin B12 are absorbed through passive diffusion (i.e. even in the absence of IF).
7
Once absorbed, vitamin B12 then binds to one of two transport proteins, transcobalamin or haptocorrin. The majority of vitamin B12 binds to haptocorrin, a glycoprotein with an unknown function, while 20–30% is bound to transcobalamin, forming holo-transcobalamin (holoTC), the metabolically active fraction of the vitamin.
8
Total body stores of vitamin B12 can reach 5 mg, with the majority stored in the liver.
9
The liver excretes around 1–10 µg/d of vitamin B12 into bile;
10
however, up to 90% of this is re-absorbed via a highly efficient enterohepatic cycle while the rest is excreted in faeces. The daily requirement for vitamin B12 is so small in comparison to body stores that it is considered to take years for vitamin B12 deficiency to develop, even when there is a complete halt in B12 absorption.
11
The absorption and transport of vitamin B12 (adapted from Andrès et al.
127
).
Analytical measurement of vitamin B12 status
Serum total vitamin B12
Measurement of serum/plasma total vitamin B12 has been the standard clinical test for vitamin B12 deficiency for many years and a variety of analytical approaches are available such as the traditional microbiological assay, competitive binding assay, radioimmunoassay and chemiluminescence assay. Early versions of the newer techniques lacked sensitivity and led to falsely elevated B12 concentrations; 12 however, there is now better agreement between the more recently developed protein binding assays and the microbiological assay; generally considered to be the gold standard assay. 13 These newer assays have a number of advantages over the microbiological assay, they are generally less time consuming, are not as sensitive to antibiotics or chemotherapy drugs, are widely available and have low running costs. 13 It has become apparent, however, that a low serum total vitamin B12 concentration does not always equate with B12 deficiency nor does a normal value necessarily exclude it. 14 There is no established cut-off value used to define vitamin B12 deficiency, with each laboratory recommended to determine its own reference range depending on its assay method. There is a large variation between laboratories with one review reporting that the lower reference limit of values cited in the literature ranged from 110 to 260 pmol/L. 15 Also, metabolic and clinical features of deficiency have been reported in individuals with serum total B12 within the ‘normal range’. 16 Furthermore, 5% of individuals with classical clinical signs of vitamin B12 deficiency were found to have serum total vitamin B12 concentrations within the normal range. 17
Serum total vitamin B12 does not reflect intracellular vitamin B12 as it is a measure of all forms of the vitamin, the majority of which is bound to haptocorrin. Haptocorrin-bound vitamin B12 is considered to be metabolically inert and has been shown to respond slowly to changes in vitamin B12 status compared with holoTC, which may explain its lack of sensitivity. 12 Furthermore, liver disorders, myeloproliferative disorders, renal failure, transcobalamin II deficiency, bacterial overgrowth and haemolysis can all lead to falsely elevated serum total B12 concentrations. False low concentrations of serum total vitamin B12 are linked with severe folate or iron deficiency, multiple myeloma, pregnancy, HIV infection and oral contraceptive use. 18
Plasma total homocysteine
The re-methylation of homocysteine to methionine is disrupted during vitamin B12 depletion (Figure 2) because the necessary enzyme, methionine synthase requires vitamin B12 as cofactor. As a consequence, plasma homocysteine concentrations accumulate with B12 deficiency thus providing a functional biomarker of vitamin B12 status. Although plasma homocysteine is highly sensitive to B12 status, it lacks specificity. This is because plasma concentrations of homocysteine are determined not only by the status of vitamin B12 19 but also by folate, 20 and to a lesser extent vitamin B6 21 and riboflavin. 22 Once folate status is optimised (as in countries with mandatory folic acid fortification) vitamin B12 emerges as the major nutritional determinant of homocysteine. 23 Apart from B vitamin status, plasma homocysteine concentrations are also influenced by other factors such as renal function, age and gender 24 and genetic polymorphisms, mainly homozygosity (TT genotype) for the 677C → T polymorphism in the gene encoding MTHFR, the enzyme required to generate 5-methyltetrahydrofolate. 25 Blood sample collection and handling can also impact considerably on homocysteine measurement. Homocysteine concentrations will increase if the sample is kept at room temperature, or if separation of the sample is delayed following collection, as homocysteine is released from red blood cells. However, this can be minimized if the sample is separated immediately or placed on ice until centrifugation. 24,26 After separation, homocysteine is extremely stable and can be stored, once frozen, for many years. 24 Several analytical methods, e.g. high-performance liquid chromatography, gas and liquid chromatography-mass spectrometry, stable-isotope dilution and immunoassay techniques have been developed for measuring plasma homocysteine and there is generally good agreement between the various techniques. 26 The assays are reliable and have become less expensive in recent years 5 but reference ranges may vary considerably between laboratories. 24 Although a plasma homocysteine concentration between 5 and 15 µmol/L is generally considered normal, 26 this reference range may not be appropriate for older adults. Despite its lack of specificity, plasma homocysteine is a sensitive and useful indicator of vitamin B12 status in situations where folate is optimized.
Serum MMA
Vitamin B12 acts as a co-factor for the enzyme methylmalonyl-CoA mutase required for the conversion of methylmalonyl-CoA to succinyl CoA. In vitamin B12 deficiency, the activity of this enzyme is reduced leading to the accumulation of the by-product MMA, which can be detected in both blood and urine (Figure 1). 18 Plasma MMA is therefore a specific, functional marker of vitamin B12 status that reflects the availability of intracellular B12, although it becomes elevated in individuals with renal impairment 27 and therefore may be less useful in older people where renal dysfunction is a common problem. MMA is more concentrated in urine and methods for its measurement have been available since the 1950s. However, it lacks sensitivity and requires a 24-hour urine collection as concentrations are more sensitive to variation in food intake than serum MMA concentrations. 28 More than 95% of patients with vitamin B12 deficiency have significantly elevated serum MMA. 27 A variety of cut-off values have been used to define vitamin B12 status; however, serum MMA value <0.30 µmol/L is generally considered indicative of no deficiency. 29 Although the development of a gas-chromatography-mass spectrometry method has increased sensitivity, the utility of MMA is compromised by high running costs, technical demands and dependence on normal renal function.
Serum holoTC
Serum total vitamin B12 and the functional assays (homocysteine; MMA) lack sensitivity and specificity, thus there has been an ongoing search for a more reliable clinical measure of B12 status. The major limitation of serum vitamin B12 is that it measures the total amount of the vitamin, while holoTC represents the metabolically active fraction of the vitamin that is involved in cellular reactions and thus may be a more reliable indicator of B12 status. It is thought that a decrease in holoTC will indicate the earliest sign of B12 depletion as it has a much shorter half-life than haptocorrin. 30 Low holoTC concentrations have been detected in populations at risk of deficiency such as vegetarians, vegans 31 and the elderly. 32 Preliminary results from an ongoing elderly Irish cohort indicate that holoTC is a considerably better indicator of vitamin B12 status than serum total vitamin B12 or MMA in an elderly population. 33 In addition, several studies have clearly demonstrated that holoTC is diagnostically superior to vitamin B12 in the detection of vitamin B12 deficiency 31,34,35 and was better correlated with symptoms of vitamin B12 malabsorption than serum total vitamin B12. 36 There is also increasing evidence that holoTC is more strongly correlated with diseases associated with low B12 status, however, this has not been thoroughly evaluated. 37
Measurement of holoTC has been an attractive prospect for more than 20 years; however, until recently, it was hampered by the lack of a good quality assay. 8 Currently, there are a number of different methods available for the measurement of holoTC including a microbiological assay, an enzyme-linked immunosorbent assay, a radioimmunoassay and a novel automated holoTC specific monoclonal assay that is suitable for routine use in clinical laboratories. 8 There is no general consensus as to what concentration of holoTC is sufficient, with the lower normal limit for holoTC varying from 19 to 42 pmol/L depending on the laboratory method used. 8 Several studies have demonstrated that holoTC increases dramatically after vitamin B12 ingestion, although the dose used in these studies was much larger than what would be typically consumed through the diet. 38,39 HoloTC is highly sensitive to altered renal function with concentrations increasing in renal impairment 31 and is influenced by a common genetic polymorphism of the transcobalamin gene (TC776C → G), liver disease and female sex hormones. 40 The future of holoTC as a first-line biomarker of vitamin B12 is promising, although further research is warranted to address the issues raised above before it can be used routinely.
Causes of low/deficient vitamin B12 status in older adults
Causes of low/deficient vitamin B12 status in older adults.

Inadequate dietary intake
Recommended daily intakes for vitamin B12 vary from country to country. 41 The UK National Diet and Nutrition Survey found that dietary intakes of vitamin B12 in those aged over 65 were more than three times greater than current dietary recommendations. 42 Thus, inadequate intakes of vitamin B12 are not a main concern as diets of older people appear to provide vitamin B12 well in excess of needs. Vitamin B12 is only found in foods of animal origin such as meats, poultry, fish, eggs and dairy products. It can also be obtained from foods such as ready to eat breakfast cereals and spreads fortified with the synthetic form of the vitamin. 10 In developed countries, inadequate dietary intakes of vitamin B12 are extremely rare, except for strict vegetarians and vegans. 30
Malabsorption
Intrinsic factor deficiency – pernicious anaemia
Pernicious anaemia is the classical form of vitamin B12 deficiency and, although more prevalent in those over 60 years, only accounts for 1–2% of vitamin B12 deficiency found in older populations. 11 It is an autoimmune disease characterized by the destruction of the gastric mucosa and subsequent loss of IF, the essential transport protein required for vitamin B12 absorption. Loss of IF prevents not only the absorption of food-bound and free vitamin B12 but also the re-absorption of vitamin B12 secreted in bile. This creates a significant negative vitamin B12 balance with a progressive and permanent depletion of body stores. 43 Although it may take a number of years for clinical deficiency to develop, pernicious anaemia would ultimately lead to death if left untreated. 44 In order to bypass the gastrointestinal tract and the requirement for IF, patients are traditionally treated with intramuscular B12 injections for life. However, there is evidence to suggest that oral vitamin B12 administered at pharmacological doses of the vitamin can be as effective. 45
Food-bound malabsorption
Food-bound malabsorption of vitamin B12 is common in older people. It arises mainly as a result of atrophic gastritis, an age-related disorder affecting up to 30% of people ≥60 years. 46 Atrophic gastritis is a chronic inflammation of the stomach leading to atrophy of the mucosa which in turn results in reduced gastric acid secretion (hypochlorhydria), and therefore diminished B12 absorption as hydrochloric acid and pepsin are essential for the release of vitamin B12 from food proteins. 11 In addition, hypochlorhydria can promote the overgrowth of bacteria in the stomach and small intestine which then utilize vitamin B12, thereby further reducing the amount of the vitamin available for absorption. 47 In particular, Helicobacter pylori (H. pylori) infection has been associated with the development of atrophic gastritis and low vitamin B12 status. 48
In theory, individuals with food-bound malabsorption of vitamin B12 should still be able to absorb free B12 because the secretion of IF remains intact and free vitamin B12 has no gastric acid requirement (because it is not intrinsically bound to food proteins). Food-bound malabsorption leads to a more subtle less profound depletion of B12 status and it remains unclear whether this will ultimately progress to clinical vitamin B12 deficiency. 44
Drug induced malabsorption – proton pump inhibitors
Proton pump inhibitors (PPIs) are a group of acid suppressant drugs intended to treat gastric acid conditions such as gastro-oesophageal reflux disorder, conditions associated with the use of non-steroidal anti-inflammatory drugs, Barrett's oesophagus and hypersecretory disorders such as Zollinger–Ellison syndrome. PPIs bind to and deactivate the gastric H+/K+ adenosine triphosphatase pump which reduces gastric acid secretion leading to profound hypochlorhydria; 49 thus PPI therapy can induce a state clinically identical to atrophic gastritis, the most common cause of vitamin B12 deficiency. This has led to the concern that long-term acid-suppression medication may lead to food-bound malabsorption and ultimately vitamin B12 deficiency especially in those vulnerable to low status such as older people. 50 Chronic acid suppressant use in older people has been associated with the initiation of vitamin B12 therapy. 51 Short-term studies demonstrated that PPIs reduced the absorption of food-bound vitamin B12. 52,53 Long-term omeprazole treatment has been associated with a decrease in serum B12 status in both Zollinger–Ellison patients and older adults; 54,55 however, results have not been entirely consistent with two recent large studies reporting conflicting results. 55,56 Future studies, using a variety of vitamin B12 biomarkers, are required to more fully investigate the effect of prolonged PPI therapy on vitamin B12 status. The impact of PPIs on B12 status is important because they are frequently misprescribed to treat less serious conditions and are in fact among the most frequently used medications, with millions of users worldwide. 57
Gastric/intestinal resection and intestinal disease
As previously described vitamin B12 absorption is dependent upon the normal functioning of the gastrointestinal tract, a function that deteriorates with normal ageing. Diseases that affect either the structure or function of the stomach or part of the small intestine will have a negative impact on the absorption of vitamin B12. Gastric resection destroys the parietal and chief cells in the stomach so the secretion of hydrochloric acid, pepsin and intrinsic factor will be reduced and thus, the absorption of vitamin B12 will be severely disrupted. 5 Furthermore, the subsequent hypochlorhydria promotes bacterial overgrowth in the stomach and small intestine which further exacerbates the problem of vitamin B12 malabsorption. 47 Ileal resection results in the loss of the IF-B12 receptors in the terminal ileum which is one of the fundamental steps in vitamin B12 absorption. A variety of other intestinal conditions such as Crohn's disease, coeliac disease, diverticulitis, intestinal blind loops and tapeworms have also been associated with vitamin B12 depletion. 5
Genetic polymorphisms
Apart from the rare inborn errors of metabolism, a number of polymorphisms in genes involved in B vitamin metabolism have been identified. The most important of these is the 677C → T variant in the gene coding for the folate metabolizing MTHFR enzyme. This polymorphism has been extensively reviewed elsewhere, but in brief, individuals homozygous for the 677C → T polymorphism (TT genotype) in MTHFR have impaired folate metabolism, lower serum and red cell folate and higher plasma homocysteine concentrations and are at increased risk of certain age-related diseases. 58 Meta-analyses of genetic studies have also shown that those individuals with the TT genotype are at a 14–21% greater risk of cardiovascular disease (CVD) compared with those without the polymorphism. 59,60 A more recent meta-analysis reported that individuals with the TT genotype are at increased risk of stroke, however, this association was limited to those with low folate status. 61 There are also several known polymorphisms of the transcobalamin protein, the most common being the 776C → G polymorphism. Individuals homozygous for this polymorphism (GG genotype) are reported to have lower concentrations of holoTC compared with individuals with the wild-type genotype, hence, may impact the use of holoTC as a routine measurement for vitamin B12. 40,62 It is thought that this polymorphism decreases the binding affinity and transport of vitamin B12. 63
Vitamin B12 diagnosis and treatment
Vitamin B12 depletion has been described in four stages ranging from mild depletion to clinical deficiency. 30 Stage 1 is early vitamin B12 depletion characterized by a decrease in holoTC concentration. Stage 2 is cellular depletion characterized by a further decrease in holoTC. Serum total B12 takes longer to respond to changes in status and can remain within the normal range in stages 1 and 2. Stage 3 is characterized by metabolic evidence of deficiency, with elevated concentrations of both plasma homocysteine and serum MMA, low holoTC and normal or below normal serum total B12 concentrations. Stage 4 is clinical deficiency where both serum total B12 and holoTC are low, the metabolic indicators are elevated and there are clinical signs of deficiency including haematological and neurological manifestations. Haematological features, which occur as a result of impaired DNA synthesis, include megaloblastic anaemia characterized by megaloblastic (large immature blood cells with low concentrations of haemoglobin) blood cells, hypersegmentation of neutrophils, leukopaenia and thrombocytopaenia. Neurological symptoms are diverse and irreversible if left untreated. The most severe form manifests as a sub-acute combined degeneration of the spinal cord and is characterized by degeneration of the posterior and lateral columns of the spinal cord. 64
Currently, there is no consensus on how best to diagnose vitamin B12 deficiency. Traditionally, diagnosis of vitamin B12 deficiency relied upon the symptoms associated with megaloblastic anaemia such as muscle weakness, fatigue and shortness of breath and the associated haematological signs. 65 However, the clinical signs of anaemia only occur in the late stages of depletion and furthermore it is now clear that there is not always a continuous progression from mild vitamin B12 depletion to clinical deficiency. 30,44 Clinical deficiency is caused by IF deficiency (i.e. pernicious anaemia) and leads to a progressive and permanent depletion of vitamin B12, but this accounts for only 1–2% of B12 deficiency. 11 Conversely, subclinical vitamin B12 deficiency is much more common and is usually as a result of food-bound malabsorption of B12. 11 Diagnosis of pernicious anaemia is relatively straightforward, patients have very low vitamin B12 concentrations and present with the typical haematological and neurological symptoms. However, diagnosis of subclinical deficiency is much more problematic as it relies on metabolic evidence of deficiency. Thus, it is recommended that the vitamin should be measured by more than one biomarker, both a direct (i.e. serum total vitamin B12 or holoTC) and indirect functional marker (i.e. plasma homocysteine or serum MMA) should be used in combination. 44
Treatment for IF deficiency is well established with patients receiving monthly intramuscular vitamin B12 injections for life. However, some studies demonstrate that oral supplementation with very high doses (i.e. 1000 µg/day) B12 may be equally effective because 1% of the vitamin can be passively absorbed without the need for IF. 66,67 Although this is an attractive prospect for clinicians, caution must be exercised as patients may not be fully compliant in the long-term. 68 Conversely, there is no consensus on how to best treat (if at all) subclinical deficiency as its clinical significance remains unclear. In the USA (but not in Europe or elsewhere), specific age-related dietary recommendations for the population have been issued by the Institute of Medicine, 69 recommending that people aged ≥50 years should consume most of their vitamin B12 from crystalline sources, i.e. fortified food and supplements. A regular consumption of foods fortified with B12 or low-dose supplements will be more than sufficient for addressing food-bound malabsorption. 70,71
Emerging roles in health of low/deficient vitamin B12 status in the older population
The potential role of homocysteine and the metabolically related B-vitamins in the development of chronic disease has attracted much interest within the scientific community. This interest was initially sparked from observations that individuals with homocystinuria, an inborn error of metabolism characterized by excessive concentrations of homocysteine, had premature vascular disease and mental retardation. 72 Since then, much scientific literature has emerged focusing mainly on homocysteine, but also on the metabolically related B vitamins, most notably folate and vitamin B12, in relation not only to CVD, 73,74 and stroke, 75 but also cognitive function, 76 fracture risk 77 and osteoporosis. 78 The possibility that B vitamin supplementation might help delay or prevent chronic disease (via correction of suboptimal status and/or lowering homocysteine) is of major interest and could have important public health implications. 79
Cardiovascular disease
CVD is the leading cause of death worldwide. Two major meta-analyses of epidemiological evidence published in 2002 predicted that a lowering of plasma homocysteine by 3 µmol/L (or by about 25% based on typical homocysteine concentrations of 12 µmol/L) would reduce the risk of heart disease by 11–16% and stroke by up to 19–24%. 73,74 As recently reviewed, the evidence from the secondary prevention trials in at-risk patients has failed to demonstrate a benefit of homocysteine-lowering therapy with B vitamins (usually using folic acid in combination with vitamin B12) on CVD events. 58 Currently, the evidence is stronger for stroke, with meta-analyses of randomized controlled trials (RCTs) showing that homocysteine lowering reduces the risk of stroke. 75,80 Few studies have investigated the independent effects of vitamin B12 on CVD risk. Subgroup analysis of the Vitamin Intervention for Stroke Prevention (VISP) Trial confined to those individuals considered to benefit from supplementation (i.e. non-supplement users and those without pernicious anaemia) suggests that higher doses of vitamin B12 may be required as they reported that individuals with higher baseline vitamin B12 and taking the high-dose B vitamin had reduced risk of recurrent vascular events while those with the lowest vitamin B12 status on the low-dose combination had the greatest risk of recurrent vascular events. 81 In addition, a number of other B vitamin RCTs have detected beneficial effects on a number of cardiac endpoints. Combined B vitamin supplementation including high-dose vitamin B12 has been associated with reduced re-stenosis and cardiovascular events in patients following coronary angioplasty. 82,83 Combined vitamin B12 and folic acid supplementation was also associated with improved coronary blood flow in patients with coronary artery disease. 84 Evidence from meta-analyses also supports a role for vitamin B12 in CVD, with a greater effect on stroke reported when folic acid was used in combination with vitamins B12 and B6 compared with folic acid alone. 80,85 Further research is warranted to more fully investigate the potential benefits of B vitamin supplementation on CVD risk, particularly in those with pre-existing vascular disease.
Cognitive function
Dementia and cognitive impairment disorders are a major public health concern in ageing populations. 86 Observations from case-control studies, that patients with Alzheimer's disease (the most common form of dementia) had significantly higher homocysteine concentrations than age-matched controls, prompted the theory that B vitamin status might be an important modifiable risk factor for cognitive dysfunction in ageing. 76,87 Cross-sectional and longitudinal studies provide a wealth of evidence to support an association between homocysteine and/or related B vitamins with cognitive function in both healthy older adults and patients with dementia. 88 A recent meta-analysis of epidemiological studies estimated that a 3-µmol/L reduction in homocysteine would reduce the risk of dementia by up to 22%. 89 Both folate and vitamin B12 have been extensively investigated in relation to cognitive performance in ageing; however, the evidence is less consistent for vitamin B12. 90–93 Many of these studies relied on serum total vitamin B12 as the sole biomarker measure of status; however, those that used MMA or holoTC are generally more supportive of a protective role for vitamin B12 in maintaining cognitive function in ageing. 79,94
A number of RCTs have investigated the potential benefits of B vitamin supplementation on cognitive function; however, many of these studies were of insufficient duration or sample size necessary to reach useful conclusions. 95–104 One large trial of long-term duration that reported no benefit was inherently flawed as the cognitive arm of the investigation was initiated 1.2 years after commencement of B vitamin treatment; therefore, any potential impact on cognitive function may have been missed. 96 Two a priori RCTs, both conducted in healthy older people for a considerable duration, have been published with conflicting results. 101,102 McMahon et al. 101 found no benefit of combined B vitamin supplementation (1000 µg folate, 500 µg vitamin B12, 10 mg vitamin B6 daily) on cognitive performance in 276 dementia-free older people (aged ≥65 years). Conversely, a supplementation trial with folic acid (800 µg/day) alone in 818 people aged 50–70 years reported significant improvements in cognition over a-three year intervention period. 102 This discrepancy is intriguing given that both studies were similar in design, interestingly however, the baseline B vitamin status between the two studies was very different. The study by Durga was conducted in the Netherlands, a country which does not permit folic acid fortification (even on a voluntary basis), while the trial by McMahon showing a null effect was conducted in New Zealand which has had an active voluntary fortification programme since 1996. Hence, the participants in Durga's study had lower baseline concentrations of folate and vitamin B12 compared with McMahon's study and therefore may have been more likely to benefit from folic acid intervention to optimize status. More recently, combined B vitamin supplementation was shown to improve cognitive performance in community-dwelling older adults with depressive symptoms. 103 The strongest evidence to date to provide support for a cause and effect relationship between B vitamin status and cognitive function is from the homocysteine and B vitamins in cognitive impairment (VITACOG) study. B vitamin supplementation (0.8 mg folic acid, 0.5 mg vitamin B12, 20 mg vitamin B6) slowed down the rate of brain atrophy (as measured using magnetic resonance imaging) 100 and improved cognitive performance 104 in patients with mild cognitive impairment. A number of RCTs conducted to investigate the effect of B vitamin intervention on CVD also include a measurement of cognitive function. While not primarily designed to examine cognitive health, the evidence from these trials should be helpful in contributing to the evidence linking optimal B vitamin status to the maintenance of cognitive function in ageing, although they will be confounded to some extent by the fact that many of the participants will have existing vascular disease.
Bone health
Osteoporosis is characterized by low bone mass and micro-architectural deterioration in bone tissue, resulting in an increased risk of fracture and affects over 75 million people in Europe, USA and Japan. Evidence from epidemiological studies has reported that elevated plasma homocysteine concentrations and/or low B vitamin status are associated with lower bone mineral density (BMD) 78,105 and a higher risk of osteoporosis 106 and osteoporotic fracture. 107 Several studies have linked low vitamin B12 status with osteoporosis and low BMD. 106,108,109 A retrospective study of postmenopausal women found that those with pernicious anaemia had a more than two-fold risk of fracture compared with those without pernicious anaemia. 110 Moreover, an increase in BMD and a reversal of osteoporosis was demonstrated in a patient with pernicious anaemia, severe osteoporosis and multiple vertebral compression fractures after two years treatment with vitamin B12. 111 Low vitamin B12 status has been associated with increased risk of fracture 77,107 and more rapid bone loss in older women; 112 however, the evidence is not entirely consistent. 107
Many RCTs investigating the effect of B vitamin supplementation showed no evidence of a beneficial effect; however, many of these interventions were conducted over too short a timeframe and typically measured bone turnover biomarkers, the validity of which has yet to be established. 113,114 One RCT of sufficient duration and with robust bone outcomes did, however, show that combined folic acid and vitamin B12 supplementation for two years was effective in reducing the risk of hip fractures in Japanese patients following stroke. 115 There is a need for further well-conducted RCTs before any firm conclusions can be made in relation to the role of vitamin B12 and related B vitamins in bone health.
Public health implications
The policy in North America and elsewhere of mandatory fortification of staple foods with folic acid for the prevention of neural tube defect (NTD) pregnancies has been an indisputable success. 116 In addition to lowering NTDs, this measure may also have had a benefit in reducing stroke mortality. 117 The implementation of a similar policy has, however, been delayed in the UK because of concerns that high-dose folic acid could have a detrimental effect on health.
Potential adverse effects of over exposure to folic acid in those with low vitamin B12
Evidence from historic case reports of vitamin B12-deficient patients mistakenly treated with folic acid have led to the concern that high doses of folic acid may exacerbate the neurological symptoms and mask anaemia in individuals with low vitamin B12. 118 Analysis of post folic acid fortification data from the USA and Canada has been useful in assessing the impact of high folic acid exposure on vitamin B12 status. Since the introduction of mandatory fortification there has been a dramatic increase in blood folate concentrations by up to 40%, 119 with unmetabolized folic acid (i.e. not the normal circulating co-factor form) now being detected in the circulation. 120 This in turn has led to an intriguing scenario where an increasing number of older adults have very high folate concentrations coupled with low vitamin B12 status. This is a relatively new phenomenon and there are concerns over potential adverse effects on health. The most widely documented concern is that folic acid at high doses may potentially ‘mask’ the anaemia of vitamin B12 deficiency, traditionally diagnosed by macrocytosis (elevated mean corpuscular volume [MCV] >100 fL). 5 This could arise as a result of folic acid being converted to tetrahydrofolate (through the activity of dihydrofolate reductase), bypassing the vitamin B12-dependent enzyme methionine synthase, thus promoting DNA synthesis and correcting signs of anaemia (independent of whether it was caused by folate or vitamin B12 deficiency) but not the neurological damage specifically caused by vitamin B12 deficiency. This is of concern in individuals with low B12 status because correction of anaemia will alleviate the symptoms associated with it and thus may delay diagnosis allowing the irreversible neurological damage to progress unabated. 5,118
An early report concluded, however, that the introduction of mandatory folic acid fortification in the USA has not had an adverse effect on the diagnosis of vitamin B12 deficiency. 121 Analysis from the National Health and Nutrition Examination Survey (NHANES) in the USA, however, observed another potential adverse interaction between folate and vitamin B12. Low vitamin B12 was, as expected, associated with elevated homocysteine and MMA. 122 Unexpectedly, however, homocysteine and MMA were highest in individuals with low vitamin B12 coupled with high folate, and furthermore, these individuals were at greater risk of both anaemia and cognitive impairment. 123 Two subsequent studies conducted in the UK and Ireland failed to detect similar adverse interactions between high folate and low vitamin B12, an inconsistency that might be explained by the absence of mandatory folic acid fortification in both countries resulting in generally lower folate, and particularly lower unmetabolized folic acid concentrations. 124,125
Apart from concerns regarding potential adverse interactions with vitamin B12, a new concern has emerged in recent years that excessively high intakes of folic acid (the synthetic vitamin form) could have a cancer-promoting effect in segments of ageing populations, but this is rather controversial. On the one hand, there is consistent epidemiological evidence that higher folate intake within the dietary range plays a protective role against cancer at various sites, on the other hand there is evidence suggesting that long-term exposure to high intakes of folic acid (greater than 1 mg/day) may promote colorectal tumorigenesis in patients with pre-existing lesions or even increase cancer risk generally. 126 Thus policy-makers worldwide (including the UK government) have delayed decisions to implement population-based folic acid fortification policies similar to those introduced in 1996 in North America.
Given the potentially serious consequences of over-exposure to folic acid some have suggested that vitamin B12 should be included together with folic acid in any new food fortification policy. Not only would this prevent any adverse outcomes in individuals with low vitamin B12 status, but would also address the issue of food-bound malabsorption of vitamin B12, the most common cause of low vitamin B12. Alternatively, a specific age-related dietary recommendation similar to that issued by the Institute of Medicine (IOM) in the USA where people aged ≥50 years are recommended to consume most of their vitamin B12 from crystalline sources (i.e. fortified food and supplements) would also address these concerns. 69 If mandatory folic acid fortification proceeds in the UK, with or without the addition of vitamin B12, it will be important to closely monitor any health effects in the older population.
Conclusion
Low vitamin B12 status is highly prevalent in older age and is much more common than generally perceived. There is a wealth of epidemiological evidence to suggest that optimal vitamin B12 status is important for healthy ageing and that low status is associated with an increased risk of chronic diseases of ageing including CVD, dementia, cognitive impairment and osteoporosis. The evidence to date from RCTs examining the effect of B vitamin supplementation on chronic disease has often failed to demonstrate beneficial effects although many of these trials were secondary prevention trials. Thus, there is a need for large scale, well-designed intervention studies to investigate the potential benefits of vitamin B12, alone or in combination with related B vitamins, in preventing disease in those without existing disease pathology and particularly in those with lower vitamin B12 status. The importance of detecting low vitamin B12 status has been highlighted in this review; however, conventional vitamin B12 assays may lack sensitivity and could mean that low/deficient B12 status remains undetected in a significant proportion of the older population. HoloTC shows promise as a sensitive biomarker of vitamin B12 status; however, further investigations are needed to determine its sensitivity (and response to vitamin B12 intervention) before its usefulness as the front-line tool in the diagnosis of vitamin B12 deficiency can be confirmed. The issue of food-bound malabsorption of vitamin B12 and consequences of mild subclinical deficiency have still not been fully explored and require further investigation. The possible adverse consequences of high folic acid exposure in older people with low vitamin B12 status will require close monitoring over the coming years. Optimization of vitamin B12 in older people should be an urgent public health priority.
Footnotes
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry.
Acknowledgements
None.
Declaration of Conflicting Interests
None.
Funding
CFH was funded through a PhD studentship provided by the Department for Employment and Learning, Northern Ireland.
Ethical approval
Not applicable.
Guarantor
HMcN.
Contributorship
CFH wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version.
