Abstract
Classical quantitative chemometrics based on absorbance spectra has been routinely performed for approximately 40 years. Since absorbance is a function of the absorption index, it is natural to extend chemometric methods to the refractive index function. This function, related to the absorption index via the Kramers–Kronig relations, is derived from corrections applied to absorbance spectra to ensure compliance with wave optics principles. In this note, we demonstrate that, at least in the quasi-thermodynamically ideal binary system of benzene and toluene, classical quantitative chemometrics performs better when based on refractive index spectra than when based on absorption index spectra. The primary reason for this difference is that the refractive index at a given wavenumber integrates all changes resulting from absorptions at higher wavenumbers. This property is particularly advantageous in non-absorbing regions, where absorption index spectra provide no information about the system's composition.
This is a visual representation of the abstract.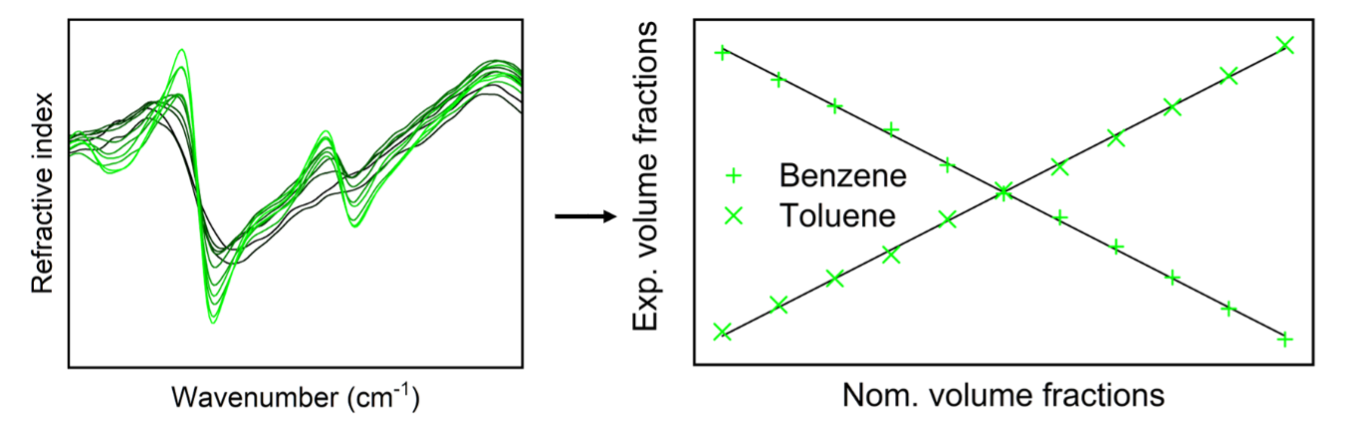
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
