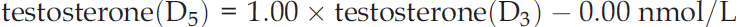Abstract

We read with interest the recent paper by Owen and Keevil 1 regarding the importance of internal standard choice for testosterone measurement by liquid-chromatography tandem-mass spectrometry (LC-MS/MS). The authors investigated the performance of three isotopically labelled internal standards for testosterone, i.e. D2-testosterone, 13C3-testostosterone and D5-testosterone, using the same protocol and a substantial number of samples covering the whole clinical range. While we agree that a proper internal standard is imperative for accurate LC-MS/MS measurements, the observed differences illustrated by their data might have clinical impact and left a few questions.
Stable isotope-dilution with 13C-isotopes to correct for losses during sample preparation is considered to be the standard because 13C-labelled compounds co-elute exactly with their endogenous counterpart. Moreover, the stability of deuterium labelled-isotopes is highly dependent on the location of the labels which might exchange with hydrogen-atoms. Nevertheless, it is generally accepted that any stable isotope could be sufficient. Comparison between D2 and 13C3 by Owen and Keevil 1 resulted in differences that are greater than the reported imprecision of the assay. It seemed fair to assume that results obtained with D2 were considered the target as these results have previously been shown to concur excellently with those obtained with a reference method. 2 However, two different D2 manufacturers are reported and it is unclear whether the same D2 was used in the comparison with the reference method 2 as in the recent paper. 1
The discrepancy between D2 and D5 is thought-provoking because, in our experience, an unexpectedly large difference was reported. According to their data, differences up to 11 nmol/L were observed when changing the internal standard from D2 to D5. Isotopically labelled components tend to elute slightly shifted from their endogenous counterpart, especially when high-resolution chromatography such as UPLC is used. Ion suppression, which is known to be sample-specific, could cause these differences in outcomes. For proper interpretation, it is essential to know to what degree D5 and endogenous testosterone co-elute in the method of Owen and Keevil, 1 and if the deviated samples show a different ion suppression profile.
We tested the differences between D3 and D5 on testosterone measurements using a method that was reported previously and also showed excellent agreement with a reference method.
3
The internal standards were tested for purity prior to the development of the method and both internal standards co-eluted with endogenous testosterone. Furthermore, ion suppression was tested on multiple samples during method validation. Samples from men and women (n = 40), covering the clinical range (0.16–50.7 nmol/L), were prepared using D3 and subsequently D5 as internal standard. To exclude possible instrumental shifts, the samples were measured alternately. Comparison by Passing–Bablok analysis resulted in the equation:
Bland–Altman bias plot of D3-testosterone compared with D5-testosterone results. The solid line at −0.09 nmol/L represents the mean difference. The dotted lines at 0.77 and −0.94 nmol/L represent the ± 1.96 SD interval, respectively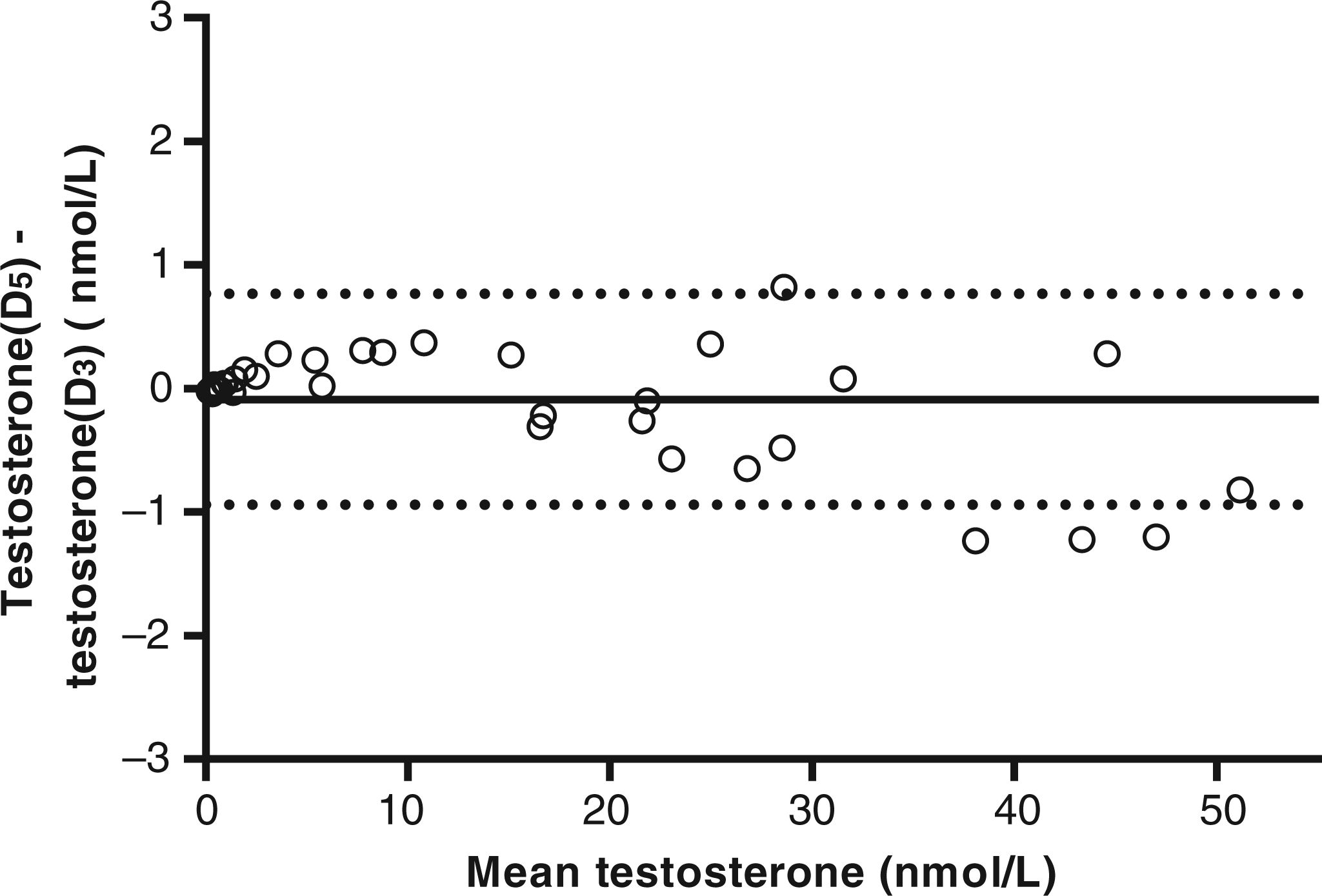
Even though our observations differ from those reported by Owen and Keevil, we agree that the choice of internal standard is crucial. We were surprised to see the differences reported. In general, the use of any internal standard should be validated. If necessary, the method could be adjusted to ensure proper functioning of the internal standard.