Abstract
Atherosclerotic peripheral artery disease (PAD) and chronic kidney disease (CKD) are highly interconnected diseases, while causes for excess mortality are not well-defined. Fibroblast growth-factor 23 (FGF-23) is elevated in mineral bone disease of CKD and was shown to be associated with higher mortality. However, it is not known if this association extends to PAD. FGF-23 was measured by ELISA in serum samples of 298 patients with stable PAD (Fontaine stage I-II) with an estimated glomerular filtration rate (eGFR) of 72 (58–85) ml/min. Mortality was assessed after a long-term follow-up of up to 10 years. FGF-23 showed significant associations with markers of metabolic syndrome (triglycerides r = .25, P < .001, high-density lipoprotein cholesterol (HDL-C) r = −.28, P < .001, c-reactive protein (CRP) r = .14, P = .016). Multivariable Cox-regression outcome analyses showed significant associations between FGF-23 and all-cause mortality (hazard ratio 1.35, 95% confidence interval 1.05–1.74) in PAD patients even after adjustment for traditional cardiovascular risk factors and renal excretory function. FGF-23 is associated with higher mortality in patients with PAD. Our findings indicate that FGF-23 has detrimental effects on patients with PAD that are independent of renal excretory function.
Background
Patients suffering from peripheral artery disease (PAD) are at excess risk both for all-cause and cardiovascular mortality. 1 Improvements in therapeutic management in recent years, especially with novel lipid-lowering therapies2,3 and better glycemic control in those with diabetes4–6 led to strong risk reductions. However, in patients with PAD fatal outcome rates are generally still high, but especially in comparison with patients with coronary artery disease (CAD). 7 Thus, further risk stratification beyond traditional risk factors is necessary to identify patients at elevated risk.
Chronic kidney disease (CKD) and disturbances in calcium/phosphate homeostasis within the context of CKD-MBD (chronic kidney disease—mineral and bone disease) lead to increased levels of vascular calcification and further atherosclerotic disease manifestations. 8 Even slightly increased levels of phosphate provoke vascular calcification and increase cardiovascular (CV) death risk. 9 Yet, the underlying mechanisms are not sufficiently understood, and adequate risk stratification markers are not established in clinical practice.
Fibroblast-growth-factor 23 (FGF-23) maintains phosphate homeostasis 10 after secretion from osteocytes. 11 FGF-23 regulates phosphate levels by three distinct mechanisms. Firstly, phosphate reabsorption is inhibited in the proximal tubuli leading to an increased urinary phosphate excretion. Secondly, via inhibition of 1,25 dihydroxy vitamin D synthesis the intestinal uptake of phosphate is reduced.12,13 Thirdly, FGF-23 inhibits parathyroid hormone (PTH) synthesis in the parathyroid glands. 13 Elevated FGF-23 levels are the first measurable alterations in CKD-MBD and are elevated in the very early stages of CKD. 14
Both intimal and medial calcification are relevant and abundantly present in atherosclerotic lesions in PAD 15 as well as in CKD. Especially patients with severe CKD are affected by pronounced vascular calcification in terms of CKD-MBD. 16 In those patients, FGF-23 levels are particularly elevated. 13 While it has been shown that FGF-23 levels are associated with incidental PAD, 17 little is known about the relevance of FGF-23 as a predictive tool for PAD outcome and severity of prevalent PAD. We thus speculated that FGF-23 is a relevant marker for mineral homeostasis dysregulation in PAD even at the early stages of CKD and tested its relevance for outcome prediction in patients with stable PAD and mild to moderate CKD with a long-term follow-up.
Methods
Patients
Patients for this study were selected from the Vascular Medicine Center (VMC) Vienna cohort. All of these patients were treated for stable PAD, either symptomatic with claudication or asymptomatic in the outpatient department of a tertiary care center for vascular medicine. Patients with critical limb-threatening ischemia or planned revascularization at inclusion were excluded. Further exclusion criteria were end-stage kidney disease, connective tissue disease, or known malign disease. A detailed study protocol was reported previously. 18 In brief, patients were followed at the outpatient ward either every 6 or 12 months and traditional CV risk factors were stringently optimized according to current clinical guidelines.19,20
All study-related procedures were performed after approval by the institutional review board of the Medical University of Vienna and complied with the Declaration of Helsinki in its current revision, Good Clinical Practice guidelines21,22 and strengthening the reporting of observational studies in epidemiology (STROBE) guidelines. All patients gave their written informed consent prior to study inclusion and procedures. Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Patient Evaluation and Data Collection
At each visit, a standardized questionnaire was completed. The evaluation included anthropometric measurements, smoking habits, and previous CV events. PAD was defined by either previous peripheral revascularization or an ankle-brachial index (ABI) <0.9. Clinical symptoms were graded by the Fontaine classification for self-reported walking impairment. 19
ABI was measured by experienced technicians with a non-invasive Doppler sonographic probe measurement (ELCAT VL5000, Wolfratshausen, Germany). Laboratory measurements included standard renal and liver parameters as well as a lipid panel and HbA1c. Calcium and phosphate levels were not measured in this study, which is a limitation, as stated in the discussion section. Blood was drawn after an overnight fasting process. Spot urine samples were collected at visits in the first year of the study. Further collected blood and urine specimens were immediately centrifuged at 1200 rpm for 15 min and stored at −80°C until further measurements.
Definition of Co-morbidities
Arterial hypertension was defined as systolic blood pressure of ≥140 mmHg and/or diastolic blood pressure of ≥90 mmHg in at least two measurements or the intake of any antihypertensive medication. 23 Type 2 diabetes mellitus was defined with a fasting plasma glucose level >125 mg/dl (7.0 mmol/l) or a plasma glucose level >200 mg/dl (11.1 mmol/l). An oral glucose tolerance test was performed in case of unknown glycemic status. According to the American Diabetes Association (ADA) guidelines, 20 type 2 diabetes mellitus was additionally defined by an HbA1c >6.5% (48 mmol/mol) or intake of anti-diabetic medication. Former smoking was defined as previous smoking of at least 100 cigarettes. Smoking one pack of cigarettes (n = 20)/day was defined as one pack-year.
Calculation of Indexes and Categorization of Deviations From the Norm
Body mass index (BMI) was calculated as body weight in kg divided by squared body height in meters (kg/m2). Spot urinary albumin-to-creatinine ratio (UACR) >30 mg/g was classified as micro-albuminuria and >300 mg/g as macro-albuminuria. Proteinuria was not evaluated in this study. The estimated glomerular filtration rate (eGFR) was calculated by the Chronic Kidney Disease Epidemiology (CKD-EPI) equation. 24 Ankle-brachial index (ABI) was calculated according to the Transatlantic Inter-Society Consensus Document (TASC) criteria 25 by dividing the higher ankle pressure by the higher brachial pressure. In cases with incompressible ankle arteries (ABI >1.4), patients were diagnosed with medial sclerosis.
Outcome Assessment
Outcome data including the ICD-10 code and the date were retrieved from the Austrian central death registry. Hospital letters or autopsy reports were further utilized for adjudication of death causes, as available. CV mortality was defined by the ICD-10 diseases of the circulatory system (I00-I99 code); 107 patients died during the 10-year observation period (56.6% of those were categorized as CV death).
Measurement of FGF-23
Serum samples were drawn at the 12-month follow-up visit, immediately centrifuged, and stored at −80°C until measurement. After a standardized overnight thawing process, serum FGF-23 (C-terminal) was measured on a precoated commercially available ELISA (Biomedica GmbH, Vienna, Austria). Calculated intra-assay and inter-assay coefficients of variation of the measured samples were 5.7% and 8.3%, respectively.
Statistics
Data are presented as mean ± standard deviation (SD) or median (25th 75th percentile), as appropriate. Student’s unpaired t-test, as well as χ2-test, were used, as appropriate. Differences between multiple groups were analyzed by analysis of variance (ANOVA) or the Kruskal-Wallis test, as appropriate. In the case of non-normal distribution, variables were log-transformed. Normal distribution was analyzed by the Kolmogorov-Smirnov test and by visual approximation for parametric statistics if needed. Bivariate correlation was estimated using the Pearson correlation coefficient. Survival curves were calculated by the Kaplan-Meier method and compared using the log-rank test. Cox regression analysis was performed to estimate the effect size and to allow for multivariable adjustment. The multivariable adjustment was performed with traditional CV risk factors (model 1: age, sex, Model 2: +eGFR CKD-EPI, UACR, HbA1c, LDL-C, hypertension, active smoking, model 3: model 1 + triglycerides, eGFR CKD-EPI, UACR, HDL-C). An alpha level of P < .05 (two-tailed) was considered statistically significant. The Kaplan-Meier graph was constructed using GraphPad Prism (GraphPad Software Inc., La Jolla, CA, USA).
Results
Baseline Characteristics
A total of 298 PAD patients were evaluated. Deceased patients were significantly older (74 ± 9 vs 67 ± 10 years, P < .001), had higher baseline systolic blood pressure measurements (140 ± 21 vs 135 ± 17 mmHg, P = .032) and worse renal function as expressed by eGFR (58 [45–73] vs 72 [58–85] ml/min/1.72 m2, P < .001) as well as albumin urine creatinine ratio (8 [4–15] vs 26 [8–90], P < .001). Deceased patients showed a higher incidence of extracranial carotid artery plaques (74, 38.7% vs 68, 63.6%, P < .001), while no difference in history of CAD was observed. Levels of C-reactive protein (CRP), as a general measurement of inflammation, did not differ between the patient groups (0.30, 0.15–0.61 vs 0.30, 0.15–0.49 mg/dl, P = .456). Details are shown in Table 1.
Baseline Characteristics of the Study Population Divided by All-cause Death.
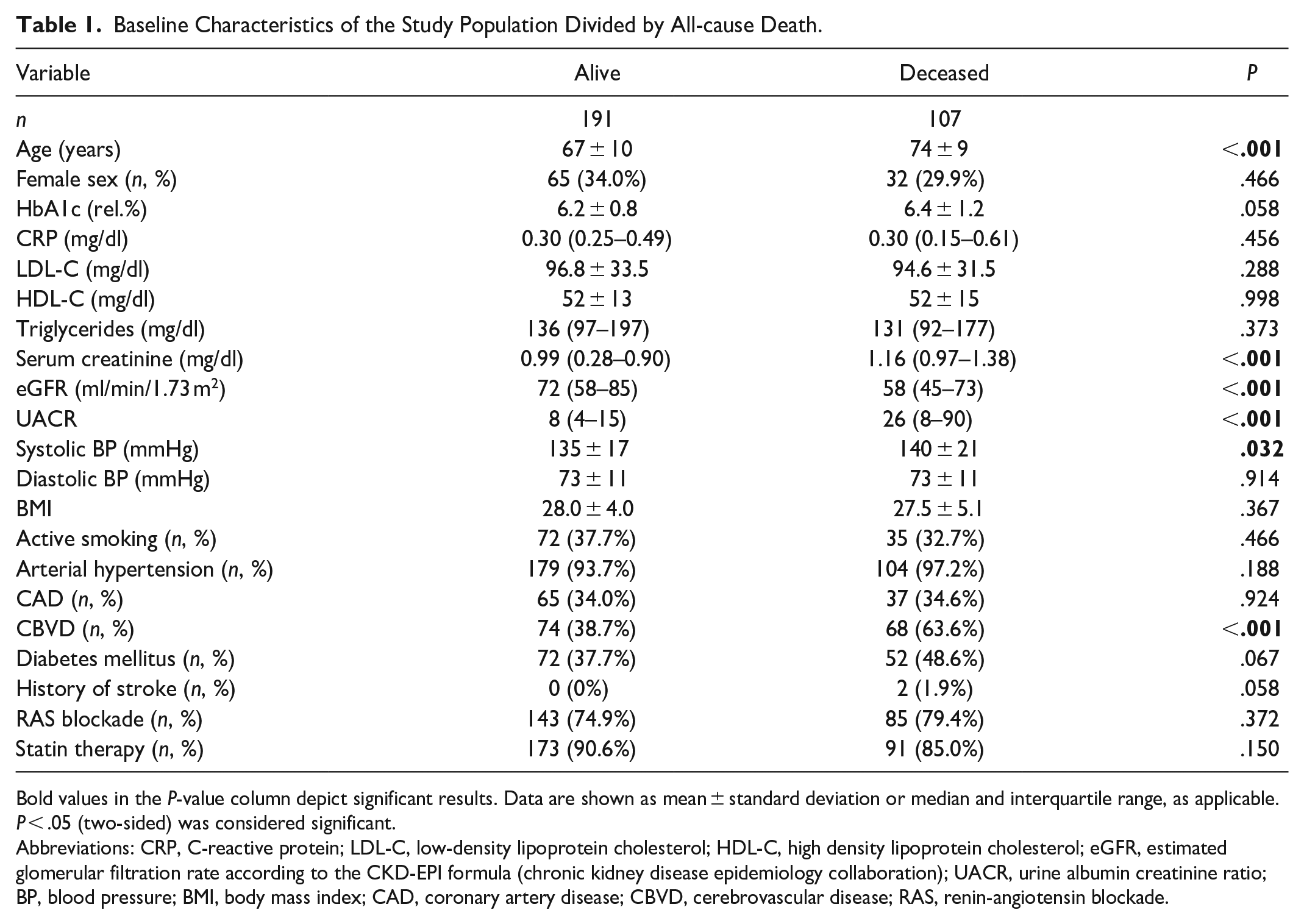
Bold values in the P-value column depict significant results. Data are shown as mean ± standard deviation or median and interquartile range, as applicable. P < .05 (two-sided) was considered significant.
Abbreviations: CRP, C-reactive protein; LDL-C, low-density lipoprotein cholesterol; HDL-C, high density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate according to the CKD-EPI formula (chronic kidney disease epidemiology collaboration); UACR, urine albumin creatinine ratio; BP, blood pressure; BMI, body mass index; CAD, coronary artery disease; CBVD, cerebrovascular disease; RAS, renin-angiotensin blockade.
Univariate Associations of FGF-23
FGF-23 significantly correlated with endogenous insulin production (C-peptide r = .22, P < .001) and traditional markers of the metabolic syndrome (triglycerides r = .25, P < .001, HDL-C r = −.28, P < .001, CRP r = .14, P = .016). A detailed overview can be seen in Table 2.
Univariate Correlation Analysis Between FGF-23 (Logarithmically Transformed) and Age and Standard Laboratory Parameters.
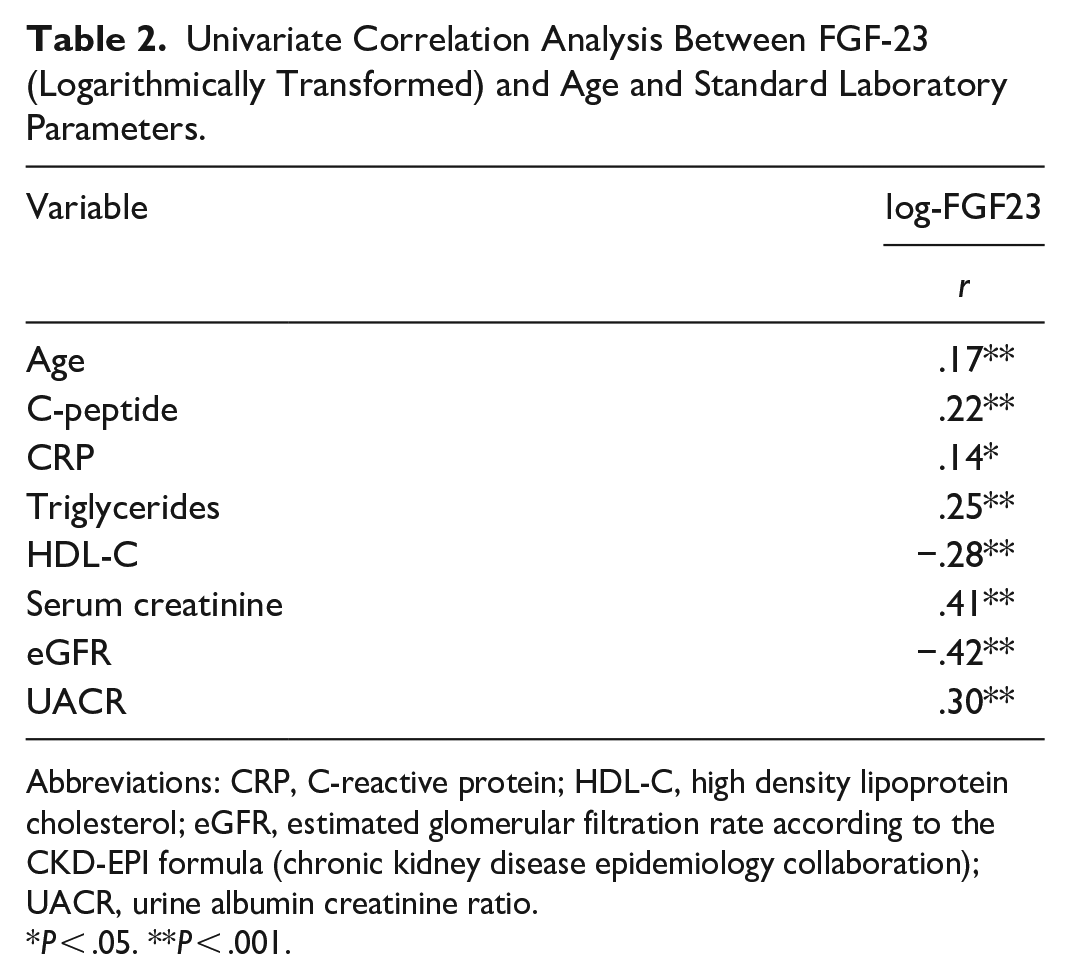
Abbreviations: CRP, C-reactive protein; HDL-C, high density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate according to the CKD-EPI formula (chronic kidney disease epidemiology collaboration); UACR, urine albumin creatinine ratio.
P < .05. **P < .001.
FGF-23 levels did not differ between patients with intermittent claudication and asymptomatic patients (1.72, 1.22–2.65 vs 1.77, 1.21–2.66, P = .868), as a marker of clinical PAD severity. Similarly, ankle-brachial index, a marker of limb perfusion, was not associated with FGF-23 (r = −.57, P = .357).
Regression Analysis
All variables significantly associated with FGF-23 in univariate correlation analyses were submitted into a multivariate stepwise backward regression analysis. All parameters (age, C-peptide, triglycerides, HDL-C, serum creatinine, eGFR CKD-EPI, and UACR) were significantly associated with FGF-23. CRP levels were borderline not associated with FGF-23 (beta .11, P = .057).
All significantly associated variables of univariate regression analyses were further subjected into a multiple backward stepwise regression analysis. In the final model triglycerides, HDL-C, eGFR CKD-EPI, and UACR were capable of explaining 21.9% of the variance of FGF-23 (R2 .219, adjusted R2 .204). A detailed overview is shown in Table 3.
Univariate and Multivariate Regression Models for an Increase of One-unit FGF-23. R2 for the Final Model is .219 with an adjusted R2 of .202.
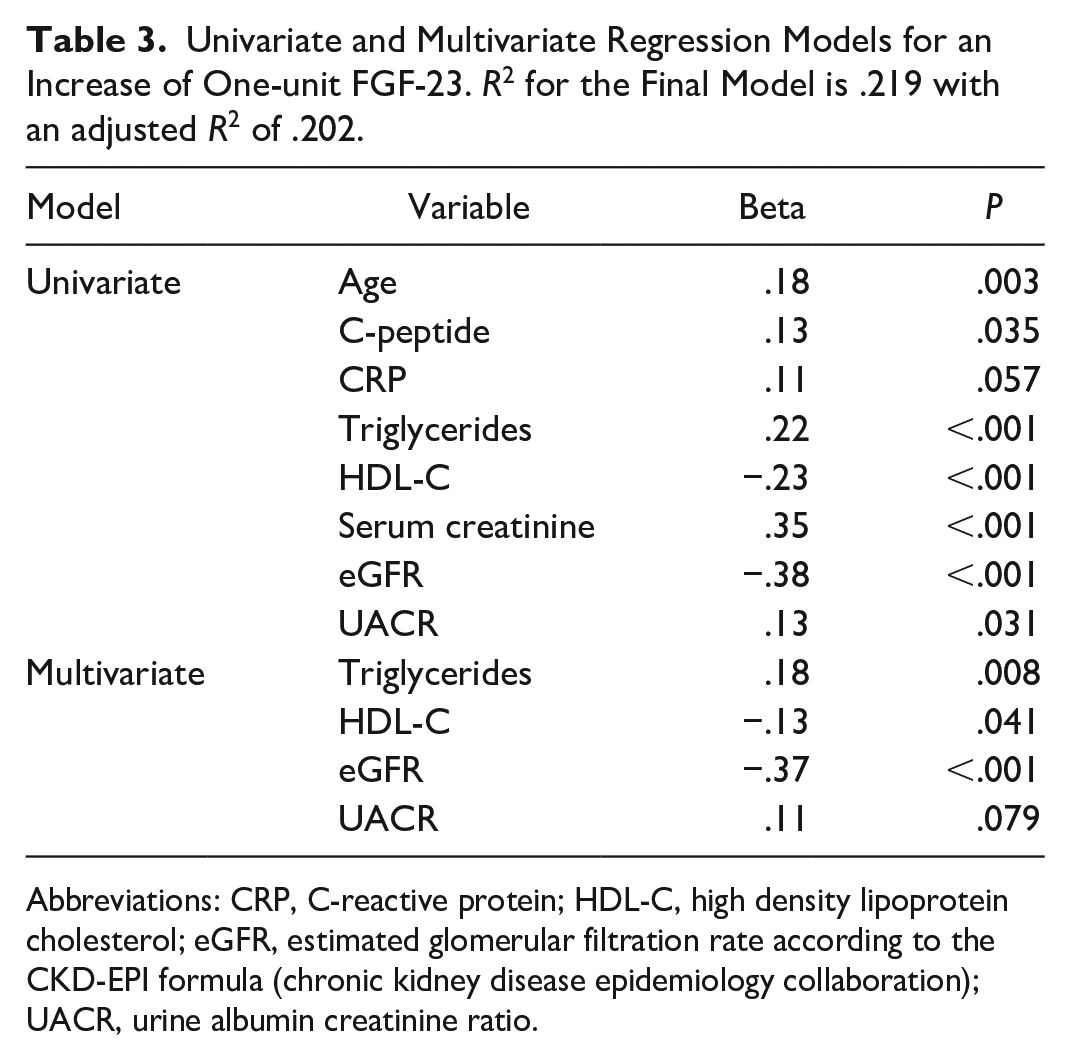
Abbreviations: CRP, C-reactive protein; HDL-C, high density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate according to the CKD-EPI formula (chronic kidney disease epidemiology collaboration); UACR, urine albumin creatinine ratio.
Outcome Analysis
During the observation time of a maximum of 10 years (median 8.06 years, 25th percentile 6.06 years, 75th 8.98 years) 107 events (35.9%) occurred; 61 events (20.4%) were identified as of CV origin. Patients were divided into tertiles for better comparison.
Patients with the highest FGF-23 tertile showed the worst outcome (log-rank P < .001). Similarly, significantly more fatal CV events were registered in the highest FGF-23 tertile (log-rank P = .014), as shown in Figure 1.
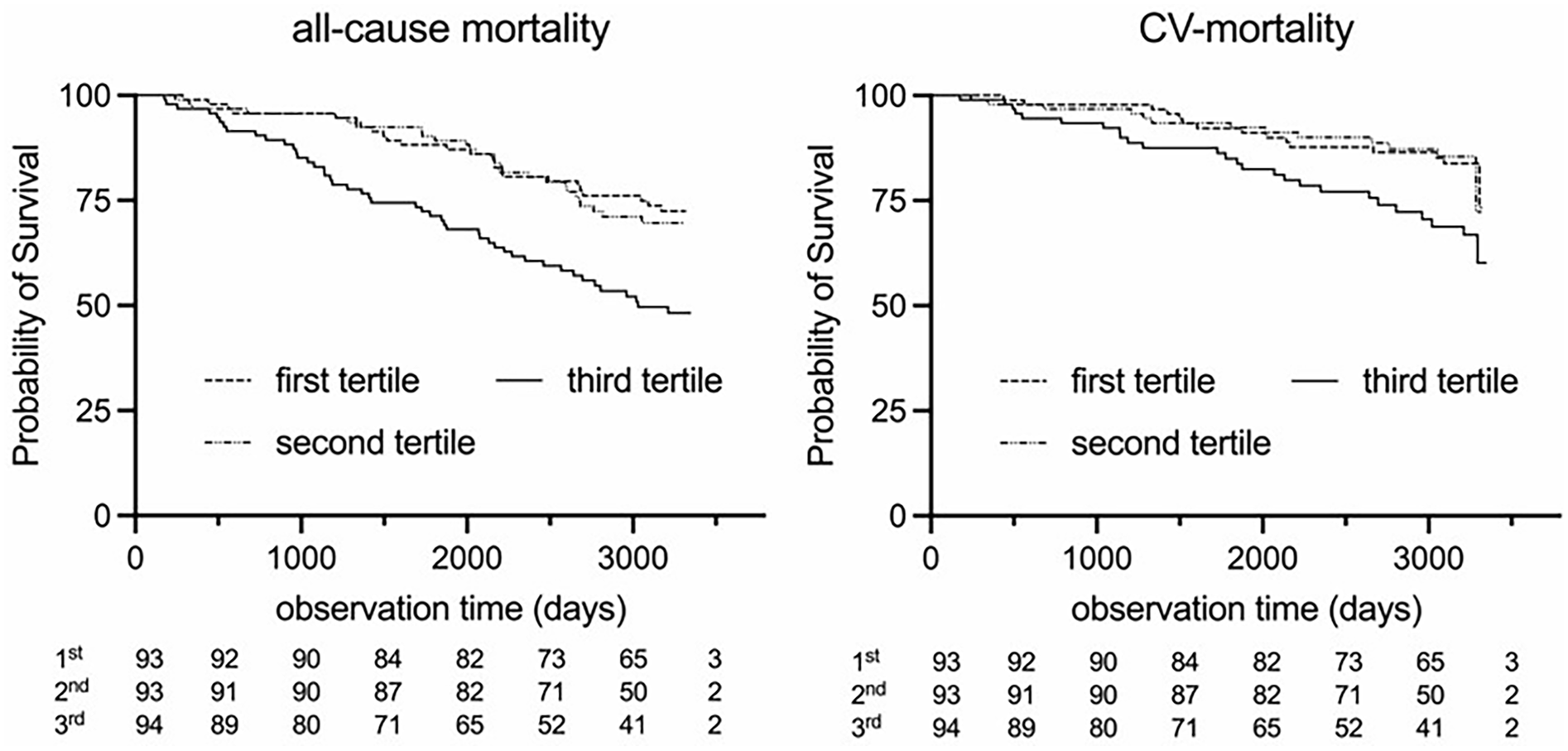
Kaplan-Meier graph for fibroblast growth factor 23 (FGF-23) tertiles regarding long-term survival. The highest tertile showed significantly worse outcome both for all-cause mortality (P < .001) as well as cardiovascular (CV) survival (P = .014). Tertile 1 n = 93; tertile 2 n = 93; tertile 3 n = 94.
Cox regression analyses both for all-cause mortality (Hazard ratio, HR, 1.56, 95% confidence interval [CI] 1.27–1.83) and CV mortality (HR 1.44, 95% CI 1.10–1.89) showed a significant association in a crude fashion. However, while the significant association was diminished in multivariable analyses for CV-death with adjustment for traditional CV-risk factors (age, sex, eGFR, UACR, HbA1c, LDL-C, hypertension and active smoking; HR 1.16, 95% CI 0.83–1.62; model 2) and significant variables of the regression analyses (age, sex, triglycerides, eGFR CKD-EPI, UACR, HDL-C; HR 1.19, 95% CI 0.85–1.67; model 3), models sustained adjustment for all-cause death (Model 2: HR 1.33, 95% CI 1.04–1.71; Model 3: HR 1.35, 95% CI 1.05–1.74). A detailed overview can be seen in Table 4.
Cox-regression Analyses for an Increase of One-unit of FGF-23.
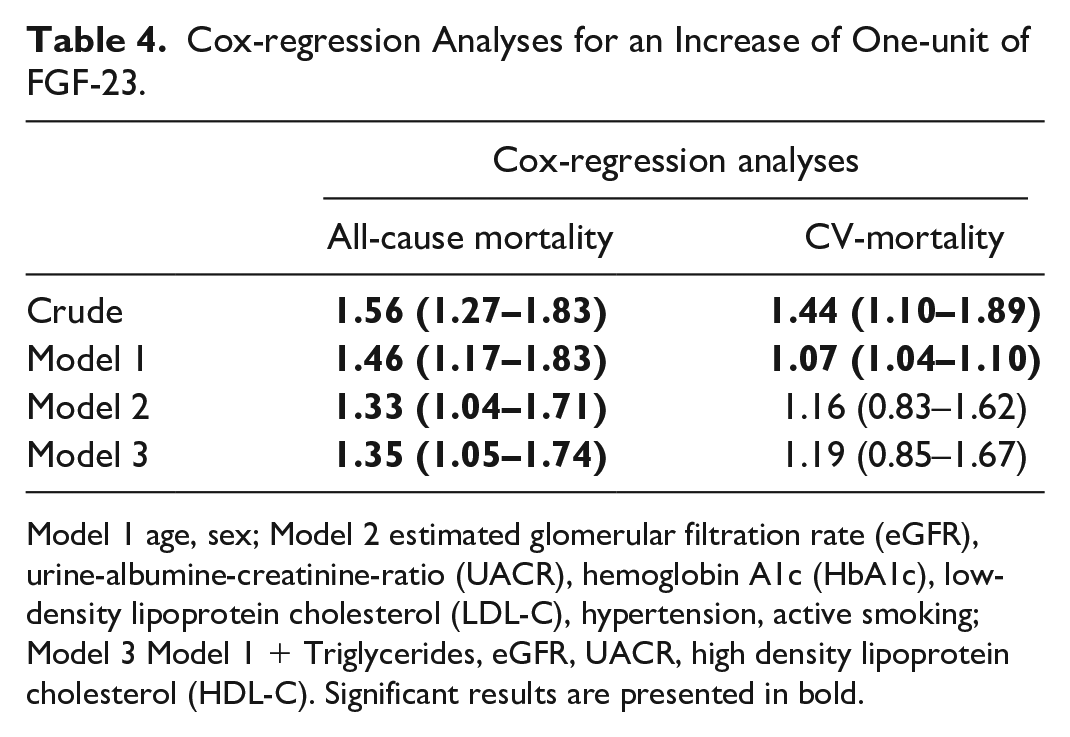
Model 1 age, sex; Model 2 estimated glomerular filtration rate (eGFR), urine-albumine-creatinine-ratio (UACR), hemoglobin A1c (HbA1c), low-density lipoprotein cholesterol (LDL-C), hypertension, active smoking; Model 3 Model 1 + Triglycerides, eGFR, UACR, high density lipoprotein cholesterol (HDL-C). Significant results are presented in bold.
Discussion
This study shows for the first time an association of all-cause mortality and FGF-23 in patients with PAD. Despite the fact that deceased patients were of older age, had worse baseline renal function and higher systolic blood pressure at baseline, Cox-regression analyses withstood adjustment for those and additional traditional CV risk factors as well as renal parameters including eGFR and UACR. Likewise, higher rates of concomitant extracranial carotid artery disease were seen in deceased patients. In addition to an association between FGF-23 and renal parameters, a further association was shown with metabolic parameters and CRP.
While it has been observed, that FGF-23 predominantly affects the cardiomyocyte, similar effects have been attributed to the vasculature. Our results are in line with previous studies evaluating the association of FGF-23 levels with the incidence of PAD in patients with diabetes.17,26 Likewise, FGF-23 was associated with the incidence of CAD 27 as well as all-cause and CV mortality in patients with CAD. 28 Comparable results were seen with associations of elevated FGF-23 levels and the presence of carotid plaques or carotid artery disease 29 as well as increased intima-media-thickness. 17 Likewise, robust data from a meta-analysis illustrated that elevated levels of FGF-23 are associated with CV morbidity and mortality in patients with CAD and CKD. 30 While these associations with CV-disease in other vascular beds are presumptive for similar findings in PAD, no such data was available previously. Additionally, risk factors and their relevance for different CV-diseases differ, which can even be seen with traditional risk factors such as sex 31 and different lipid parameters. 32 Thus, we present the first data for an association of outcome and PAD in a long-term follow-up.
Vascular calcification remains a challenging clinical situation. 33 Calcified atherosclerotic plaques are in general more complex to treat from an endovascular perspective and optimal medical therapies are not established. 34 The fact that mechanisms of vascular calcification are not completely understood further contributes to this issue. However, as observed in several in vivo and in vitro models, the underlying pathophysiologic process is predominantly facilitated by elevated phosphate levels. 34 Hyperphosphatemia may induce vascular calcification trough several distinct mechanisms, one of which is the induction of osteogenic transdifferation of vascular smooth muscle cells (VSMCs). 35 Eventually, the loss of contractile properties and the gain of osteogenic features leads to ossification. The calcification process is furthermore accelerated by a disbalance of calcification promotors and inhibitors.34,36
Vascular calcification can be divided into intimal and medial vascular calcification.33,37 Independent of the different pathophysiologic processes, both variants are of high prevalence in PAD, as seen in histopathologic studies.37,38 Animal models showed that FGF-23 induced vascular calcification, 39 while others did not find an association with FGF-23 in the vascular wall. 40 However, elevated FGF-23 levels were initially described in the context of CKD-MBD and are associated with worse outcome in CKD patients. 41 Our data shows that FGF-23 levels are associated with outcome independent of kidney function parameters. Yet, while this is in line with previous studies,42,43 no clear explanation can be given for this observation. We hypothesize that FGF-23 levels could be a surrogate marker representing excessive vascular calcification before the onset of severe kidney function decline, which is associated with worse outcome on a systemic level.
Several limitations of this study have to be considered. Firstly, it was prospectively conducted, but FGF-23 measurements were performed retrospectively, only associations can be shown. Secondly, the sample size of our cohort is only modest in comparison to larger CAD cohorts. Thirdly, only fatal events are shown, and no further conventional outcome parameters are available due to the study design. Fourthly, by study design no further markers on CKD-MBD like phosphate, calcium or klotho are available. However, this study also has several strengths to consider. Firstly, diagnosis of PAD was made in a specialized tertiary care center with frequent follow-up visits. Secondly, optimization of traditional CV risk factors was performed stringently. Thirdly, the analysis was made with a long-term follow-up.
Conclusion
This study shows a novel association between FGF-23 and mortality in patients with stable PAD. A further association was seen with higher rates of extracranial carotid artery disease, which additionally highlights FGF-23 as a potential marker for systemic atherosclerosis. Further insights into vascular calcification and PAD as well as the potential of FGF-23 as a possible biomarker for outcome assessment are warranted.
Footnotes
Acknowledgements
The authors thank Daniela Seidinger for her service in laboratory work.
Author Contributions
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published.
Data Availability Statement
Data of this manuscript can be inquired upon reasonable request to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Basic institutional funding was provided by Medical University Vienna, Austria.
