Abstract
Patients with acute coronary syndromes (ACS) and atrial fibrillation (AF) have more complex conditions, making risk stratification crucial. The added discriminatory power of including anemia in the CHA2DS2-VASc ([congestive heart failure, hypertension, age [>65 = 1 point, >75 = 2 points], diabetes, previous stroke/systemic embolism/transient ischemic attack [2 points], vascular disease, age 65–74 years, and sex category]) score for poor prognosis in this population remains unclear. This study investigates the impact of anemia on 1166 patients with ACS and AF from 2017 to 2019. Multivariate Cox regression analysis showed anemia significantly increased the risk of all-cause mortality (Hazard ratio [HR] = 1.935, 95% CI: 1.106–3.384, P = .021) and major adverse cardiovascular events (MACE; HR = 1.857, 95% CI: 1.171–2.945, P = .009), but not thromboembolic events (HR = 0.651, 95% CI: 0.210–2.022, P = .458). Receiver operating characteristic (ROC) curves showed that hemoglobin’s area under the curve (AUC) for mortality and MACE was 0.722 and 0.710, respectively, improving to 0.770 and 0.772 when combined with the CHA2DS2-VASc score. Integrated discrimination improvement (IDI) and net reclassification improvement (NRI) metrics showed significant improvement in predicting mortality (NRI = 0.258, P = .004; IDI = 0.037, P < .001) and MACE (NRI = 0.232, P = .002; IDI = 0.026, P = .004). Adding hemoglobin to the CHA2DS2-VASc score enhances its predictive performance.
Introduction
Acute Coronary Syndrome (ACS) is a common critical condition in cardiovascular disease, with >7 million people diagnosed with ACS each year. 1 Atrial fibrillation (AF) is the most common cardiac arrhythmia that shares many risk factors with ACS, increasing mortality risk and adverse prognosis in patients with ACS.2 –4 It is critical to identify patients at high risk for ACS combined with AF and perform a comprehensive risk assessment to optimize treatment. The CHA2DS2-VASc ([congestive heart failure, hypertension, age [>65 = 1 point, >75 = 2 points], diabetes, previous stroke/systemic embolism/transient ischemic attack [2 points], vascular disease, age 65–74 years, and sex category]) score is an important clinical tool for evaluating thromboembolic risk in patients with AF. 4 Many researchers have investigated its broader applicability, especially in the context of cardiovascular diseases.5,6 Current guidelines recommend that the CHA2DS2-VASc score be used to assess risk and guide clinical practice in patients with ACS and AF.4,7 However, some risk factors or predictors associated with poor prognosis are not included in this score. In addition, the score lacks laboratory parameters that reflect the condition of the patients in vivo.
According to the World Health Organization definition, 8 anemia depends on the hemoglobin value. Low hemoglobin levels have been shown to be associated with poor prognosis in ACS patients.9 –11 Compared with ACS, clinical researches focusing on the relationship between hemoglobin and the AF prognosis are limited. A few studies have found anemia to be associated with poor prognosis in patients with AF.12 –15 They were consistent that anemia increases the risk of death in patients with AF, but whether it increases the risk of stroke/embolism is uncertain.12,14 –16 Limited clinical evidence provides uncertainty about the relationship between anemia and adverse outcomes in patients with ACS and AF. This study focused on whether anemia was an independent predictor of poor prognosis in patients with ACS and AF and discussed whether hemoglobin can improve the predictive ability of the CHA2DS2-VASc score.
Materials and Methods
Study Population and Data Collection
The study population was derived from an observational, prospective, single-center registry study conducted at Fuwai Hospital (Chinese Academy of Medical Sciences) that enrolled patients with ACS and AF from January 2017 to December 2019. Inclusion criteria were age ≥18 years, with ACS and AF diagnosed by reviewing clinical records and electrocardiograms and those who survived at discharge and completed 1 year of follow-up. Exclusion criteria were post mechanical valve replacement and moderate-to-severe mitral stenosis. Patients with incompletely documented clinical data were also excluded. The study was approved by the Ethics Committee of Fuwai Hospital in accordance with the Declaration of Helsinki. All patients provided a signed informed consent.
Trained investigators collected baseline information on all participants by communicating with patients, consulting with physicians, and surveying medical records. The CHA2DS2-VASc ([congestive heart failure, hypertension, age [>65 = 1 point, >75 = 2 points], diabetes, previous stroke/systemic embolism/transient ischemic attack [2 points], vascular disease, age 65–74 years, and sex category]) and HAS-BLED ([uncontrolled hypertension [systolic blood pressure >160 mm Hg], abnormal renal function, abnormal hepatic function, previous ischemic or hemorrhagic stroke, bleeding history or predisposition, elderly [age >65 years], concomitant use of antiplatelet or NSAID, excessive alcohol intake per week, and labile international normalized ratio (INR) for patients receiving warfarin with >3 INR measurements]) scores of the patients were calculated. ACS is a series of conditions that include unstable angina (UA), non-ST-segment elevation myocardial infarction (STEMI), or ST-segment elevation myocardial infarction (NSTEMI). Fasting venous blood samples were collected from anterior elbow veins. According to the reference values in the report card of Fuwai Hospital, admission troponin over 0.02 ng/mL was defined as elevated troponin. Hemoglobin levels were measured using an automated hematology analyzer (XT-1800i, Sysmex, Japan), and samples were evaluated by standard of care.
Clinical follow-up visits were scheduled in months 1, 6, and 12 after each individual patient was discharged from the hospital and were conducted by telephone, on-site visits, and medical record review. The primary endpoint was defined as 1-year all-cause death and the occurrence of thromboembolic events (ischemic stroke and systemic embolism). The secondary end point was defined as 1-year major adverse cardiovascular events (MACE), including a composite of all-cause death, myocardial infarction, and stroke. All clinical end points were determined by a clinical events committee that was unaware of the patient’s clinical characteristics.
Statistical Analysis
The population was divided into two groups according to anemia and non-anemia. Anemia was defined as hemoglobin <130 g/L for men and <120 g/L for women. 8 Continuous variables were expressed as median and interquartile range. Categorical variables were expressed as frequencies and percentages. Comparisons between groups were analyzed using t-test, Mann-Whitney U-test, Pearson’s chi-square test, and Fisher’s exact test as appropriate. Multivariate Cox regression analyses were used for all-cause mortality and MACE, which incorporated baseline factors that were significantly different (P < .05) in relation to mortality between the two groups in univariate analyses, as well as clinically significant variables. For the analysis of thromboembolic events, anemia/hemoglobin and CHA2DS2-VASc score variables were incorporated into the multivariate Cox regression model. The presence of anemia was assessed for relative risk to the endpoint outcome using Kaplan-Meier curves and log-rank tests. A receiver operating characteristic (ROC) curve was plotted to evaluate the discriminatory ability of different parameters, and the corresponding area under the curve (AUC) was calculated. The incremental predictive value from adding hemoglobin to the CHA2DS2-VASc score to predict all-cause mortality and MACE was analyzed from these predicted probabilities using several measures of improvement in discrimination: net reclassification improvement (NRI) and integrated discrimination improvement (IDI). Statistical significance required a two-sided P < .05. Data were analyzed using SPSS version 26.0 software (IBM Corporation, Chicago, IL, USA), R version 4.0.2 software (Vienna, Austria) and GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA) for statistical analysis and plotting.
Results
Comparison of Baseline Characteristics for Anemia and Non-Anemia patients
This study investigated 1166 patients with ACS with AF, and the baseline characteristics of the population were categorized into 2 groups based on the presence or absence of anemia. In this study, 231 patients (19.8%) were in the anemia status. Compared with the no-anemia group, the anemia group was significantly older and had a higher prevalence of heart failure, diabetes mellitus, chronic obstructive pulmonary disease, peripheral arterial disease, and prior myocardial infarction. In addition, median hemoglobin, albumin, and LVEF (Left ventricular ejection fraction) were lower in the anemia group than in the non-anemia group, whereas median creatinine values, blood urea nitrogen, and NT-proBNP (N-terminal pro-B type natriuretic peptide) were significantly higher than in the non-anemia group (Table 1).
Baseline Characteristics of Included Patients.
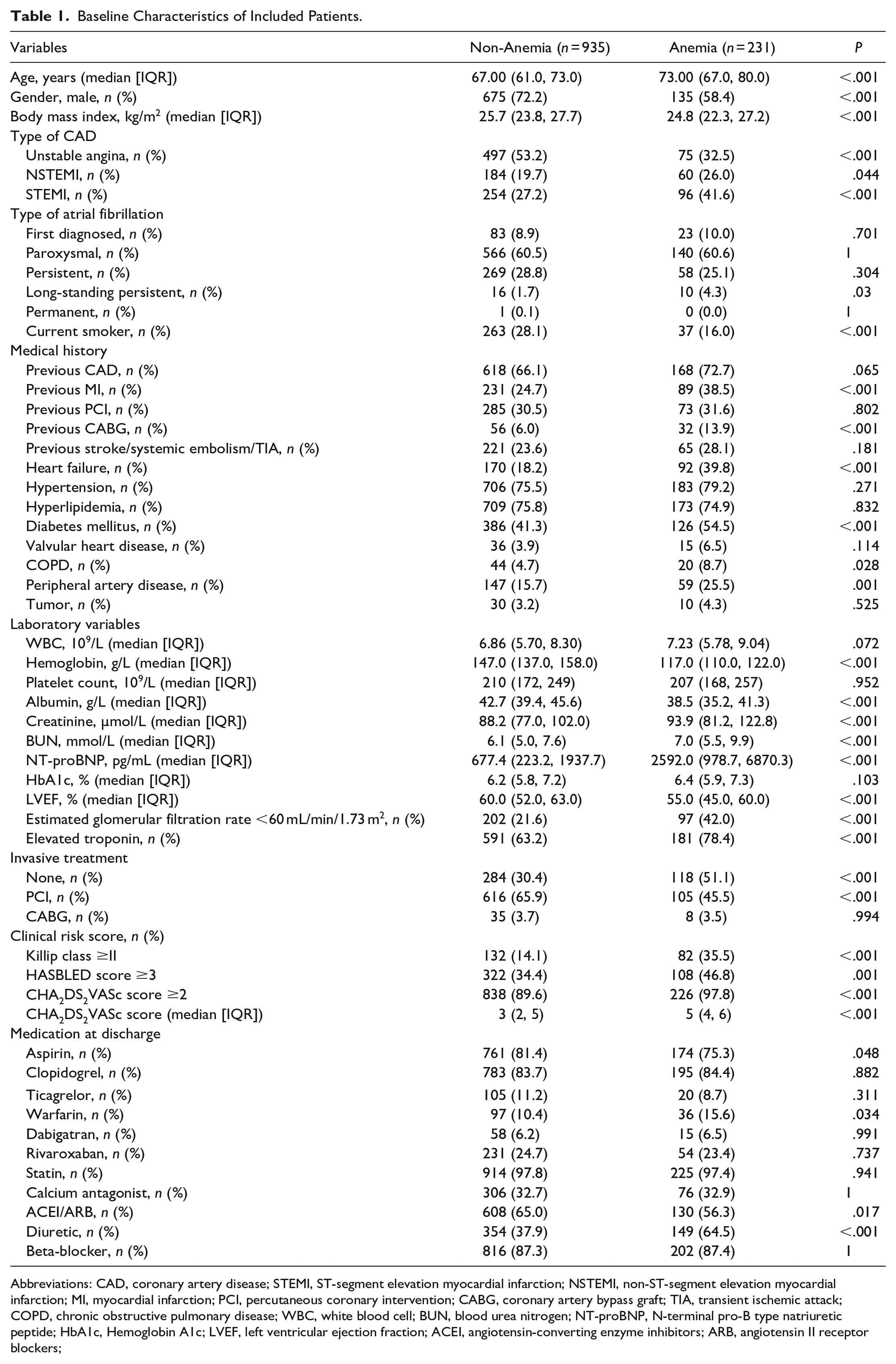
Abbreviations: CAD, coronary artery disease; STEMI, ST-segment elevation myocardial infarction; NSTEMI, non-ST-segment elevation myocardial infarction; MI, myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; TIA, transient ischemic attack; COPD, chronic obstructive pulmonary disease; WBC, white blood cell; BUN, blood urea nitrogen; NT-proBNP, N-terminal pro-B type natriuretic peptide; HbA1c, Hemoglobin A1c; LVEF, left ventricular ejection fraction; ACEI, angiotensin-converting enzyme inhibitors; ARB, angiotensin II receptor blockers;
Relationship Between Hemoglobin and Clinical Outcomes in Patients With ACS and AF
For the primary outcome of all-cause mortality, we found that hemoglobin was associated with an increased risk of mortality both as a continuous variable and as a categorical variable (anemic and non-anemic; Table 2). Univariate Cox regression analysis showed that the HR (Hazard ratio) of hemoglobin for all-cause mortality was 0.959 per unit increase (95% CI: 0.949–0.970, P < .001). After adjustment for sex, age, BMI (Body mass index), current smoker, type of CAD (Coronary artery disease), heart failure, diabetes mellitus, valvular heart disease, tumor, previous MI (Myocardial infarction), previous CABG (Coronary artery bypass graft), previous stroke/systemic embolism/TIA (Transient ischemic attack), WBC (White blood cell), albumin, creatinine, BUN (blood urea nitrogen), NT-proBNP, estimated glomerular filtration rate <60 mL/min/1.73 m2 (n%), LVEF, elevated troponin, Killip class ≥II, HAS-BLED score ≥3, CHA2DS2VASc score, aspirin, diuretic, and invasive treatment, the HR of hemoglobin for all-cause mortality was 0.979 per unit increase (95% CI: 0.965–0.993, P = .004). This association remained consistent when hemoglobin was categorized by standard principles into anemic and non-anemic groups. The multivariate Cox regression showed the HR of the anemic group was 1.935 (95% CI: 1.106–3.384, P = .021) compared with the non-anemic group. A similar trend was observed for the secondary outcome of MACE. Kaplan-Meier survival curves are shown in Figures 1 and 2.
Associations Between Hemoglobin and Clinical Outcomes by Cox Analysis.
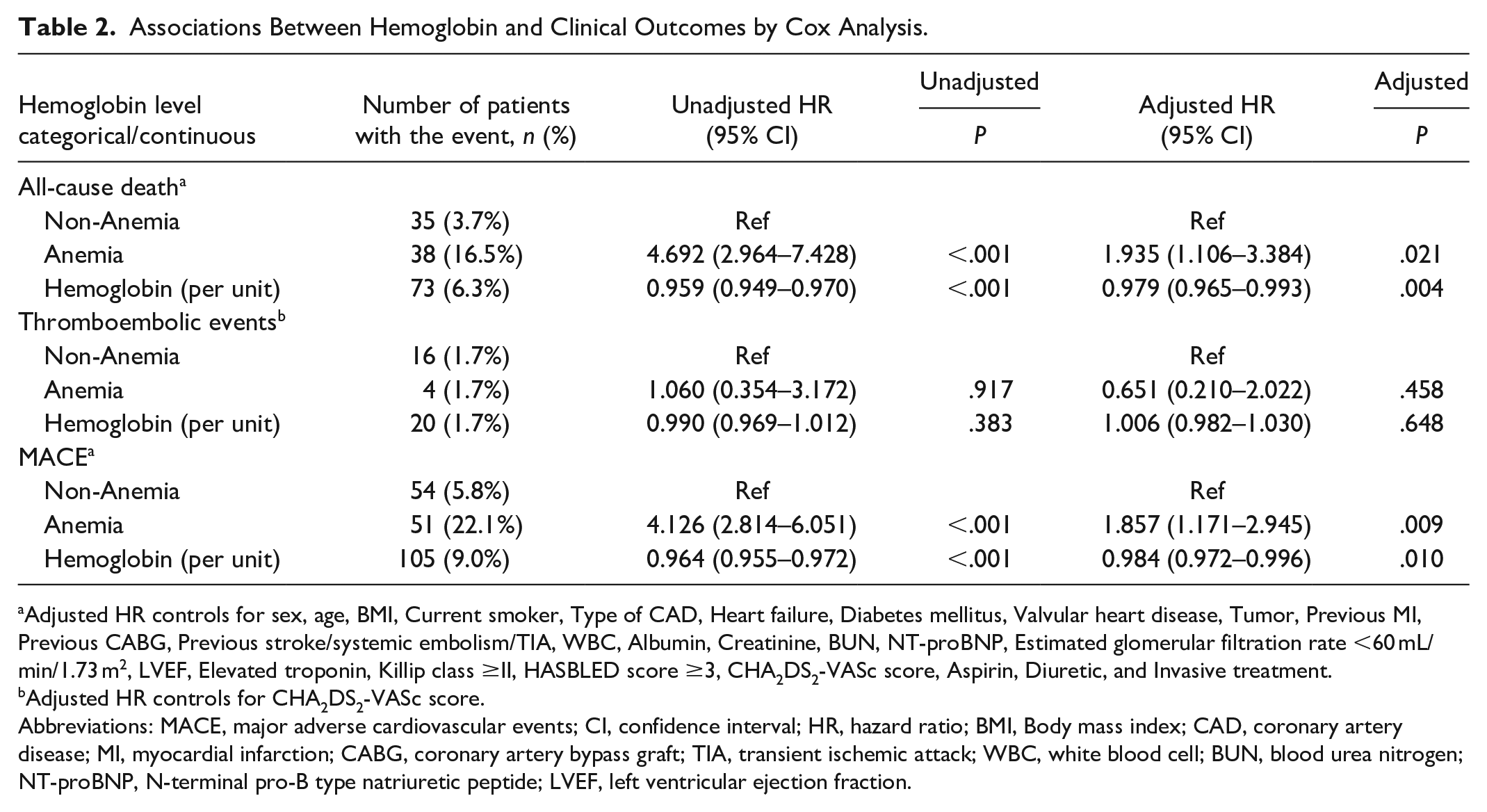
Adjusted HR controls for sex, age, BMI, Current smoker, Type of CAD, Heart failure, Diabetes mellitus, Valvular heart disease, Tumor, Previous MI, Previous CABG, Previous stroke/systemic embolism/TIA, WBC, Albumin, Creatinine, BUN, NT-proBNP, Estimated glomerular filtration rate <60 mL/min/1.73 m2, LVEF, Elevated troponin, Killip class ≥II, HASBLED score ≥3, CHA2DS2-VASc score, Aspirin, Diuretic, and Invasive treatment.
Adjusted HR controls for CHA2DS2-VASc score.
Abbreviations: MACE, major adverse cardiovascular events; CI, confidence interval; HR, hazard ratio; BMI, Body mass index; CAD, coronary artery disease; MI, myocardial infarction; CABG, coronary artery bypass graft; TIA, transient ischemic attack; WBC, white blood cell; BUN, blood urea nitrogen; NT-proBNP, N-terminal pro-B type natriuretic peptide; LVEF, left ventricular ejection fraction.
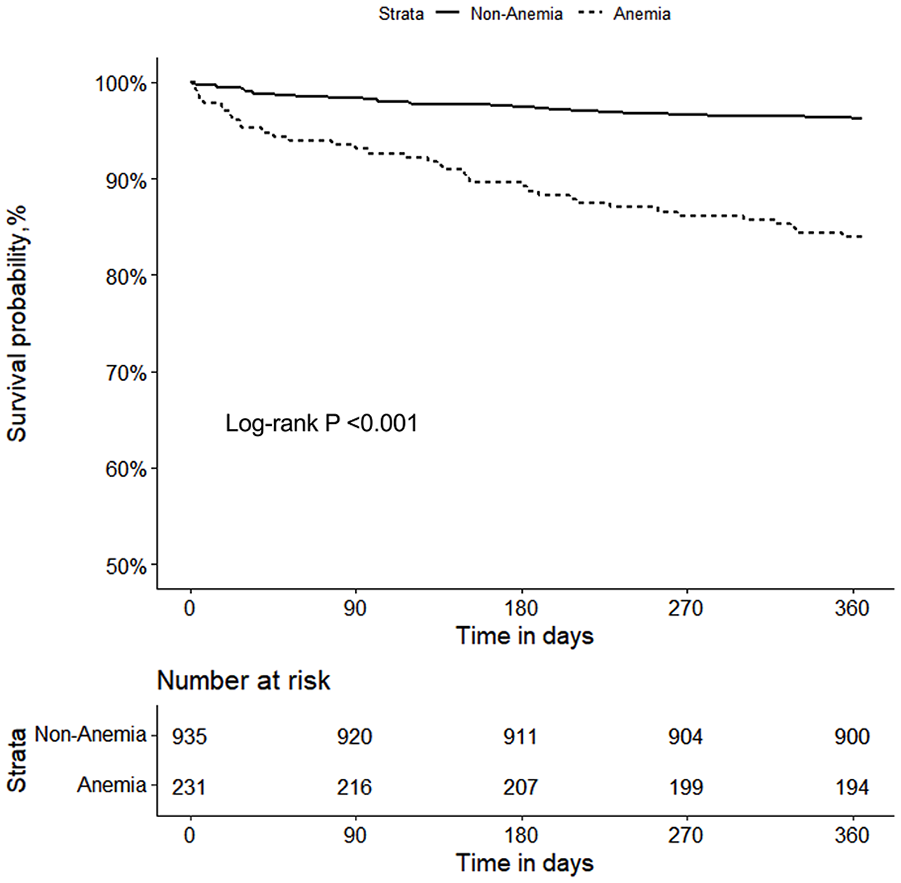
Kaplan-Maier curves for all-cause mortality between the anemia and non-anemia groups.
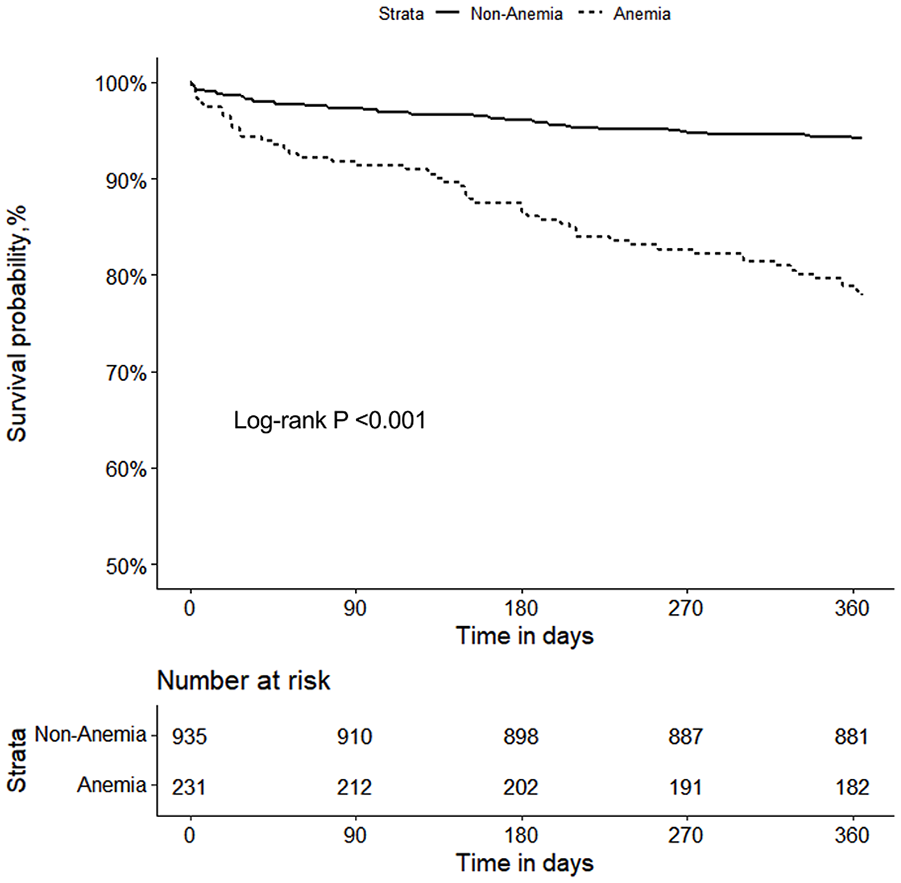
Kaplan-Maier curves for MACE between the anemia and non-anemia groups.
However, for the primary outcome of thromboembolic events, both as a continuous variable and a categorical variable (anemia vs non-anemia), hemoglobin was found to be unrelated to the risk of thromboembolic events (Table 2).
Predictive Performance of Hemoglobin
We examined the ability of hemoglobin as predictors of all-cause mortality and MACE in patients with ACS and AF using ROC curves, and the AUC values of these curves indicated that hemoglobin demonstrated good predictive performance for all-cause mortality (AUC = 0.722, 95% CI: 0.652–0.792, P < .001) and MACE (AUC = 0.710,95% CI: 0.653–0.768, P < .001; Figure 3). In addition, the ROC curves showed that the CHA2DS2-VASc score combined with hemoglobin compensated for the relatively deficient regions of hemoglobin and CHA2DS2-VASc score (Figures 4 and 5). Next, we evaluated the potential incremental prognostic value of hemoglobin for the addition of hemoglobin to the CHA2DS2-VASc score. We found that differentiation and reclassification of all-cause mortality prediction (NRI = 0.258, 95% CI: 0.120–0.381, P = .004; IDI = 0.037, 95% CI: 0.008–0.084, P < .001) and MACE (NRI = 0.232, 95% CI: 0.110–0.312, P = 0.002; IDI = 0.026, 95% CI: 0.005–0.062, P = .004) prediction were significantly improved when hemoglobin was combined with the CHA2DS2-VASc score (Table 3).
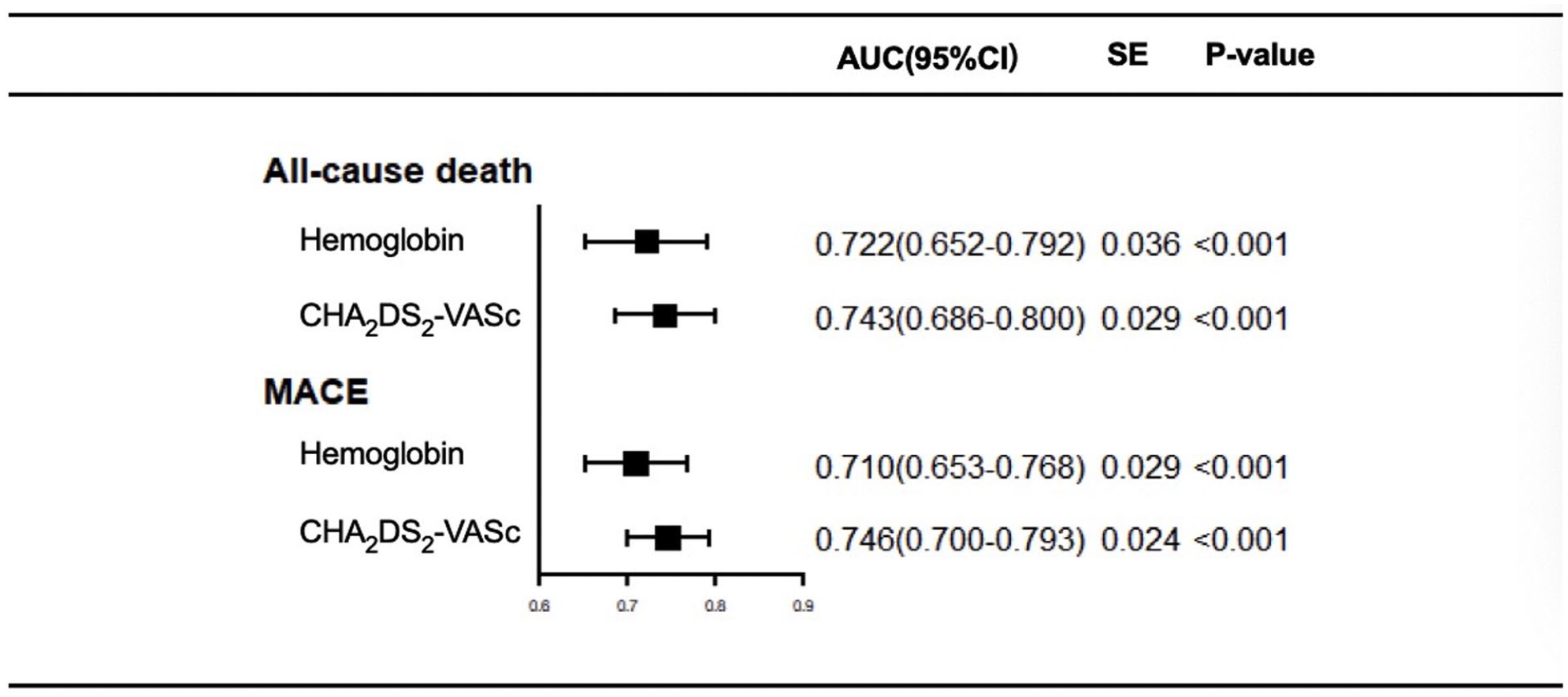
AUC of hemoglobin and CHA2DS2-VASc score for clinical outcomes.
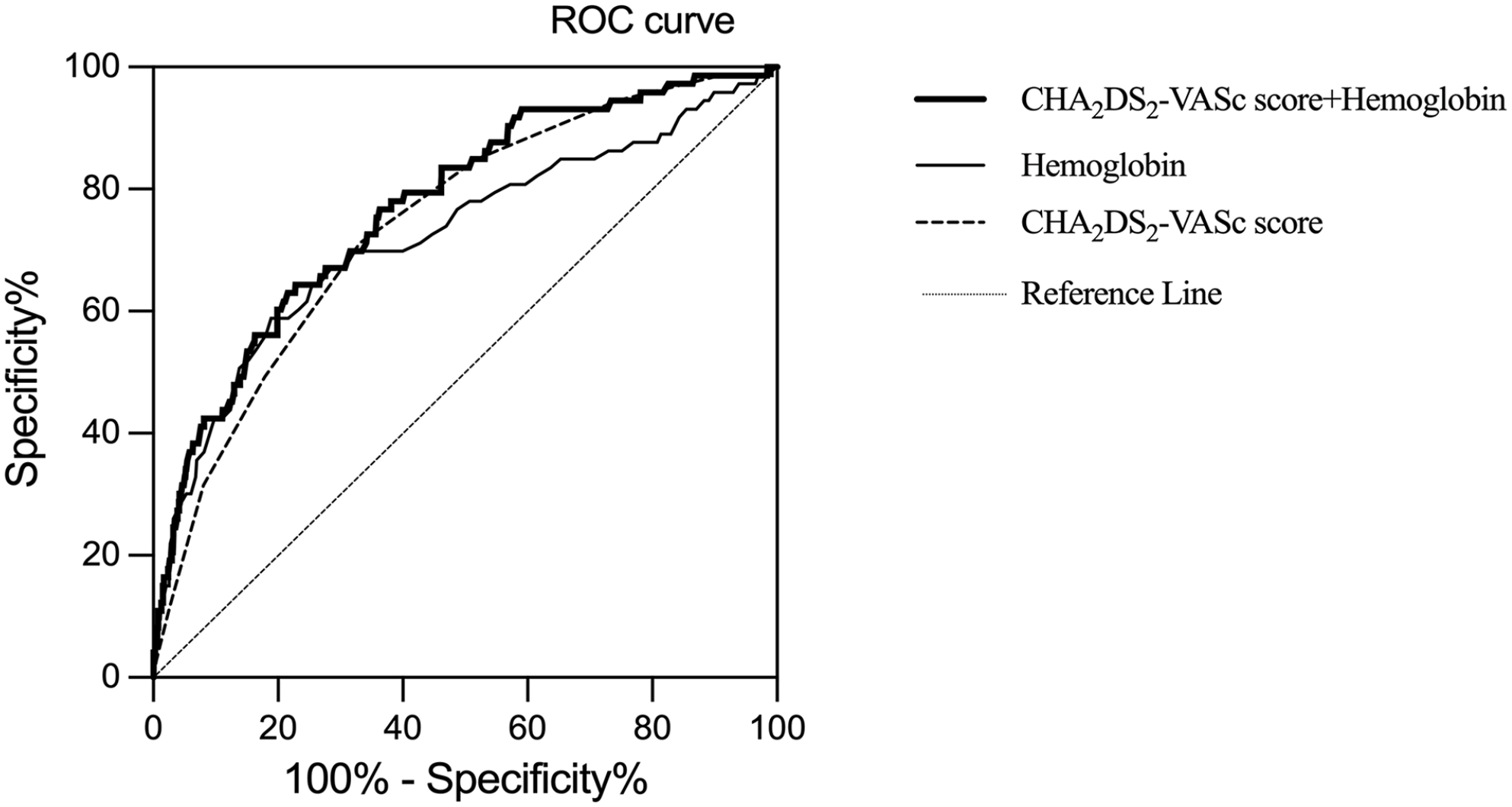
ROC curves of the CHA2DS2-VASc score and addition of hemoglobin to the CHA2DS2-VASc score for all-cause death.
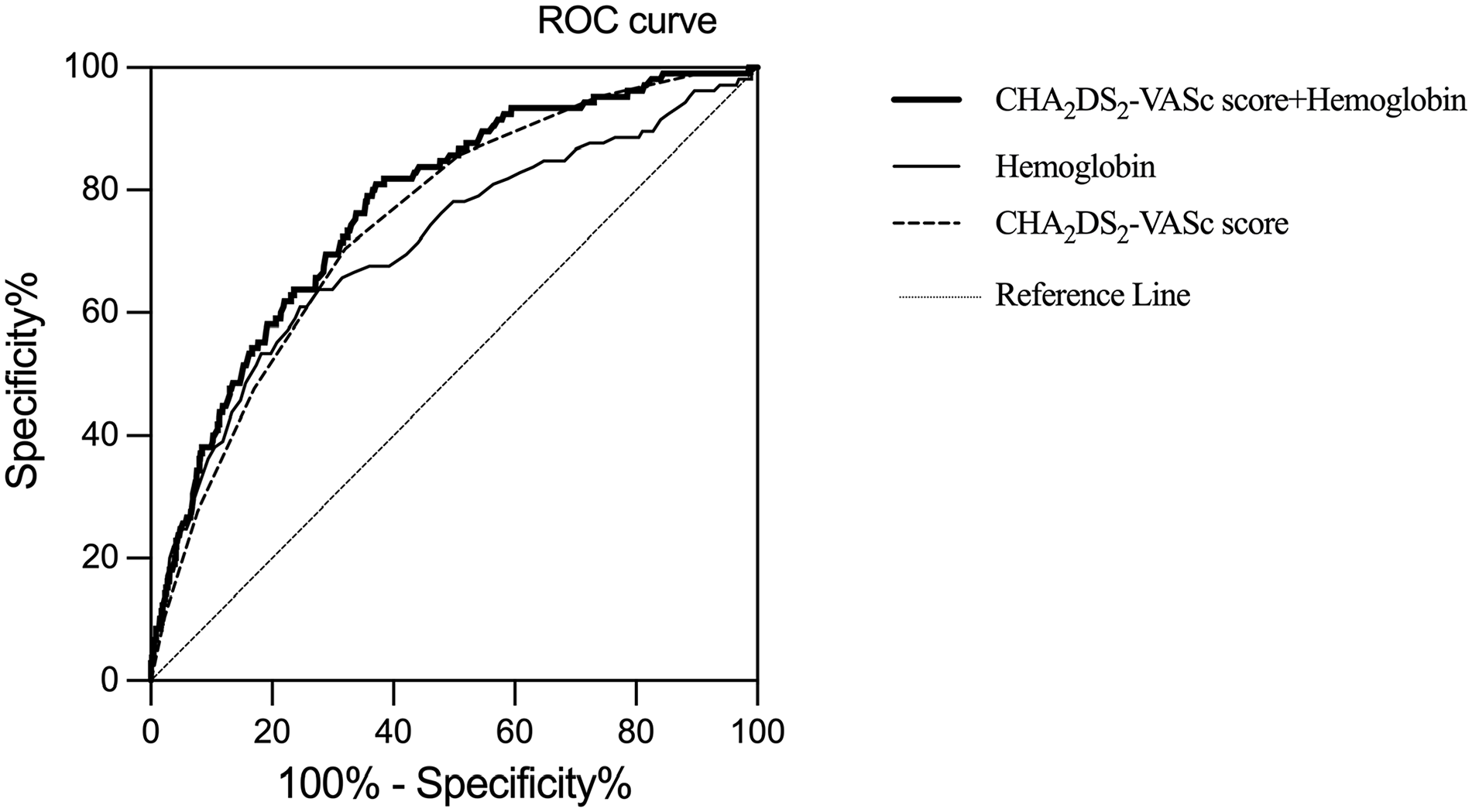
ROC curves of the CHA2DS2-VASc score and addition of hemoglobin to the CHA2DS2-VASc score for MACE.
Assessment the Incremental Prognostic Value in the Addition of Hemoglobin to the CHA2DS2VASc Score to Predict Clinical Outcomes.

Abbreviations: MACE, major adverse cardiovascular events; NRI, net reclassification improvement; IDI, integrated discrimination improvement; CI, confidence interval; CHA2DS2-VASc,Congestive Heart Failure, Hypertension, Age ≥75 [Doubled], Diabetes Mellitus, Prior Stroke/systemic embolism/Transient Ischemic Attack [Doubled], Vascular Disease, Age 65–74, Female.
Discussion
The present study found that low hemoglobin levels at admission were significantly associated with 1-year all-cause mortality and MACE in patients with ACS and AF. Additionally, the presence of anemia was associated with a significantly increased risk of all-cause mortality and MACE in our population compared with absent anemia. Moreover, adding hemoglobin to the CHA2DS2VASc score can improve the predictive value of long-term adverse cardiovascular events in patients with ACS and AF, contributing to subsequent management decisions. However, hemoglobin, whether considered as a continuous variable or a categorical variable (anemia and non-anemia), showed no association with the risk of thromboembolism.
ACS is a common cardiovascular emergency and AF is a common arrhythmia. 1 These two conditions share many risk factors, such as diabetes, hypertension, and advanced age, which often coexist in patients.2 –4 The interaction between coronary artery disease and AF forms a vicious cycle. 17 Besides shared risk factors, mechanisms such as inflammation, atrial ischemia, and left ventricular remodeling further complicate the clinical scenario in patients with both ACS and AF, increasing their risk. 18 Additionally, compared with patients with ACS alone, those with non-valvular AF (NVAF) are often older and have a higher burden of comorbidities. 3 Previous studies have shown that patients with ACS and NVAF have worse short-term and long-term prognosis compared with those with ACS alone.3,19 In conclusion, patients with ACS and AF present more complex and challenging characteristics in clinical practice. When exploring the complex management challenges of this population, identifying valuable biomarkers or factors exacerbating their adverse prognosis is particularly crucial.
Hemoglobin is usually obtained from blood routine examination after admission to the hospital. It is an important indicator of the presence of anemia. As with AF and coronary heart disease, the prevalence of anemia increases with age. 20 Previous studies have shown that anemia is associated with increased mortality in patients with AF.12,14 –16 Anemia is also linked to various adverse cardiovascular prognoses in patients with ACS, including all-cause mortality and adverse cardiovascular events.9,21 An earlier study suggested that the higher 1-year mortality rate in patients with acute infarction and anemia compared with non-anemic patients might be due to older patients and higher comorbidities. 22 However, subsequent studies and clinical trials have indicated that anemia is an independent risk factor for mortality in ACS.9,23 –25 In our study, anemia can predict all-cause mortality in this population. Anemia also can predict composite cardiovascular adverse events, including all-cause death, myocardial infarction, and stroke. In this study, anemic patients similarly faced the complexities of advanced age and higher comorbidities. However, even after adjusting for comorbidities and age, anemic patients still had worse clinical outcomes. Furthermore, considering the complex relationship between chronic kidney disease and anemia, this study included low glomerular filtration rate, blood urea nitrogen, and creatinine in the multivariate Cox analysis for all-cause mortality and MACE, and still observed an independent correlation between anemia and poor prognosis in this population. The association of low hemoglobin levels with poor prognosis in patients with ACS and AF may be explained by several points. First, hemoglobin can reflect body states including malnutrition and inflammation. In ACS patients, low hemoglobin may be linked to in vivo inflammatory responses, which are relevant to poor prognosis during follow-up. 26 Second, Anemia may reduce the deformability and increase the viscosity of microcytic red blood cells, which may contribute to abnormal blood flow patterns within the vessels, thereby increasing the risk of thromboembolism. 27 Third, the relative ischemic status of the body due to anemia may cause changes in cardiac electrophysiology and sympathetic activation.28,29 Lastly, the limitations of our study prevented us from fully exploring the interplay between anemia and disease, as well as between AF and ACS, which may have influenced our results. Future studies are needed to explore these mechanisms more deeply.
The correlation between anemia and increased thromboembolic events/strokes in patients with AF remains controversial.12,14 –16 This study tries to answer this question. It should be clear that our study focused on ACS and AF patients. However, we did not observe that anemia was independently associated with an increased risk of thromboembolism in this population of patients. This finding aligns with the conclusions of a large registry study focused on AF patients. 12 Puurunen et al. 16 explored the impact of anemia on clinical outcomes in patients undergoing PCI and found no statistically significant difference in the 1-year stroke/transient ischemic attack incidence between the anemic and non-anemic groups. Unfortunately, this study did not further investigate whether anemia is an independent risk factor for thromboembolic events. Our results suggest that the relationship between anemia and thromboembolic risk observed in some studies may not be generalizable to patients with both AF and ACS. The interaction between AF and ACS could contribute to this discrepancy. Additionally, the limited number of thromboembolic events in our study may have influenced our findings. Future studies with larger sample sizes and more comprehensive data are needed to validate our findings and clarify the potential role of anemia as an independent risk factor for thromboembolism in patients with AF and ACS.
Another important finding of this study is that incorporating hemoglobin into the CHA2DS2-VASc score can enhance its ability to predict all-cause mortality and MACE in patients with AF and ACS. The CHA2DS2-VASc score is a valuable tool in clinical practice for assessing the risk of thromboembolic events in patients with AF. 4 Researchers are continually exploring the value of the CHA2DS2-VASc score in various diseases, such as predicting all-cause mortality in patients with chronic kidney disease and COVID-19.30,31 More studies have been conducted in the field of cardiovascular diseases.5,6,32 –35 Previous studies indicate that CHA2DS2-VASc effectively predicts coronary atherosclerosis in ACS patients and long-term MACE risk in ACS patients undergoing Percutaneous Coronary Intervention (PCI).36,37 A multicenter study indicated that CHA2DS2-VASc effectively predicts all-cause mortality and MACE in AF patients undergoing coronary stent treatment. 38 Current guidelines recommend that the CHA2DS2-VASc score be used to assess risk and guide clinical practice in patients with ACS and AF.4,7 This study found that the CHA2DS2-VASc score demonstrated good predictive performance for long-term all-cause mortality and MACE in AF patients with concurrent ACS. The addition of hemoglobin improved its predictive value and presented robust results. Although the improvement in the area under the curve (AUC) was modest, the receiver operating characteristic (ROC) curve showed that the combined CHA2DS2-VASc score with hemoglobin compensated for the relatively deficient regions of hemoglobin and CHA2DS2-VASc score. It is important to note that the CHA2DS2-VASc score already incorporates several significant clinical risk factors, meaning the contribution of additional variables may be somewhat limited. However, the CHA2DS2-VASc score lacks laboratory parameters that reflect the internal status of the patient, and hemoglobin can partially address this gap. Incorporating anemia as an additional variable into the scoring system may enable more precise risk stratification. Furthermore, hemoglobin is typically obtained through routine blood tests upon hospital admission, providing a quick and simple method of assessment. Its low cost, high accessibility, and potential value in predicting mortality make it a highly applicable tool in clinical practice. These characteristics suggest that hemoglobin has strong potential for widespread use in clinical settings.
To our knowledge, this study is the first to explore the relationship between hemoglobin levels and all-cause mortality, thromboembolic events, and MACE in patients with ACS and AF. By identifying anemia as a significant predictive factor, healthcare providers can implement more personalized treatment plans. For example, actively addressing and managing anemia may reduce the overall mortality risk in patients with AF and ACS. Furthermore, we found that adding hemoglobin to the CHA2DS2-VASc score demonstrated robust predictive performance improvement. The modified CHA2DS2-VASc score incorporating anemia contributes to better risk stratification of AF patients and ACS. Clinicians can more accurately identify high-risk patients, ensuring that they receive appropriate monitoring and interventions, potentially improving patient outcomes and optimizing the use of healthcare resources.
This study has some limitations. First, only admission hemoglobin levels were used for assessment. This approach does not account for dynamic changes in hemoglobin over time, which could provide additional insights into patient outcomes. However, dynamic monitoring of hemoglobin changes increases the economic cost and complexity of clinical practice. Future studies should explore the impact of these dynamic changes to provide a more comprehensive understanding. Second, although we used multivariate Cox regression to adjust for potential confounding factors, it is impossible to completely eliminate the influence of all variables affecting adverse outcomes. This limitation is inherent in most observational studies and emphasizes the need for cautious interpretation of our results. Finally, this study is a post hoc analysis of a large, observational, prospective, single-center registry, which may affect the generalizability of the results. Larger sample sizes and multicenter studies are needed to further validate these findings.
Conclusions
Anemia is closely associated with an increased risk of all-cause mortality and MACE in patients with ACS and AF. Moreover, adding hemoglobin to the CHA2DS2-VASc score improves its predictive performance for 1-year all-cause mortality and MACE. Attention to anemia in patients with AF and ACS is necessary to improve prognosis.
Footnotes
Author Contribution
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Capital’s Funds for Health Improvement and Research (No. 2018-2-4031), High-Level Hospital Clinical Research Funding (ID: 2022-GSP-GG-26), and Beijing key clinical specialty construction project.
