Abstract
Acute myocardial infarction is a major cause of mortality in individuals with obstructive airway disease. The impact of inpatient care quality following non-ST elevation myocardial infarction (NSTEMI) on long-term mortality among those with chronic obstructive pulmonary disease (COPD) and asthma remains poorly understood. We analysed 499 318 adults with NSTEMI from the Myocardial Ischaemia National Audit Project registry between 2005 and 2019, linked with Hospital Episode Statistics for airway disease diagnosis and Office for National Statistics data for mortality outcomes. Inpatient care quality was measured using the opportunity-based quality-indicator (OBQI) score. Long-term outcomes were evaluated using multivariable Cox regression and Kaplan-Meier analyses. Individuals with COPD and asthma received lower quality of care (OBQI score: no airways disease: 83.5 vs COPD: 78.1, asthma: 80.8, P < .001). Percutaneous coronary intervention was less frequent in COPD patients (22%) than in those without airway disease (30%) or with asthma (31%), P < .001. COPD was associated with higher 10-year mortality (hazard ratio [HR]: 1.58, 95% CI 1.56-1.60), whereas those with asthma had lower risk (HR: 0.97, 95% CI 0.95-0.98). COPD was associated with increased adjusted cardiovascular mortality (sub-distribution HR: 1.89, 95% CI 1.84-1.95). Individuals with COPD received lower-quality inpatient care and fewer coronary interventions, which was associated with higher long-term mortality.
Keywords
Introduction
Obstructive airways disease encompasses a group of conditions characterised by airways obstruction and inflammation 1 most notably asthma and chronic obstructive pulmonary disease (COPD). It is estimated that around 800 million people worldwide live with COPD 2 and at least 260 million are affected by asthma. 3 Global trends suggest that asthma prevalence is declining, whereas the global burden of COPD is projected to rise by nearly 50% by 2050.4,5 Both COPD and asthma have been associated with an increased risk of acute myocardial infarction (AMI), largely attributable to overlapping cardiopulmonary risk profiles. 4 Meta-analyses indicate that individuals with COPD have a relative risk of AMI between 1.84 and 2.43 6 whereas those with asthma have a relative risk of ~1.28 compared with the general population. 7
Individuals with COPD who experience AMI face substantially worse outcomes, with reported mortality rates up to 26% at 6 months. 8 This excess mortality is thought to be related to lower rates of transfer for invasive coronary procedures and reduced utilization of guideline-directed medical therapies, including antiplatelet and lipid-lowering agents.9 -11 In contrast, evidence regarding outcomes among individuals with asthma is limited. A large contemporary cohort study of over 300 000 AMI admissions found no significant association between asthma and mortality up to 6 months 12 ; however, data on longer-term outcomes remain scarce. Furthermore, many studies have analysed AMI as a composite outcome, combining ST-elevation myocardial infarction (STEMI) and non-ST-elevation myocardial infarction (NSTEMI). 13 These conditions differ markedly in their pathophysiology, severity of presentation and management strategies, analysing them together limits both the interpretability and the clinical relevance of the findings.
To address these gaps, we used data from the Myocardial Ischaemia National Audit Project (MINAP) registry (UK), linked to the Office for National Statistics (ONS) mortality data and Hospital Episode Statistics (HES), to compare in-hospital care quality and long-term mortality following NSTEMI among patients with asthma or COPD.
Methods
Study Design
This population-based, retrospective cohort study utilises data obtained from MINAP, HES and National Deaths Registry from the ONS. MINAP is a prospective national registry of patients admitted to UK hospitals with an acute coronary syndrome (ACS). It is the UK’s largest AMI registry containing ~130 variables, including baseline demographics, comorbidities, management strategies, pharmacotherapy, in-hospital clinical outcomes and discharge diagnosis. 14 Data are submitted by hospital clinical staff, and ~90 000 pseudonymised records annually are uploaded to the National Institute for Cardiovascular Outcomes Research.15,16 HES is a national registry obtaining data from National Health Service (NHS) Hospitals in England. 17 Data is recorded at the person level as a consultant episode for admitted patients, >17 million consultant episodes are added yearly. Each HES record contains clinical information including diagnosis and co-morbidities. Mortality information is recorded via ONS data which collects data regarding every death registered in the UK, coding deaths according to the international classification of diseases (ICD-10) and cause of death from the Medical Certificate of Cause of Death. Mortality follow-up was available up to July 31, 2021.
Study Population
We identified all patients admitted with an index diagnosis of NSTEMI to hospitals in England and Wales between January 2005 and March 2019, recorded in MINAP and linked to HES. Asthma and COPD diagnoses were identified using ICD-10 codes: J44.0, J44.1, J44.8, J44.9 (COPD) and J45.0, J45.1, J45.8, J45.9, J46 (asthma). NSTEMI diagnoses were determined locally in accordance with the Joint European Society of Cardiology (ESC) and American College of Cardiology (ACC) consensus criteria. 18 Patients were excluded if they had missing data for key variables (in-hospital or cardiac mortality), missing NHS number or inconsistent mortality dates. Only the index NSTEMI admission per patient was included.
Outcomes
The primary outcome was all-cause mortality, at 30 days, 1, 5 and 10 years (where available). To assess the quality of inpatient care, we calculated the opportunity-based quality indicator (OBQI) score, which comprises inpatient prescription of: aspirin, P2Y12 inhibitors, statins, β-blockers and angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) and referral to cardiac rehabilitation on discharge.19,20 These represent elements of the European Society of Cardiology (ESC) quality metrics and are reflected in the 2023 ESC AMI Guidelines. 21 OBQI scores were categorised as excellent (≥90), good (80-89), fair (70-79) or poor (<70).
We also evaluated the 2020 ESC Association for Acute Cardiovascular Care NSTEMI quality indicators, 22 including invasive coronary angiography within 72 hours, dual antiplatelet therapy at discharge, low molecular weight heparin (LMWH) or fondaparinux and assessment of left ventricular function. Quality of care metrics were assessed only for patients that survived beyond 30 days given that these quality-of-care metrics are predominately assessed at the point of discharge.
Statistical Analysis
Continuous variables such as age at admission, and body mass index (BMI) were summarised using mean and standard deviation if normally distributed and median and interquartile ranges (IQR) if data was not normally distributed. Normality of distribution was assessed using the Shapiro-Wilk test. These data were compared using Student’s t-test if normally distributed, and Wilcoxon rank sum test or Kruskal-Wallis equality-of-populations rank test if not normally distributed. Categorical variables were compared using the Pearson chi-square (χ2) test and summarised as percentages (%). Missing data were imputed using multiple imputation with chained equations (MICE), incorporating all covariates from the analytical model. MICE is considered best practice for handling missing data, providing unbiased estimates even when missingness is substantial or not completely random. 23
Unadjusted survival was evaluated using Kaplan-Meier curves for patients with COPD, asthma and no airways disease. Cox proportional hazards models were then fitted on 10 imputed datasets to estimate hazard ratios (HRs) for mortality, adjusted for; age, gender, year, ethnicity, heart rate on admission, blood pressure on admission, region, comorbidities (previous stroke, peripheral arterial disease, chronic kidney disease, hypertension, smoking status, diabetes mellitus, previous AMI), medication strategy (aspirin, P2Y12 inhibitor, low molecular weight heparin, statin, warfarin, ACE inhibitor and β-blockers), clinical outcomes (left ventricular ejection fraction and cardiac arrest), invasive coronary angiography (ICA) or revascularisation by percutaneous coronary intervention (PCI) and coronary artery bypass graft surgery (CABG). Separate models were run to generate Hazard Ratios (HR) for 30-day, 1-, 5-, 10-year and overall mortality during the study period. To evaluate post-discharge outcomes, additional Kaplan-Meier curves stratified by airway disease status and OBQI score were plotted after excluding 30-day mortality to avoid bias from inpatient quality indicators. Proportional hazards assumptions were verified using Schoenfeld residuals and presented graphically in Supplemental Figure 1. Competing risks between cardiovascular and non-cardiovascular death were analysed using the Fine and Grey sub distribution hazards model, adjusted for the same covariates. Our temporal trend analysis of 1-year mortality included patients admitted between January 2005 and March 2019. Only complete study years are presented in the temporal trends analysis up to and including 2018. These are organised biennially, into 2005 to 2006, 2007 to 2008, 2009 to 2010, 2011 to 2012, 2013 to 2014, 2015 to 2016 and 2017 to 2018 for ease of presentation.
Stata 18.0 (StataCorp 2025, Stata Statistical Software: Release 18; StataCorp LLC, College Station, TX) was used for all analyses and a 2-sided alpha level of .05 was used throughout.
Results
After application of exclusion criteria, a total of 499 318 patients were admitted with NSTEMI to hospitals in England and Wales between 1 January 2005 and 30 March 2019 (Figure 1). Of these, 77 832 (16%) had a diagnosis of obstructive airways disease: 45 658 (59%) with COPD and 32 174 (41%) with asthma. The mean follow-up duration was 5.57 ± 4.34 years.
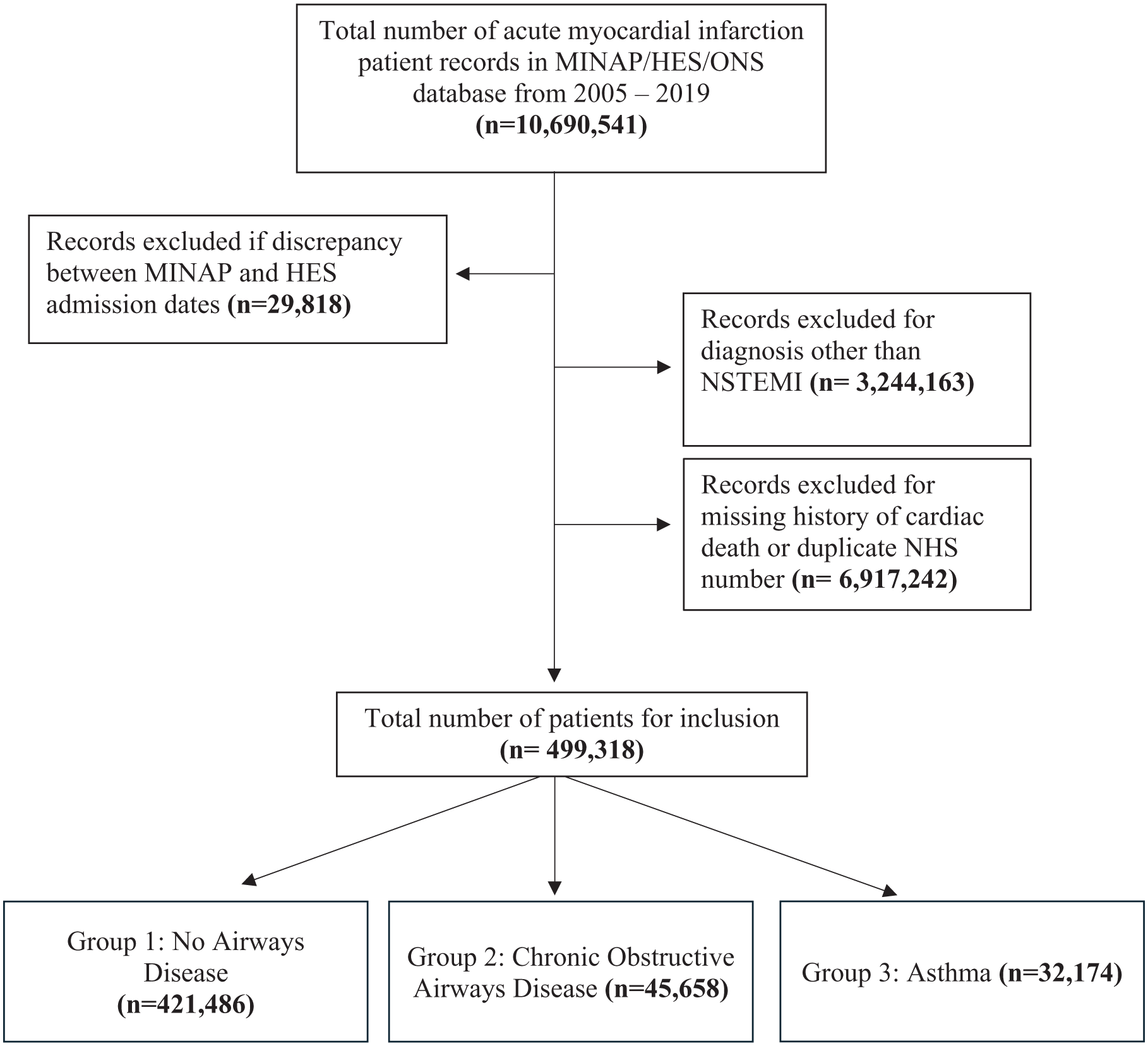
STROBE diagram detailing exclusion criteria.
Demographic Comparison
Individuals with COPD were older, whereas those with asthma were younger than individuals without airways disease (median age: no airways disease: 72.8, interquartile range [IQR] 20.8 vs COPD: 76.1, IQR 14 vs asthma: 71.6, IQR 20.3, P < .001). Individuals with airways disease were more likely to be female (no airways disease: 36% vs COPD: 40%, asthma: 47%, P < .001; Table 1).
Demographic, Outcome and Treatment Comparison Between Non-ST-Elevation Myocardial Infarction Patients With and Without Airways Disease.
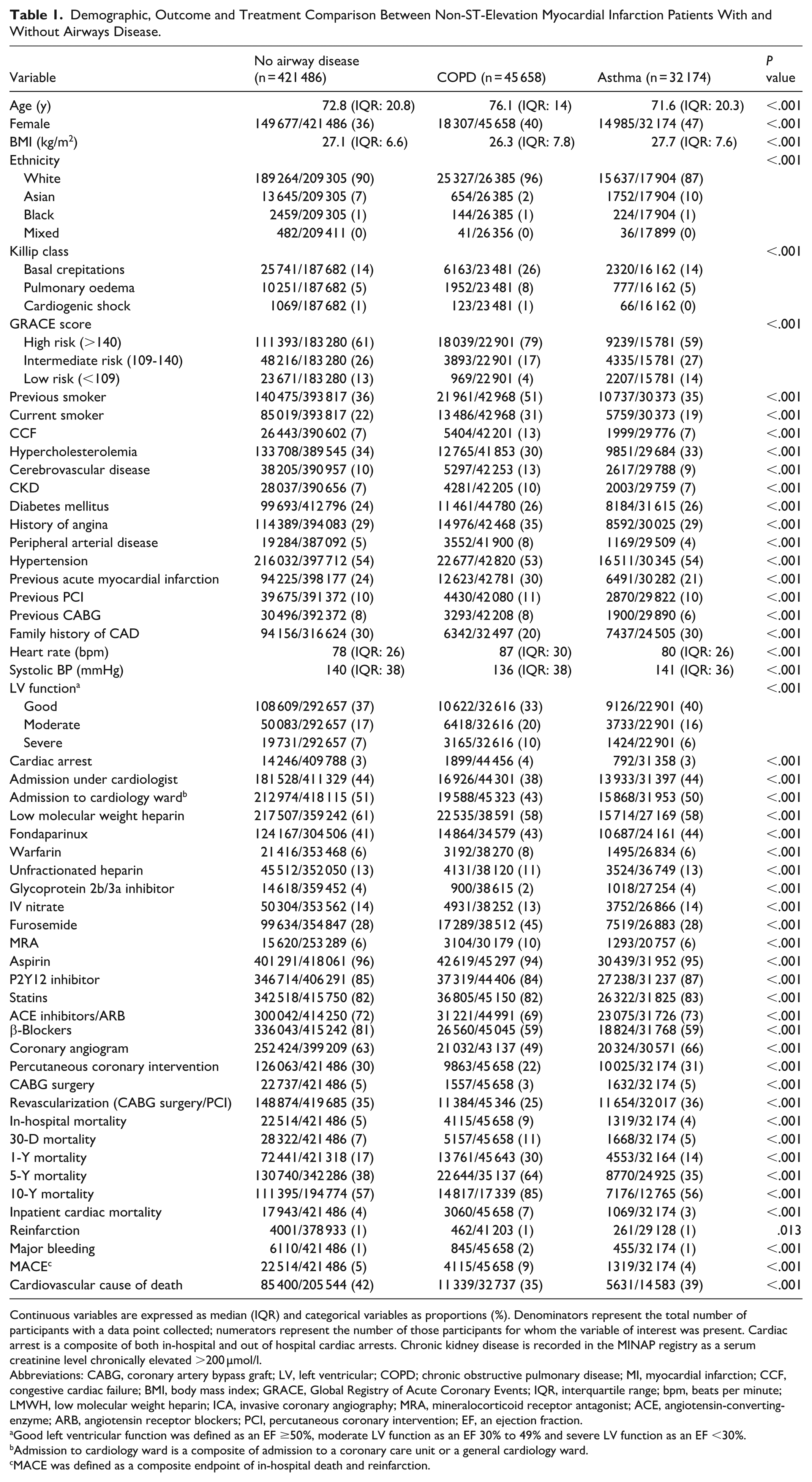
Continuous variables are expressed as median (IQR) and categorical variables as proportions (%). Denominators represent the total number of participants with a data point collected; numerators represent the number of those participants for whom the variable of interest was present. Cardiac arrest is a composite of both in-hospital and out of hospital cardiac arrests. Chronic kidney disease is recorded in the MINAP registry as a serum creatinine level chronically elevated >200 µmol/l.
Abbreviations: CABG, coronary artery bypass graft; LV, left ventricular; COPD; chronic obstructive pulmonary disease; MI, myocardial infarction; CCF, congestive cardiac failure; BMI, body mass index; GRACE, Global Registry of Acute Coronary Events; IQR, interquartile range; bpm, beats per minute; LMWH, low molecular weight heparin; ICA, invasive coronary angiography; MRA, mineralocorticoid receptor antagonist; ACE, angiotensin-converting-enzyme; ARB, angiotensin receptor blockers; PCI, percutaneous coronary intervention; EF, an ejection fraction.
Good left ventricular function was defined as an EF ≥50%, moderate LV function as an EF 30% to 49% and severe LV function as an EF <30%.
Admission to cardiology ward is a composite of admission to a coronary care unit or a general cardiology ward.
MACE was defined as a composite endpoint of in-hospital death and reinfarction.
Individuals with COPD were more likely to present with cardiovascular risk factors including, chronic renal failure (no airways disease: 7% vs COPD: 10%, asthma: 7%), previous angina (no airways disease: 29% vs COPD: 35%, asthma: 29%) and previous acute myocardial infarction (no airways disease: 24% vs COPD: 30%, asthma: 21%; all P < .001; Table 1). They also had higher Global Registry of Acute Coronary Events (GRACE) risk scores (>140: 79% vs 61%) and more advanced Killip class at presentation (basal crepitations, no airways disease: 14% vs COPD: 26%, asthma: 14%; pulmonary oedema, no airways disease: 5% vs COPD: 8%, asthma: 5%, all P < .001). COPD patients were less likely to be admitted under the care of a cardiologist (38% vs 44%; P < .001; Table 1).
Management Strategies and Unadjusted Clinical Outcomes
Individuals with COPD were less likely to undergo invasive coronary angiography (no airways disease: 63% vs COPD: 49%, asthma: 66%, P < .001) or revascularisation by PCI or CABG (no airways disease: 35% vs COPD: 25%, asthma: 36%, P < .001). Individuals with COPD and asthma were less likely to receive β-blockers (no airways disease: 81% vs COPD: 59%, asthma: 59%, P < .001; Table 1).
Individuals with COPD had higher 30-day (no airways disease: 7% vs COPD: 11%, asthma: 5%), 1-year (no airways disease: 17% vs COPD: 30%, asthma: 14%), 5-year (no airways disease: 38% vs COPD: 64%, asthma: 35%) and 10-year (no airways disease: 57% vs COPD: 85%, asthma: 56%; all P < .001) mortality (Table 1). Individuals with COPD had a lower crude rate of cardiovascular mortality (no airways disease: 42% vs COPD: 35%, asthma: 39%, P < .001; Table 1). Figure 2 shows the unadjusted Kaplan-Meier survival curves of these patients.
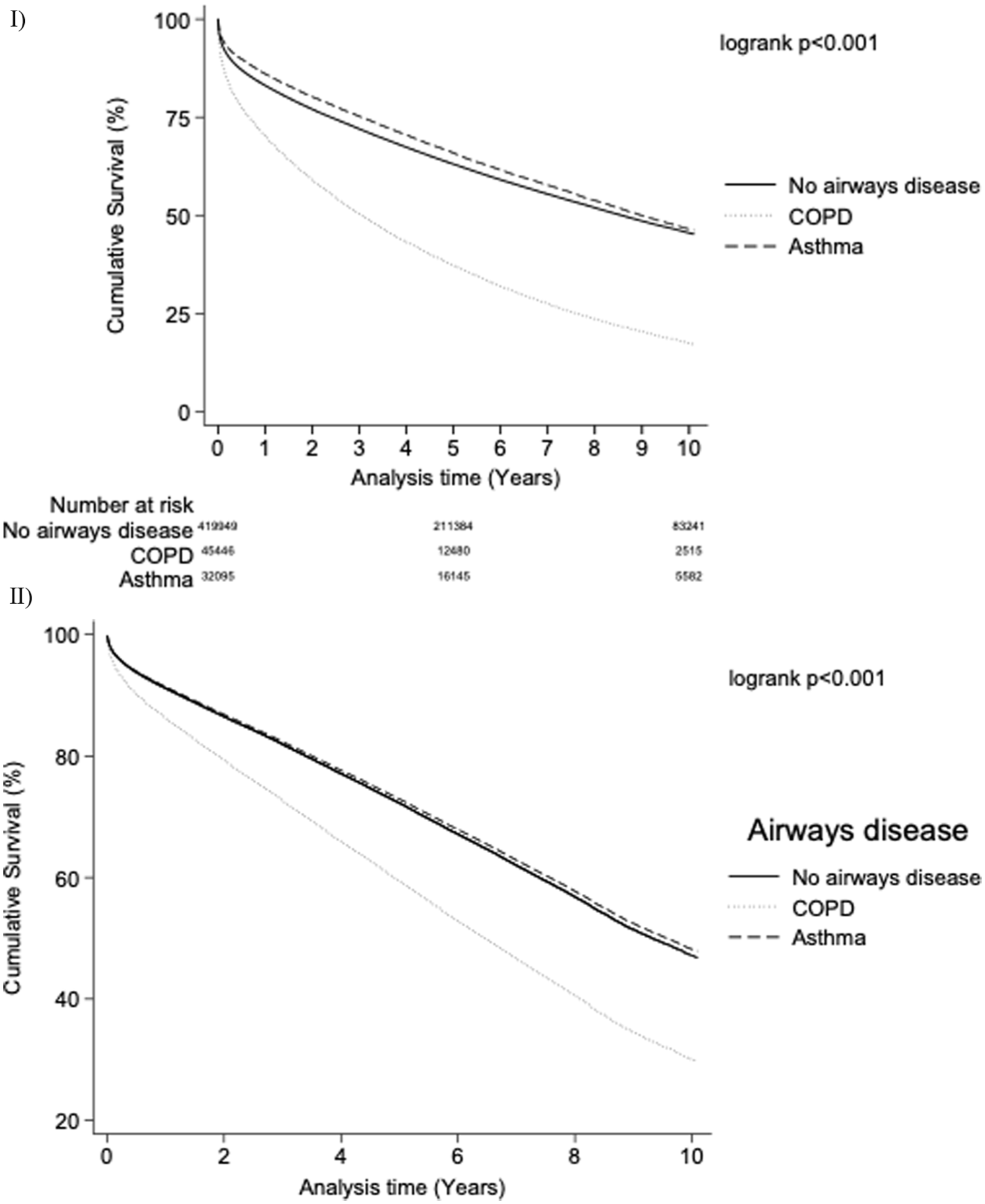
Kaplan-Meier survival curves for patients with airways disease compared to those without. (I) Unadjusted Kaplan-Meier survival curves. (II) Adjusted Kaplan-Meier survival curves adjusted for: age, gender, year, ethnicity, heart rate, blood pressure, region, comorbidities (previous stroke, peripheral vascular disease, chronic kidney disease, hypertension, smoking status, diabetes mellitus, previous acute myocardial infarction), medication strategy (aspirin, P2Y12 inhibitor, low molecular weight heparin, statin, warfarin, angiotensin converting-enzyme inhibitor and β-blockers), clinical outcomes (left ventricular ejection fraction and cardiac arrest), ICA or revascularisation by PCI and CABG surgery.
Quality Indicators
Individuals with COPD were less likely to receive a coronary angiogram within 72 hours (no airways disease: 59% vs COPD: 48%, asthma: 57%, P < .001) and referral to cardiac rehabilitation (no airways disease: 79% vs COPD: 72%, asthma: 80%, P < .001) than those without airways disease. Among patients with moderate or severe LV systolic dysfunction, those with COPD or asthma were less likely to receive β-blockers (no airways disease: 85% vs COPD: 70%, asthma: 68%, P < .001).
The overall quality of inpatient care, as assessed by the OBQI score, was lower for individuals with COPD compared with both those without airways disease and asthma (mean OQBI score; no airways disease: 83.5 vs COPD: 78.1, asthma: 80.8, P < .001; Table 2). In adjusted linear regression, both COPD (β = −2.16, 95% CI −2.45 to −1.87) and asthma (β = −2.52, 95% CI −2.86 to −2.18) were independently associated with lower care quality (Supplemental Table 1).
Quality Indicators for Non-ST-Elevation Myocardial Infarction Patients With and Without Airways Disease (ESC, ACVC and OBQI).
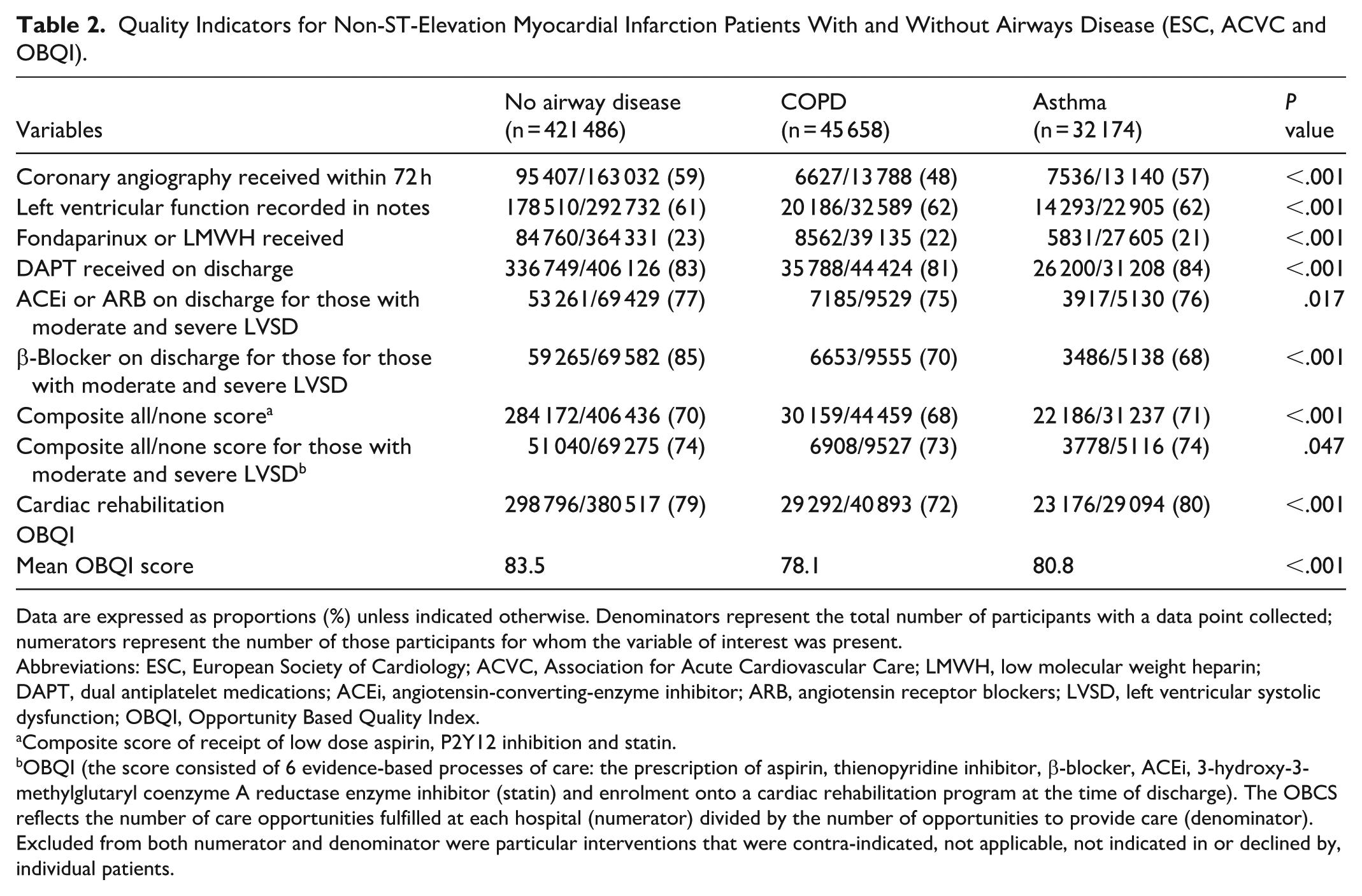
Data are expressed as proportions (%) unless indicated otherwise. Denominators represent the total number of participants with a data point collected; numerators represent the number of those participants for whom the variable of interest was present.
Abbreviations: ESC, European Society of Cardiology; ACVC, Association for Acute Cardiovascular Care; LMWH, low molecular weight heparin; DAPT, dual antiplatelet medications; ACEi, angiotensin-converting-enzyme inhibitor; ARB, angiotensin receptor blockers; LVSD, left ventricular systolic dysfunction; OBQI, Opportunity Based Quality Index.
Composite score of receipt of low dose aspirin, P2Y12 inhibition and statin.
OBQI (the score consisted of 6 evidence-based processes of care: the prescription of aspirin, thienopyridine inhibitor, β-blocker, ACEi, 3-hydroxy-3-methylglutaryl coenzyme A reductase enzyme inhibitor (statin) and enrolment onto a cardiac rehabilitation program at the time of discharge). The OBCS reflects the number of care opportunities fulfilled at each hospital (numerator) divided by the number of opportunities to provide care (denominator). Excluded from both numerator and denominator were particular interventions that were contra-indicated, not applicable, not indicated in or declined by, individual patients.
As shown in Figure 3, survival improved stepwise with increasing inpatient care quality. Transition from poor to good or excellent care was associated with the largest relative mortality reduction among patients with asthma (good care: adjusted hazard ratio [aHR] 0.82, 95% CI 0.69-0.85; excellent care: aHR 0.73, 95% CI 0.62-0.85).
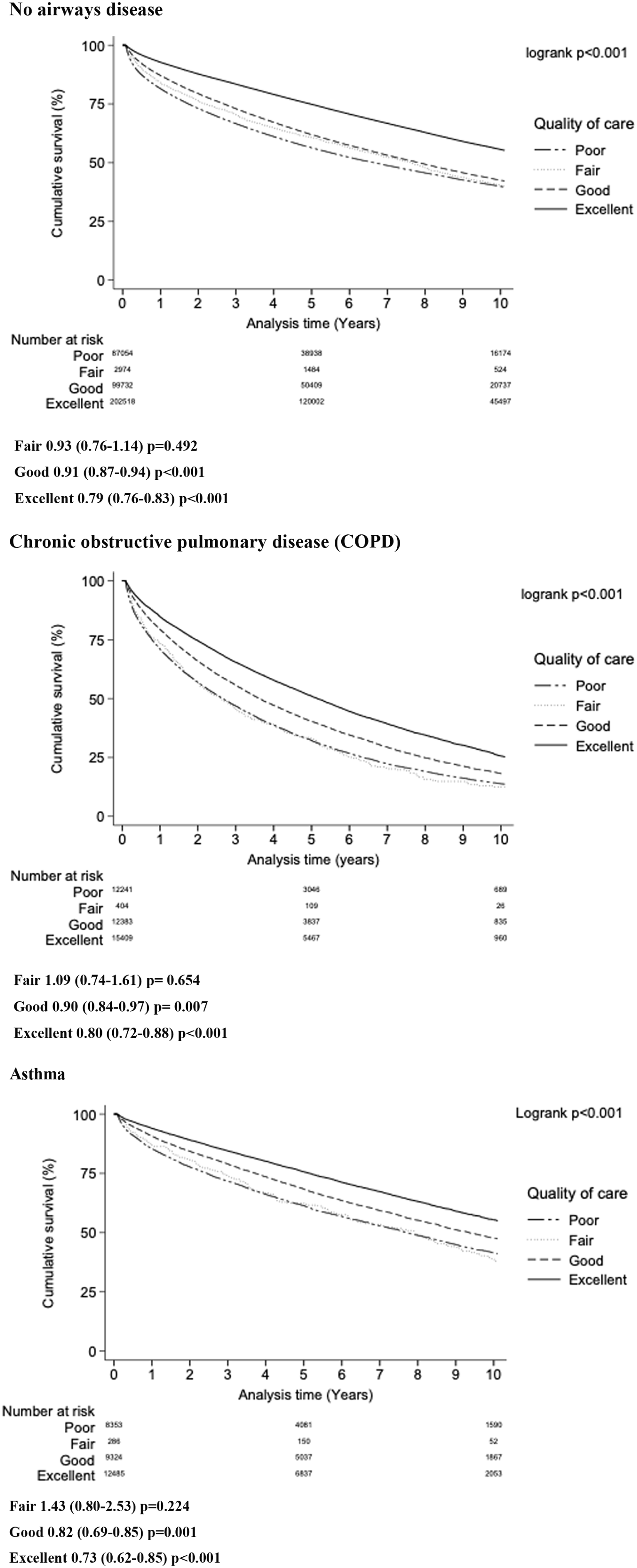
Impact of inpatient quality of care according to OBQI score on clinical outcomes of patients with and without airways disease. Results landmarked to exclude mortality within 30 days of admission to reflect aspects of OBQI score that are assessed on discharge.
Long-Term Mortality Analysis
In our multivariable model, COPD was associated with higher mortality across all follow-up intervals, from 30 days to up to 10 years (30 days; aHR 1.19, CI 1.15-1.22, 10 years; aHR 1.58, CI 1.56-1.60). Conversely, individuals with asthma had a lower risk of mortality up to 10 years of follow up (30 days; aHR 0.82, CI 0.78-0.87, 10 years; aHR 0.97, CI 0.95-0.98; Table 3).
Survival Analysis for Non-ST-Elevation Myocardial Infarction Mortality Outcomes Depending on Diagnosis of Obstructive Airways Disease.
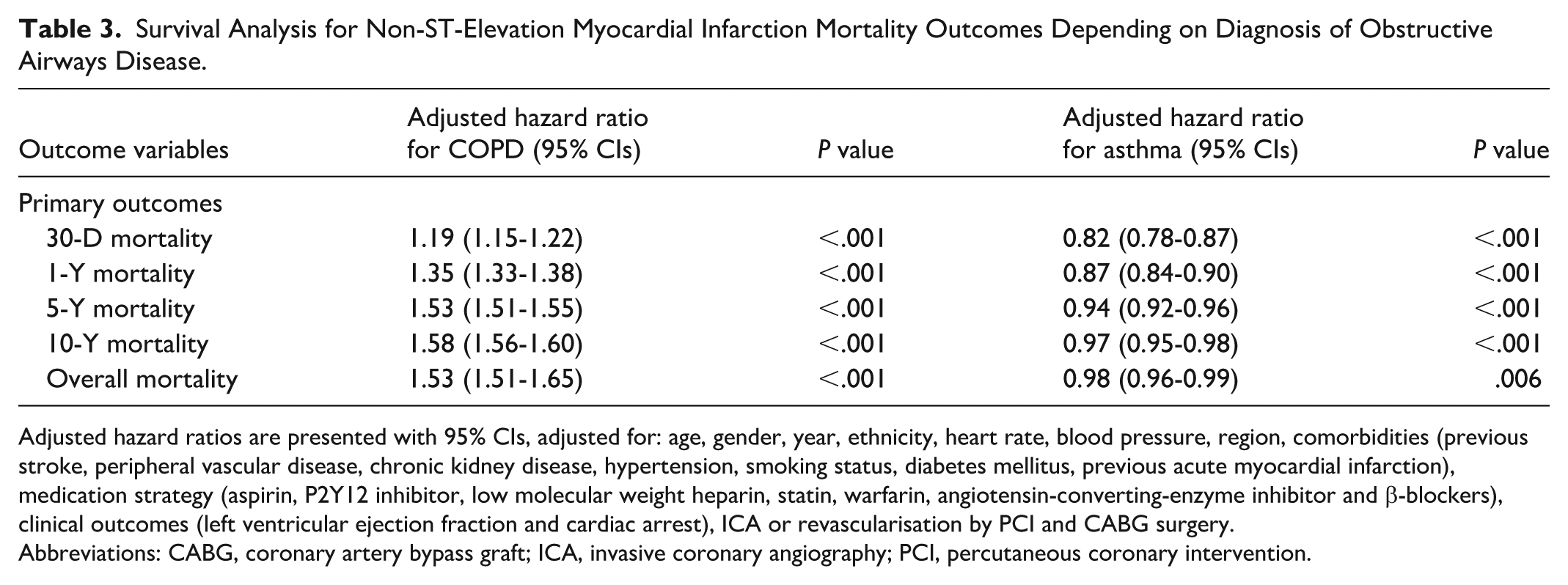
Adjusted hazard ratios are presented with 95% CIs, adjusted for: age, gender, year, ethnicity, heart rate, blood pressure, region, comorbidities (previous stroke, peripheral vascular disease, chronic kidney disease, hypertension, smoking status, diabetes mellitus, previous acute myocardial infarction), medication strategy (aspirin, P2Y12 inhibitor, low molecular weight heparin, statin, warfarin, angiotensin-converting-enzyme inhibitor and β-blockers), clinical outcomes (left ventricular ejection fraction and cardiac arrest), ICA or revascularisation by PCI and CABG surgery.
Abbreviations: CABG, coronary artery bypass graft; ICA, invasive coronary angiography; PCI, percutaneous coronary intervention.
We performed a Fine-Grey regression for cardiovascular mortality, with non-cardiovascular mortality treated as a competing event. COPD was associated with higher cardiovascular mortality (sub-distribution hazard ratio [sdHR] 1.89, 95% CI 1.84-1.95), while asthma showed no significant difference (sdHR 1.03, 95% CI 0.99-1.08; Supplemental Table 2).
Temporal trends in absolute 1-year mortality demonstrated progressive improvement across all groups; however, COPD remained associated with the highest mortality throughout the study years. Between 2005 to 2006 and 2017 to 2018, 1-year mortality improved from 21% to 14% in patients without airways disease, 36% to 25% in COPD and 17% to 11% in asthma (P < .001; Supplemental Figure 1).
Discussion
In this large, contemporary registry of >400 000 patients with NSTEMI, we demonstrate marked disparities in care quality and outcomes among individuals with obstructive airways disease. Patients with COPD were less likely to undergo invasive coronary angiography or revascularisation, received lower-quality inpatient care, and were less frequently referred to cardiac rehabilitation at discharge. These individuals experienced the highest adjusted mortality risk at all follow-up intervals up to 10 years and had a greater burden of cardiovascular mortality, after accounting for competing non-cardiovascular risks. Interestingly individuals with asthma demonstrated a lower adjusted mortality risk following NSTEMI compared with both those without airway disease and COPD.
Cardiovascular disease remains a leading cause of morbidity and mortality among individuals with COPD or asthma. 24 In our analysis, COPD was associated with substantially lower rates of invasive management for NSTEMI, and when undertaken, was often delayed beyond the ESC guideline-recommended <72-hour target. 22 The lower use of invasive therapy likely reflects a combination of technical challenges, comorbidity burden and clinician bias against intervention in patients with severe respiratory disease.
Individuals with COPD often present more acutely unwell on arrival with NSTEMI as reflected in this study by their higher Killip class and pulmonary oedema on admission. These factors have been shown to complicate NSTEMI diagnosis and procedural tolerance. 25 Pulmonary congestion can hinder supine positioning and delay angiography, while lower peak troponin levels in COPD may also obscure the diagnosis of AMI. 26 Given that Killip class is an independent predictor of mortality in NSTEMI 25 these factors collectively contribute to delayed or withheld revascularisation and the observed adverse outcomes.
Moreover, individuals with COPD presented more commonly with concurrent cardiovascular risk factors such as chronic kidney disease (CKD) and diabetes mellitus (DM), both of which influence clinical decision-making and prognosis. CKD is associated with increased bleeding risk and concern for contrast-induced nephropathy, often discouraging invasive management,27,28 whilst DM confers higher long-term mortality following NSTEMI. 29
This clustering of high-risk characteristics including advanced age, frailty and multimorbidity, likely contributes to the underuse of guideline-directed invasive strategies. The pattern observed aligns with the well-described risk-treatment paradox, whereby patients at highest baseline risk receive the least aggressive care. 30 This may, paradoxically, deprive those most likely to benefit from revascularisation of potentially life-saving therapy. 31
Current ESC guidelines advocate early invasive therapy in high-risk NSTEMI patients identified by risk stratification tools such as the GRACE score.21,32 However, emerging data suggests that the GRACE score underestimates risk among individuals with COPD. Rothnie et al demonstrated that up to one-third of COPD patients classified as “low risk” are of moderate risk, and nearly two-thirds of “moderate-risk” patients are high risk. 33 Within our cohort, over one-fifth of COPD patients were classified as low to moderate risk, highlighting substantial potential for misclassification. This may partly explain the lower rates of early invasive therapy in this group and represents a critical gap in current risk assessment paradigms. Revision or recalibration of risk tools for comorbid respiratory disease populations may therefore improve the equity of invasive management.
Frailty is likely an important, unmeasured determinant of outcomes in this cohort. Patients with COPD were older and had a greater burden of comorbidity than those with asthma or no airways disease, consistent with increased physiological frailty. Although the crude rates of cardiovascular mortality were lower in those with COPD and asthma, our competing-risk analysis showed that COPD was associated with a markedly higher subdistribution hazard ratio of cardiovascular death (sdHR 1.89; 95% CI 1.84-1.95), whereas asthma was not (sdHR 1.03; 95% CI 0.99-1.08). This pattern indicates that excess non-cardiovascular mortality, particularly from respiratory causes, obscures the true cardiovascular vulnerability of patients with COPD when raw mortality rates are considered in isolation.
Frailty may also contribute to under-recognition and under-treatment of coronary artery disease in advanced lung disease. 34 Prior AMI registries show that frail patients experience longer admissions, undergo fewer invasive procedures and experience higher in-hospital mortality. 35 Clinicians may therefore favour conservative management in those perceived to have limited life expectancy or functional reserve. Together, these factors may lead to more conservative management in COPD, despite their substantially elevated adjusted risk of cardiovascular death. In COPD, this approach persists despite a substantially elevated adjusted risk of cardiovascular death, which becomes apparent only after accounting for competing non-cardiovascular mortality such as infection and malignancy. Recognition of this excess risk is critical, as patients with COPD are likely to benefit from evidence-based cardiovascular therapies. Treatment decisions should therefore explicitly balance procedural risk against the demonstrated long-term survival advantage of revascularisation.
Our findings also highlight suboptimal discharge planning within patients with COPD. Patients with COPD were less frequently referred for cardiac rehabilitation, despite strong evidence that participation reduces mortality following AMI. 36 The lower referral rate may reflect a clinician perception that respiratory disease limits physical participation, 37 yet evidence indicates that rehabilitation confers similar benefits irrespective of underlying lung disease. 38 Improving access to, and uptake of, tailored cardiac rehabilitation programs for patients with COPD represents a key opportunity to reduce long-term cardiovascular risk.
Interestingly, individuals with asthma demonstrated a lower adjusted mortality risk following NSTEMI compared with both those without airway disease and COPD. Several mechanisms may explain this finding. First, individuals with asthma were younger and had fewer comorbidities than those with COPD, which may confer a survival advantage. Second, many individuals with asthma receive long-term corticosteroid or β₂-agonist therapy, which may exert anti-inflammatory and cardioprotective effects, potentially attenuating atherothrombotic risk. 39 Finally, a diagnostic bias may exist in this study, whereby some patients coded as having asthma have milder airway symptoms or overlap syndromes, representing a comparatively healthier cohort.
Limitations
This study has several limitations, the MINAP/HES/ONS linked dataset lacks data on frailty, clinical rationale for treatment decisions, angiographic findings and the full spectrum of comorbidities. MINAP also has strict definitions for comorbidities; for example, CKD is defined as creatinine >200 µmol/L, preventing subclassification by kidney disease severity. Data on pulmonary function, steroid use and recent exacerbations were unavailable, preventing stratification by airways disease severity.
Airways disease classification relied on ICD-10 coding within HES. Misclassification between COPD and asthma, or omission of milder cases, is possible due to absence of spirometry confirmation. We were also unable to examine the influence of specific COPD treatment regimens, including inhaled therapies, which may modify cardiovascular risk.
Within our use of the OBQI score to assess quality of care, individuals were excluded if medications were recorded as clinically contraindicated or not applicable. Given the nature of data coding at the end of patients’ admissions, it is possible that medications were omitted appropriately, but without clear clinical reasoning documented which would have negatively affected our assessment of their quality of care.
Although we present prospective data and our modelling adjusted for many important confounding variables, these observational data have potential for residual confounding within variables that we are unable to assess such as frailty, socioeconomic status and severity of lung disease such as spirometry or exacerbation frequency and therefore there should be caution in making causal inferences. We acknowledge that in presenting data from 2005 to 2019, this is a wide range of years of study, over which AMI, COPD and asthma treatment has evolved significantly. Unfortunately, we did not have access to more contemporary data from 2019 onwards, and our results must be interpreted in this context.
Interpretation
Individuals with COPD represent a particularly high-risk population following NSTEMI, characterised by more severe clinical presentation, lower rates and delays in invasive management and poorer inpatient quality of care. These disparities were associated with a higher adjusted mortality risk extending up to 10 years. In contrast, individuals with asthma experienced more favourable long-term outcomes. Targeted strategies to improve risk recognition, optimise evidence-based care and ensure equitable access to invasive therapies and cardiac rehabilitation are essential to reduce the persistent excess mortality observed in patients with COPD.
Supplemental Material
sj-docx-1-ang-10.1177_00033197261416660 – Supplemental material for Assessing Disparities in Long Term Outcomes in Non-ST Elevation Myocardial Infarction According to Presence of Obstructive Airways Disease
Supplemental material, sj-docx-1-ang-10.1177_00033197261416660 for Assessing Disparities in Long Term Outcomes in Non-ST Elevation Myocardial Infarction According to Presence of Obstructive Airways Disease by Andrew Cole, Nicholas Weight, Mohamed Dafaalla, Thomas Shepherd, Richard Partington, Evangelos Kontopantelis, Muhammad Rashid and Mamas A. Mamas in Angiology
Footnotes
Acknowledgements
This study was conducted with support from the National Institute for Health and Care Research (NIHR) Birmingham Biomedical Research Centre (BRC). All authors made substantial contributions to the conception or design of the work; or the acquisition, analysis or interpretation of data; and drafting or reviewing the article critically for important intellectual content. All authors approved the final version to be published. MAM is responsible for the integrity of the work as a whole.
Ethical Considerations
Secondary use of anonymised Myocardial Ischaemia National Audit Project (MINAP) dataset for research purposes is authorised under United Kingdom (UK) National Health Service (NHS) research governance arrangements and further supported under section 251 of NHS act 2006 (NIGB: ECC1-06[d]/2011), which allows researchers to use patient information collected within the dataset for medical research without patient consent. Therefore, formal ethical approval was not sought for this study. Approval of the linkage of MINAP, Hospital Episode Statistics (HES) and Office for National Statistics (ONS) registries, all UK-based institutions, was granted by the Health and Care Research Wales and the Health Research Authority (Research Ethics Committee reference 20/WA/0312). Additionally, approval was obtained by the Confidentiality Advisory Group, an independent body providing expert advice on the use of confidential patient information for research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research is funded by the National Institute for Health and Care Research (NIHR) Birmingham Biomedical Research Centre (BRC). M.R. is funded by a NIHR Development and Skills Enhancement award, and Academy of Medical Sciences (AMS) grant (SGL025/1064). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: N.W. research fellowship salary is funded by Abbot (Abbott House, Vanwall Business Park, Vanwall Road, Maidenhead, Berkshire SL6 4XE, UK).
Data Availability Statement
The data underlying this article were provided by the National Institute for Cardiovascular Outcomes Research (NICOR). Data will be shared on reasonable request to the corresponding author with the permission of NICOR.
Supplemental Material
Supplemental material for this article is available online.
