Abstract
Background:
Bevacizumab is a vascular endothelial growth factor (VEGF) inhibitor that is used off-label for select cases of recurrent respiratory papillomatosis (RRP) that are severe, involve the distal airway or lung parenchyma, and refractory to other forms of adjuvant therapy. However, there is limited safety data for the use of bevacizumab in children and VEGF inhibitors are reported to have a range of adverse renal effects, including hypertension, proteinuria, and thrombotic microangiopathy (TMA).
Case-Diagnosis/Treatment:
This report describes a case of severe juvenile-onset RRP that had an exceptionally high operative burden that was refractory to several adjuvant treatment strategies (including intralesional cidofovir and subcutaneous pegylated interferon). Bevacizumab treatment resulted in a dramatic and sustained improvement in disease control over a 5-year period. However, after 3 years of treatment, the patient developed hypertension and proteinuria and was found to have evidence of a glomerular TMA on kidney biopsy. These complications were successfully managed with a reduction in bevacizumab frequency and angiotensin-converting enzyme inhibitor initiation.
Conclusions:
Clinicians caring for children treated with VEGF inhibitors should be aware of the potential renal complications and their management.
Keywords
Introduction
Bevacizumab is a recombinant vascular endothelial growth factor (VEGF) inhibitor with anti-angiogenic effects, thereby limiting growth of malignancies. Indications have expanded and in the past decade it has been utilized off-label to treat recurrent respiratory papillomatosis (RRP). Analysis of pediatric RRP biopsy specimens demonstrates increased expression of VEGF protein and mRNA compared to controls, providing evidence for a role of VEGF in RRP pathogenesis. 1 However, there are limited safety data for pediatric bevacizumab use, 2 and even less in the RRP treatment context. 3
VEGF inhibitors can have various kidney side-effects, including hypertension, proteinuria, acute kidney injury, thrombotic microangiopathy (TMA), and rarely glomerulonephritis.4-8 Hypertension and proteinuria are common (up to 20%-50% of patients) and are dose- and duration-dependent. 9 TMA has been reported with VEGF inhibitor use, and may respond to drug cessation or eculizumab treatment. 5 Experts in RRP management were recently surveyed to establish a consensus for treatment with bevacizumab, 10 but formal guidelines do not yet exist. This case demonstrates a need for these guidelines since bevacizumab treatment is not curative and has long-term safety implications.
Case Presentation
The patient presented at 7 months old with respiratory distress requiring intubation. Laryngeal biopsy confirmed juvenile-onset recurrent respiratory papillomatosis (RRP) and identified human papillomavirus (HPV) type 11. The patient subsequently required frequent microlaryngoscopy with microdebridement for disease control (mean 13.9 procedures/year within 3-years post-diagnosis; Figure 1). Over 12 years since diagnosis, the patient required 65 microdebridement procedures. The papillomas have always been confined to the glottis and supraglottis, causing airway obstruction (Figure 2). Due to the frequent surgeries, the patient developed supraglottic stenosis.
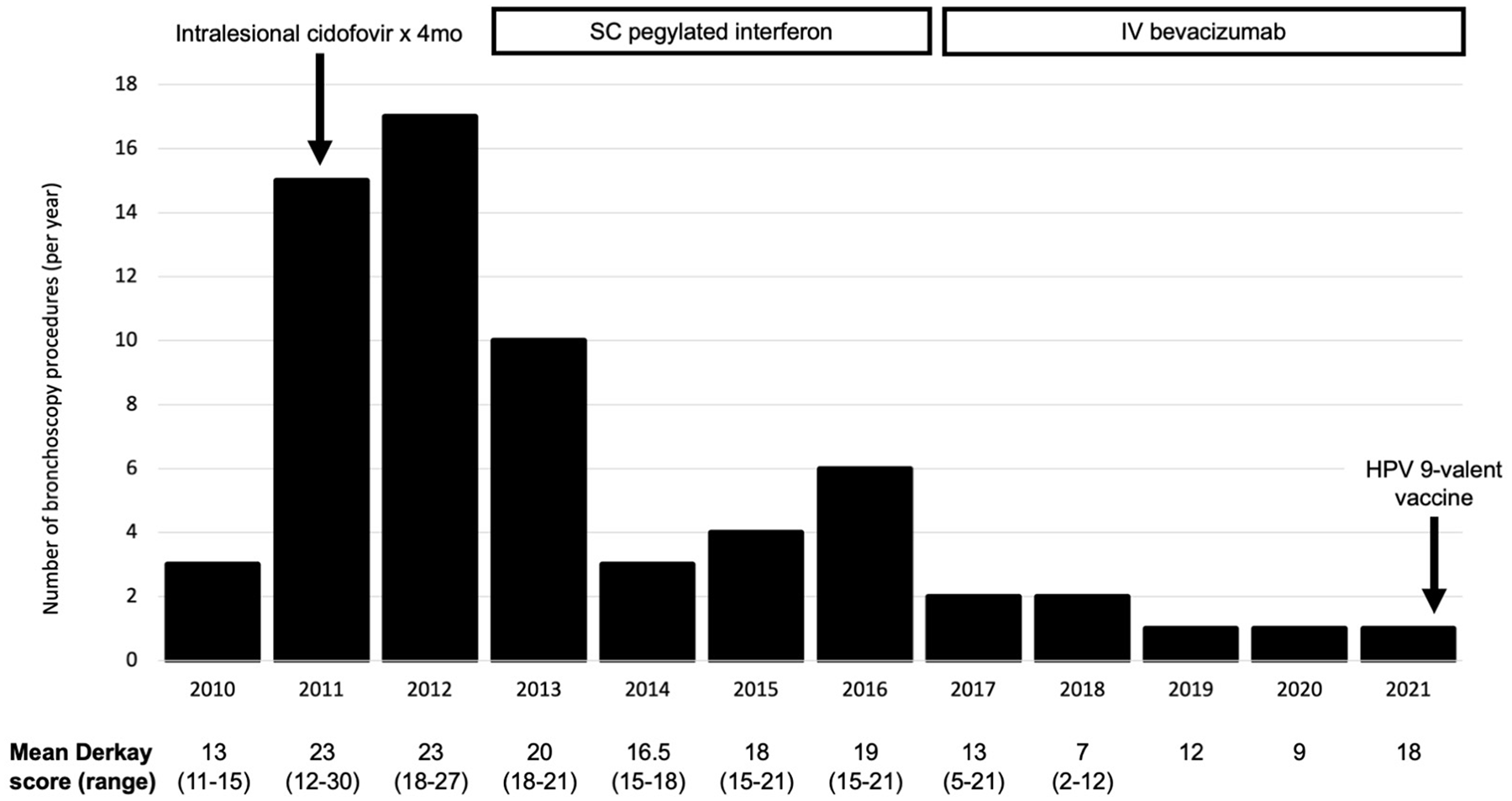
Number of bronchoscopy procedures per year, with patient treatment timeline and Derkay scoring (disease severity index). This figure illustrated the number of bronchoscopy debridement procedures performed each year, with the mean Derkay score below (a staging system for recurrent respiratory papillomatosis severity) and adjuvant treatments above. Following initiation of pegylated interferon, the frequency of procedures initially decreased but gradually increased again. Since systemic bevacizumab was started, the frequency of procedures and mean Derkay score have improved.
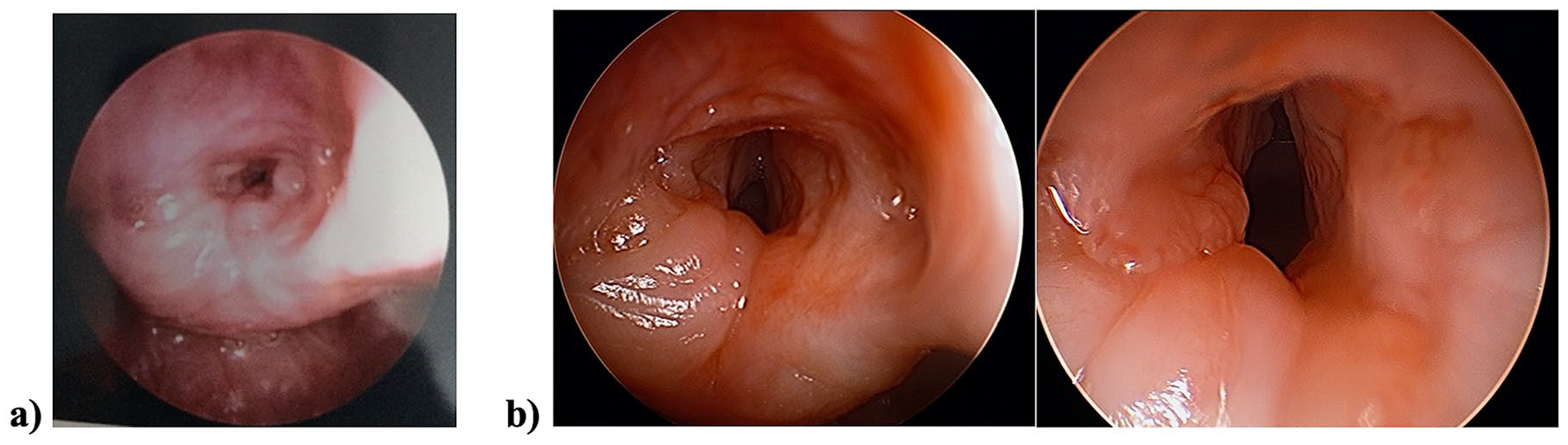
Bronchoscopy images of the recurrent respiratory papillomatosis lesions, prior to bevacizumab treatment (a) and after bevacizumab treatment (b). This figure shows bronchoscopy images prior to bevacizumab treatment (a) and after treatment (b), showing a significant improvement in disease severity and airway patency following bevacizumab.
Several adjuvant treatments were attempted (Figure 1). An initial 4-month course of intralesional cidofovir was unsuccessful. Weekly subcutaneous pegylated interferon alpha decreased microlaryngoscopy frequency from every 3-weeks to 3-months. After 4-years of pegylated interferon treatment, the efficacy waned and microlaryngoscopy frequency increased. Therefore, systemic bevacizumab was initiated and pegylated interferon stopped. Initially, bevacizumab (10 mg/kg) was administered every 3-weeks. The treatment interval was progressively increased to every 12-weeks, and subsequently adjusted based on symptoms and intraoperative disease burden. Overall, bevacizumab resulted in a significant, sustained reduction in microlaryngoscopy frequency (Figure 1).
Initially, bevacizumab was well tolerated without adverse events. Over the first 3 years the patient maintained normal blood pressure (BP; <95th percentile for age-height). There was no proteinuria at bevacizumab initiation (protein:creatinine ratio [PCR] 14 mg/mmol). During their third consecutive year of bevacizumab treatment, the patient developed stage 1 hypertension (office BP 122/86 mmHg) and proteinuria (PCR 80 mg/mmol, albumin:creatinine ratio [ACR] 30 mg/mmol). The renal function was normal (estimated GFR 168 mL/min/1.73 m2 by Bedside Schwartz equation), and there was no hematuria. Kidney doppler ultrasound found structurally normal kidneys with normal arterial flow.
The patient was started on enalapril (0.08 mg/kg/day), with slight improvement in the proteinuria (PCR 51-65 mg/mmol, Figure 3) but persistent hypertension. A 24-hour urine collection showed non-nephrotic range proteinuria (protein excretion rate 9.9mg/m2/hour; normal <4 mg/m2/hour). A kidney biopsy was performed, which demonstrated signs of glomerular TMA (including dilated glomerular capillaries, hyaline pseudothrombi, and double-contour appearance of the glomerular basement membrane), focal foot process effacement (~10%), but no globally sclerosed glomeruli (Supplemental Figure 1). There were no laboratory signs of a systemic TMA process (including anemia, thrombocytopenia, schistocytes, or elevated lactate dehydrogenase). Complement analysis showed low-grade complement system activation (soluble C5b-9 251 ng/mL; normal 58-239 ng/mL). A decision was made to lengthen the bevacizumab treatment interval to every 14 weeks. The enalapril dose was increased to 0.15 mg/kg and amlodipine was added temporarily, then stopped after 5 months once adequate BP control was achieved. Within the year following angiotensin-converting enzyme inhibitor (ACEi) initiation and bevacizimab reduction, the proteinuria normalized (PCR 12-19 mg/mmol; Figure 3), the BP remained well-controlled on enalapril monotherapy, and there was no change in serum creatinine. Unfortunately, this reduction in bevacizumab frequency resulted in a RRP exacerbation and the bevacizumab frequency was necessarily increased back to every 6 to 10 weeks.
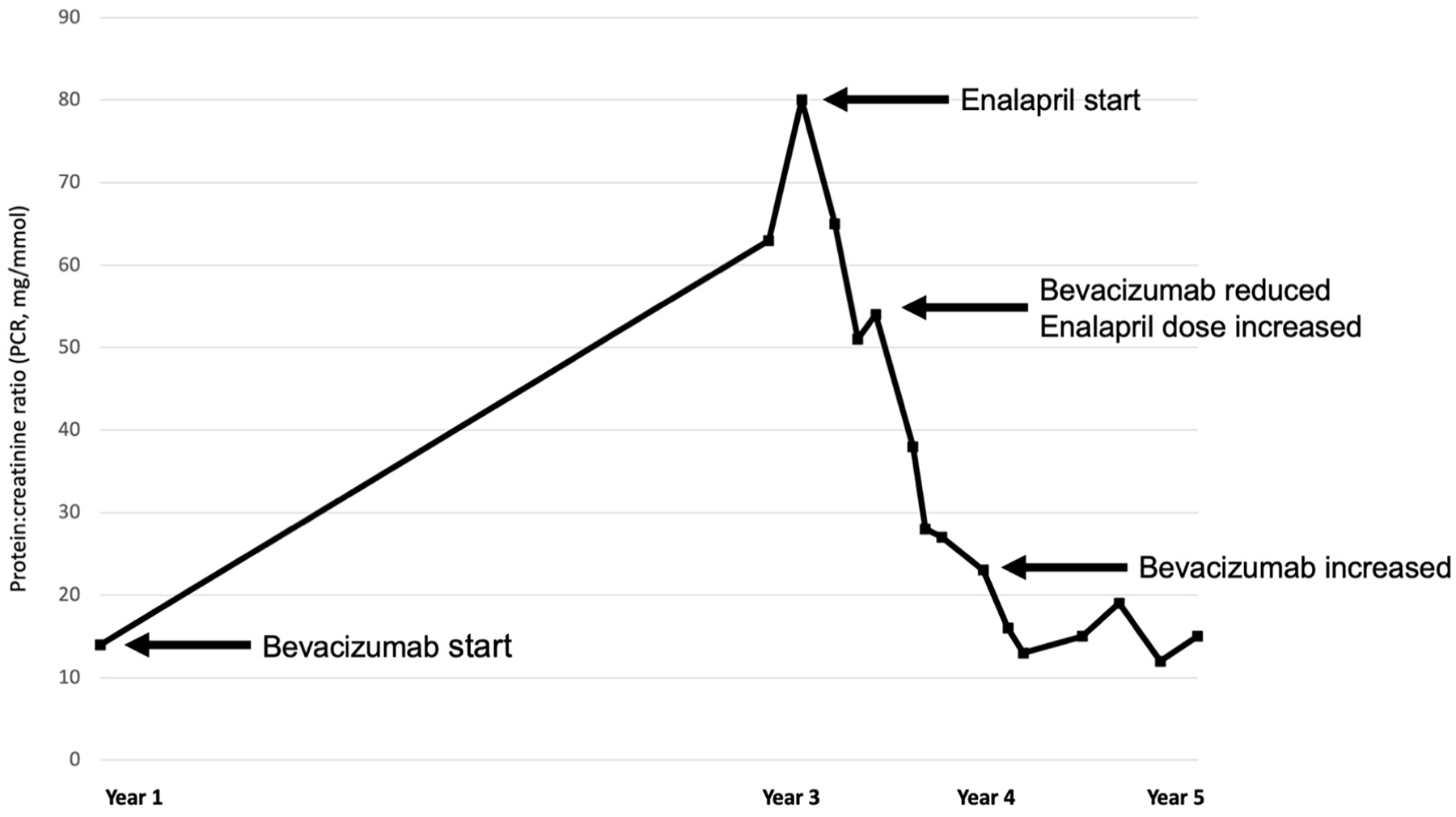
Proteinuria trend during Bevacizumab treatment course. This figure shows the trend in proteinuria after starting systemic bevacizumab, and the response to treatment with an angiotensin-converting enzyme inhibitor and reduction in the frequency of bevacizumab administration.
Discussion
Bevacizumab administration for RRP was initially reported in 5 patients with progressive, life-threatening RRP involving the distal airway or lung parenchyma. 11 One of these patients was an 8-year-old female that had required >30 debulking procedures before bevacizumab initiation. Best, Mohr, and Zur subsequently published a series of American children treated with bevacizumab for RRP, identified by surveys of the RRP Task Force of the American Society of Pediatric Otolaryngology and the American Broncho-Esophagological Association. 12 Eight children with aggressive papillomatosis, including 7 with pulmonary disease were identified. Regression or stabilization of disease was achieved in all children, although one developed malignant transformation, one had hemoptysis, and one had proteinuria during treatment.
The promising responses to systemic bevacizumab in patients with advanced RRP challenged the established dogma that adjuvant therapy should be reserved for severe, life-threatening disease. The consequences of repeated surgery on quality of life, psychological wellness, voice outcomes, and lack of alternative effective adjuvant treatments has prompted reconsideration of the timing and criteria for systemic bevacizumab. This has driven efforts to collect data systematically and establish consensus statements regarding the indication and best practices for dosing, monitoring, and discontinuation of systemic bevacizumab for RRP. As with any new treatment, a vigilant approach is recommended to ensure the safety of children, especially when little is known about the long-term treatment impact.
In this case of severe juvenile-onset RRP, the risks of life-threatening airway obstruction and the need for frequent microlaryngeal debridement warranted trials of several adjuvant treatments. Following bevacizumab initiation, disease control markedly improved and has been sustained. However, the glomerular TMA process development with hypertension and proteinuria highlights some of the potential risks of this treatment. VEGF inhibitors should be used judiciously for the management of juvenile-onset RRP, with appropriate patient counseling and regular surveillance.
VEGF inhibitors have been associated with various kidney sequelae. Proteinuria and hypertension are both common with bevacizumab use and are typically mild. 9 However, up to 4% of patients develop nephrotic range proteinuria (relative risk 7.8 vs chemotherapy alone).9,13 VEGF plays important roles in the maintenance of a healthy glomerular filtration barrier, with effects on the glomerular endothelium and the podocyte. In animal studies, VEGF helps in maintenance of fenestrated endothelium, and in repair following glomerular endothelial injury.14,15 Mice that are heterozygous for VEGF-A develop a loss of endothelial fenestrations of the glomerular capillaries, podocyte dropout, and proteinuria, 16 whereas VEGF knockout mice develop proteinuria and endothelial injury resulting in TMA. 17 In humans, VEGF inhibition also results in a loss of endothelial fenestrations and down-regulation of nephrin production (a protein integral to the podocyte slit diaphragm).18-20 Other cases of VEGF inhibitor-associated TMA are reported; most are “renal-limited” and do not develop a systemic TMA process. 5
Izzedine et al reported a series of 100 patients treated with VEGF inhibitors for various malignancies that developed biopsy-proven kidney disease. 5 Of these, 73 patients had evidence of TMA on kidney biopsy and 21 had minimal change disease or collapsing focal segmental glomerulosclerosis. TMA was more common in females, bevacizumab treatment (vs other VEGF inhibitors), and hypertensive patients. It typically occurred within 1-year after treatment initiation, 100% of patients developed proteinuria, 74% hypertension, and 40% impaired kidney function (eGFR <60 mL/min/1.73 m2). Nearly half of these patients had laboratory evidence of systemic TMA. In all patients, proteinuria and BP improved following antihypertensive treatment and VEGF inhibitor discontinuation. However, as in our case, certain patients cannot discontinue VEGF inhibitors. In the Izzedine study, 4 renal TMA patients continued VEGF inhibitors and 3 developed systemic TMA (one with severe disease). 6 When VEGF inhibitors must be continued, kidney biopsy should be considered to confirm the diagnosis and extent of kidney injury. In our case, there was no evidence of a systemic TMA, and the proteinuria and hypertension both improved with a reduction in bevacizumab frequency and ACEi introduction. In cases of severe VEGF inhibitor-associated TMA, successful treatment with eculizumab has been reported, although the role for complement blockade in secondary forms of TMA remains uncertain.21,22
Conclusion
There is a need for effective adjuvant treatment options for juvenile-onset RRP, with favorable side-effect profiles. In the case presented, systemic bevacizumab was highly effective at improving disease control and reducing microlaryngoscopic debridement frequency. However, the development of a glomerular TMA process highlights one of the potential risks of VEGF inhibitor use. Appreciation of such toxicities is particularly relevant when agents such as bevacizumab are used in the long-term and for non-malignant conditions. A high index of suspicion for a glomerular TMA process is warranted if hypertension and proteinuria develop while on VEGF inhibitor treatment, since the classic laboratory markers of TMA may be absent.
Evidence-based treatment guidelines for juvenile-onset RRP are required, with recommendations for adjuvant treatments and surveillance for associated complications. Although systemic bevacizumab will not cure RRP, it is emerging as a favorable early adjuvant treatment option that may mitigate the surgical sequelae and control disease until a definitive treatment, immune-based or otherwise, is discovered. In the meantime, a judicious and scientifically-informed approach that considers disease and patient specific factors is strongly recommended to achieve optimal patient outcomes.
Supplemental Material
sj-docx-1-aor-10.1177_00034894231184942 – Supplemental material for Renal Implications of Long-Term Systemic Bevacizumab for Recurrent Respiratory Papillomatosis
Supplemental material, sj-docx-1-aor-10.1177_00034894231184942 for Renal Implications of Long-Term Systemic Bevacizumab for Recurrent Respiratory Papillomatosis by Cal H. Robinson, Michael Hart-Matyas, Daniel A. Morgenstern, Damien Noone and Paolo Campisi in Annals of Otology, Rhinology & Laryngology
Footnotes
Author Contributions
Cal H. Robinson, Michael Hart-Matyas, and Paolo Campisi contributed to study conception and design, literature search and review, data collection and analysis, drafting the manuscript, formatting and revising the manuscript for submission. Daniel A. Morgenstern and Damien Noone contributed to study design, literature review, critical appraisal and revisions to the manuscript. All authors have approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
