Abstract
This study evaluated the association between no-reflow (NR) and serum uric acid/albumin ratio (UAR) in 360 consecutive patients with non-ST-elevation myocardial infarction (NSTEMI) undergoing primary percutaneous coronary intervention. The study population was divided into two groups as follows: reflow (n = 310) and NR (n = 50) group. The thrombolysis in myocardial infarction (TIMI) flow score was used to describe NR. High UAR (Odds Ratio: 3.495, 95% CI; 1.216–10.048; P < .001) was found to be an independent predictor of NR. Additionally, UAR was positively correlated with the SYNTAX score and neutrophil/lymphocyte ratio; UAR was negatively correlated with left ventricular ejection fraction. The highest cut-off ratio of UAR predicting NR was found to be 1.35 with 68% sensitivity and 66.8% specificity. The area under the curve (AUC) for UAR was .768 (95% CI: .690-.847) after receiver operating characteristic (ROC) curve assessment. The AUC for UAR was found to be higher than for its components: serum uric acid (AUC: .655) and albumin (AUC: .663) (P < .001 for each evaluation).
Keywords
Introduction
Primary percutaneous coronary intervention (pPCI) is a reperfusion procedure used in patients with acute coronary syndrome. 1 In some cases, adequate tissue perfusion may not be achieved, even with epicardial coronary patency.2,3 This situation is called no-reflow (NR), which is linked with short- and long-term mortality and morbidity. 3 Although many treatment strategies have been proposed for the management of NR, there is no complete consensus on the issue as the pathophysiology of NR is still not clearly understood. The main possible causes of this phenomenon include distal microembolization, inflammation caused by microvascular spasm, ischemic endothelial damage, microvascular leukocyte plug formation, the effect of reactive oxygen species, and intravascular thrombus formation. 4
Albumin has several biological activities. 5 It is a negative acute phase reactant, so a systemic inflammatory response causes decreased albumin synthesis and increased albumin degradation. 6 In turn, decreased albumin levels can lead to higher blood viscosity and endothelial dysfunction. 7 In addition, albumin is an inhibitor of platelet activation 8 and a crucial regulator of coronary artery constriction triggered by platelets. 8
Serum uric acid (UA) is a possible predictor of coronary artery disease (CAD). 9 UA has been shown to be a regulator of vascular disease, inflammation and endothelial dysfunction, 10 just as hyperuricemia is associated with higher coronary atherosclerosis burden. 11
Studies have shown that serum the UA/albumin ratio (UAR), which includes two easily accessible parameters, plays a prognostic role in determining worse cardiovascular outcomes in patients with ST-elevation myocardial infarction (STEMI). 12 Moreover, this index has been shown to detect the level of CAD more effectively compared with the C-reactive protein/albumin ratio (CAR) in patients with non-STEMI (NSTEMI). 13
Since we did not come across any study showing that UAR had a relationship with NR in patients with NSTEMI who underwent pPCI, we proposed that UAR would predict NR better than serum UA or albumin alone in these patients. We also investigated the relationship with the SYNTAX score (SS)—(SYNergy between percutaneous coronary intervention with TAXus and cardiac surgery) which grades the complexity of CAD.
Material and Methods
Data Collection
We evaluated 360 consecutive NSTEMI patients who underwent pPCI in the cardiology department of our hospital between January 2020 and August 2022 as part of a retrospective cohort study. The diagnosis of NSTEMI was made according to current standards. 14 All clinical findings of NSTEMI patients who underwent pPCI were included within the study. We excluded patients with unavailable serum UA or albumin levels, <18 years of age, with severe inflammation, autoimmune or hematological diseases, patients who received thrombolytic therapy, those with malignancies, acute liver and kidney disease, use of medicine which may influence rates of serum UA, or patients with gout or high UA. Former medications, comorbidities, and baseline characteristics of all cases were obtained from the hospital database. The left ventricular (LV) ejection fraction was measured for each patient using the modified Simpson technique. The study protocol was approved by the Bozok University Ethics Committee. We obtained written informed consent from each patient, in accordance with the 1975 Declaration of Helsinki.
Laboratory Analysis
Subsequent to obtaining blood samples from peripheral veins during admission, the hematological coefficients were evaluated using a hematology analyzer (Beckman Coulter LH 750; Beckman Coulter Inc, USA). Conventional methods were used for assessing biochemical factors. Then, albumin concentrations were identified using the bromocresol green technique before pPCI. The standard reference value in our hospital for serum albumin was 3.5–5.2 mg/dL and 1.5–7.06 mg/dL for serum UA concentration. UAR was measured by dividing the serum UA level by the albumin level.
Angiographic Analysis
Coronary angiography was carried out using the Standard Judkins technique (Expo; Boston Scientific Corporation, Natick, MA, USA) and Siemens Axiom Sensis XP device (Munich, Germany). Each coronary artery was visualized in at least two perpendicular planes, and all coronary angiographic images were digitally recorded for quantitative analysis. pPCI was performed according to objective implementation standards using iopromide (low osmolarity and nonionic contrast agent). Before the intervention, each patient received an oral loading of 180 mg of ticagrelor or 600 mg of clopidogrel and 300 mg of acetylsalicylic acid, and all patients were given an intravenous bolus of unfractionated heparin 40–70 U/kg. The use of the stent type (bare metal or drug-coated) and the glycoprotein IIb/IIIa receptor inhibitor tirofiban was left to the discretion of the surgeon, while pPCI processes were conveyed in accordance with the latest NSTEMI guidelines published by the European Society of Cardiology. 14 Angiographic recordings were examined by two qualified operators who were unaware of the clinical findings. The thrombolysis in myocardial infarction (TIMI) score was used to measure flow following pPCI. For angiographic NR, TIMI flows were defined as 0, I, and II without dissection or coronary spasm. Advanced NR was mostly treated with adenosine, calcium channel blockers, nitroprusside, inhibitors of glycoproteins IIb/IIIa, or a combination of these drugs in our clinic.
Two independent experienced interventional cardiologists reviewed digital angiograms and calculated SS scores. These scores were not significantly different when compared with the findings of interventional cardiologists. Each lesion in epicardial arteries ≥1.5 mm in diameter and producing ≥ 50% lumen narrowing was measured through the online SS Calculator version 2.1 (www.syntaxscore.com).
Statistical Analysis
We used the SPSS software version 22.0 for Windows (SPSS Inc, Chicago, IL, USA) to analyze the study and the Kolmogorov–Smirnov test to identify the distribution patterns of the variables. We presented categorical variables in numbers and percentages and continuous variables as mean ± standard deviation or median with the interquartile range (IQR), based on patterns of distribution. Non-parametric continuous variables were compared using the Mann–Whitney U-test, whereas categorical variables—shown as percentages—were evaluated using Pearson’s chi-square or Fisher’s exact test. The correlation between UAR and SYNTAX score and NLR was evaluated by means of Spearman’s correlation analysis. In order to analyze the multivariate regression, we made use of the logistic regression analysis in examining the variables with a P < .05 in univariate analysis and detecting the independent predictors of NR. In addition, we assessed whether the model fits the Hosmer–Lemeshow goodness of fit statistics. We used receiver operating characteristic (ROC) curve analysis to detect the optimal cut-off value of UAR in defining NR and investigated specificity and sensitivity by using the Youden index. The findings of ROC curve analysis relating to albumin, NLR, uric acid, and UAR were evaluated by means of the DeLong test on the MedCalc program (MedCalc Software Ltd, Oostende, Belgium). A two-sided P < 0.05 was considered statistically significant.
Results
Of 360 NSTEMI patients, 71% (n = 220 patients) were male and 29% (n = 140 patients) were female, and 50 (13.8%) patients developed NR following pPCI.
Baseline Characteristics and Laboratory Parameters of the Study Groups.
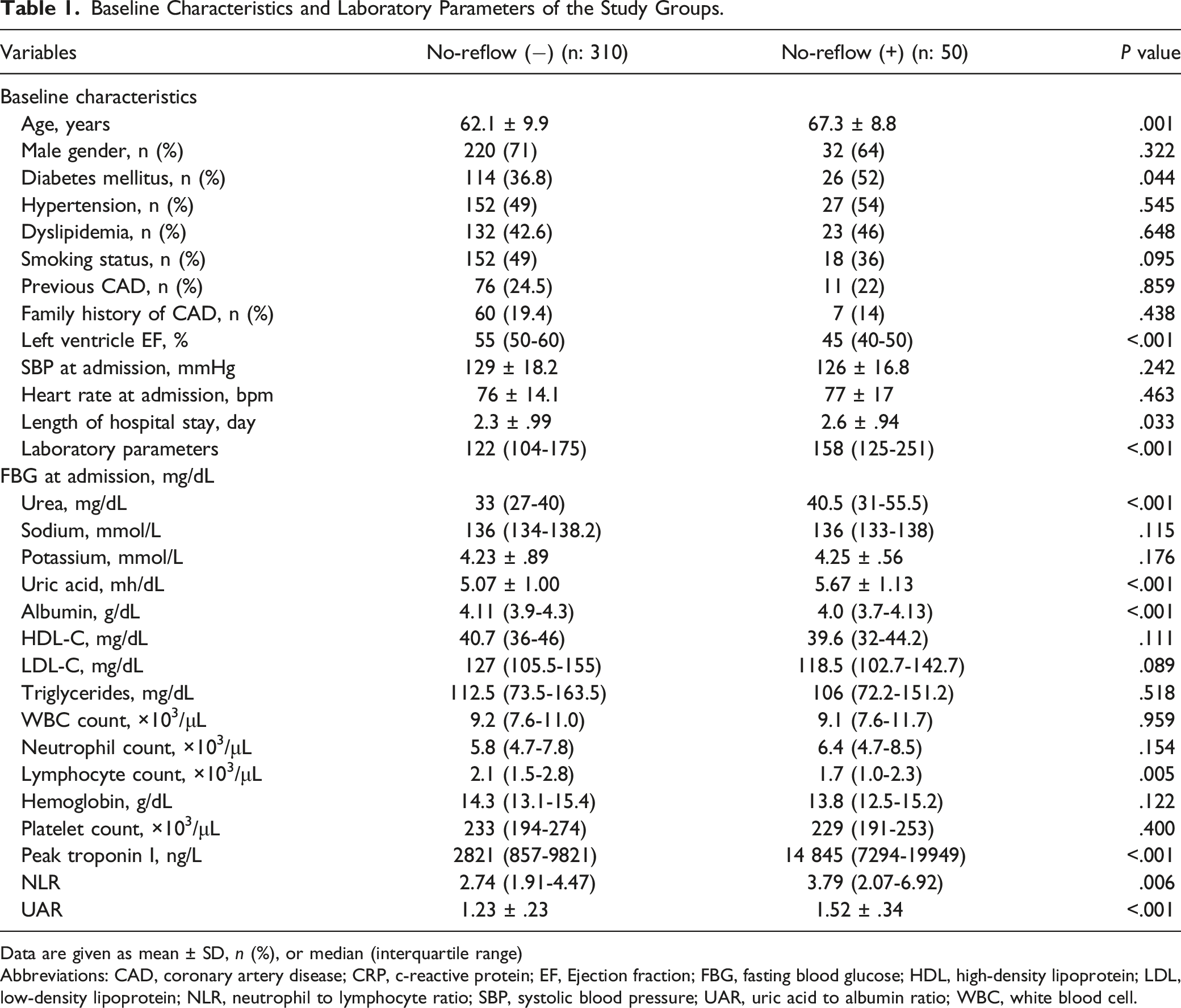
Data are given as mean ± SD, n (%), or median (interquartile range)
Abbreviations: CAD, coronary artery disease; CRP, c-reactive protein; EF, Ejection fraction; FBG, fasting blood glucose; HDL, high-density lipoprotein; LDL, low-density lipoprotein; NLR, neutrophil to lymphocyte ratio; SBP, systolic blood pressure; UAR, uric acid to albumin ratio; WBC, white blood cell.
Spearman Correlation Analysis between UAR, SYNTAX Score, LVEF, and NLR.
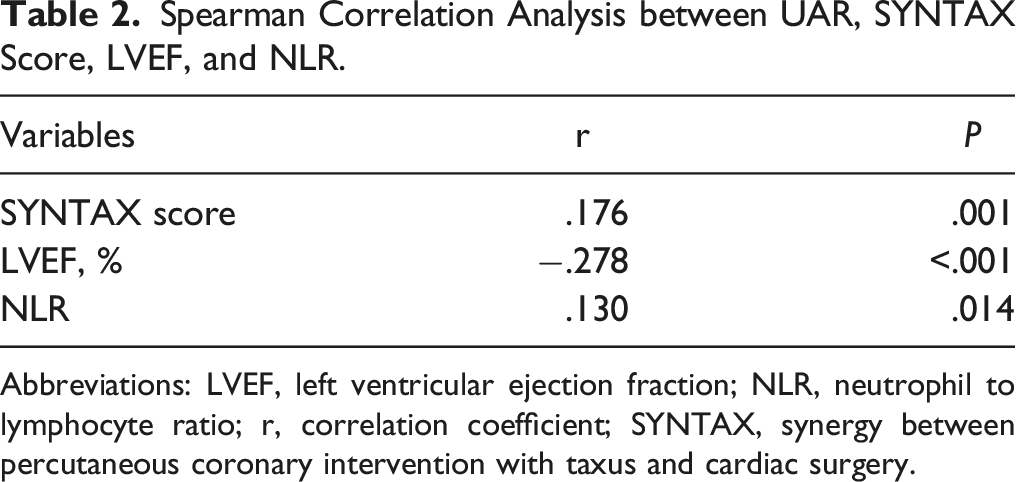
Abbreviations: LVEF, left ventricular ejection fraction; NLR, neutrophil to lymphocyte ratio; r, correlation coefficient; SYNTAX, synergy between percutaneous coronary intervention with taxus and cardiac surgery.
Angiographic Features of All Patients According to the Presence or Absence of No-Reflow.
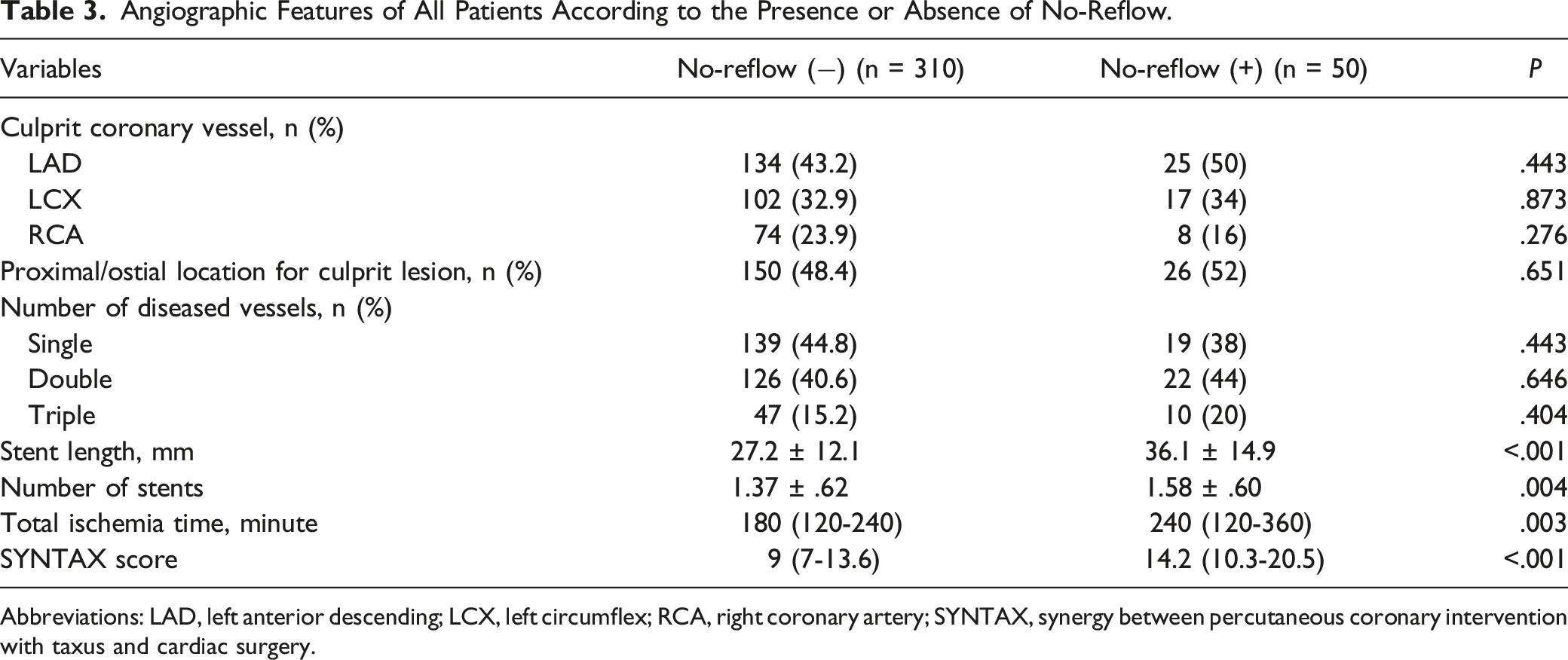
Abbreviations: LAD, left anterior descending; LCX, left circumflex; RCA, right coronary artery; SYNTAX, synergy between percutaneous coronary intervention with taxus and cardiac surgery.
Univariate and Multivariate Logistic Regression Analysis for Detecting No-Reflow.
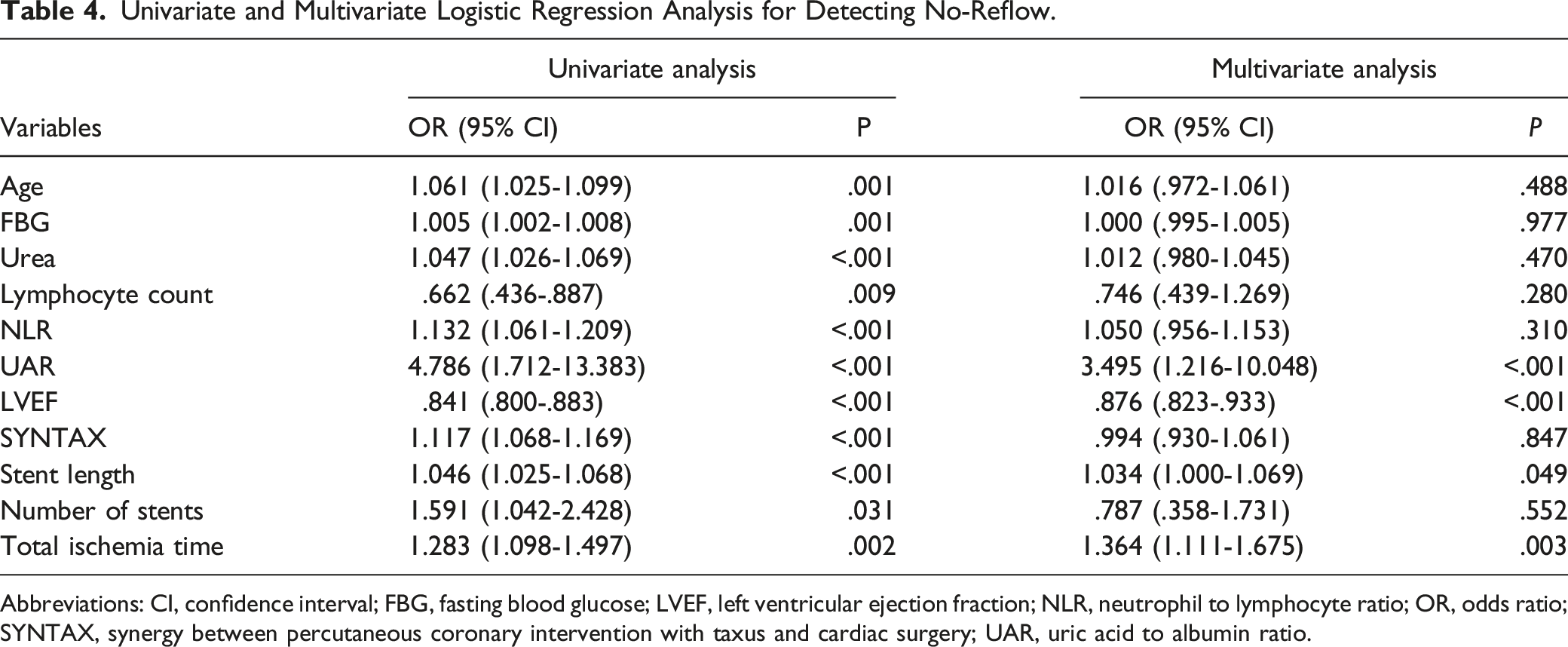
Abbreviations: CI, confidence interval; FBG, fasting blood glucose; LVEF, left ventricular ejection fraction; NLR, neutrophil to lymphocyte ratio; OR, odds ratio; SYNTAX, synergy between percutaneous coronary intervention with taxus and cardiac surgery; UAR, uric acid to albumin ratio.
Regarding ROC assessment, the area value beneath the curve (AUC) of the UAR was .768 (95% CI: .690-.847; P < .001) (Figure 1). Notably, the AUC of UAR was greater compared with its components, albumin (AUC: .663; 95% CI: .690-.847) and serum UA (AUC: 0.655; 95% CI: .690-.847) (P < .05 for both comparisons). In addition, when UAR and NLR were compared, the AUC of UAR was found to be higher than the AUC of NLR (AUC for NLR: .620; 95% CI: .529-.711; P = .006). In patients with NSTEMI, UAR was predictive for NR with a sensitivity of 68%, a specificity of 66.8% at a cut-off of 1.35. Systemic receiver operating characteristic (ROC) curves analysis of no-reflow prediction using the uric acid to albumin ratio (UAR), albumin, uric acid, and neutrophil-lymphocyte ratio (NLR).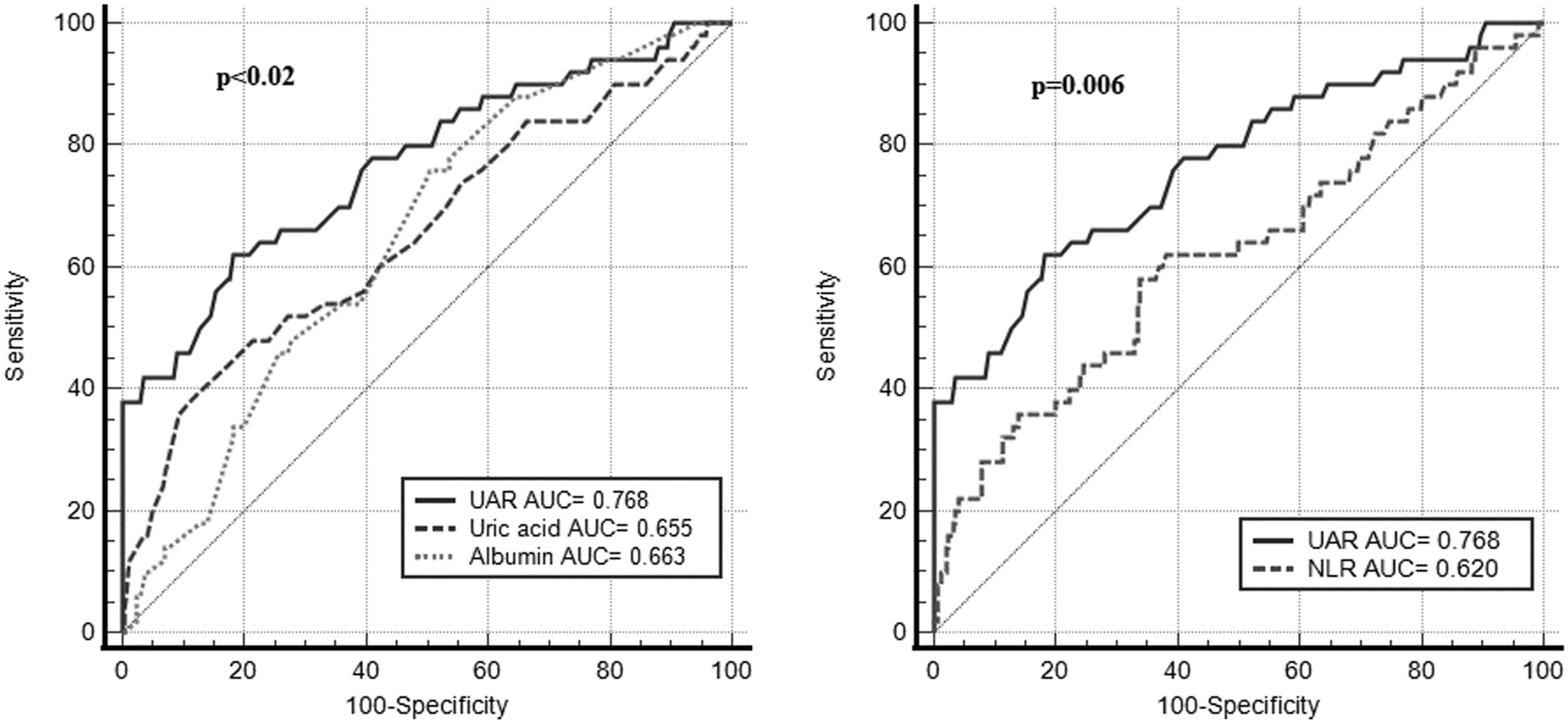
Discussion
To the best of our knowledge, this study is the first to show that UAR is an independent predictor of NR in patients with NSTEMI who underwent pPCI.
No-reflow linked to pPCI is associated with mortality and a higher risk of fatal arrhythmias in patients with STEMI and its frequency can range from 3% to 15%.1,15 In our study, the prevalence of NR was 13.8%. Although many tests have been processed to determine risk variables associated with NR, we still need accurate risk assessment tools that provide definite results.15,16 Being aware of such deficiency, we proposed a simple index to predict NR in patients with NSTEMI.
Albumin has several functions, such as the transport of different physiologically active materials, and pH buffering, as well as anti-inflammatory and antioxidant properties.5,6 Studies have shown that lower blood albumin levels following pPCI are associated with a high chance of causing NR and a lower degree of myocardial flushing in patients with NSTEMI. 17 Lower blood albumin levels can indicate chronic inflammation, and low blood albumin levels are associated with poor cardiovascular outcomes in patients with constantly recurring coronary syndrome. 18 Low levels of serum albumin are largely associated with coronary atherosclerosis. 19 In addition, a shortage of albumin may worsen injury caused by reperfusion in the myocardium, and lack of antioxidant properties may increase the risk of coagulation in the coronary capillary lumen.20,21
Uric acid can crystallize and initiate local inflammatory responses by forming monosodium urate crystals in different organs. 22 It has been reported that these local inflammatory activities involving UA crystals are significantly increased in patients with CAD. 23 Human atherosclerotic plaque contains significant amounts of UA, and high serum UA can promote thrombus formation—via purine metabolism.24,25 Additionally, UA can induce oxidative stress, inflammation, vasoconstriction, and endothelial dysfunction. 26 As observed in patients with hyperuricemia, each of the above factors is well known to play a significant role in the progression of atherosclerosis and may potentially lead to the progression of CAD. A hypoxic coronary microvascular state can trigger off purine degradation pathways, resulting in a rise in the production of UA. 27
Another study compared the prognostic value of various inflammatory indicators such as NLR, the C-reactive protein/albumin ratio (CAR), and platelet-lymphocyte ratios (PLR) and reported that CAR is a better marker of moderate-to-high SS than NLR in patients with NSTEMI. 28 Çakmak et al. 13 have reported that UAR is a better marker of CAD and a better inflammatory marker than NLR and CAR, while UAR is a stronger independent predictor for moderate-to-high SS compared with CAR. In a recent study, the perceived stress level was found to be an independent predictor of a high SS. 29 Another recent study, reported that CAR was independently associated with the development of no-reflow in STEMI patients. 30
Uric acid/albumin ratio has been studied as a new indicator of inflammation and oxidative stress in patients with cardiovascular issues. As suggested by some studies, UAR has been linked to CAD and SYNTAX score in patients with NSTEMI and it has also been reported to be an independent marker of the risk of contrast-induced nephropathy after pPCI in STEMI patients.13,31 A prospective study 32 showed that NR was independently associated with low ejection fraction in young STEMI patients. Additionally, long-term mortality was found to be significantly higher in patients with decreased LVEF. 32 The superiority of UAR over UA and albumin levels in reflecting systemic inflammation has been demonstrated, which has led to the investigation of a possible correlation between the development of UAR and NR in systemic inflammation. We observed through this research that UAR is an independent marker in the emergence of NR, thus we argue that UAR can predict NR better than serum UA or album alone. Since these measurable factors can easily be collected, it is possible to use UAR for NR estimation in patients with NSTEMI. In addition, as the risk of NR is higher in patients with high UAR, precautions can be taken against complications in NSTEMI patients undergoing pPCI.
The present study has some limitations. It had a retrospective, single-center design and only included patients with NSTEMI. Therefore, its findings may not apply to patients with other severe coronary syndromes. Blood samples were obtained on admission and an assessment of the extent of NR was carried out without using more sensitive methods (eg coronary flow reserve, contrast echocardiography or cardiac MRI).
Conclusion
The present study demonstrated that UAR is an independent predictor of NR in patients with NSTEMI. Moreover, the AUC of the UAR is more effective in detecting the incidence of NR compared with each of its components alone. This result may lead to a more aggressive pharmacoinvasive treatment strategy by predicting the risk of NR in patients with high UAR levels.
Footnotes
Author Contribution
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and, (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
