Abstract
This study aimed to evaluate the utility of the systemic immune-inflammation index (SII) in predicting the development of Cardiac Implantable Electronic Device (CIED) infections. A retrospective analysis was performed using the medical records of 2185 patients who underwent CIED (including de novo, revision, or upgrade) implantation at our institution from January 2012 to December 2019. We recorded CIED infections and risk factors according to the patient, device, and procedural characteristics during the median 28.7 months (6.2–56.8) follow-up. CIED infections were identified in 52 patients. Diabetes, chronic renal disease, SII before implantation, new cardiac resynchronization therapy (CRT) implantation, CRT-battery replacement, revision or upgrade, and the number of previous procedures were independent predictors of CIED infections. The area under the curve (AUC) of SII to predict CIED infection was .733 (95% CI: .654-.811). A raised SII may be a useful predictor of CIED infection.
Introduction
Cardiac Implantable Electronic Devices (CIEDs) were defined in the 1930s as aiming to monitor and treat heart contraction or rhythm disturbance by acting on electrical activity and heart muscle contraction. 1 Although CIED (pacemakers [CPM], implantable cardioverter defibrillator [ICD], and cardiac resynchronization therapies [CRT]) implantation procedures are safe and with a low risk of severe complication rates (infection, thrombosis, lead failure, and pneumothorax), 2 CIED infections remain a problem causing morbidity and mortality. The infection rate is reported as .5%–5% in different studies. 3 In Turkey, this rate was 2.5% after 7 years of follow-up. 4
The European and American Infective Endocarditis and Pacemaker Guidelines recommend antibiotic prophylaxis to prevent infection before implantation of CIED.5-7 It is important to identify high-risk patients so as to minimize infection risk (e.g., by using an antibiotic envelope, a prolonged perioperative antibiotic regime, and other actions).
Recently, the systemic immune-inflammation index (SII) has been developed based on the platelet counts and neutrophil/lymphocyte ratio (NLR) to simultaneously consider inflammatory and immunothrombosis status. The SII was related to adverse outcomes in chronic heart failure and coronary artery disease.8-10 However, the relationship between SII and the development of CIED infection in patients who underwent CIED implantation remains unclear. Thus, we evaluated the predictive value of SII for CIEDs infections.
Materials and Methods
We retrospectively examined 2185 patients admitted to our hospital for CIED implantation, replacement, upgrade, or revision between January 2012 and December 2019. The patients were divided into 2 groups: those with CIED infections (n = 52) or without CIED infection (n = 2133). Our center in Izmir, Turkey, provides tertiary referral health care. In the cardiology unit, approximately 400 CIED procedures are performed annually. These procedures include new pacemakers, new defibrillators, new CRT implantation, battery replacement for ICDs/CPMs, or revision and upgrade procedures. The patients received cefazolin or vancomycin/clindamycin in case of allergy to beta-lactam agents as prophylaxis before procedures in our hospital. The study was approved by the Local Ethics Committee of Izmir Katip Celebi University (approval number: 1040 date: 2020) and complied with the Declaration of Helsinki.
Blood samples were obtained from antecubital veins of all patients after admission, before the procedure. Age, sex, pacemaker type, laboratory parameters as hemoglobin, creatinine, estimated glomerular filtration rate, white blood cell count levels; co-morbid diseases such as chronic kidney disease and diabetes mellitus; isolated microorganism from microbiological culture, susceptibility of the isolated microorganism; the infection type as the pocket infection or endocarditis; and the result of extraction were collected. These parameters were obtained from an institutional database. In our final analysis, we only included patients with pacemaker pocket infection and infective endocarditis as device infections. Infection was confirmed via positive cultures from a pocket swab or tissue sample obtained at the time of device removal, from a lead tip, from blood, or with clinical criteria. The Duke criteria for diagnosis of endocarditis were applied to systemic infections related to implantable cardiac devices. 11
Definitions
The SII was defined as follows: SII = platelet count × neutrophil count/lymphocyte count. The mean value and corresponding 95% reference interval for the SII were 459 (189-1168) in a previous study. 12 The neutrophil-lymphocyte ratio (NLR) was defined as the ratio of the neutrophil count to the lymphocyte count. CKD (chronic kidney disease) was defined as estimated glomerular filtration rate (eGFR) <60 mL/min/1.73 m2 which was calculated using the Modification of Diet in Renal Disease (MDRD) formula. 13
Initial CIED implantation was defined as a de novo procedure. Generator replacement, lead revision, and system upgrades were considered revision procedures. These groups within the procedure type are mutually exclusive. For example, if a patient is undergoing an upgrade to CRT, then he or she is counted in the revision/upgrade group. An immunocompromised status was defined as having at least 1 of the following conditions: (1) immunosuppression (defined as viral immunosuppression, neoplastic disease, immunosuppressive drugs including steroids, chemotherapy, or congenital immunosuppression), (2) active hematologic malignancy (i.e., still requiring treatment), or (3) active neoplasm (i.e., a neoplasm that has not been resected, still requires treatment or with metastasis).
Time of Infection
Early infection
The time between implant or revision procedure and CIED infection diagnosis was 1 month or less.
Late infection
The time between implant or revision procedure and CIED infection diagnosis was 1-12 months.
Delayed Infection
When CIED infection occurred 12 months after implant or new procedure.
Statistical Analysis
Statistical tests were carried out using SPSS version 19 (SPSS Inc, Chicago, IL, USA). Continuous variables were provided as mean ± standard deviation, and categorical variables were presented as the number of patients, with the percentage of the total number. Either the Student’s t-test or the Mann–Whitney U test was used to compare values between the 2 groups, as appropriate. The Chi-squared test was used when comparing categorical variables. A multivariate Cox regression analysis was used to identify independent predictors for CIED infection. Variables with a P < .1 and known predictive values from the analysis without variables were entered into the multivariate model. The estimation of predictive values of the multivariable model and multivariable model plus SII were made by the areas under the receiver operating characteristic curve. We used the DeLong test to compare the area under the curve (AUC) with each of these parameters. 14 Moreover, the increased discriminative value of the multivariable model was also estimated using net reclassification improvement (NRI) and integrated discrimination improvement (IDI). 15 A 2-sided P < .05 was considered statistically significant.
Results
Baseline Characteristics of the Study Population.
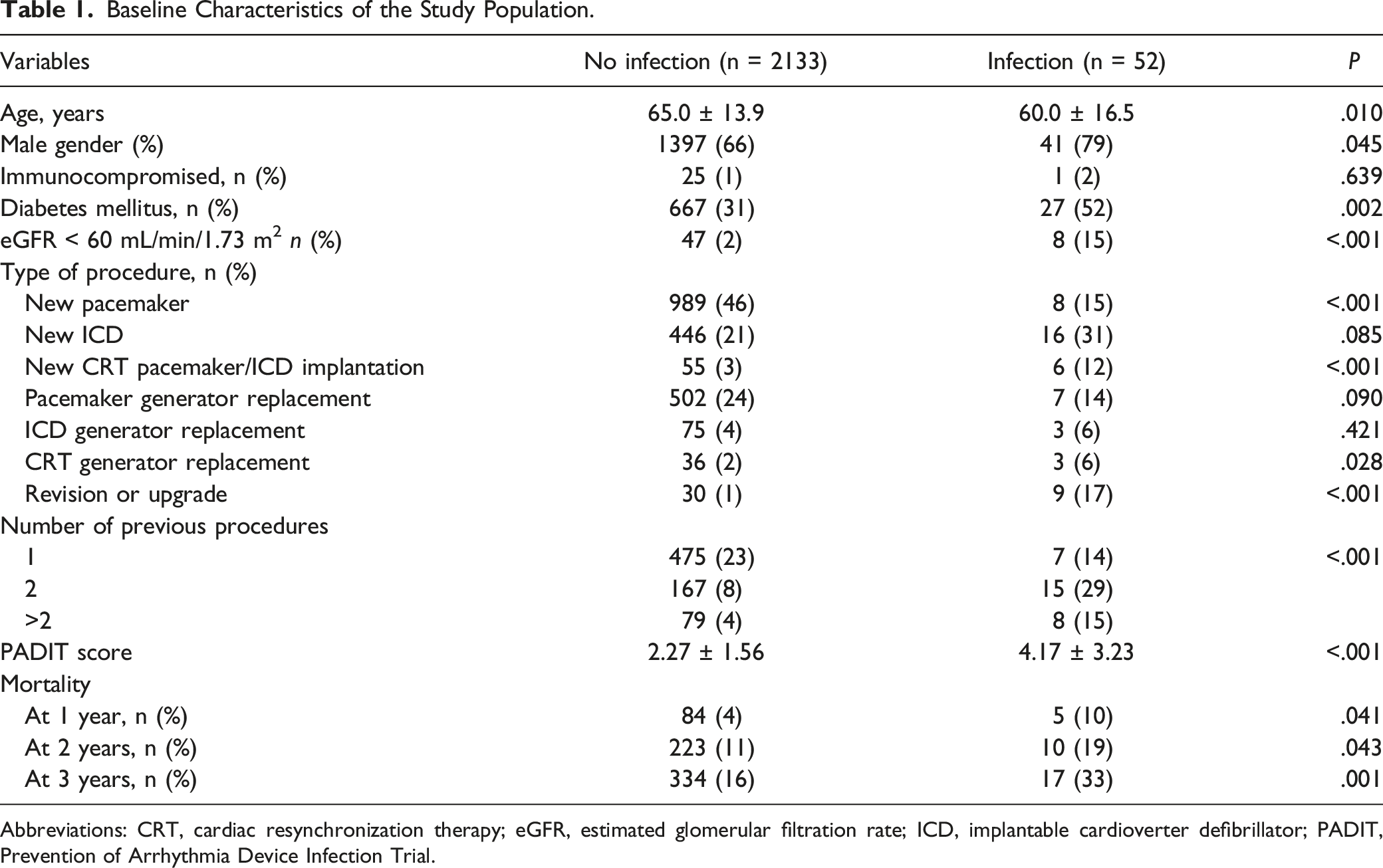
Abbreviations: CRT, cardiac resynchronization therapy; eGFR, estimated glomerular filtration rate; ICD, implantable cardioverter defibrillator; PADIT, Prevention of Arrhythmia Device Infection Trial.
The median follow-up time was 28.7 months (6.2-56.8). Among the 2185 patients, CIED infection was present in 52 (2.4%) patients. Of the 52 CIED infection patients, 13 (25%) had an early infection (<1 month after the procedure), 18 had (35%) late infection (1-12 months after the device-related procedure), and 21 had (40%) delayed infection (>12 months after the procedure).
The CIED infection rate was lower with initial pacemaker implantation vs repeat procedures (15% vs 46%, P < .001). New CRT pacemaker (CRT-P) or defibrillator (CRT-D) implantation and CRT generator replacement, revision, or upgrade were more common in CIED infection patients compared with patients without (each P < .05, Table 1). The number of previous procedures was higher in the infection group than in the non-infected group.
Laboratory Findings of the Patients.
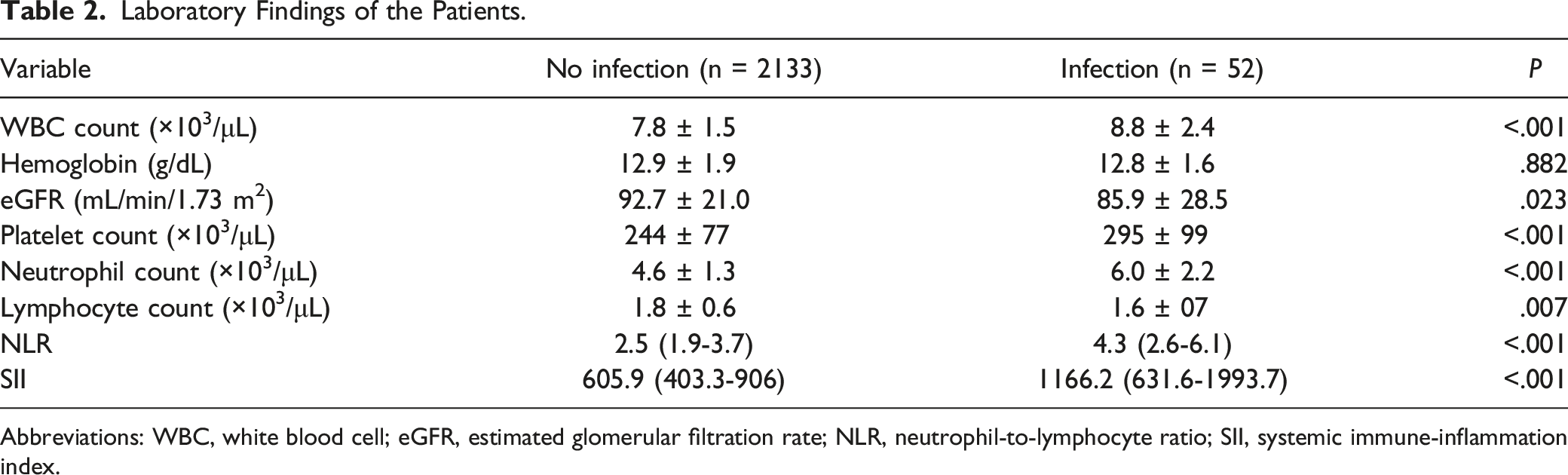
Abbreviations: WBC, white blood cell; eGFR, estimated glomerular filtration rate; NLR, neutrophil-to-lymphocyte ratio; SII, systemic immune-inflammation index.
Univariate and Multivariate Analysis for Prediction of CIED Infections.
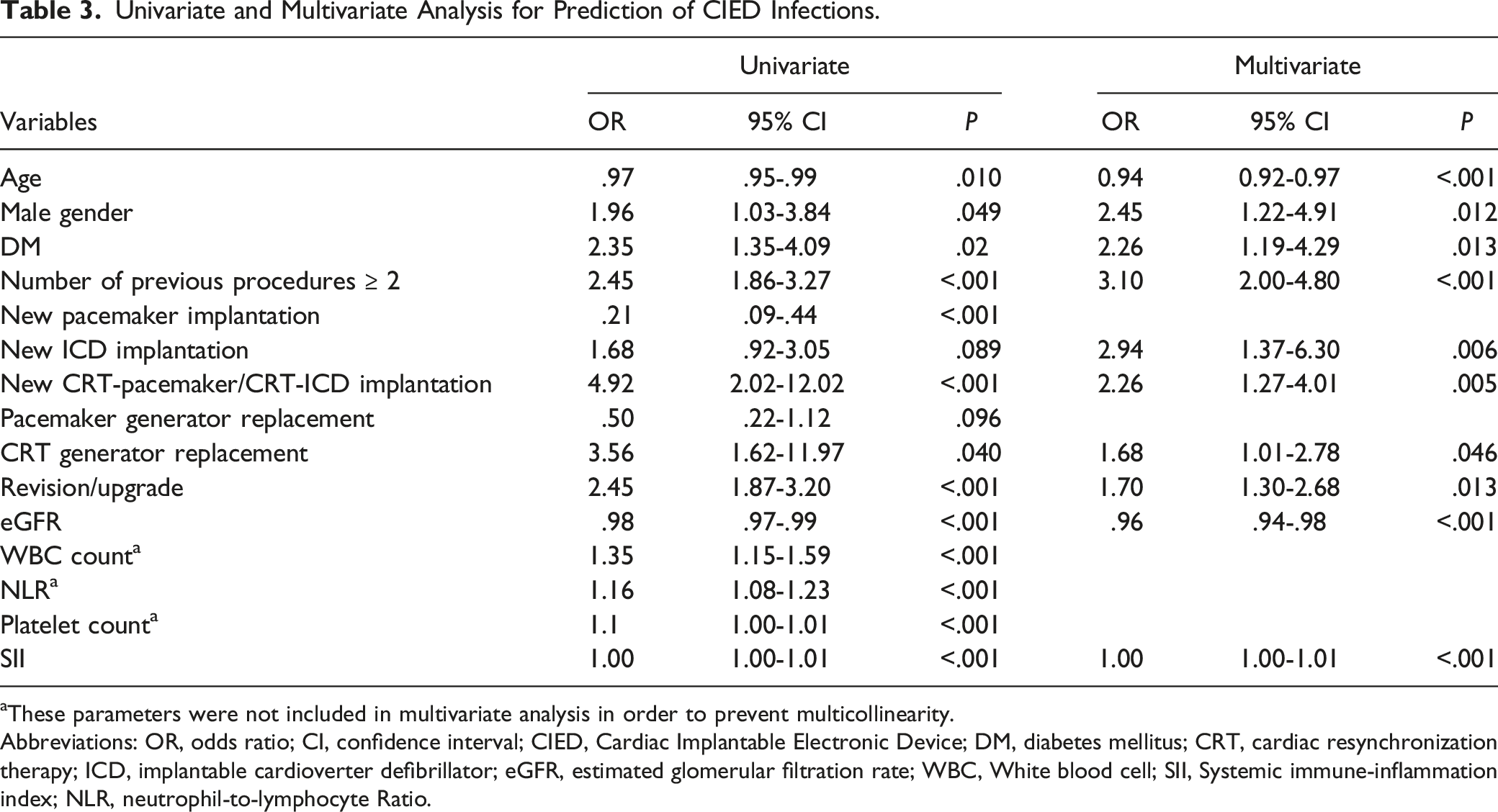
aThese parameters were not included in multivariate analysis in order to prevent multicollinearity.
Abbreviations: OR, odds ratio; CI, confidence interval; CIED, Cardiac Implantable Electronic Device; DM, diabetes mellitus; CRT, cardiac resynchronization therapy; ICD, implantable cardioverter defibrillator; eGFR, estimated glomerular filtration rate; WBC, White blood cell; SII, Systemic immune-inflammation index; NLR, neutrophil-to-lymphocyte Ratio.
The AUC of SII in the prediction of CIED infection was .733 (95% CI: .654-.811, P < .001, Figure 1). We found that SII had higher accuracy in predicting CIED infection compared with platelet count alone (SII vs platelet: AUC: .733 vs .646, z = 2.421, P = .0155, Figure 1). Although SII and NLR had similar accuracy for predicting CIED infection (SII vs NLR: AUC: .733 vs .715, z = .981, P = 0.326), SII was related to a significant NRI of 51.4% (z = 3.663, P = .0002) and an IDI of .014 (P < .05) compared with NLR alone (Figure 1). Receiver operating characteristic (ROC) curves of platelet count, neutrophil-to-lymphocyte Ratio (NLR), and the systemic immune-inflammation index (SII) in predicting Cardiac Implantable Electronic Device (CIED) infection.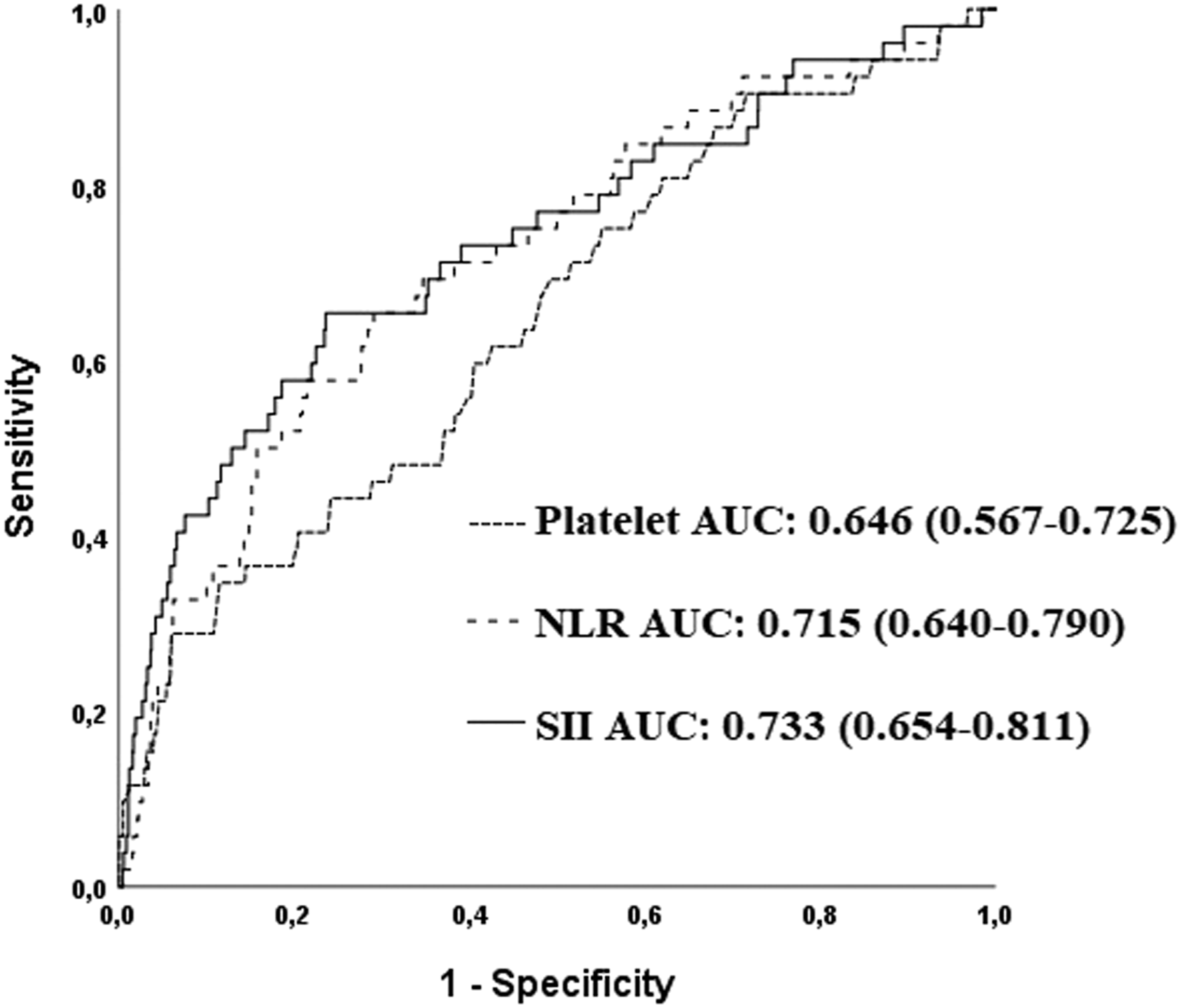
The adding of SII to a multivariable model including age, male gender, diabetes mellitus, eGFR, new ICD implantation, new CRT-P or CRT-D implantation, CRT-battery replacement, revision, or upgrade, and number of previous procedures provided a better predictive value than multivariable model alone (AUCs: .824 vs .749, z = 3.762, P < .001; NRI: 80.2%, P < .001; IDI: .085, P < .05, Figure 2). The predictive values of combination of SII with Prevention of Arrhythmia Device Infection Trial (PADIT) score
16
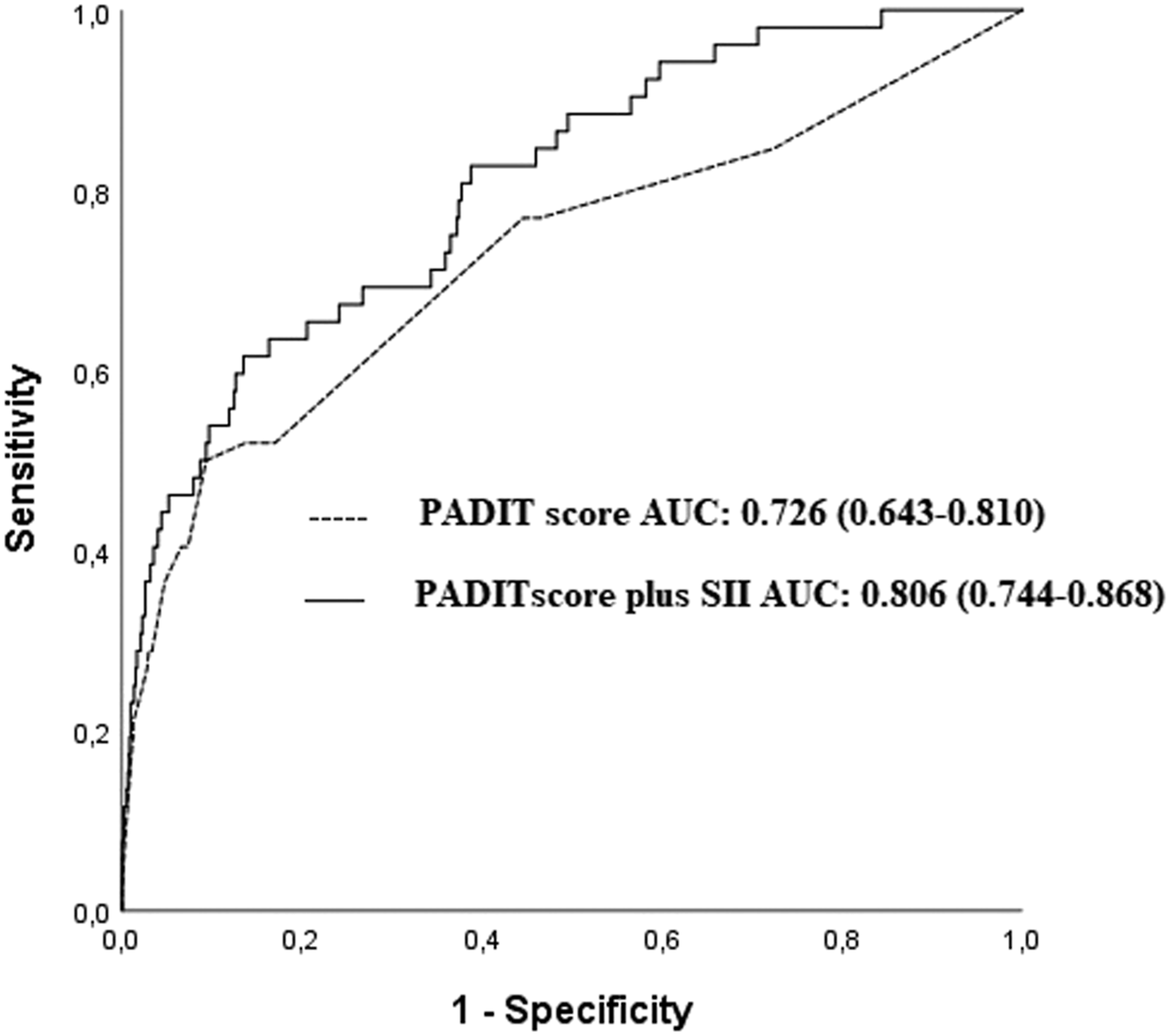
are provided in Figure 3 (PADIT plus SII vs PADIT; AUCs: .806 vs .726, z = 2.008, P = .044; IDI: .054, P < .05; NRI: 55.4%, z = 3.951, P < .001). Receiver operating characteristic (ROC) curves of the multivariable model and multivariable model plus systemic immune-inflammation index (SII) in predicting Cardiac Implantable Electronic Device (CIED) infection. Receiver operating characteristic (ROC) curves of the Prevention of Arrhythmia Device Infection Trial (PADIT) score and PADIT score plus systemic immune-inflammation index (SII) in predicting Cardiac Implantable Electronic Device (CIED) infection.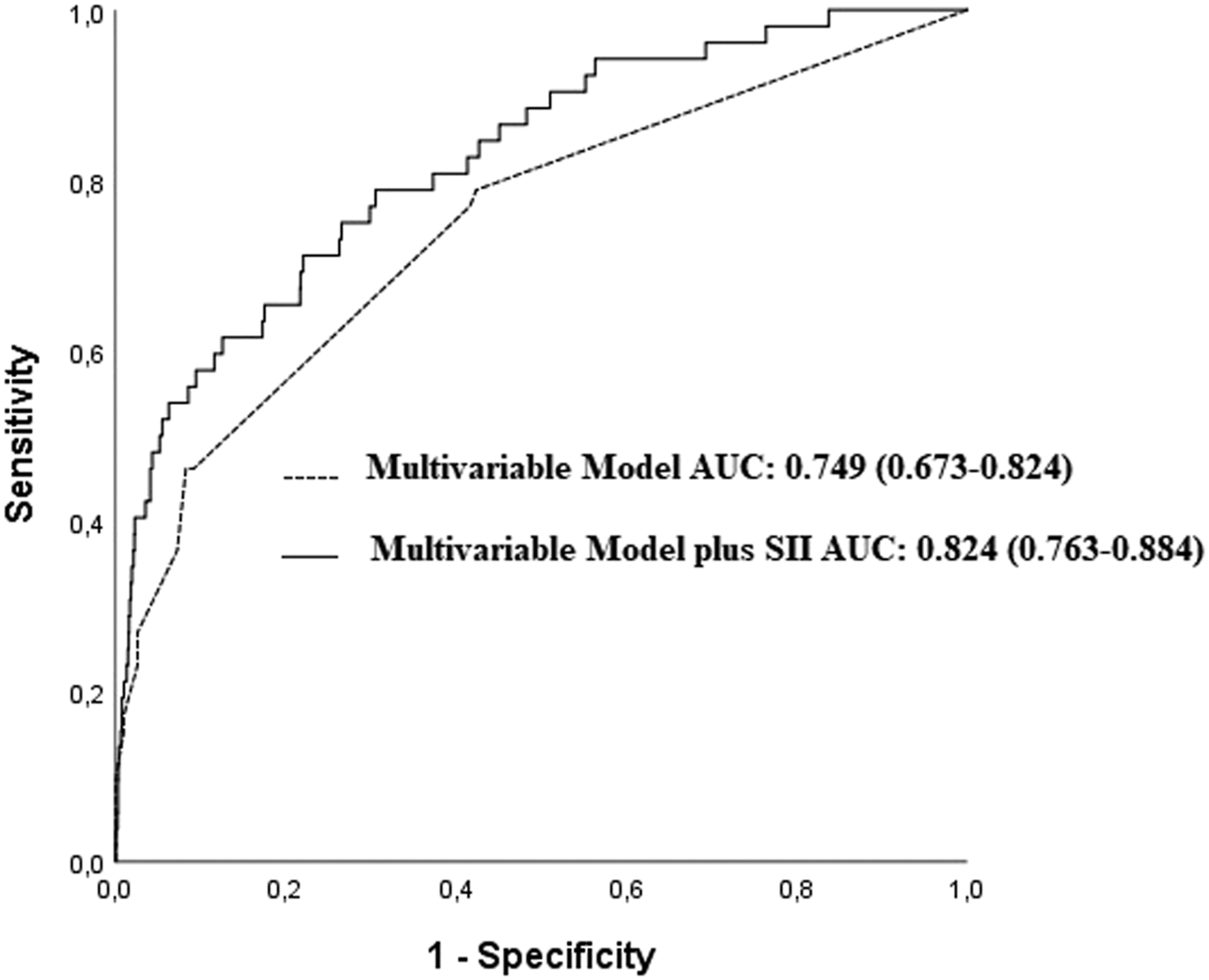
Microorganisms Isolated From Blood Culture.
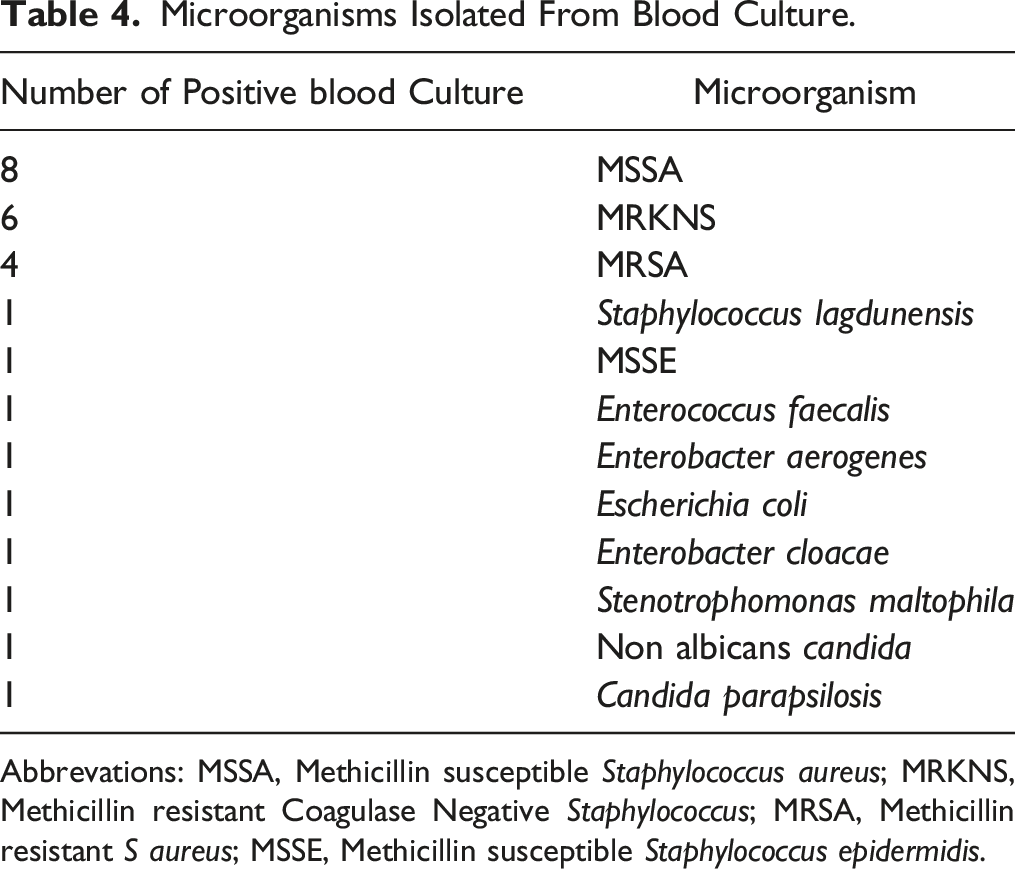
Abbrevations: MSSA, Methicillin susceptible Staphylococcus aureus; MRKNS, Methicillin resistant Coagulase Negative Staphylococcus; MRSA, Methicillin resistant S aureus; MSSE, Methicillin susceptible Staphylococcus epidermidis.
Of the bacteremic patients, 4 (14.8) had lead or valvular endocarditis and 23 (85%) had a pocket infection. In the lead-infected group, no mortality was recorded, whereas in the pocket-infected group 1 death due to heart failure occurred. Also, 1 death was recorded due to pneumonia, 2 years after pocket infection.
Discussion
The present study found that SII was independently associated with the development of CIED infection. Also, SII improved risk stratification, and enabled a more accurate prediction of CIED infection compared with the PADIT score alone.
The most important factors in preventing CIEDs are sterilization of the procedure area and obeying the rules of antisepsis. The infection rate is predicted to be low with formally educated and experienced clinicians since the duration of the process is expected to be shorter. Antibiotic prophylaxis is recommended by American and European Guidelines17,18 but there are fluctuating data and there is no decision about the duration of antibiotic therapy after the implantation process. Dwivedi et al 19 compared 2 groups that had antibiotic therapy for 48 h and 7 days/procedure; they found no significant difference in infection rate between the 2 groups. The authors usually use 1 dose antibiotic therapy before implantation and one dose afterward. In different centers, the study groups did not obtain any significant difference between prolonged antibiotic use for 1 day or 2 days after the procedure. According to these data, there is still a debate on a prophylactic antibiotic dose, duration, and type to prevent infection.
White blood cell and its subgroups are traditional inflammatory markers and have been widely used in clinical practice. Lymphocytes are the smallest cells among WBC. They mainly include T lymphocytes and B lymphocytes, which participate in cellular immunity and humoral immunity, respectively. Lymphocytes may secrete specific antibodies or produce cytotoxicity to those participating in immune inflammatory reactions. 20 It has been shown that a decreased lymphocyte count is associated with poor outcomes in cardiovascular disease. 21 Neutrophils play an important role in the early stage of vegetation formation. 22 Neutrophils also have an essential role in the development of immunothrombosis. 23 As platelets release numerous inflammatory mediators, they play a pivotal role in inflammation. 24 A previous study has shown that pathogens can promote vegetation formation by inducing intravascular neutrophil extracellular traps through activated platelets. 25 Bacterial adhesion to the damaged or inflamed structure such as valve and device may be promoted by platelets. 25 Therefore this may lead to the infection of sterile platelet-fibrin clots and the expanded vegetation. In a synergetic manner, the NLR can reflect more about disease severity than either of its constituent leukocyte counts. 26 Previous studies have demonstrated the prognostic value of immune biomarkers such as NLR in many diseases including coronary artery disease, and pneumonia.27-29 NLR has been shown to have predictive value for infective endocarditis.11,30 NLR was an independent predictor of adverse outcomes including in-hospital mortality or an intra-cerebral event (cerebrovascular event, meningitis, embolism, or brain hemorrhage) in patients with infective endocarditis. 26 In the present study, both platelet count and NLR were independent predictors of the development of CIED infection in patients who underwent CIED implantation. SII as a novel marker consists of the neutrophil count, lymphocyte count, and platelet count, and indicates the inflammatory and immune response simultaneously in patients. Furthermore, increased SII may represent the impaired balance of inflammatory and immunologic status. SII was found to be useful in predicting in-hospital mortality in patients with infective endocarditis. 24 Having higher SII was associated with embolic events in these patients. 31 In the present study, patients with CIED infection had higher SII values before the procedure compared with those without CIED infection. Higher SII before the procedure may be a sign of infection or inflammation, especially in cases having early infection risk.
Several risk scores have been developed for the pre-operative assessment of CIED infection risk, combining device-related and patient factors.16,32 PADIT study has an important place in this area. 16 Five independent predictors of CIED infection for PADIT are prior procedures [P], age [A], depressed renal function [D], immunocompromisation [I], and procedure type [T]. This risk score system did not include markers of inflammation such as WBC, NLR, and SII. In our study group, being young aged males, the primary application of CIED, procedures containing multi-lead like the implementation of CRT were found independently statistically significant for infection. In addition to having comorbidity as DM, CKD also increased SII before the implantation procedure was obtained as independent risk factors for post-procedure infections. Our study showed that SII can be used in addition to the PADIT score in predicting CIED infection in patients who underwent CIED implantation. The present study has some limitations. This is a retrospective, single-center study, including selection bias. As some patients were transferred from other hospitals to our hospital, there were no data regarding the presence of postoperative pocket hematoma, procedure time, anticoagulant use, or operator experience which are associated with the development of CIED infection. The lack of inflammatory markers, such as C-reactive protein, was another limitation. In the present study, SII was applied to single-center data. Thus, its performance should be externally validated in future studies.
Conclusion
We found that platelet count, NLR, and SII were predictive markers for CIEDs infection in patients who underwent CIED implantation. Early detection of SII may assist in early diagnosis, risk stratification, and management of these patients.
Footnotes
Author Contribution
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and, (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
