Abstract
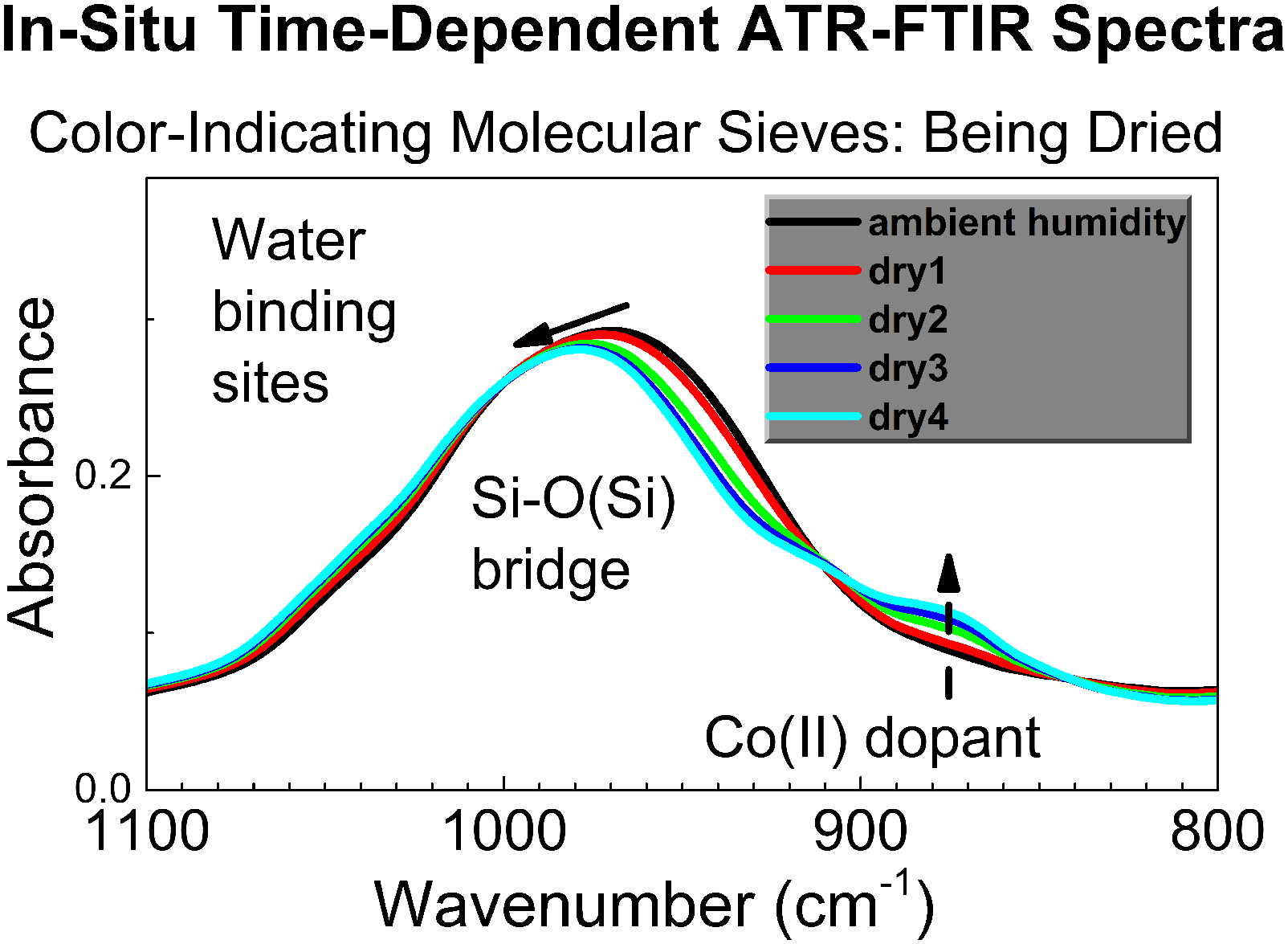
Attenuated total reflection Fourier transform infrared (ATR FT-IR) spectroscopy is a powerful instrumental method of chemical analysis of solids and liquids. The majority of published studies by in situ ATR FT-IR spectroscopy describe analysis of homogeneous samples, such as liquid solutions under circulation, or films on the ATR crystal that react with the gas of interest. The in situ ATR FT-IR spectroscopic studies of specimens in physical shape of crystals or powder that react with a gas or vapor are rare. This work describes a modification of in situ time-dependent ATR FT-IR spectroscopy to allow monitoring heterogeneous reaction “solid-gas” of powder in controlled atmosphere and in the time domain. Also, we describe a new facile gas flow chamber attachment to ATR FT-IR spectrometer which allows creating controlled atmosphere surrounding the specimen on the ATR crystal. Additionally, the capabilities of the described in situ time-dependent ATR FT-IR spectroscopy experiment in controlled atmosphere are enhanced by the sensor for in situ time-dependent monitoring the relative humidity (RH) of air surrounding the specimen. The operation of the setup for in situ time-dependent ATR FT-IR spectroscopy in controlled atmosphere is demonstrated by monitoring reaction of gradual desorption of water vapor from color-indicating molecular sieves under controlled low air humidity. Further, the described spectroscopic method and apparatus is applied to monitor the reverse process, namely sorption of water vapor by color-indicating molecular sieves under controlled elevated air humidity. Water molecules are found to reversibly interact with two distinct sorption sites in the sorbent: the Si–O backbone and the color-indicating Co(II) centers. The reported variant of in situ time-dependent ATR FT-IR spectroscopy in controlled atmosphere is powerful, yet facile and straightforward. It is promising for mechanistic, in situ studies of sorption, desorption, chemosensing, heterogeneous catalysis and photocatalysis, and analysis of chemical kinetics of various “solid-gas” reactions.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
