Abstract
Background:
Patency of the pedal-plantar arch limits risk of amputation in peripheral artery disease (PAD). We examined patients without chronic kidney disease (CKD)/diabetes mellits (DM) [PAD-control], those with DM without CKD, and those with CKD without DM.
Method:
Uni- and multivariate logistic regression was used to assess association of CKD with loss of patency of the pedal–plantar arch and presence of tibial or peroneal vessel occlusion. Multivariate models adjusted for age, sex, hypertension, hyperlipidemia and smoking.
Results:
A total of 419 patients were included [age 75.2 ± 10.3 years, 288 (69%) male]. CKD nearly doubled the unadjusted odds ratio (OR) for loss of patency of the pedal–plantar arch. After adjustment, association remained significant for severe CKD [estimated glomerular filtration rate (eGFR) ≤ 29 ml/min compared with eGFR ≥ 60 ml/min, adjusted (adj.) OR 8.24 (95% confidence interval {CI} 0.99–68.36, p = 0.05)]. CKD was not related to risk of tibial or peroneal artery occlusion [PAD-control versus CKD, adj. OR 1.09 (95% CI 0.49–2.44, p = 0.83)] in contrast to DM [PAD–control versus DM, adj. OR 2.41 (95% CI 1.23–4.72, p = 0.01), CKD versus DM, adj. OR 2.21 (95% CI 0.93–5.22); p = 0.07)].
Conclusions:
Below the knee (BTK) vascular pattern differs in patients with either DM or CKD alone. Severe CKD is a risk factor for loss of patency of the pedal–plantar arch.
Introduction
Lower limb peripheral artery disease (PAD) is a common comorbidity in patients with diabetes mellitus (DM) and chronic kidney disease (CKD).1–3 Atherosclerotic disease pattern in PAD is influenced by cardiovascular risk factors.4,5 CKD and DM are known to be the leading cause for vascular calcification and share similar pathomorphological aspects. 6 DM was shown to be a significant predictor for below the knee (BTK) artery involvement 5 and small vessel disease defined as the highest 10% of toe brachial index decline with limited ankle brachial index. 4 Major cardiovascular disease trials frequently exclude patients with CKD and do not provide adequate information on renal function of enrollees or effect of interventions on patients with CKD without DM. 7
DM and CKD are associated with critical limb ischemia (CLI)8–10 that goes along with a high rate of minor and major amputations.11,12 The importance of patency of the pedal–plantar arch on the rate of amputations was suggested in early series derived from patients with DM 13 or end stage renal disease (ESRD). 14 It is rather more recently that patency of the pedal–plantar arch as well as obstructive burden of pedal arteries has become a dedicated focus of interest for the understanding of poor clinical limb outcome after distal bypass or endovascular treatment.3,15–19
Kidney disease is a frequent complication in DM. 20 Differentiation between DM and CKD as leading risk factors for marked distal atherosclerotic disease pattern in studies of PAD is missing, and in patients with CKD, it is mainly limited to patients with ESRD.2,3,16,21,22
In this retrospective, single-center PAD patient group analysis, patency of the arterial pedal–plantar arch and occlusion of BTK arteries were assessed with respect to the coexistence of DM or CKD. The hypothesis was that loss of patency of the pedal–plantar arch is more frequent in patients with CKD compared with patients without CKD, independent of the presence of DM.
Patients and methods
Study design
For a retrospective analysis, patients were selected from a consecutive, prospectively collected, cross-sectional database of patients undergoing digital subtraction angiography (DSA) for symptomatic PAD. The time span was from January 2010 to June 2015 for the patient’s first visit to the Swiss Cardiovascular Center, Division of Vascular Medicine at Bern University Hospital, Switzerland, a tertiary referral center responsible for peripheral vascular service in a population of about one million subjects. Noninvasive Doppler examination and duplex scan are routine first-line assessments followed by DSA if an indication for revascularization is given. It can be assumed that <5% of symptomatic patients referred were excluded from analysis because of an other-than-angiographic assessment (Figure 1). Patients with DM and CKD were consecutively selected, the PAD-control group was randomly selected to match in number of patients included. Selection of patients was regardless of their clinical status of PAD and was undertaken in a two-step procedure. In a first step, patients had to have a known and determined status of cardiovascular risk factors. In a second step, DSA had to have adequate readability of the pedal–plantar artery arch as quoted by two independent reviewers. With focus on assessing whether there is an independent influence of CKD as compared with DM on patency of the pedal–plantar arch, inclusion criterion was symptomatic PAD to prevent selection bias, for example, patients with predominant BTK disease often seen in CLI. Inclusion criteria were: patients aged > 18 years with symptomatic chronic PAD as defined according to international guidelines,23,24 ankle–brachial index < 0.9 or toe–brachial index < 0.6, undergoing DSA without previous revascularization, and readable DSA for analysis of the atherosclerotic burden of the lower limb and pedal arteries. Exclusion criteria were low-quality DSA not suitable for analysis of atherosclerotic burden, that is, unknown inflow, low-contrast filling, inadequate pedal arch documentation, nonatherosclerotic disease, acute limb ischemia, or previous revascularization. Overall, 419 patients met the inclusion criteria. Patients were stratified according to the following prespecified groups: patients without DM and without CKD (PAD-control, n = 149), patients with DM without CKD (DM, n = 183). Patients with CKD without DM (CKD, n = 87). The research protocol was approved by the institutional review board of cantonal ethic committee according to the guideline of the 1975 Declaration of Helsinki and its amendments. As this was a retrospective study, the need to obtain written informed consent was waived and patients’ records were anonymized before analysis.

Flow chart of patient enrollment.
Angiographic scoring
DSAs were read in a random order by two experienced readers. Both readers were blinded to clinical data, especially blinded to the groups. Patency of the pedal–plantar arch was defined according to the definition of Higashimori and colleagues 17 with arterial flow through either lateral branch of the plantar–pedal artery or dorsal–pedal artery reaching the opposite artery through connection via the deep penetrating artery (Figure 2). A consensus had to be found regarding patency of the pedal–plantar loop.
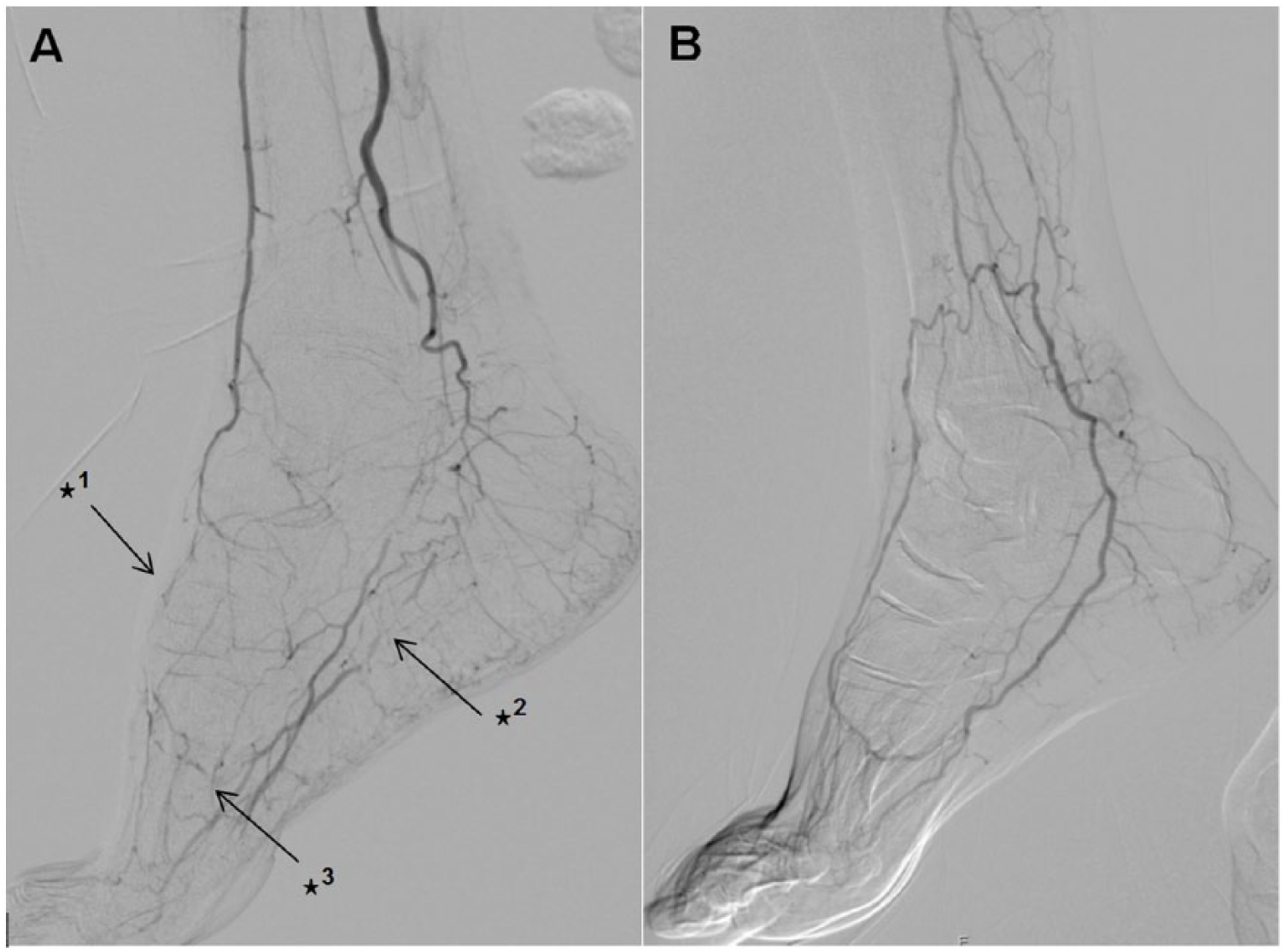
Interrupted dorsal–pedal artery (✶1), interrupted lateral branch of plantar–pedal artery (✶2) and interrupted pedal–plantar arch (✶3) (A); communicating dorsal–pedal artery with open lateral-branch plantar of the plantar–pedal artery with a patent pedal–plantar arch (B).
Definition of risk factors and comorbidity
Presence of DM was defined by glycosylated hemoglobin > 6.5% or if the patient was on insulin therapy or oral antidiabetics. 25 CKD patients were identified by calculation of the estimated glomerular filtration rate (eGFR) using the Modification of Diet in Renal Disease (MDRD) formula < 60 ml/min. 26 CKD was classified according to severity of renal failure as moderate CKD with an eGFR of 30–59 ml/min and severe CKD with an eGFR < 29 ml/min. ESRD was defined as GFR < 15 ml/min or on dialysis. 27 Arterial hypertension was defined by systolic blood pressure > 140 mmHg or diastolic blood pressure > 80 mmHg, or if any antihypertensive drug was consumed. Hyperlipidemia was defined by total cholesterol level > 5 mmol/l, or triglycerides > 2 mmol/l or high-density lipoprotein cholesterol < 1 mmol/l, or if any lipid-lowering drug was consumed. Patients were classified as ever-active smokers or nonsmokers, according to records.
Statistics
Descriptive analyses of sociodemographic characteristics were based on patient characteristics at the time of the performed angiogram. Demographic and clinical characteristics of the study population are reported as mean [±standard deviation (SD)] for continuous variables and as number (percentage) for categorical variables.
Binary outcomes were analyzed using logistic regression and presented as odds ratios (OR) and 95% confidence intervals (CI). p values are from Wald tests. Discrete outcomes with three or more levels were analyzed using χ2 tests. Where indicated, models were adjusted for age, sex, hypertension, hyperlipidemia and smoking status. Interrater reliability was measured by kappa coefficient for nominal categorical variable (occlusion of tibial and peroneal arteries or loss of patency of pedal–plantar arch). Intrarater reliability was not evaluated. Statistical analyses were performed using Stata 14.2 (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX: StataCorp LP.).
Results
Baseline characteristics
Overall 419 patients fulfilling inclusion criteria and were analyzed for pedal–plantar arch patency and atherosclerotic occlusion of BTK arteries. The three groups differed statistically significant in patient number, mean age and active smoking status. Demographic data are shown in Table 1.
Demographic data on 419 patients enrolled into the study.
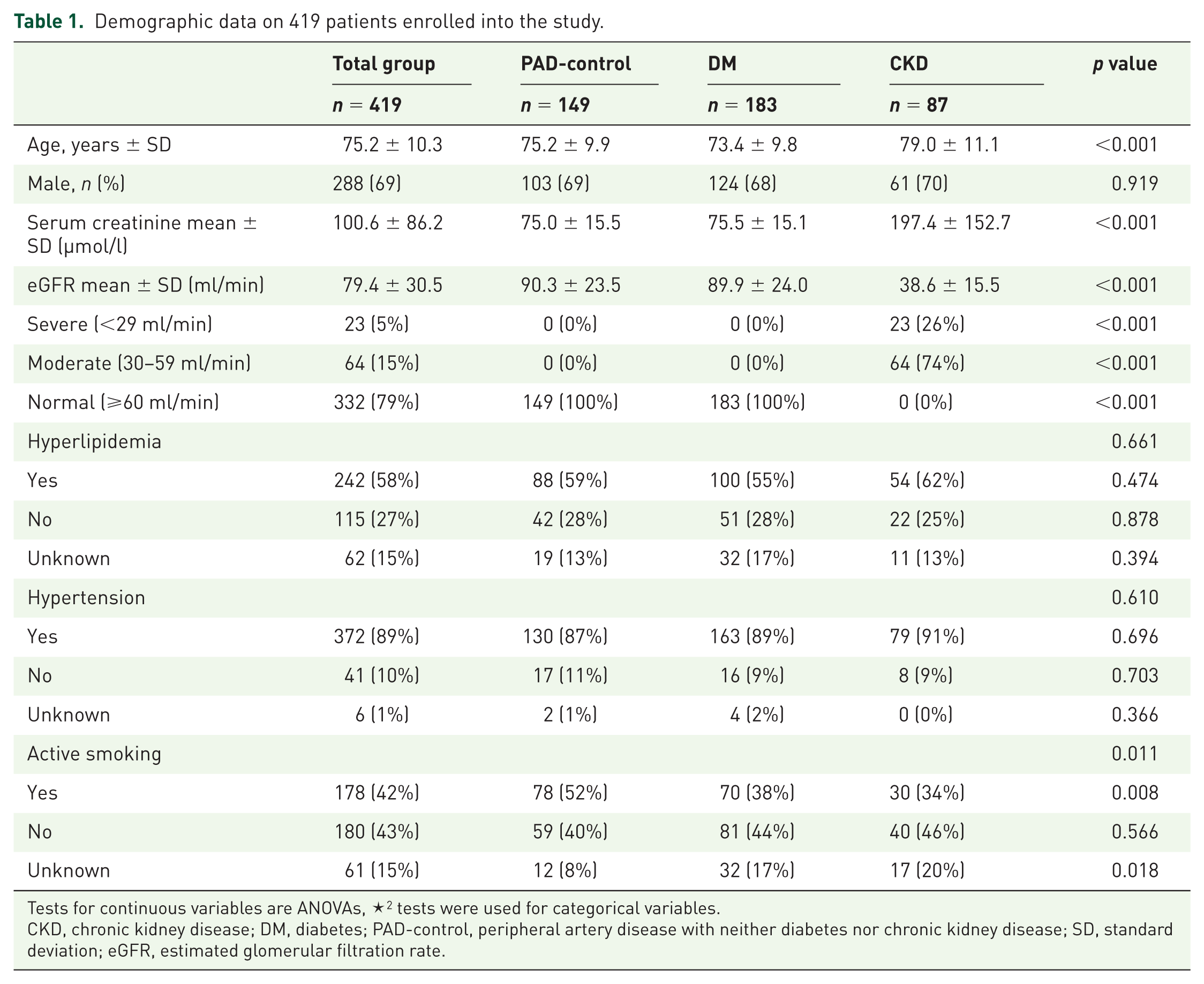
Tests for continuous variables are ANOVAs, ✶2 tests were used for categorical variables.
CKD, chronic kidney disease; DM, diabetes; PAD-control, peripheral artery disease with neither diabetes nor chronic kidney disease; SD, standard deviation; eGFR, estimated glomerular filtration rate.
Loss of patency of the pedal–plantar loop
The adjusted ORs for loss of patency of pedal–plantar loop were higher in CKD above both PAD-control [OR 1.39 (95% CI 0.67–2.87), p = 0.37], diabetics [OR 1.32 (95% CI 0.64–2.72), p = 0.46] and both [OR 1.35 (95% CI 0.69–2.64), p = 0.38]. DM did not show a higher OR above PAD-control [OR 1.05 (0.61–1.83), p = 0.85]. However, the ORs were not statistically significant (Table 2).
Loss of patency of the pedal–plantar arch.

Adjusted models included age, sex, hypercholesterolemia, arterial hypertension and smoking.
CKD, chronic kidney disease; DM, diabetes; PAD, peripheral artery disease; OR, odds ratio; CI, confidence interval.
Within the subgroup of CKD, patients’ loss of patency of the pedal loop was related to the severity of CKD. Moderate CKD (eGFR 30–59 ml/min) did not show higher ORs after controlling for age, sex, hypercholesterolemia, arterial hypertension and smoking [OR 0.93 (95% CI 0.43–2.01), p = 0.85]. Instead, patients with severe CKD (eGFR ≤29 ml/min) showed significantly increased adjusted ORs compared with patients with normal kidney function, even after adjustment for baseline factors [OR 8.24 (95% CI 0.99–68.36), p = 0.05; Table 3].
Association of severity of chronic kidney disease and loss of patency of pedal-plantar loop.

Adjusted models included age, sex, hypercholesterolemia, arterial hypertension and smoking.
No CKD eGFR ⩾ 60 ml/min (n = 332); moderate CKD eGFR 30–59 ml/min (n = 64); severe CKD eGFR <29 ml/min (n = 23).
CKD, chronic kidney disease; OR, odds ratio; CI, confidence interval; eGFR, estimated glomerular filtration rate.
Burden of atherosclerotic disease in below-the-knee arteries
CKD was not related to the risk of tibial or peroneal artery occlusion, whereas the risk of an occlusion of at least one BTK artery was 2.5-fold increased in patients with DM as compared with PAD-controls and CKD patients. The adjusted OR for occlusion of BTK arteries in PAD-control versus CKD was 1.09 (95% CI 0.49–2.44, p = 0.83), PAD-control versus DM was 2.41 (95% CI 1.23–4.72, p = 0.01), and CKD versus DM 2.21 (95% CI 0.93–5.22, p = 0.07), respectively.
Consistency of our interrater agreement was good to very good for rating on occlusion of tibial and peroneal arteries and moderate for rating on loss of patency of the pedal–plantar arch (Supplementary Table 1). As there was lowest interrater agreement within the subgroup of CKD, data shown refer to a consensus between two raters. For those patients without agreement with loss of patency of the pedal–plantar arch, a consensus between the two readers was found. Analysis showed comparable results if CKD was defined as plasma creatinine level > 105 µmol/l as compared with eGFR < 60 ml/min.
Discussion
Focus of this first evaluation of the pedal–plantar arch in patients with PAD was to study whether there is an independent influence of CKD and DM on patency. Severe CKD with an eGFR less than 29 ml/min seems to be an important vascular risk factor for loss of patency of the pedal-plantar arch in patients with symptomatic PAD. Moreover, analysis shows that BTK vascular disease pattern differs in patients with either DM or CKD alone. Patients with DM seem to have pronounced occlusive disease of tibial and peroneal arteries, whereas patients with severe CKD have pronounced pedal artery disease.
Studies on PAD patients providing data on patency of pedal arteries including the pedal arch are rare and include a limited number of patie-nts.2,13,16,17,28–30 Various risk factors influence the pattern of lower limb atherosclerosis in PAD patients, with DM and CKD typically associated with pronounced distal disease4,5 and vascular calcification. 6 Comparative data on the disease patterns in pedal and BTK arteries with strict differentiation of patients with PAD suffering from either DM or CKD are missing.
There is growing evidence showing that even moderate renal insufficiency is associated with arterial calcification,31–33 and that coronary artery calcification, its progression and impact on a rise in cardiovascular events is associated with CKD at predialysis status.34,35 Thereby, progression rate of CAC among patients with ESRD is higher compared with diabetes patients without and also with a predialysis status. 36 In accordance, loss of kidney function is also associated with an increase of calcification in PAD. 22 Furthermore, it is described that a decrease in GFR level by 10 ml/min per 1.73 m2 is associated with a relative risk ratio of 1.08 for the presence of distal limb artery disease involvement with ESRD as an independent predictor for a centrifugal arterial lesion pattern (relative risk ratio 7.72, 95% CI 3.0–19.88, p < 0.0001). 37 Data presented that severe CKD is associated with pedal arch occlusion are well in accordance with these findings.
Data on the atherosclerotic disease pattern in PAD with a distinction of diabetes and CKD are scarce. In a small-sized group of patients with critical limb ischemia (CKD group n = 15, DM group = 25), Diehm and colleagues have shown that patency rate of at least one pedal artery is 72% in patients with DM compared with 47% in patients with CKD and 48% for patients suffering from both CKD and DM. 2 In another analysis from Brosi and colleagues, a complete occlusion of both the plantar–pedal arch and dorsal–pedal arch was found in 58% in those PAD patients with ESRD. 21 Kawarada and colleagues analyzed three different types of pedal-arch patency in 85 patients with critical limb ischemia. Of those, 51% had ESRD and 78% had diabetes, showing that patency of both pedal arteries and pedal–plantar arch was present in 16%, patency of one pedal artery in 64% and loss of patency in both pedal arteries in 20% of patients. 15
Analysis on patency of the pedal–plantar arch and pedal arteries is an ongoing focus for understanding poor clinical outcome after distal bypass or endovascular treatment. More than 20 years ago, it was considered that only modest success could be expected with pedal bypass grafts in patients with ESRD. 14 Today, advanced endovascular techniques by transmetatarsal retrograde access of the transplantar arch can help improve clinical outcome in CLI.38,39 However, findings differ between series. Meyer and colleagues found no impact of quality of pedal arch on wound healing or survival in a cohort of 32 patients with ESRD after BTK angioplasty. 18 Matsukaru and colleagues looked at perimalleolar atherosclerotic burden in PAD patients, 77% with diabetes and CKD, 60% with ESRD, that were treated with infrapopliteal (tibial or paramalleolar) arterial bypass surgery for critical limb ischemia. Those patients showing a higher atherosclerotic burden of perimalleolar arteries had a statistically significant worse outcome for amputation-free survival and overall survival rate. 3 Patency of both dorsalis pedis and plantar artery results in a higher wound healing rate over patients with no or single patent pedal artery. 15 Other studies found no impact of pedal arch quality on amputation-free survival but an improved outcome in wound healing for complete or incomplete pedal arch over no pedal arch.16,19 None of the studies distinguished between DM and CKD as potentially independently acting vascular risk factors. Results show an important difference between DM and CKD regarding the distal vascular disease pattern of patients with symptomatic PAD. It underlines that CKD, often coincident with diabetes, is a serious independent risk factor for pedal vascular disease, suggesting that patients with severe CKD are prone to revascularization failure and poor clinical outcome. If this hypothesis can be proven in a larger sized, adequately powered study, it might even be justified to define this as nephropathic foot syndrome.
The main limitations of our study include its retrospective design, limited generalizability due to sample size, and the possible selection bias that might contribute to results. Exclusion of patients with a pedal angiogram defined as unreadable for our analysis can be related to several factors. We assume that the lack of adherence to a standardized protocol for high-quality angiograms is likely a result of the intention to save contrast media volume, particularly as in this case the foot lesions had no clinical requirement for selective pedal angiogram, carbon dioxide contrast medium was used and fretful foot movement was apparent (Figure 1).
The study is not powered to assess the influence of patency of the pedal–plantar arch on clinical outcome, but results show an important difference in peripheral vascular disease pattern that might explain the particular poor outcome in patients with severe CKD. Moreover, no direct implication to clinical relevance can be undertaken. Duration of diabetes and CKD might have had a relevant contribution to the disease pattern as shown. However, this information was only available in a fragmentary form and a meaningful statistical computation was not feasible. That duration of diabetes and ESRD per se is an independent risk factor for vascular calcification is well described.33,40–42 Methods being used were not meant to answer the question of influence on extent of vessel wall calcification. Even using a rough semiquantitative calcification score, the study was not powered to answer whether calcification was related to patency. Moreover, adequate plain films were not always available to allow reliable, systematic post hoc analysis. Also, there is no systematic information on the comedication of patients available, in particular, long-term corticosteroid use that is associated to infragenicular vascular calcification, was unknown. 43 Certain aspects on definition of the pedal arch have to be considered if data are compared with clinical outcome studies. We used the definition as previously described by Higashimori and colleagues, 17 however, there is some variability in the literature.16,21,44
In conclusion, results indicate severe CKD as a vascular risk factor defining a specific subgroup of PAD patients at risk for pronounced below-the-ankle artery disease. The specific atherosclerotic disease pattern of patients with CKD and ‘nephropathic’ foot ischemia might have relevant implications with regard to contemporary endovascular revascularization strategies that needs further evaluation.
Footnotes
Acknowledgements
All authors have made substantial contributions to all of the following: (1) the conception and design of the study, or acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; (3) final approval of the version to be submitted.
Conflict of interest statement
All authors declare no competing interests with respect to this manuscript. In 2016, IB received grant support from Abbott, Cook, Terumo, Sanofi and Optimed.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
