Abstract
This paper presents the results of study of titanium–magnesium catalysts often used in polymerization processes, by photoluminescence spectroscopy (PL) in combination with diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS). The interaction of dibutyl phthalate (DBP) with MgCl2 was studied at DBPadded/Mg = 0–1 (mol/mol). The luminescence spectra with excitation at 278 nm and the excitation spectra for main emission bands were recorded. It was shown that DBP adsorbed on magnesium chloride, both in the form of donor–acceptor complexes (D+A–) and in the form of molecular complexes. At DBPadded/Mg <0.15, the formation of D+A– complexes occur predominantly; with an increase in DBPadded/Mg, the fraction of molecular complexes increases. Molecular complexes are destroyed during the treatment of the support by TiCl4. In this case, the structure of magnesium chloride is disordered and new coordination–unsaturated sites are formed. This work is a first attempt to apply PL spectroscopy in combination with DRIFTS spectroscopy to study titanium–magnesium Ziegler–Natta catalysts. The application of PL spectroscopy to such systems made it possible to detect interactions within and between donor molecules, which would be particularly challenging to achieve using other spectroscopic methods. Both spectroscopic methods provided crucial information about the existence of two types of complexes on the sample surface which is important for tuning the synthesis procedure of the titanium–magnesium catalysts for olefin polymerization.
Keywords
Introduction
Titanium–magnesium catalysts (TMCs) for the stereospecific polymerization of ethylene have been known for over 60 years. TMCs are usually composed of magnesium chloride, electron donor base (phthalates, for example, dibutyl phthalate (DBP), malonates, alkoxysilanes, succinates, 1.3-diesters), and TiCl4 (Scheme 1).
General scheme for the synthesis of TMC (black balls: Mg, light balls: Cl).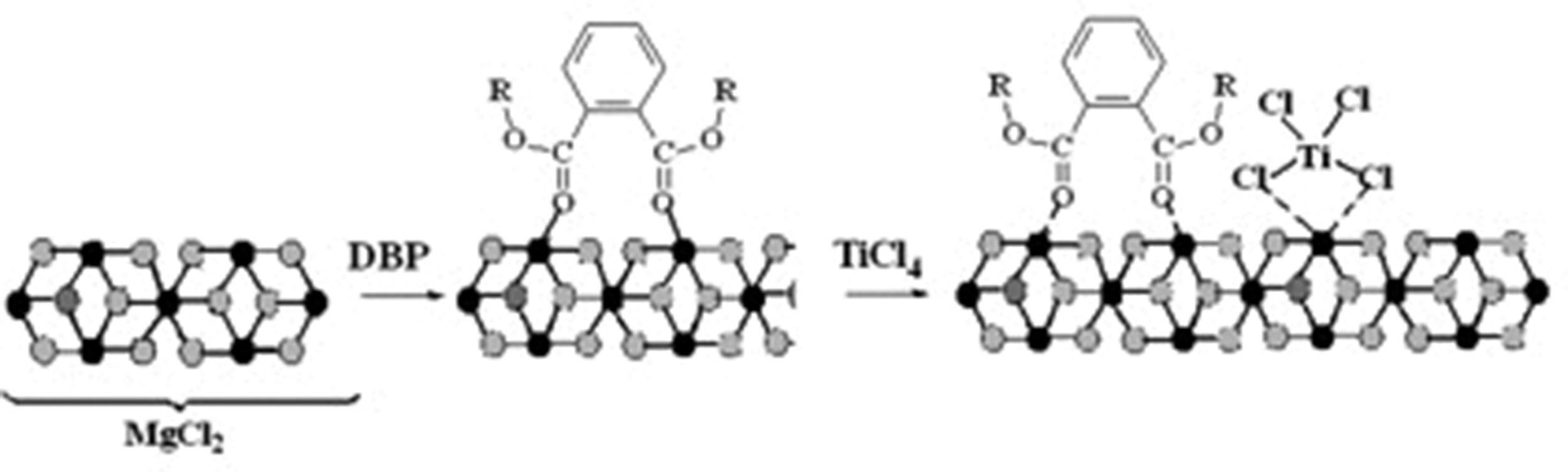
Despite numerous studies, the formation of TMCs is still debatable, and this is primarily due to unresolved questions about the nature of the interaction of the Lewis bases (LB) with magnesium chloride and TiCl4. The specificity of the catalysts for olefin polymerization does not allow the contact of samples with air at any stages of their preparation, and the catalysts themselves operate at temperatures of 40–90 ℃. Therefore, exploring such systems is challenging.
Infrared (IR) spectroscopy and in particular diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) is one of the common physicochemical methods of analysis.1–4 It allows one to study the surface groups of a catalyst, evaluate acid–base properties, and study reaction mechanisms. IR spectroscopy is actively used to study TMCs. In the study of catalysts for olefin polymerization, the DRIFTS approach has an advantage over other modes of measurements in IR spectroscopy because it is very suitable for studies catalysts as powder and without contact of the catalysts with the atmosphere, i.e., in situ. Thus, the acid sites of magnesium chloride obtained from Mg and BuCl were studied using DRIFTS with CO as a probe molecule. 5 Three bands were observed in the DRIFTS spectrum of CO adsorbed at room temperature: 2210, 2190, and 2170 cm–1, which the authors attributed to three-, four-, and five-coordinated Mg2+ ions. Later, low-temperature IR spectroscopy of CO adsorbed at MgCl2 obtained from the MgCl2 · 6C2H5OH adduct also revealed three bands at 2194, 2182, and 2163 cm–1. 6 The authors attributed the band at 2182 cm–1 to CO complexes formed on four-coordinated magnesium ions of the (110) face, the band at 2163 cm–1 to five-coordinated magnesium ions of the (104) face, and the band at 2194 cm–1 to (012) and (015) faces containing five-coordinated magnesium ions. The higher ν(CO) value of CO complexes on five-coordinated magnesium ions of the (012) and (015) faces compared to CO complexes on four-coordinated magnesium ions of the (110) face was explained as the effect of chlorine ligands belonging to the neighboring layer on the CO molecule.
The interaction of DBP and ethyl benzoate (EB) with MgCl2 at LB/MgCl2 <0.3 (LB = DBP, EB) was studied by the IR spectroscopy.7,8 It was shown that esters are fixed on the surface of MgCl2 due to the interaction of the carbonyl group with the Lewis acid site (LAS) of magnesium chloride. In this case, surface complexes of the donor–acceptor type (D+A–) are formed. For malonates, alkoxysilanes, phthalates, succinates, 1,3-diesters, the existence of various forms of adsorption on the surface of magnesium chloride was shown by quantum chemical calculations. On the (104) face (sometimes referred to as (100)), both monodentate and bidentate (bridge) donor coordination are possible, and on the (110) face, bidentate (bridge), chelate, and interlayer (zip) donor coordination are possible.9–11 Depending on the O–O distance, a preferred form of donor adsorption on the (110) or (104) face is plausible.
It is worth noting that [MgCl2 · (LB) n ]x molecular complexes can be obtained from magnesium chloride, for example, the complexes with n = 2–3, where LB is HCOOC2H5, CH3COOC2H5, C2H5OH, dialkyl ketones.12–15 As was shown using X-ray diffraction (XRD), such adducts are characterized by a chain structure resembling the polymer [MgCl2 · nLB]x chains.12–15 After chemical treatment, using TiCl4 or thermal activation, such structures are destroyed with the formation of δ-MgCl2, and the donor content becomes very low (n <0.3).12,16 Such molecular complexes can play a key role in the formation of the magnesium chloride, and thus studies investigating the structure of such [MgCl2 · (LB) n ]x adducts are of significant interest. Unfortunately, the molecular complexes were not detected using IR spectroscopy in TMC, they were observed using XRD in titanium adducts, e.g., [MgCl2 · (HCOOC2H5) k ·(TiCl4) m ]x, 0.286 <k <0.5 and m/k ≈ 0.16. 12 However, we do not exclude the presence of such adducts in ready-made TMCs. At the same time, IR spectroscopy does not allow the separation of molecular complexes and D+A– complexes, since they have close absorption bands of stretching vibrations of carbonyl groups (νС=О).
It is known that with γ irradiation of a magnesium chloride, F sites are formed that are characterized by 3.35–3.40 eV (400–394 nm) in the electronic spectrum, and by a signal of g = 2.0 with a half width of 50 ± 5 G in the EPR spectrum. 17 Quantum chemical calculations show that in a well-ordered MgCl2 crystal, F sites are formed as a result of the removal of one Cl* atom, one or two Cl– ions from (001) surface. 18 In these works, it was suggested that such F sites can play an important role in the formation of TMCs. Furthermore, “activated” MgCl2 with a large number of defects on the surface is used for the synthesis of TMC.
Cheng et al. 19 showed by DFT method the possibility of the existence of at least 12 defects on the (110) and (104) faces. These defects can contribute to the formation of F sites in MgCl2/LB samples and can possibly be identified by electron spectroscopy and photoluminescence (PL) spectroscopy.
Photoluminescence spectroscopy is one of the most sensitive research methods for determining low concentrations of luminescent compounds. The process of fluorescence and phosphorescence of organic compounds in solutions is usually described by the energy level diagram proposed by Kinno and Oblanka. 17 Since nonradiative energy transfer depends on the electronic structure of the molecule and on the interaction of the molecule with the environment at short molecular distances, the PL spectra of organic molecules provide information on their interaction with neighboring molecules, which is usually not available for other spectral methods. In this case, phosphorescence spectra are especially informative. Phosphorescence is a slow process and so nonradiative processes of energy dissipation, for example, nonradiative deactivation, compete with it. Therefore, phosphorescence of organic molecules usually manifests itself in the gas phase, solution, or when the molecules are “rigidly” fixed in solution at low temperatures in an inert atmosphere, when interaction with the environment is minimized.20,21 In some cases, phosphorescence is observed without freezing, at room temperature. For example, 7-bromo-9,9-didodecylfluorene-2-carbaldehyde shows phosphorescence in a solution of CHCl3 and a polymethylmethacrylate (PMMA) matrix at 298 K in argon. 22 It was shown that weakly photoluminescent 2,5-dihexyloxy-4-bromobenzaldehyde introduced into PMMA is capable of phosphorescing at 298 K. 23
Phosphorescence is also characteristic of molecular complexes of the donor–acceptor D+A– type. For example, there are complexes with charge transfer hexamethylbenzene–tetracyanbenzene; durene and mesitylene with phthalic or tetrachlorophthalic anhydrides; pyrene, perylene, and anthracene with amines.20 PL spectroscopy allows the study of the intermolecular interaction between adsorbed molecules on the surface. Nishikiori et al. managed to isolate the monomeric, dimeric, and larger associates of the 9-aminoacridine molecule on the surface of silica gel, since hydrogen bonds or other interactions lead to a bathochromic shift of the observed bands in the PL spectra. 24
The study of TMCs is a challenging task, and this is related to the preparation of samples for analysis. These systems are unstable in the air, and minor changes in synthesis (temperature, solvent, DBP/Mgadded ratio, etc.) can extremely change the catalytic properties of the produced catalyst. Besides, real catalysts may differ greatly from the model systems. DRIFTS and PL spectroscopy allows exploring the real catalysts and controlling the process of their synthesis. These methods are available, simple to use, and allows identifying the reasons of uniqueness of a particular catalyst. Analysis of the literature showed that the PL spectroscopy was not previously used for the study of TMCs. However, the presence of coordination–unsaturated sites in magnesium chloride suggests the possibility of its use. We anticipated that this novel approach will allow us to establish fine details of the interaction of donor molecules with the surface of magnesium chloride.
The aim of this work was to identify various D+A– complexes of the donor–acceptor type, as well as molecular complexes on the surface of magnesium chloride using PL spectroscopy in combination with IR spectroscopy. The formation of such complexes can be expected upon DBP adsorption as a result of the interaction of the oxygen of the carbonyl group of the donor with the LAS of the MgCl2 support. The results of this study provide a molecular-level insight for further developments in the synthesis of titanium–magnesium Ziegler–Natta catalysts.
Experimental
All of the chemical reagents and solvents used in this study, heptane, chlorobenzene (PhCl) and DBP, buthyl chloride (BuCl), were dried over molecular sieves.
Synthesis of MgCl2(BuCl)
An activated MgCl2 sample was synthesized accordingly to described procedure by reaction of magnesium with BuCl (at a molar ratio BuCl/Mg = 3) in n-heptane at 98 ℃, and then washed twice with the same solvent. 25 MgCl2(BuCl) sample (SBET = 70 m2/g) contained ∼10 wt.% of organic products.
Synthesis of MgCl2/nDBP
As a standard procedure, a suspension of MgCl2(BuCl) in PhCl containing 1 g MgCl2/L was prepared in a reactor purged with dry argon by addition of PhCl to the MgCl2 powder under vigorous stirring. The suspension was then treated with a solution of donor (DBP) in PhCl (Cdonor = 0.2 M), at a molar ratio of donor/MgCl2 = 0.1 or 1 at 115℃ for 1 h under a continuous flow of argon. After this reaction, chlorobenzene was decanted and the sample was washed twice with hot (115℃) PhCl and three times with heptane at room temperature, in portions of 20 mL per 1 g of MgCl2.
Synthesis of MgCl2/DBP/TiCl4
As a standard procedure, a suspension of MgCl2/D in PhCl containing 40 g MgCl2/L was prepared in a reactor purged with dry argon by addition of PhCl to the MgCl2 powder under vigorous stirring. The suspension was then treated with a solution of TiCl4 in PhCl, at a molar ratio of Mg/Ti = 100 at 115℃ for 1 h under a continuous flow of argon. After this reaction, chlorobenzene was decanted and the sample was washed twice with hot (115℃) PhCl and three times with heptane at room temperature, in portions of 20 mL per gram of MgCl2.
Chemical analysis
The Mg and Ti contents of the samples were determined using inductively coupled plasma -- atomic emission spectroscopy (ICP-AES) on an Optima 4300 DV (PerkinElmer) spectrometer, while the contents of DBP were determined by high-performance liquid chromatography in isocratic mode by using standard solutions of the compounds in acetonitrile. The measurements were made on an LC-20 Prominence (Shimadzu) liquid chromatograph.
Diffuse Reflectance Infrared Fourier Transform Spectroscopy
All DRIFTS spectra were acquired on samples under an inert Ar atmosphere. All samples were dried under vacuum to a residual pressure of 2 × 10–2 mbar before measurements. Samples (0.2–0.3 g) were transferred, under an inert atmosphere, to a cell suitable for DRIFTS measurements. DRIFTS spectra were acquired with an FTIR-8400S (Shimadzu) spectrometer equipped with deuterated lanthanum α alanine-doped triglycine sulfate detector and a DRS-8000 attachment in the range of 400–6000 cm–1 with a resolution of 4 cm–1. All spectra are presented in F(R) Kubelka–Munk scale: F(R) = (1 − R) 2 /2R, where R is the reflection coefficient.
Photoluminescence Measurements
PL spectra (λexcitation = 278 nm) and photoluminescence excitation (PLE) spectra of the samples were obtained using an Eclipse (Cary) spectrofluorimeter.
Results and Discussion
MgCl2/nDBP Samples Study Using DRIFTS
Chemical analysis data of MgCl2/DBP supports and corresponding MgCl2/DBP/TiCl4 catalysts.
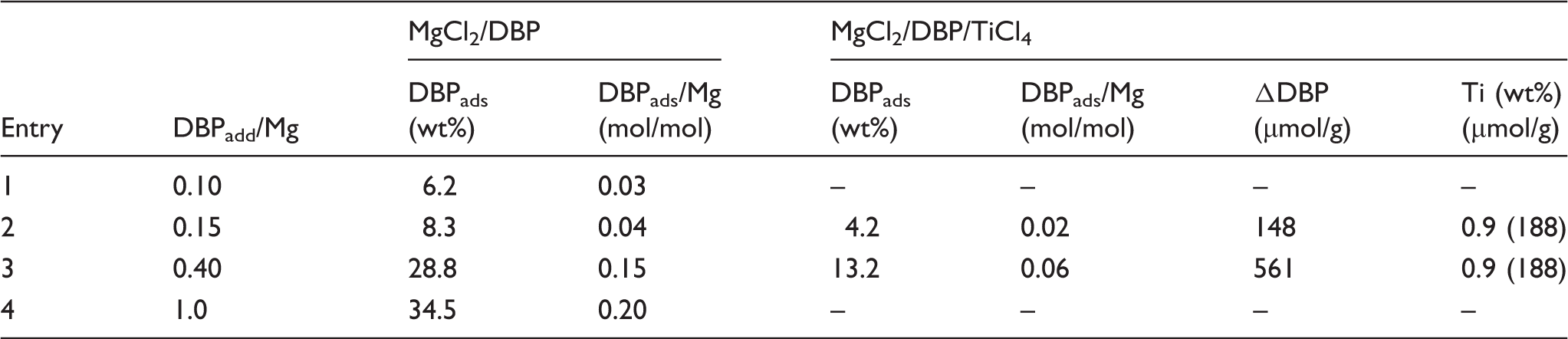
It can be seen that the amount of adsorbed donor in MgCl2/nDBP samples increases with an increase in DBPadded/MgCl2. The amount of adsorbed donor (DBPads) is less than the introduced DBP and is 20–30% of DBPadded in all samples. This is indicated by a lower DBPads/Mg value in the samples. A similar result was obtained for TMC prepared from Mg(OEt)2, TiCl4, and DBP at DBPaddeed/Mg = 0.15. 26 Such TMC had a molar ratio of DBPads/Mg equal to 0.067 and the content of DBP of 13.5 wt%.
Figure 1 shows the effect of the DBPadded/Mg molar ratio on the amount of fixed DBP (DBPads). It can be seen that as the DBPadded/Mg molar ratio increased, there was an increase in the donor content in the samples. Moreover, in the samples obtained at DBPadded/Mg <0.15 (region I), the adsorbed DBPads increases linearly, and in the samples obtained at DBPadded/Mg >0.15 (region II), it changes drastically. The data suggest that the interaction of DBP in these regions with magnesium chloride occurs in different ways. It is possible that in the region I, the donor is adsorbed onto the LASs of the MgCl2 support with the formation of D+A– donor–acceptor complexes, and in the region II, the formation of molecular complexes takes place.
Dependence of the amount of fixed DBP on the molar ratio of introduced DBP to magnesium for MgCl2(BuCl)/nDBP samples.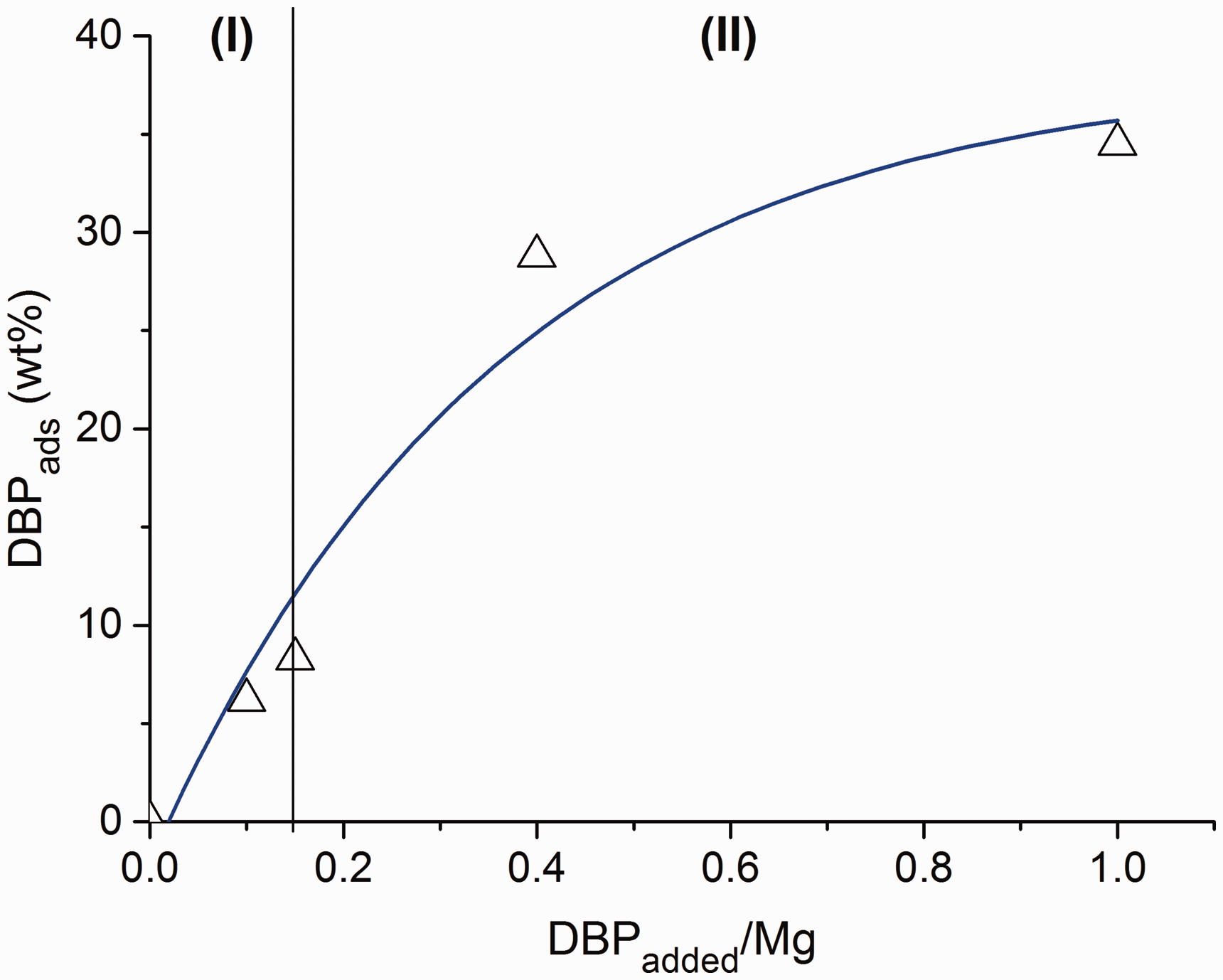
Figure 2 shows the DRIFTS spectra acquired from MgCl2/nDBP samples in the vibrational region of DBP carbonyl groups. It can be seen that the shape of the spectra of the samples at all molar ratios of DBPadded/Mg are similar.
DRIFTS spectra of MgCl2(BuCl)/nDBP samples obtained at various DBPadded/Mg ratios.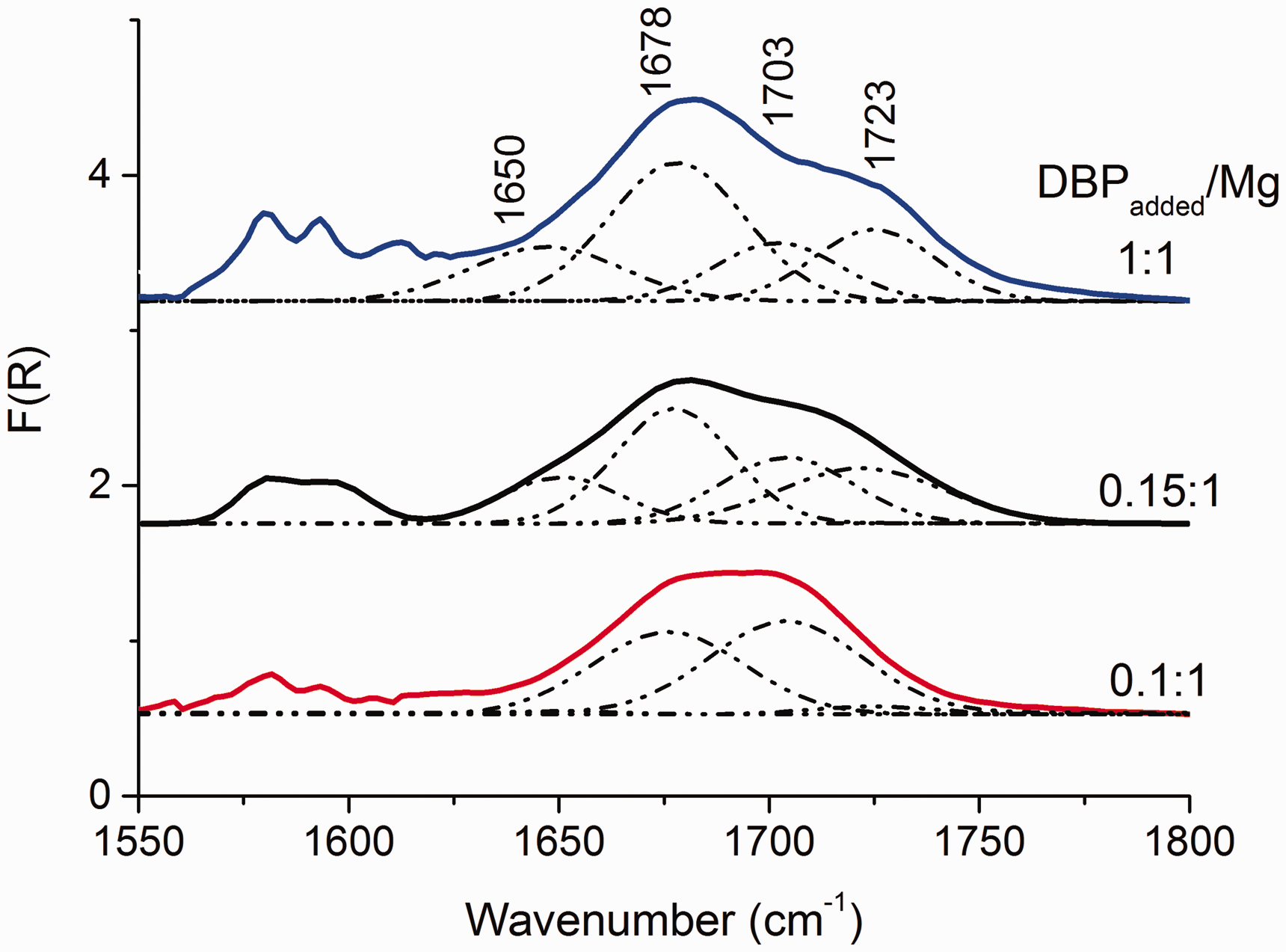
It is known that the spectral bands of the IR spectrum of MgCl2/nLB supports is a superposition of a large number of components characterizing the state of an adsorbed donor. 27 The deconvolution of the MgCl2/nDBP spectra (Fig. 2) into four components was made based on the assumption of DBP adsorption on three types of LAS and physical adsorption. It is a simplified model that allows, as a first approximation, one to estimate the surface state of an adsorbed donor. As a result of the deconvolution of the spectra into four components, the bands at 1648, 1672, 1702, and 1723 cm–1, characterizing the stretching vibrations (νС=О) of adsorbed phthalate, were revealed.
The bands attributed to DBP adsorbed onto Mg ions are shown in 1640–1670 cm–1 region from three-coordinated Mg ions, 1670–1690 cm–1 from four-coordinated Mg ions of the (110) face, 1690–1710 cm–1 from five-coordinated Mg ions of the (104) face, and the bands above 1710 cm–1 were weakly bound carbonyl groups. 28
Furthermore, three-coordinated Mg ions are stronger LAS than sites formed on four- and five-coordinated magnesium ions; therefore, the bands of molecules adsorbed on these sites should appear in the DRIFTS spectra first.5,29 However, the band at 1650 cm–1 appears later, and its intensity increases with increase in DBP concentration. This suggests that 1650 cm–1 characterizes the adsorption of DBP on other LAS, for example, on four-coordinated magnesium ions.
The formation of [MgCl2 · (LB)x]n molecular complexes during the synthesis of the MgCl2/LB support has been suggested previously.30,31 Polymeric [MgCl2 · (LB)x]n complexes were synthesized and characterized in many works. For example, MgCl2 · (LB)2, where LB is ethyl acetate, ethyl propionate, EB, ethyl crotonate, acetone or 2-butanone, ethanol, THF, [Mg2Cl4(1,3-dimethoxypropane)2(H2O)].12,32–34
The DFT calculations also show the possibility of the formation of molecular complexes upon adsorption of donors (1,3-diethers, alkoxysilanes, and succinates) on magnesium chloride. 35 In such complexes, two oxygen atoms of one or two LB are bound to a four-coordinated magnesium ion.
Since donor adsorption is insular in nature, we can hypothesize the formation of similar complexes with DBP in MgCl2/nDBP samples. However, for a site containing two adsorbed LB molecules, two absorption bands should be observed in the DRIFTS spectrum (νs and νas).
Probably, after adsorption of the second DBP molecule, the cleavage of the band at 1678 cm–1 into two bands should occur, with one of the bands being at 1650 cm–1, and the second band in the region above 1780 cm–1. However, the spectral bands in the 1730–1800 cm–1 region are complex, so it is not yet possible to isolate it.
The DFT calculations show that bidentate donors, DEP, DIBP, p-iso-propoxy ethyl benzoate, p-ethoxy ethyl benzoate, and p-tert-butyl ethyl benzoate may have interlayer (zip) coordination on the (110) MgCl2 face. 11 All this suggests the formation of molecular complexes in MgCl2/nDBP samples obtained with DBPadded/Mg >0. With high donor content, such adsorption can lead to the destruction of the structure of magnesium chloride. Thus, it can be assumed that in region I (Fig. 1), DBP adsorption occurs on the LAS with the formation of ionic complexes, and molecular complexes begin to form in region II.
MgCl2/nDBP Samples Study by Photoluminescence
The source MgCl2 was studied by photoluminescent spectroscopy. Analysis of the literature data showed that λexcitation = 278 nm can be used to measure the PL spectra of DBP.20,22 The PL spectrum of the MgCl2 sample obtained at λexcitation = 278 nm is shown in Fig. 3. The PL spectrum contains a wide band with a maximum at 488 nm and a shoulder at 421 nm. The obtained PLE spectrum for λmax = 488 nm is characterized by strong bands in the ultraviolet spectral region with maxima at 230 and 281 nm. The nature of this PL is most likely related to possible organic impurities, as well as the possible manifestation of anionic defects in the form of Cl− vacancies.
18
The kinetics of PL decay with λmax = 488 nm is described by a biexponential dependence and has lifetimes of 0.17 and 2.3 ms typical for phosphorescence (τ1 and τ2).
(a) PL (1) and PLE (2) spectra and (b) decay kinetics of MgCl2.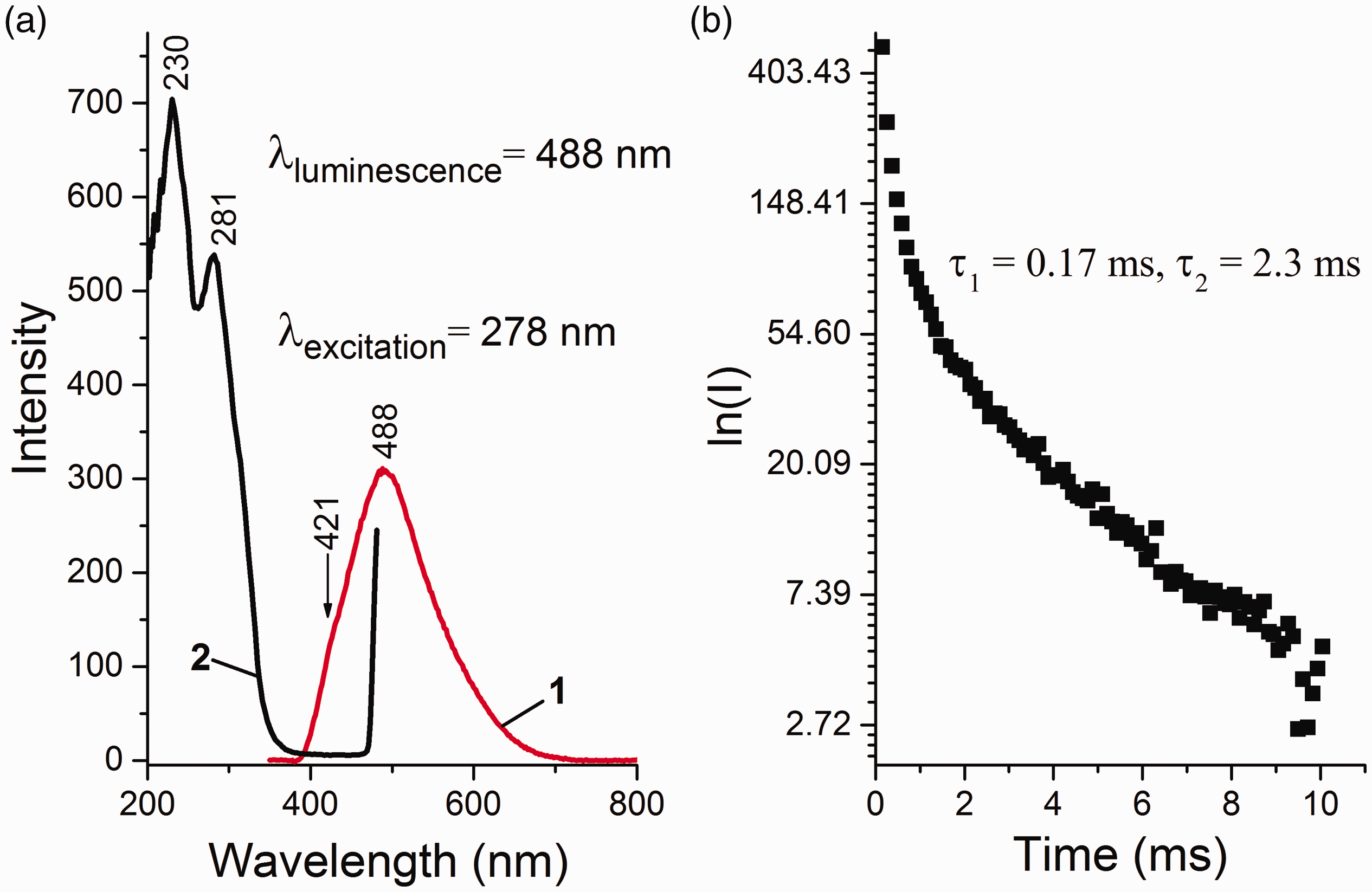
The PL spectrum of the DBP heptane solution contains the bands 371, 414, 436, and 470 nm (Fig. S1, Supplemental Material). Since luminescence decay occurs at a high rate, we can conclude that they characterize S1* → S0, i.e., only fluorescence is observed in a DBP solution at room temperature.
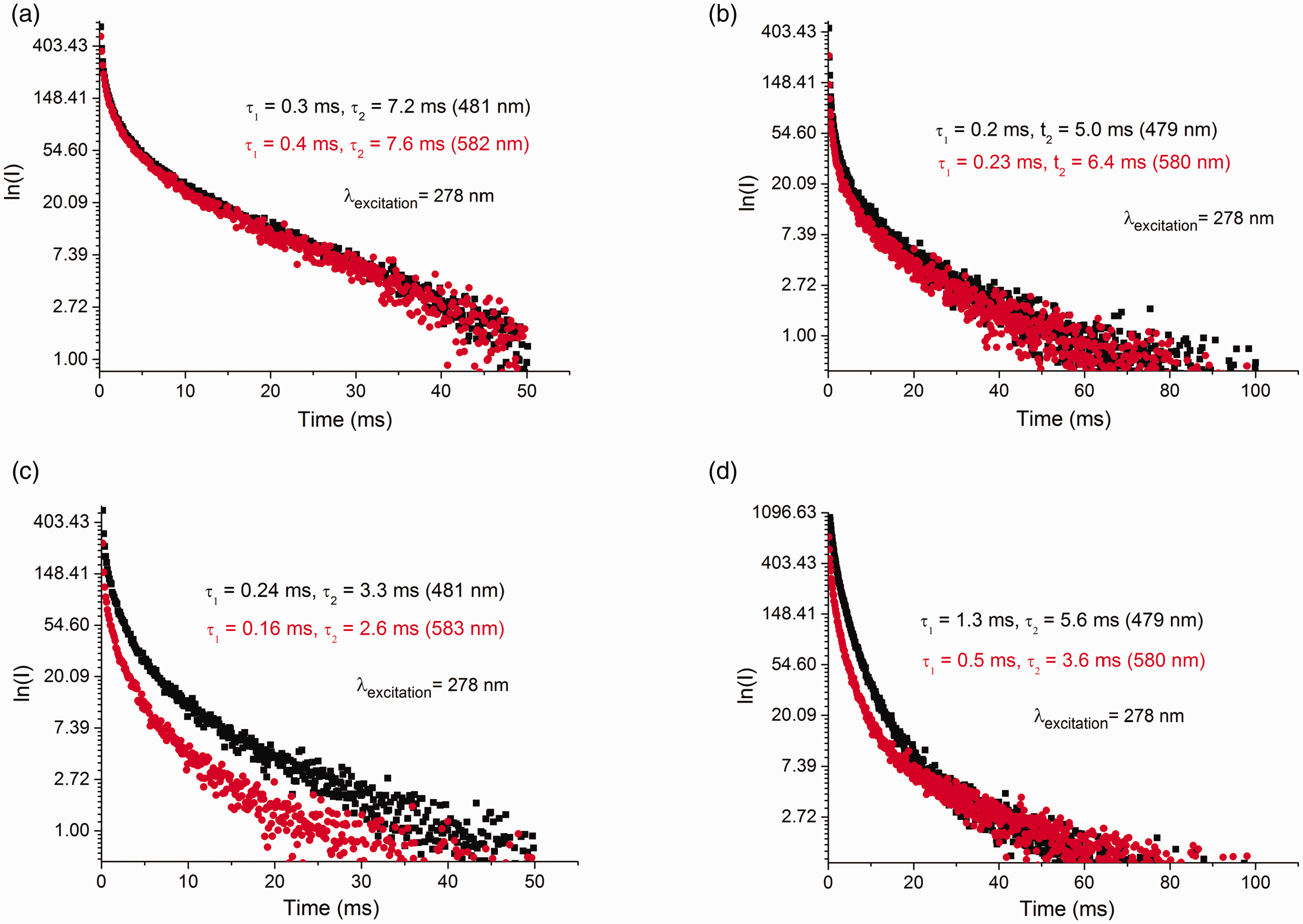
The PL spectra of MgCl2/nDBP samples with n = 0.1, 0.15, 0.4, and 1.0 are shown in Fig. 4 and Fig. S2. The spectra of all samples contain two bands with maxima near 479–482 and 579–583 nm. The decay time (τ) of these bands is shown in Fig. 5. The decay time is in the millisecond interval that is characteristic of phosphorescence. The values of τ for the samples are close to each other and significantly differ from τ for MgCl2. This indicates that the PL sites with maxima of 479–482, 579–583, and 620 nm are of a different nature from that of MgCl2 and characterize adsorbed DBP. A decrease of τ with a rise in DBPadded/Mg indicates an increase in the concentration of “quenching” atoms, i.e., the amount of DBP adsorbed on the support surface. The decay kinetics of all the above bands are described by their biexponential dependence. τ depends on many parameters: on energy relaxation processes and on external conditions (e.g., temperature, pressure, concentration of luminescent atoms and molecules, concentration of “quenching” atoms and molecules, etc.). It can be assumed that the biexponential dependence τ is associated with the existence of various defects on the surface of magnesium chloride that act as electron “traps” upon excitation.
PL (1) and PLE (2) spectra of samples at λexcitation = 278 nm: (a) MgCl2/0.15DBP, (b) MgCl2/0.4DBP, (c) MgCl2/0.15DBP/TiCl4, and (d) MgCl2/0.4DBP/TiCl4. Decay kinetics for samples at λexcitation = 278 nm: (a) MgCl2/0.15DBP, (b) MgCl2/0.4DBP, (c) MgCl2/0.15DBP/TiCl4, and (d) MgCl2/0.4DBP/TiCl4.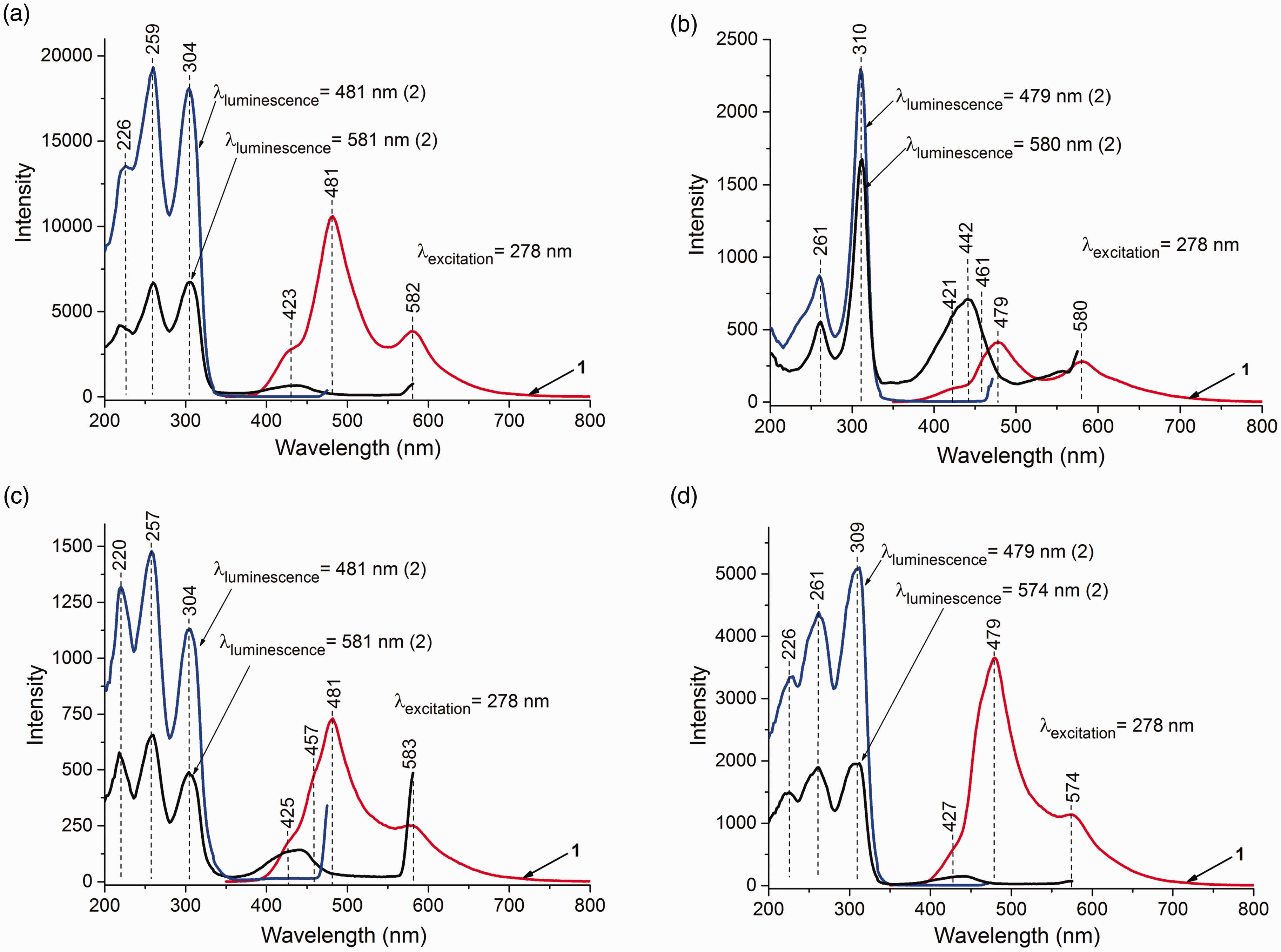
In the PLE spectra of the samples, bands similar in structure with maxima at 259 (261) and 301 (310) nm, characterizing DBP, are observed. The PLE spectra of the samples were similar in their positions to the DR ultraviolet–visible spectra (Fig. S3). The band at 259 (261) nm characterizes π–π transitions of the aromatic ring, and 301 (310) nm characterizes n–π transitions of the carbonyl group. Higher values of the band characterizing the n–π transitions in MgCl2/nDBP samples compared to the DBP solution (290 nm) may indicate the interaction of DBP carbonyl groups with the LAS of the support with the formation of D+A– donor–acceptor surface complexes. 36 An increase in the donor content in the samples resulted in a bathochromic shift of the band in the PLE spectrum which characterized the n–π transition of the carbonyl group. This indicated a weakening of the bond between the carbonyl group of the donor and the LAS support. The intensity ratio Iπ–π/ In–π decreases from 1.3 to 0.2. This shows that the number of D+A– ion pairs is reduced. This was probably due to a change in the structure of magnesium chloride and the formation of polymer molecular complexes.
Thus, analysis of the PL and PLE spectra of MgCl2/nDBP samples indicated that the bands at 479–482 and 579–583 nm are similar in nature and characterized ionic complexes of adsorbed DBP with the transition energy of T1* → S0 equal to 2.82 and 2.34 eV, respectively. Surface D+A– complexes characterized by a band at 479–482 nm are stronger than the complexes characterized by a band at 579–583 nm.
The presence of two bands in the PL spectra of MgCl2/nDBP samples indicated the formation of two different D+A– complexes of DBP. Since there are four and five coordinatively unsaturated magnesium ions on the surface of magnesium chloride, it could be assumed that the appearance of two bands in the PL spectrum is associated with this. On the other hand, the long-wavelength shift of the bands in the PL spectra of these samples can be associated with interactions with neighboring molecules, for example, chlorine atoms or OH groups, always present in magnesium chloride due to the trace amounts of water.25,37
Study of the Interaction of MgCl2/nDBP with TiCl4
It has been shown that the polymer [MgCl2 · (LB)x]n molecular complexes with n = 2, 3 are destroyed by interaction with TiCl4 or thermal activation with the formation of δ–MgCl2.13,38 Therefore, for a more accurate identification of molecular complexes, the interaction of MgCl2/0.15DBP samples (region I) and MgCl2/0.4DBP (region II) with a TiCl4 solution at Ti/Mg = 100 was studied.
The chemical analysis is summarized in Table I. The application of TiCl4 leads to a decrease in the donor content by 49.50–54.15%. The number of magnesium atoms in these samples per one DBP molecule increases from 25 to 50 and from 6.6 to 17, respectively. The TiCl4 content in the samples is small (0.9 wt.%), and it differs from the amount of desorbed DBP. In the MgCl2/0.15DBP/TiCl4 sample, the amount of adsorbed titanium tetrachloride is 30% more than the amount of removed donor, while in the MgCl2/0.4DBP/TiCl4 sample, the amount of adsorbed TiCl4 is three times higher than that of desorbed DBP.
Figure 6 shows the DRIFTS spectra of MgCl2/0.15DBP and MgCl2/0.4DBP samples before and after treatment with a TiCl4 solution. The deposition of TiCl4 on the supports leads to the spectrum shape change. In the DRIFTS spectrum of the MgCl2/0.15DBP/TiCl4 sample, the relative intensity of bands at 1650 and 1678 cm–1 decreases and the band at 1723 cm–1 disappears (Fig. 6a). In the spectra of the MgCl2/0.4DBP/TiCl4 sample, a decrease in the relative intensity of bands at 1650, 1678, and 1723 cm–1 is observed (Fig. 6b). In addition, new bands at 1640, 1660, and 1715 cm–1 appear. A sharp decrease in the relative intensity of bands at 1678 and 1723 cm–1 indicates the removal of DBP adsorbed on four-coordinated Mg ions.
DRIFTS spectra of (1) MgCl2/nDBP supports and (2) corresponding MgCl2/nDBP/TiCl4 catalysts: (a) n = 0.15 and (b) n = 0.4.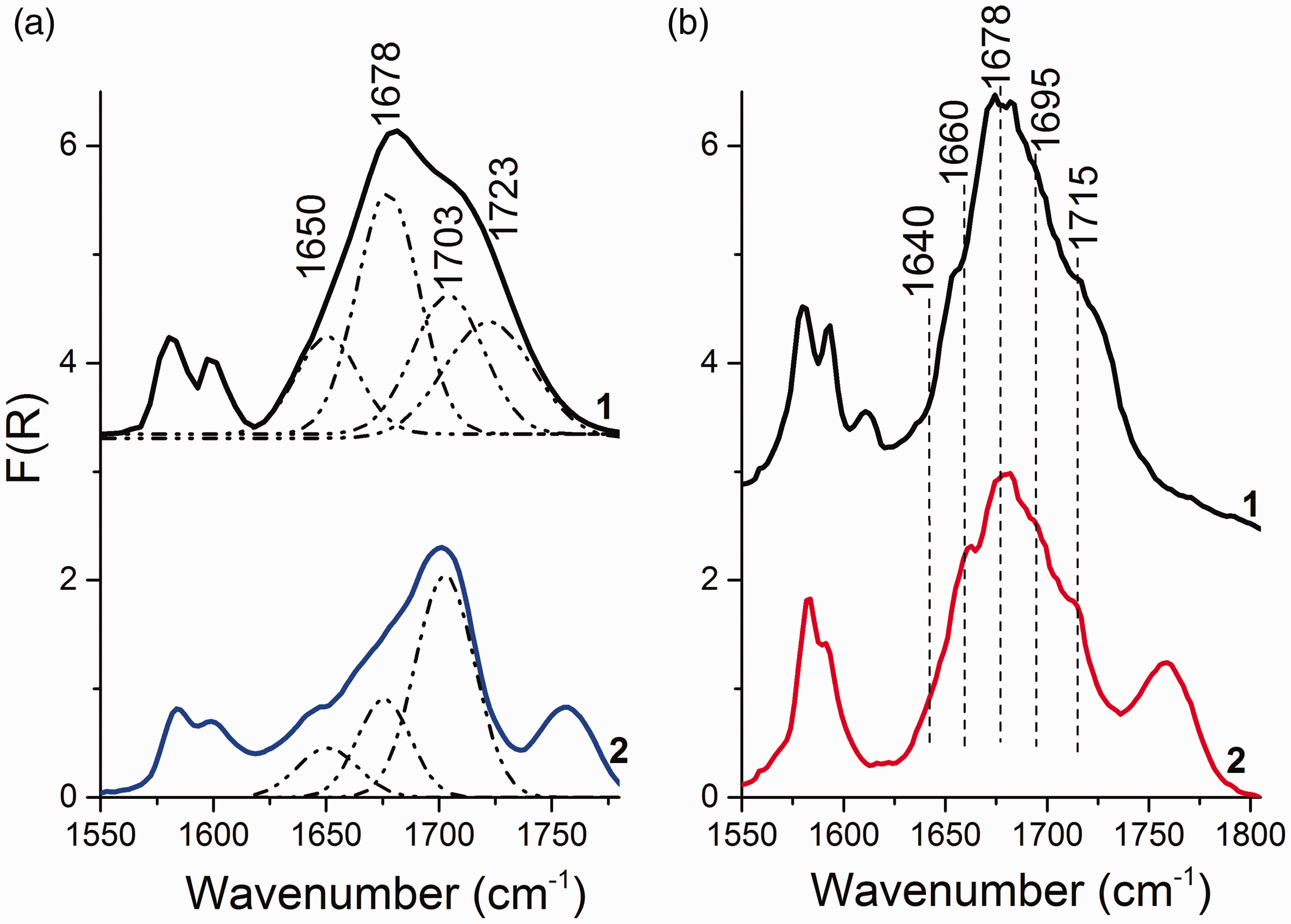
Figure 4 shows the PL spectra of the studied catalysts. Two bands with maxima at 480–482 and 580–582 nm can also be distinguished in the spectra. The PL spectra of the different samples were very similar.
It is noteworthy that the decrease in DBP concentration (Fig. 4) in the MgCl2/0.15DBP/TiCl4 sample leads t a decrease in PL band intensity (12 000 → 750 for λ481) and to an increase in that for MgCl2/0.4DBP sample (400 → 3800 for λ481). This shows that the number of D+A– ion complexes in the MgCl2/0.15DBP/TiCl4 sample decreases. The number of D+A– ion complexes was found to increase for the MgCl2/0.4DBP/TiCl4 sample, probably as a result of an increase in the number of defects in the MgCl2 support.
Numerous XRD studies of [MgCl2 · (LB)x]n molecular complexes with n = 2–3 containing monodentate donors (e.g., COOC2H5, CH3COOC2H5, C2H5OH) show that such complexes have well-ordered crystalline structure with donors in axial position.12,14,15 However, chemical or temperature activation removes most of LB, which leads to the destruction of the complexes structure. In this case, δ-MgCl2 is formed. Our data show that the treatment of the MgCl2/0.4DBP support with a TiCl4 solution results in increase in the number of LAS capable of adsorbing DBP with the formation of D+A– ion complexes and this was probably due to the destruction of molecular complexes.
The phosphorescence lifetime τ of the observed bands at 482 and 582 nm also changes when TiCl4 is deposited on MgCl2/0.15DBP and MgCl2/1.0DBP supports. The values of τ1 and τ2 decrease. The reason for this may be an increase in the rate of nonradiative conversion of the absorbed radiation energy and the rate of quenching of the triplet state by “impurities” which are adsorbed TiCl4 molecules. This suggested that the adsorbed TiCl4 molecule was located in the first coordination sphere of DBP, i.e., it is a part of the precursor of the active site.
Conclusion
The interaction of DBP with MgCl2 was studied by PL spectroscopy in combination with DRIFTS. DRIFTS showed how the DBP interacted with the LAS of the magnesium chloride support. PL spectroscopy revealed the formation of both D+A– donor–acceptor complexes and molecular complexes on the surface of magnesium chloride. Part of the ionic D+A– complexes probably interacted with neighboring surface Cl− or OH− groups. Their amount depended on the DBPadded/Mg molar ratio used for the synthesis of the support. Molecular complexes began to form in MgCl2/nDBP samples at DBPadded/Mg >0.1.
Photoluminescence spectroscopy revealed that treatment of MgCl2/nDBP supports with a TiCl4 not only led to the removal of the donor from the surface of magnesium chloride and to the formation of new LAS capable of adsorbing titanium or an organoaluminum activator, but also resulted in the destruction of molecular complexes. This method allowed us to explain why the synthesis of the TMC for olefin polymerization is preferable to perform at a molar ratio of DBP/Mgadded below 0.2.
Supplemental Material
sj-pdf-1-asp-10.1177_0003702820927434 - Supplemental material for New Insight into Titanium–Magnesium Ziegler–Natta Catalysts Using Photoluminescence Spectroscopy
Supplemental material, sj-pdf-1-asp-10.1177_0003702820927434 for New Insight into Titanium–Magnesium Ziegler–Natta Catalysts Using Photoluminescence Spectroscopy by Valentina N. Panchenko, Anton I. Kostyukov, Anton Yu Shabalin, Evgeniy A. Paukshtis, Tatiana S. Glazneva and Sergei G. Kazarian in Applied Spectroscopy
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation, grant number 075-15-2019-1876.
ORCID iDs
Anton Y. Shabalin https://orcid.org/0000-0002-3706-1291 Tatiana S. Glazneva https://orcid.org/0000-0001-8366-9698 Sergei G. Kazarian ![]()
Supplemental Material
The supplemental material mentioned in the text, consisting of Figs. S1 to S3, is available in the online version of the journal.
