Abstract
Objective:
Despite vaccination and early antibiotic treatment, pneumococcal meningitis remains a disease with significant mortality and morbidity. The resulting inflammatory response can lead to cochlear fibrosis, ossification where cochlear implant surgeries are far challenging. Our study aimed to investigate the preventive effect of controlled-release dexamethasone implant in such cases in terms of structural integrity.
Methods:
Twenty-four rats were induced with pneumococcal meningitis and randomized into study (n = 16) and control (n = 8) groups. Controlled-release dexamethasone implants were placed transbullarly into the right round window of the study group. Bilateral cochleas underwent histological examination 3 months post-infection.
Results:
In the study, cochlear effects of pneumococcal meningitis were evaluated. The basal turn was significantly more affected by fibrosis and ossification (P = .013 and .010, respectively). Compared with control ears, the dexamethasone implant group showed less fibrosis in all turns and less ossification in the basal turn (P = .014, .003, .044, and .035, respectively).
Conclusion:
In pneumococcal meningitis, fibrosis and ossification occur more intensively in the basal turn of the cochlea. Controlled-release dexamethasone implants are effective in preventing cochlear ossification and fibrosis. The prevention from the structural damage indicates the potential role of these dexamethasone implants in post-meningitic hearing loss and easing cochlear implant surgeries.
Keywords
Introduction
The inner ear comprises the cochlea which is crucial for hearing and the vestibular system which is essential for maintaining balance. The organ of Corti where auditory transduction occurs is located within the scala media. The organ of Corti is composed of specialized sensory cells which convert sound vibrations into electrical signals that are interpretable by the brain. These sensory cells are supported by the tectorial membrane and supporting cells. In case of pneumococcal meningitis, a bacterial infection can infiltrate the inner ear, leading to inflammation and subsequent structural damage. This inflammation can result in cochlear sclerosis, fibrosis, and disruption of structures associated with the organ of Corti, potentially resulting in permanent hearing loss.1 -5
Pneumococcal meningitis is a bacterial infection that affects the brain and spinal cord, a potentially life-threatening condition. Among survivors, permanent hearing loss is one of the most common neurological sequelae.2,5 Findings suggest that treatment modalities developed by adding glucocorticosteroids, which reduce the expression of inflammatory cytokines in meningitis treatment, may be effective against hearing loss and neurological sequelae. 6 Recent studies indicate that adjunctive use of dexamethasone in children with pneumococcal meningitis can significantly reduce the incidence and severity of hearing loss. 7 According to meta-analyses of randomized controlled trials, early administration of dexamethasone provides significant protection against severe hearing loss in children with pneumococcal meningitis. 8 This finding is supported by guidelines from the Infectious Diseases Societies which recommend adjunctive dexamethasone as standard therapy for suspected or confirmed pneumococcal meningitis patients. 9
Dexamethasone can be administered orally, intravenously, or by intratympanic injection. Systemic use can lead to adverse effects such as hypertension, hyperglycemia, avascular necrosis of bones, and susceptibility to infections. However, short-term systemic use has a low side effect profile, though potential side effects cannot be ignored. Intratympanic applications have been developed to avoid off-target effects and to utilize lower doses with higher efficiency.10 -12 For this purpose, intratympanic dexamethasone is used as a targeted delivery method in Meniere’s disease, idiopathic sudden hearing loss, and other inner ear disorders.
A safety study of intratympanic administration demonstrated that 5 intratympanic injections of 4 mg/mL dexamethasone were safe and had no adverse effects on outer hair cell function. 13 Another study on rats found that intratympanic dexamethasone administration provided significant protection against hearing loss both histopathologically and audiologically. 14 Additionally, a study on patients with sensorineural hearing loss confirmed by audiometry suggested that transtympanic dexamethasone injections could be used to prevent systemic side effects by providing a higher drug concentration locally in the inner ear. 3 However, the duration of dexamethasone therapy poses the most significant limitation to its effectiveness in preventing hearing loss, and this is the major challenge in intratympanic injections. Due to its liquid form, it can drain through the Eustachian tube, making the steroid effect insufficient, and precise dose and duration control of intratympanic steroid therapy cannot be achieved.12,15 To overcome this problem, several solutions have been proposed in the literature. Among these, placing dexamethasone in gel or powder forms into the middle ear through a tympanostomy tube has been suggested.4,7,16 These methods have shown to be effective in retaining the drug within the inner ear, reducing systemic exposure, and enhancing the effectiveness of treatment. However, they are not sufficient in cases requiring long-term treatment. For this purpose, controlled-release dexamethasone implants are being investigated as an alternative intratympanic application and hold promise since they last 5 to 6 months.15,17,18
In clinical practice, a biodegradable dexamethasone implant(Ozurdex®, Allergan Inc., Irvine, CA, United States) is used to manage diabetic macular edema. This implant, Approved by the FDA, provides sustained concentrations of dexamethasone in the vitreous of the eye for up to 6 months. 17 Therefore, its applicability has been considered as a potential prophylactic measure against cochlear structural damage resulting from pneumococcal meningitis.15,17 In this present study, we aimed to investigate the preventive efficacy of a poly(D, L-lactic-co-glycolic acid) (PLGA)-based drug delivery system in meningitis-induced inner ear damage.
Materials and Methods
The study was conducted after obtaining approval from the Necmettin Erbakan University Animal Experiments Local Ethics Committee (Approval no: 2021-012), and the entire study process was carried out following the Principles of Care and Use of Laboratory Animals of the National Institutes of Health. Experimental animals were obtained from the KONUDAM Experimental Medicine Application and Research Center. The study design was based on the power analysis conducted using G*Power software and was planned to account for the expected loss rate due to lethal meningitis. A total of 24 rats with weights ranging from 200 to 250 g with normal tympanic membranes were acquired for the study.
The animals were randomly divided into 2 groups: the study group (n = 16) and the control group (n = 8). To induce the meningitis model as described in the literature, all animals underwent intracisternal administration of 30 µL suspension of Streptococcus pneumoniae type 3 bacteria using a 25-G needle. Pneumococcal meningitis was confirmed by quantitative cultures of cerebrospinal fluid, 18 hours after infection when the animals typically became symptomatic. However, 6 animals from the study group and 2 animals from the control group died within the first 24 hours post-procedure. The surviving animals received a 3-day treatment of 125 mg/kg ceftriaxone daily, starting from the first day of inoculation.
On the first day after bacterial inoculation, a 0.46 mm × 3 mm controlled-release dexamethasone implant (Ozurdex®, Allergan Inc., Irvine, CA, United States) implant was transbullarly placed into the right round window of the study group animals. The hole in the bulla was then sealed with bone wax. In the control group, a transbullar approach was used to administer a saline injection into the right round window, followed by sealing the bulla hole with bone wax. No procedures were performed on the left ears, which were considered as untreated control ears (Figure 1). All animals were euthanized 3 months after bacterial inoculation, by intraperitoneal administration of sodium thiopental (Pentothal, Abbott, U.S.A, 100 mg/kg) under deep anesthesia and cochleae’s were harvested bilaterally for histological examination.
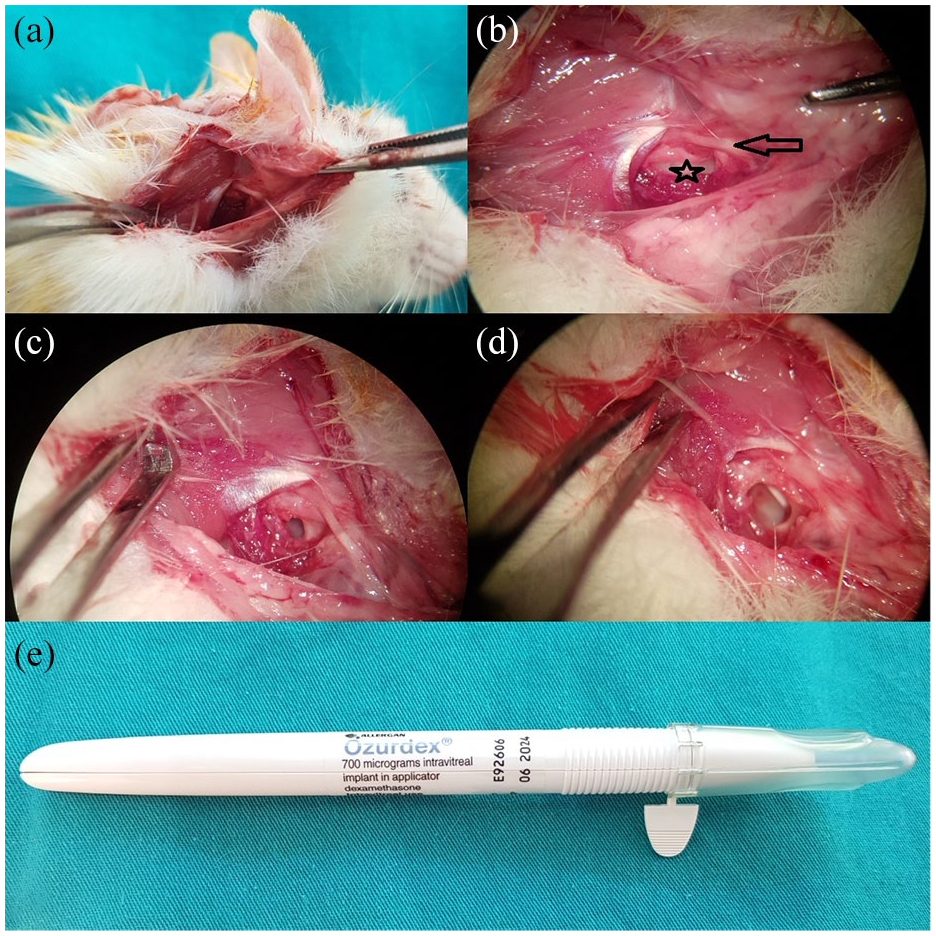
Transbullar controlled-release dexamethasone implant application in the experimental rat model. (a) Macroscopic photograph of the right retroauricular approach. (b) Microscopic photograph of the right retroauricular approach showing the bulla (star) and facial nerve (arrow). (c) Creation of an opening on the bulla for controlled-release dexamethasone implant. (d) Application of controlled-release dexamethasone implant onto the round window. (e) controlled-release dexamethasone implant applicator.
Tissues fixed with 10% neutral buffered formalin were decalcified with 4% ethylenediaminetetraacetic acid (EDTA) solution for histopathological examination. Following decalcification, tissues were processed through routine tissue tracking and paraffin embedding, and multiple sections with an average thickness of 4 µm and 5 to 7 sections per turn were obtained using a microtome. Sections were then prepared for microscopic evaluation by applying Hematoxylin/Eosin and histochemical Masson Trichrome staining. Histological changes that were observed were recorded, and microscopic images were captured using a digital camera (Olympus SC50, Tokyo, Japan) attached to a light microscope (Olympus BX53, Tokyo; Figures 2–4).
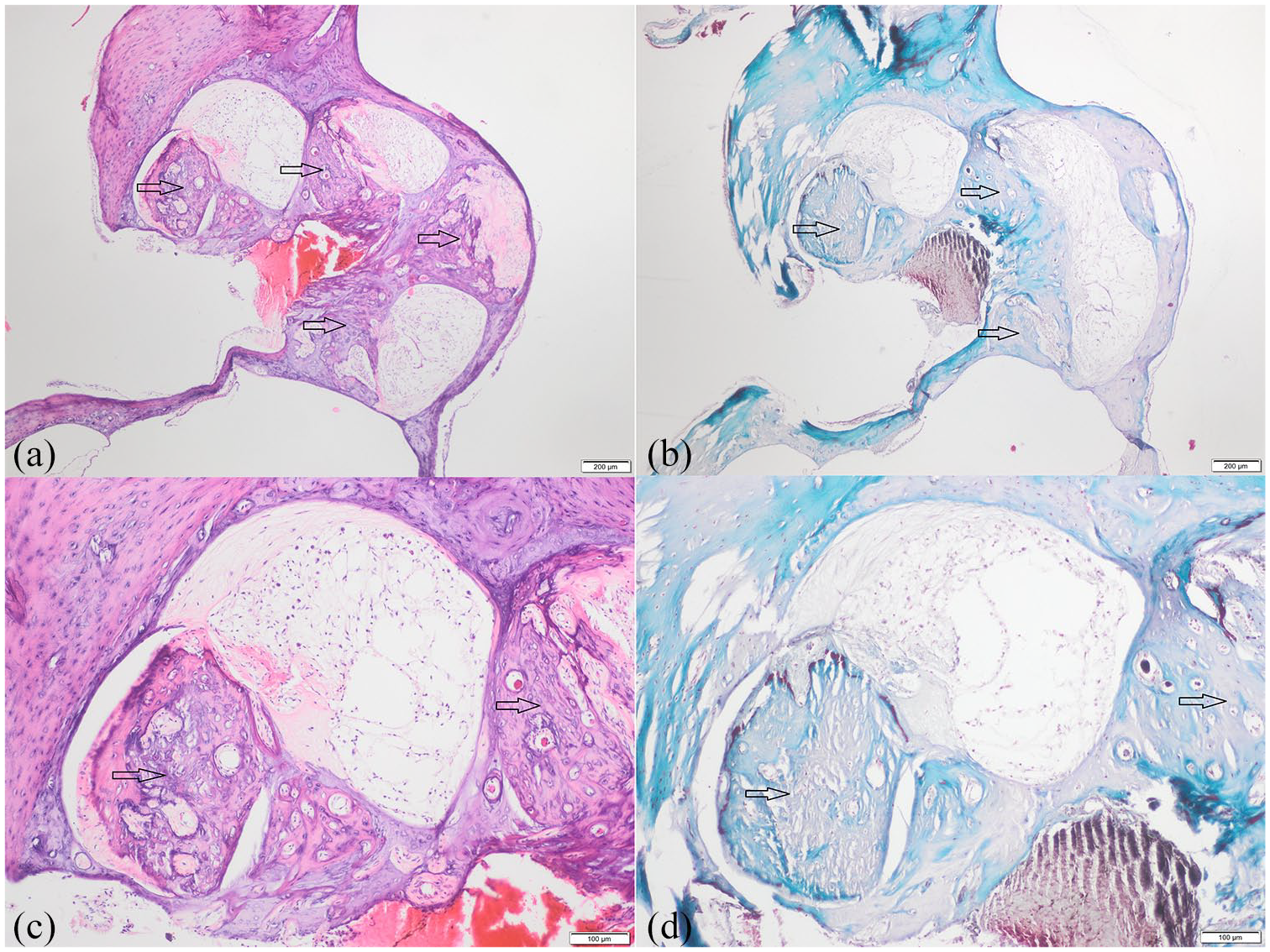
Example of cochlea specimen in the control ear group where ossification in the basal and middle turns completely obliterates the scala tympani (a) Hematoxylin/Eosin, 40× magnification, scale bar = 200 µm; (b) Masson’s Trichrome, 40× magnification, scale bar = 200 µm; (c) Hematoxylin/Eosin, 100× magnification, scale bar = 100 µm; (d) Masson’s trichrome, 100× magnification, scale bar = 100 µm).
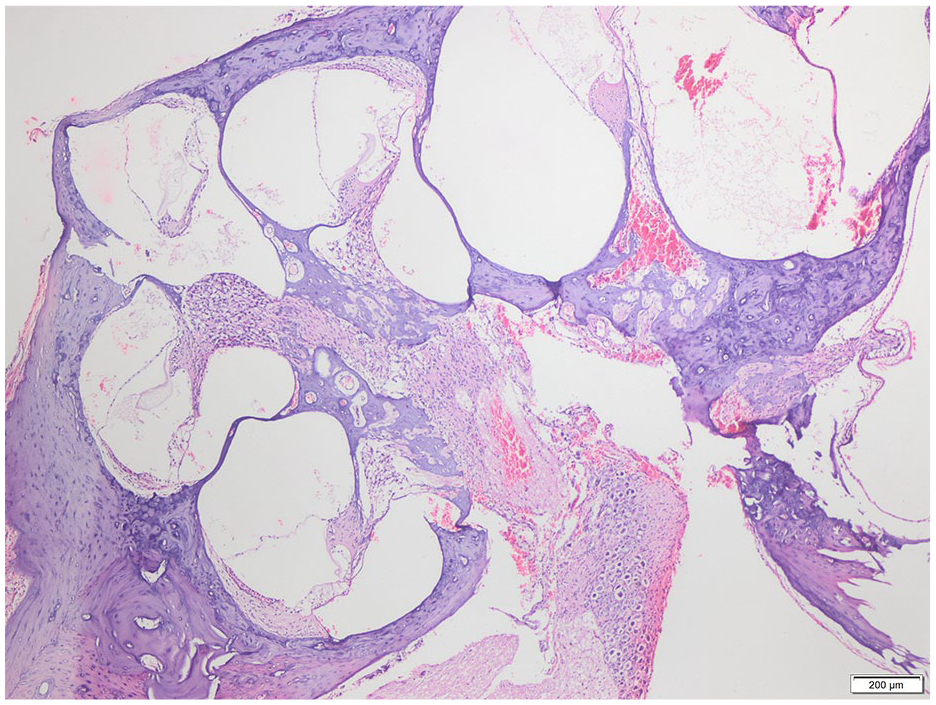
Histological demonstration of all turns of cochlea in one of the rats in the controlled-release dexamethasone group. Histological structure is largely preserved (Hematoxylin/Eosin, 40× magnification, scale bar = 200 µm).
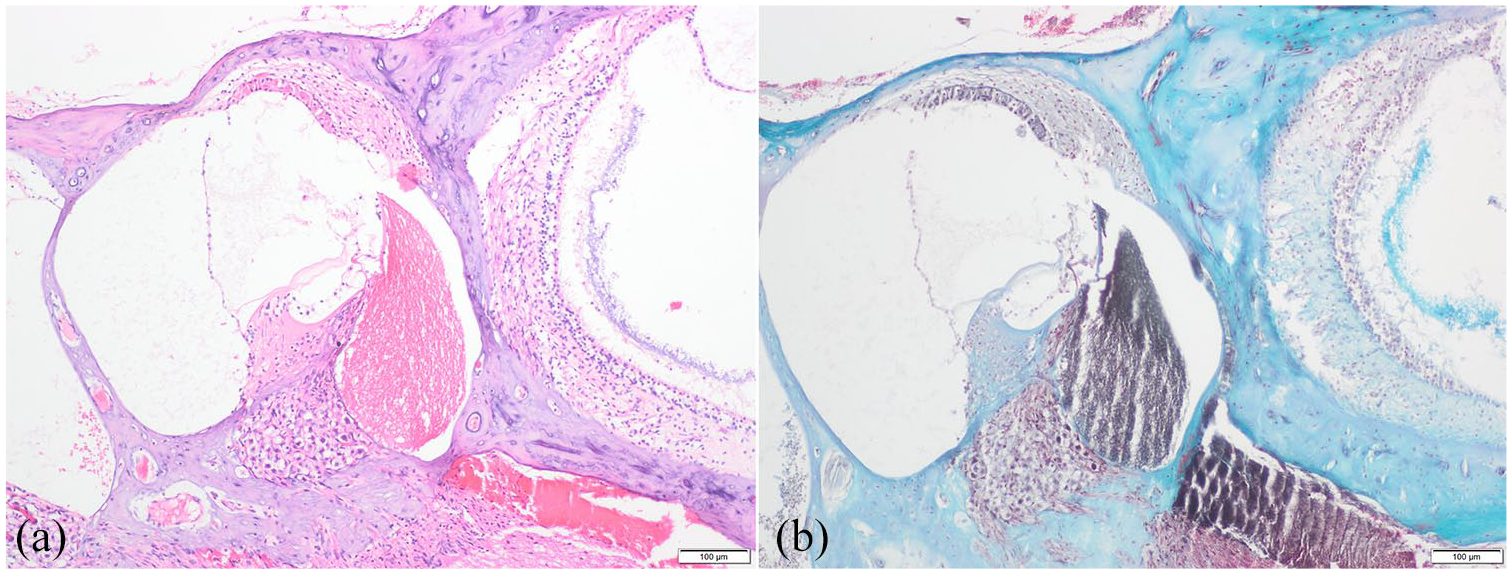
Cochlear specimen in the controlled-release dexamethasone group showing increased cellularity in the stria vascularis (a) Hematoxylin/Eosin, 100× magnification, scale bar = 100 µm; (b) Masson’s Trichrome, 100× magnification, scale bar = 100 µm).
Histologically, the basal, mid, and apical turn regions of the cochlea were identified, and for each turn, the structural damage level of the organ of Corti (hair cell condition, structural integrity, surrounding basilar membrane, and tectorial membrane), stria vascularis (cell density and morphology), spiral ligament (cell density and morphology), spiral ganglion; and the severity of fibrosis, ossification, and inflammation were evaluated. Each examination was categorized as normal, mild, moderate, or severe. We used the same percentage categories as the referenced study: 75% to 100% for severe, 25% to 75% for moderate, <25% for mild, and none for normal. 19 Two experienced pathologists evaluated all slides. Each pathologist examined the slides and recorded their findings independently. In cases of disagreement between the pathologists, the final decision was made through mutual discussion and consensus.
Statistical analysis was performed using SPSS software (version 22.0; SSPS, Inc., Chicago, IL). Pearson’s chi-square test was used to compare categorical parameters (structural damage of the organ of Corti, stria vascularis, spiral ganglion; presence of inflammation, fibrosis, and ossification) between groups. A P-value of <.05 was considered statistically significant in all statistical analyses.
Results
A total of 24 rats were included in the study, 8 (6 animals from the study group and 2 animals from the control group) were lost within the first 24 hours following intracisternal pneumococcal application. The left ears of the remaining 16 rats were compared in terms of the effects of pneumococcal meningitis on the cochlea, categorized into basal, middle, and apical turns, and these findings are presented in Table 1. Similar findings were observed in terms of spiral ganglion, the organ of Corti, stria vascularis damage, and inflammation across turns (P > .05). However, in the control group (n = 6) and the contralateral ears of the study group (n = 10), it was determined that fibrosis and ossification were more severe in the basal turn (P = .013 and .010, respectively).
Structural Damage of the Organ of Corti, Stria Vascularis, Spiral Ganglion; and Presence of Inflammation, Fibrosis, and Ossification in the Basal, Middle, and Apical Turns of the Left Cochlear Structures After Pneumococcal Meningitis.

Chi-square test.
The ears treated with controlled-release dexamethasone implants in the study group (n = 10) were compared with the contralateral ears in the control group to assess the effect of dexamethasone implants and these are summarized in Table 2. In the comparison between the right and left ears, similar degrees of impairment were found in terms of the organ of Corti, stria vascularis, spiral ganglion damage, and inflammation (P > .05); however, fibrosis was found to be less in all turns (basal, middle, and apical), and ossification was less in the basal turn in the dexamethasone implant group (P = .014, .003, .044, and .035, respectively). In the comparison between the dexamethasone implant and saline groups, similar degrees of damage were observed in terms of the organ of Corti, stria vascularis, spiral ganglion damage, and presence of inflammation (P > .05); however, fibrosis was less in all turns (basal, middle, and apical), and ossification was less in the basal turn in the dexamethasone implant group (P = .037, .001, .047, and .034, respectively).
Structural Damage of the Organ of Corti, Stria Vascularis, Spiral Ganglion; and Presence of Inflammation, Fibrosis, and Ossification in the basal, Middle, and Apical Turns of the Cochlear Structures in the Controlled-release Dexamethasone Group (Right Ear Receiving Controlled Release Dexamethasone Implantation, Left Ear Undergoing No Intervention) Following Pneumococcal Meningitis.

Chi-square test
Discussion
Pneumococcal meningitis, caused by Streptococcus pneumoniae, remains a disease with significant mortality and morbidity despite widespread vaccination and prompt initiation of antibacterial therapy upon early diagnosis. Hearing loss (HL) is a common sequel of this disease, affecting 5% to 35% of individuals, with bilateral involvement in 37% of cases. 2 In the literature, the hypothesis of developing suppurative labyrinthitis has been proposed due to meningitis reaching the inner ear as the cause of hearing loss. In an experimental meningitis model, serofibrinous exudate was observed in perilymphatic spaces after infection, followed by the development of labyrinthitis ossificans at later stages, with the basal turn being the most affected region. 20 Similarly, Trakimas et al 21 reported fibrosis and gradual ossification in temporal bones after meningitis, even 1.5 weeks post-infection. Moreover, ossification was predominantly observed in the scala tympani of the basal turn and the round window niche. Consistent with the literature, our study also revealed that the basal turn was the most affected turn in terms of ossification and fibrosis.
Cochlear implantation procedures can be performed immediately in individuals with post-meningitic hearing loss to provide auditory rehabilitation. Considering the necessity of having a minimum of 3500 spiral ganglion cells for successful cochlear implantation, it is noteworthy that some studies have highlighted the loss of spiral ganglion cells due to meningitis.21,22 It has been suggested that acute bacterial meningitis may lead to the involvement of spiral ganglia through the cochlear aqueduct or cochlear nerve via subdural spread. In contrast to previous studies in the literature, we did not observe differences among cochlear turns in terms of spiral ganglion loss. In post-meningitic cochlear implantation, fibrosis and ossification pose challenges for electrode insertion. If it’s in the early grades, scala tympani or scala vestibuli insertions are possible. In some cases to address this challenge drill-out surgeries and the use of rigid electrodes are suggested. However, outcomes of post-menengitic cochlear implantation users have been inferior to those with a normal patent cochlear canal. 23 Therefore, inhibiting the inflammatory response in the inner ear during meningitis is crucial for preventing post-meningitic hearing loss and labyrinthitis ossificans.
The literature suggests that anti-inflammatory therapy in pneumococcal meningitis, in addition to antibiotic treatment, is effective in preventing cochlear inflammation. 1 Previous studies have shown that concomitant intratympanic steroid and antibiotic administration, contributed to cochlear function and preserved spiral ganglion integrity. 14 Similarly, Rappaport et al 24 found that anti-inflammatory agents such as dexamethasone and ketorolac minimized cochlear damage in a rabbit model when used alongside antibiotics. Although clinical studies report conflicting results, a meta-analysis emphasizes the importance of the timing of steroid therapy in preventing hearing loss in pneumococcal meningitis. 8 This efficacy is attributed to the reduction of proinflammatory cytokines and inhibition of reactive oxygen radical production during the early stages of inflammation. 6 Moreover, evidence suggests the presence of corticosteroid receptors in the human cochlea. Nevertheless, the precise mechanism of corticosteroids in the inner ear remains elusive.14,25
Corticosteroids are the main medication for reversing hearing loss in various indications, such as idiopathic sudden hearing loss.8,14 Due to the variable effects of corticosteroids on the inner ear, studies have focused on local applications as salvage therapy for cases resistant to systemic treatment. 3 While local applications offer the advantage of reaching high doses with a low side effect profile, they may require repeated doses and have disadvantages such as pain, transient dizziness, and tympanic membrane perforation. 26 Proposed solutions include high-dose single applications and the use of controlled-release biodegradable polymers (gels or solid implants).15,17,18,26 Our study focused on evaluating the efficacy of controlled-release dexamethasone implants in post-meningitic inner ears.
Controlled-release dexamethasone implants, approved for intravitreal use, consist of sterile rods containing poly(
Our study histopathologically demonstrates the intracochlear effects of meningitis. Consistent with the literature, fibrosis and ossification were more severe in the basal turn of the cochlea and dexamethasone implants were considered for their prevention. Although controlled-released dexamethasone implants were effective in preventing fibrosis and ossification, there was not any effect on spiral ganglion loss prevention. However, further research is needed to investigate loading higher doses of dexamethasone into biodegradable polymers.
When assessing the strengths of our study, it is the first article to evaluate the use of controlled-release dexamethasone implants for preventing structural damage in cochlea caused by meningitis. It demonstrated that these implants have a preventive effect on fibrosis and ossification, the most significant structural changes complicating cochlear implant surgeries. This significant result provides important support for potential clinical use in post-meningitic patients and might give more time for cases before implantation rather than an otologic emergency. However, the limitations of the study include the absence of functional audiometric assessments in the animals. The inability of CR-Dex to prevent spiral ganglion neuron (SGN) loss limits its efficacy in preventing hearing loss and suggests that post-meningitic patients may experience reduced cochlear implant efficiency compared to individuals without meningitis. Nevertheless, the primary objective of this study was to prevent structural damage to the cochlea, which could facilitate cochlear implant surgery following meningitis. Additionally, combining CR-Dex with systemic steroids, the mainstay treatment for hearing loss after meningitis, may enhance treatment effectiveness. Another comparison that should be performed is to evaluate systemic steroids (oral or IV/IP in an animal) against local delivery. Therefore, future studies incorporating audiometric tests and a systemic steroid group are anticipated to better address the functional aspects of treatment and provide valuable insights. This could be the subject of future studies.
Conclusion
In cases of acute pneumococcal meningitis, fibrosis and ossification tend to occur more intensely in the basal turn of the cochlea. Controlled-release dexamethasone implants have shown to be effective in preventing the development of fibrosis and ossification. This indicates the potential role of these implants in post-meningitic hearing loss and cochlear implant surgeries.
Footnotes
Acknowledgements
The first author thanks to Prof. Dr. Metin Yılmaz, for his contribution to the design of the study through the advice given in respect of researching Ozurdex in the inner ear, during the residency at Gazi University ENT Department.
Author’s Note
This study was presented at the 44th National Otorhinolaryngology Congress as an oral presentation, Antalya/Turkey, November 15 to 19th, 2023.
Authors Contributions
Idea/Concept: Fakih Cihat Eravci. Design: Fahriye Kilinç, Metin Doğan, and Ömer Kaplan. Control/Supervision: Fakih Cihat Eravci, Fahriye Kilinç, and Hamdi Arbağ. Data Collection and/or Processing: Fahriye Kilinç, Metin Doğan, Ömer Kaplan, and Fakih Cihat Eravci. Analysis and/or Interpretation: Fakih Cihat Eravci and Miyase Orhan. Literature Review: Ömer Kaplan and Miyase Orhan. Writing the Article: Fakih Cihat Eravci, Miyase Orhan. Critical Review: Hamdi Arbağ, Fahriye Kilinç, and Metin Doğan. References and Fundings: Fakih Cihat Eravci and Miyase Orhan. Materials: Ömer Kaplan, Fahriye Kilinç, and Metin Doğan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Projects of Necmettin Erbakan University under project number 211218025.
Ethics Committee Approval
Ethics committee approval was received for this study from Necmettin Erbakan University Animal Experiments Local Ethics Committee (Approval number: 2021/012) and the entire study process was carried out following the Principles of Care and Use of Laboratory Animals of the National Institutes of Health.
