Abstract
The associations between several blood inflammatory indicators and risk of vascular plaques remain inconclusive. A total of 4596 native rural residents in Southeast China were enrolled from the Fuqing cohort study. Blood cell counts and their composite indexes including the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and 2 novel indicators (systemic immune inflammation index (SII) and systemic immune inflammation response index (SIRI)) were considered as inflammatory indicators. Common carotid and femoral intima-media thickness (IMT) and plaques were assessed using B-mode ultrasound. Unconditional or multinomial logistic regression was used to evaluate potential associations. The prevalence of multiple femoral plaques (defined as IMT ≥1.5 mm) was significantly higher among participants with the highest tertile of total leukocyte count (odds ratio, 1.78), neutrophil count (1.88), monocyte count (2.51), platelet count (1.68), NLR (1.93), PLR (1.57)
Keywords
Introduction
Inflammation plays a key role in the development of atherosclerosis and future cardiovascular events.1-5 Indicators of inflammation, including platelet counts, lymphocyte counts, neutrophil-to-lymphocyte ratio (NLR), and platelet-to-lymphocyte ratio (PLR), have been shown to be associated with an increased risk of cardiovascular outcomes as well as the severity of coronary artery disease.6-12 Carotid artery plaque has been widely used as a marker of carotid and systemic atherosclerosis, while carotid intima-media thickening (IMT) is a surrogate marker of atherosclerosis.13-16 Some studies have investigated the associations between blood cell counts and IMT or plaque formation. Several studies have shown that levels of leukocytes and its subtypes may be associated with inflammation level at the carotid intima-media;13,17,18 however, previous findings are inconsistent. 19 Other leukocyte indicators such as monocyte count and platelet count may also be correlated with the presence of carotid plaque.20,21
A growing number of studies have reported that levels of several systemic inflammatory markers such as NLR and PLR can partly reflect the development of inflammation-related diseases, including subclinical or carotid atherosclerotic plaques.22-24 Two relatively novel indexes, systemic immune inflammation index (SII) and systemic immune inflammation response index (SIRI), which also are calculated based on the counts of the leukocytes, neutrophils, lymphocytes, and monocytes, have been applied in the prediction of cancer survival; 25 however, the relationship between these 2 inflammation indexes and the development of atherosclerosis as indicated by presence of atherosclerotic plaques is unknown. Cardio-ankle vascular index has been typically used as a marker for atherosclerosis of the lower extremities.26,27 Recent studies suggested that measurement of femoral artery by duplex ultrasound may be a better screening method than ankle brachial index for identifying the participants at risk of atherosclerosis of the lower extremities.28,29 Combined femoral and carotid plaque assessment might also have a better capacity to identify populations at high risk of coronary heart disease.28,30
There are limited data available for assessing the association between inflammatory factors and increased IMT or atherosclerotic plaques especially based on ultrasound measurement of the femoral and carotid arteries. Therefore, in the present study, we used baseline data collected from first wave of Fuqing cohort study, a population-based cohort study, to assess the potential associations between several inflammation indicators, including leukocyte count, platelet count and composite inflammatory indexes, and the presence of atherosclerotic plaques in the carotid and femoral arteries.
Materials and Methods
Study Design and Participants
We used baseline survey data of 7218 participants recruited into Fuqing cohort from the general population of Fuqing city in Southeast China from March of 2019 to November of 2019. Participants, aged 35–75 years who were native residents of Fuqing, were eligible to participate in the study. All enrolled participants were invited to receive a face-to-face questionnaire interview, basic physical examination, laboratory tests for blood cell counts, fasting serum biochemical profile, HbA1c, B-mode ultrasound examination of the carotid and femoral arteries, and other clinical examinations. This study was approved by the ethical committee of Fujian Medical University (approval number, 2017-07), and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and all participants provided written informed consent.
Quality Control of Data Collection
Logic errors in the dataset were checked and corrected for the stored electronic records. Ultrasound data of the carotid and femoral arteries were doubly entered and cross-checked using Epidata (EpiData Association, Odense, Denmark) and Office Access software (Microsoft, Silicon Valley, USA). Quality control for laboratory testing was performed by regular internal quality control procedures and an external quality assessment scheme. Participants who did not participate in the questionnaire interview, or were outside the eligible age range, or had missing data on blood lipid or glucose levels, blood cell counts and IMT data, or had acute inflammation (total leukocyte count ≥14×109/L), or had been using lipid-lowering drugs were excluded from the study. Finally, 4596 participants were left for the analysis of the associations between blood inflammatory indicators and IMT or atherosclerotic plaques (Supplementary Figure 1).
Measurement of IMT and Atherosclerotic Plaque
IMT and presence of atherosclerotic plaques were measured at the far wall of the left and right carotid arteries and femoral arteries using B-mode ultrasonography (portable ultrasonic diagnostic instrument, EDGEII, SonoSite, Bothell
Carotid or femoral plaques were scanned in the carotid sinus, common carotid artery, and common femoral artery. The presence of atherosclerotic plaque was defined as a focal structure that encroaches into the arterial lumen by at least .5 mm, or >50% of the surrounding IMT value, or in presence of thickening ≥1.5 mm of IMT. The number of atherosclerotic plaques in each participant was estimated by summing the number of plaques identified by doctors and the number of arteries with IMT ≥1.5 mm (defined to be plaque) from all IMT measurement sites. Estimated stenosis severity was classified into no stenosis or stenosis (mild, moderate, or severe) based on B-mode imaging of identified plaques.
Assessment of Inflammatory Indicators
Blood cell counts were measured using standard laboratory procedures (Fully automated blood cell analyzer, Xs-1000i, Sysmex, Osaka
Measurement of Potential Confounding Factors
Information for well-known or other potential confounders and mediators for atherosclerotic disease included demographics, lifestyle, and status of chronic diseases.17,31-35 Information regarding age, gender, education level, smoking, alcohol drinking, and family history of cardiovascular diseases was collected by trained interviewers using electronic, structured questionnaires. Cigarette smoking was defined as smoking ≥1 cigarette/day in the previous half a year. Drinking alcohol was defined as drinking at least once per week during the past year. Height (cm) and weight (kg) were recorded by anthropometric measurements, and body mass index (BMI) was calculated and classified into 4 groups according to the WHO standard for the Asian (<18.5, 18.5-23.8, 23.9–27.9, or ≥28.0 kg/m2). Resting systolic and diastolic blood pressures were measured twice. Serum lipid profile and fasting glucose level were measured using standard laboratory procedures (Toshiba automatic biochemical analyzer, TBA-120FR, Tokyo
Hypertension (yes or no) was defined as the average systolic blood pressure ≥140 mmHg, or average diastolic blood pressure ≥90 mmHg, or self-reported ever diagnosis, or use of antihypertensive medication. Hyperlipidemia was defined as present if ≥1 of the following criteria are satisfied: total cholesterol >6.2 mmol/L, triglyceride >2.3 mmol/L, high-density lipoprotein cholesterol <1.0 mmol/L, or low-density lipoprotein cholesterol >4.1 mmol/L. Diabetes was defined as fasting glucose ≥7.0 mmol/L, HbA1c ≥6.5, being diagnosed by doctors, or on anti-diabetes treatment.
Statistical Analysis
Differences in distribution of demographics and medical histories between participants with and without atherosclerotic plaques at any of the measurement sites were compared using chi-squared test. All inflammation indicators were divided into 3 levels according to the tertiles among the participants with IMT of <1.0 mm. Fully adjusted unconditional or multinomial logistic regression models were employed to examine the associations between inflammation indicators (counts of 5 types of blood cell and 4 inflammatory indexes) and the presence of carotid plaques or femoral plaques. The odds ratio (OR) and the corresponding 95% confidence intervals (CIs) were reported. Fully adjusted models included age, sex, education level, current occupation, smoking, alcohol drinking, hypertension, diabetes, hyperlipidemia, and family history of cardiovascular diseases (stroke or ischemic heart disease). Linear trend of dose-response relationship between inflammation indicators and risk of plaques was tested using logistic regression by treating the categorical inflammation indicators with 3 levels as continuous variables.
Stratified analysis by sex was performed, and the interaction P value was derived from likelihood ratio test based on 2 logistic regression models with or without the interaction terms. All analyses were performed with SAS 9.4 statistical software
Results
Characteristics of Participants with and without Carotid or Femoral Plaques
Characteristics of Study Subjects, Overall and by the Presence of Carotid or Femoral Plaques.
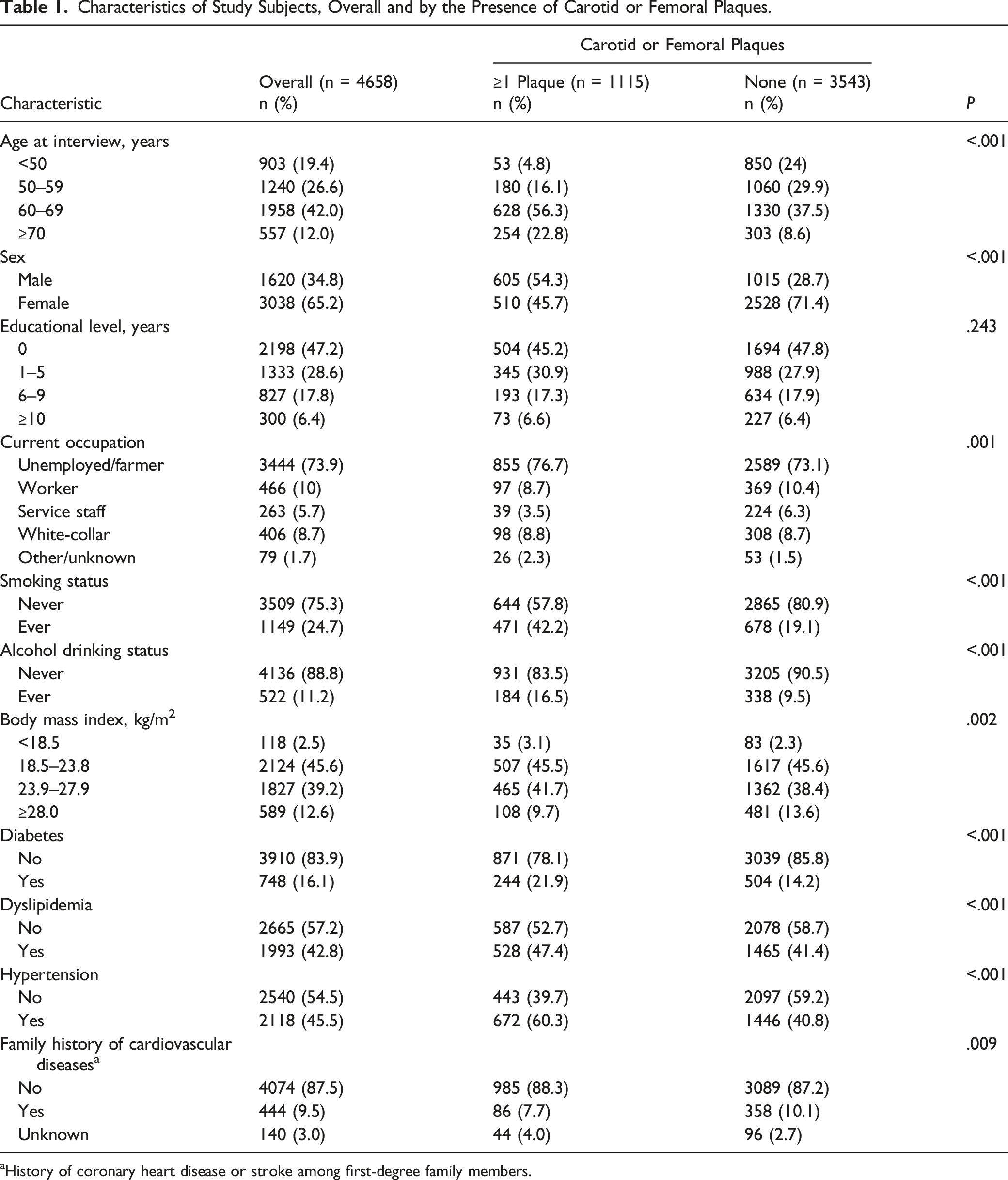
aHistory of coronary heart disease or stroke among first-degree family members.
Detection Rate of Carotid or Femoral IMT ≥1.5 mm or Stenosis due to Plaques
Figure 1 shows overlap of the detection of carotid or femoral plaques, IMT ≥1.5 mm and stenosis due to plaques. A total of 559 participants (detection rate of 12.0%) with carotid plaques and 792 (17.0%) with femoral plaques were detected. There was lower detection rate of plaques defined by IMT ≥1.5 mm in the carotid arteries (1.6%, 74/4658) than in the femoral arteries (5.3%, 247/4658). For participants with carotid or femoral plaques, 133 with mild or worse stenosis (47 carotid stenosis, 69 femoral stenosis, and 17 carotid and femoral stenosis) were further ascertained, and the detection rate of stenosis in the carotid arteries was a bit higher than that in the femoral arteries. Venn diagram of overlaps of the detected intima-media thickness thickening (≥1.5 mm) (A) and stenosis due to plaques (B), and carotid or femoral plaque. Dark gray in part A and part B indicates presence of carotid plaque or femoral plaque; white in part A indicates carotid intima-media thickness thickening (≥1.5 mm) or femoral intima-media thickness thickening (≥1.5 mm); white in part B indicates carotid stenosis or femoral stenosis.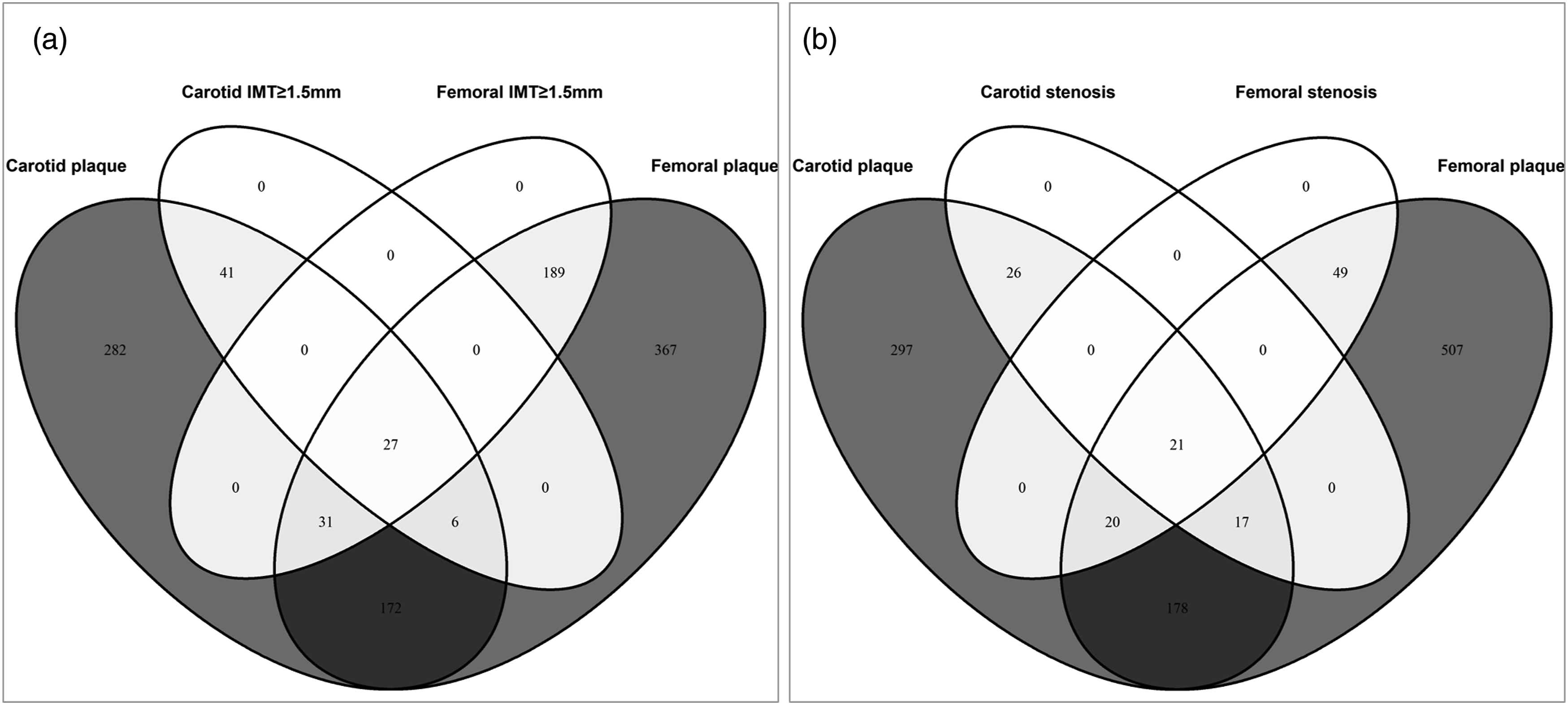
Blood Inflammatory Indicators and Risk of the Plaques
Odds Ratios and Corresponding 95% Confidence Intervals for Risks of Carotid Plaque and Femoral Artery Plaque in Association with Inflammatory Indexes.
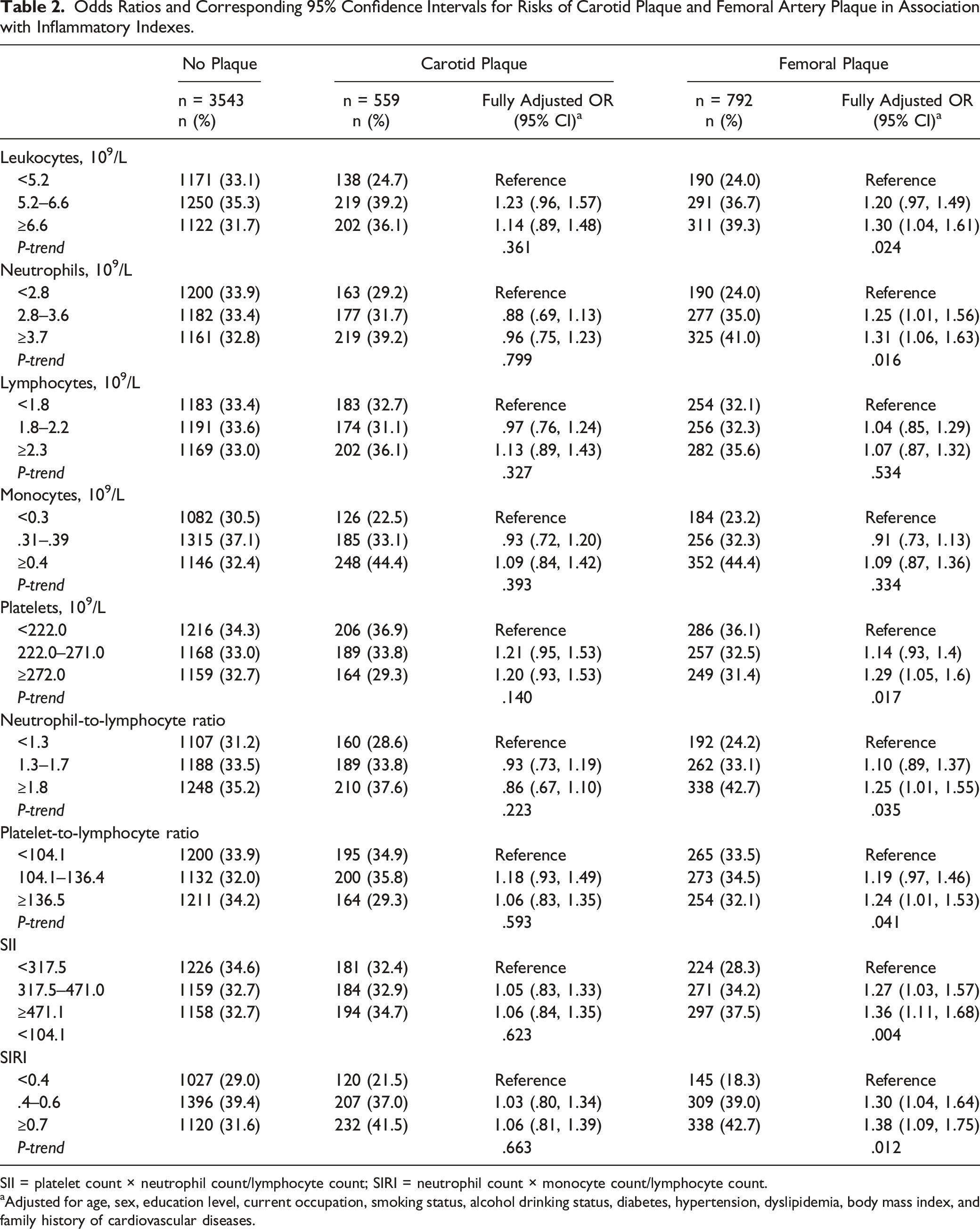
SII = platelet count × neutrophil count/lymphocyte count; SIRI = neutrophil count × monocyte count/lymphocyte count.
aAdjusted for age, sex, education level, current occupation, smoking status, alcohol drinking status, diabetes, hypertension, dyslipidemia, body mass index, and family history of cardiovascular diseases.
Blood Inflammatory Indicators and Risk of Femoral Plaques Defined by IMT ≥1.5 mm
Odds Ratios and Corresponding 95% Confidence Intervals for Association Between Inflammatory Indexes and Femoral Artery Plaque Defined by IMT ≥1.5 mm.
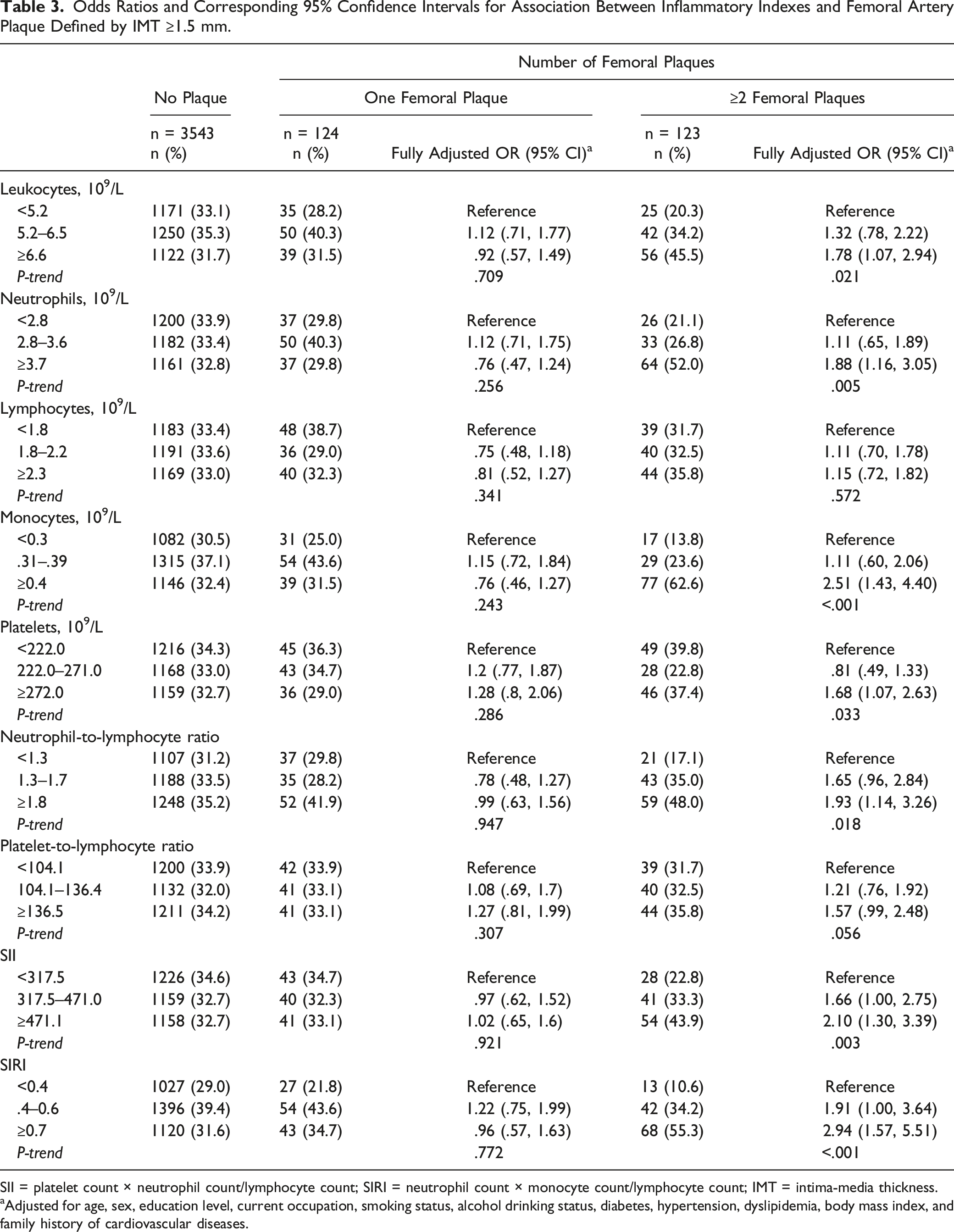
SII = platelet count × neutrophil count/lymphocyte count; SIRI = neutrophil count × monocyte count/lymphocyte count; IMT = intima-media thickness.
aAdjusted for age, sex, education level, current occupation, smoking status, alcohol drinking status, diabetes, hypertension, dyslipidemia, body mass index, and family history of cardiovascular diseases.
Blood Inflammatory Indicators and Risk of Carotid or Femoral Stenosis due to Plaques
Odds Ratios and Corresponding 95% Confidence Intervals for Risks of Femoral Stenosis Due to Plaques, and Carotid or Femoral Stenosis, in Association with Inflammatory Indexes.
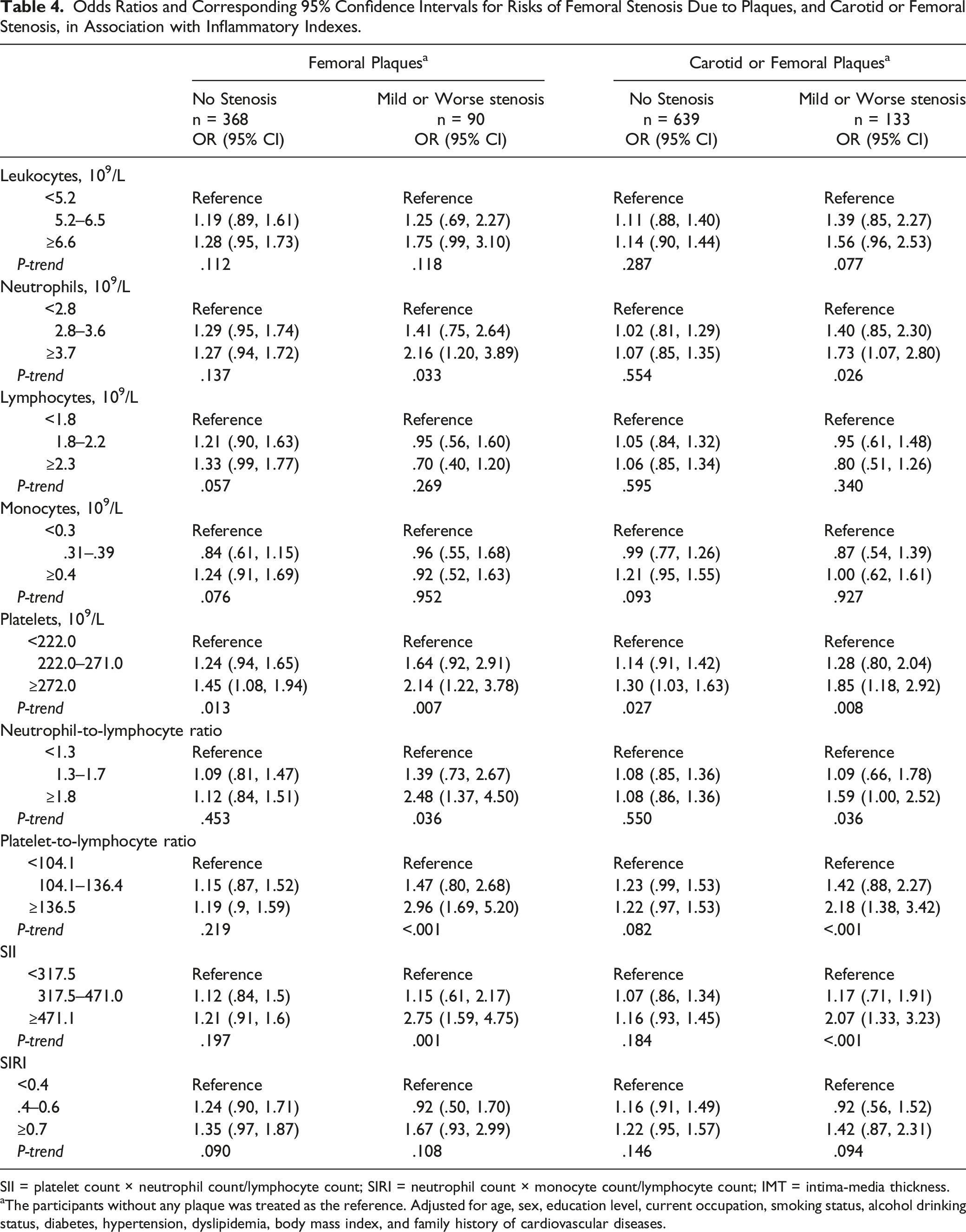
SII = platelet count × neutrophil count/lymphocyte count; SIRI = neutrophil count × monocyte count/lymphocyte count; IMT = intima-media thickness.
aThe participants without any plaque was treated as the reference. Adjusted for age, sex, education level, current occupation, smoking status, alcohol drinking status, diabetes, hypertension, dyslipidemia, body mass index, and family history of cardiovascular diseases.
Stratified analyses by sex showed that the increased levels of lymphocyte count and platelet count were significantly associated with risk of presence of femoral plaques among males but not among females (Supplementary Table 1).
Discussion
This study investigated the association between counts of multiple types of blood cells including total leukocytes, neutrophils, lymphocytes, monocytes, and platelets; 4 composite inflammatory indexes (NLR, PLR, SII, and SIRI); and the prevalence of plaques in carotid and femoral arteries. It was found that increased levels of inflammatory indicators were associated with significantly higher prevalence of plaques in the femoral arteries. This phenomenon was more obviously observed among the participants with multiple femoral plaques defined as IMT ≥1.5 mm or those with carotid or femoral stenosis, and there were significant linear dose-response relationships between these inflammatory indicators and risk of presence of ≥2 femoral plaques and the prevalence of stenosis.
Plaques especially in the carotid arteries have been widely accepted as predictive indicators for coronary heart disease or stroke.4,14-16,36,37 The relationship between blood leukocytes or its subtypes and risk of plaques in carotid artery has been extensively investigated; however, there is limited data on the associations between these inflammatory indicators and atherosclerosis in femoral arteries. Positive associations were reported between leukocytes (most frequently reported), monocytes, and risk of the presence of carotid atherosclerosis or plaques.17,18,20,31,38 Similar with our results, 2 studies also showed that severity of carotid atherosclerosis was associated with concurrently increased count of multiple blood inflammatory indicators including total leukocytes, neutrophils, and monocytes.39,40 However, there were also inconsistent results, 21 for example, one population-based medical screening study in Japan (3738 participants) reported that the leukocyte count did not correlate with carotid atherosclerosis, 19 and it was weakly correlated with cardio-ankle vascular index in men.
Among studies on the composite inflammatory indexes (NLR and PLR), a high NLR was associated with increased carotid IMT or plaques, or it was a significant predictor (NLR >2.4 was associated with an average presence of 2.86 carotid plaques) of the degree of carotid stenosis.23,38,41 However, there was also a study reporting that NLR was not related to carotid atherosclerosis progression. 42 A high PLR may be associated with vulnerable plaque features. 43 Our study also first reported that the presence of both multiple plaques and IMT ≥1.5 mm or severity of stenosis was accompanied by increased levels of 2 novel indicators (SII and SIRI).
To our knowledge, there was some evidence supporting why inflammatory biomarkers had stronger correlation with femoral plaques than carotid plaques. Similar with us, several studies including 1 cross-sectional study 44 and 2 autopsy studies45,46 demonstrated that the presence of femoral plaques showed stronger associations with traditional cardiovascular risk factors, coronary plaques, and coronary deaths than plaques in the common carotid arteries, which may partly support our conclusions. Additionally, the differential outcome definition might cause confusion when comparing results across different studies, although most studies used the maximal IMT or maximal thickness of the plaques as the outcome of carotid atherosclerosis. In our study, the limited sample size (n = 74) of common carotid plaque defined by ≥1.5 mm prevented us for further evaluation. Thus, more studies are needed to verify the findings and to explore potential biological mechanisms.
Moreover, the stratified analysis also found the increase in lymphocyte count was associated with the presence of femoral plaques among males, but not among females, which may be related with different distribution of confounding factors across sex or attributable to the residual unexpected confounding factors. Another population-based study also reported similar results. 39
We excluded the participants who were likely to have acute infection (leukocytes ≥14×109/L), and our study was more likely to evaluate chronic blood inflammatory status and current presence of atherosclerosis. In view of physiological mechanisms, differential monocyte subpopulations coupled with platelets, leukocyte, and lymphocyte subpopulations may be involved in the infiltration into the vessel wall or inflammatory reactions, or act as indicators of vascular repair activity in the atherosclerosis.47,48 Increased levels of inflammatory markers may lead to inducing oxidative stress on the vascular walls, then both local and systemic inflammatory actions promote the development of atherosclerosis plaques and further cause arterial stenosis.49-51 One recent review pointed out that NLR plays a central role as a reliable indicator of inflammatory status and adaptive immunity. 52
The main strength of our study was a relative large-scale population-based study with strict study settings and good quality control. Bilateral carotid and femoral IMT and plaques were measured concurrently in our study, which was uncommon in studies on the same topic. While most of the studies focused on carotid artery plaque measurement, the femoral plaques may play a more important role for the evaluation of the lower extremity atherosclerosis. However, our study had several limitations. First, this cross-sectional study did not observe the onset of the exposure and the outcome, which limits the temporal interpretation or causal inference. Second, the misclassification bias might exist for measurement of the common carotid or femoral plaques because the evaluation of ultrasound imaging was dependent on doctors’ judgment and not all plaques had the measurement of thickness. But the carotid or femoral ultrasound measurement and blood cells test were performed independently, which to some extent reduced the possibility of false association. Third, the response rate of Fuqing cohort study at baseline in 2019 was about 20%; thus, selection bias may exist and influence the study conclusion, although a comprehensive list of potential confounders for atherosclerosis were adjusted in the analysis. The results obtained from our single population limit the generalizability of the findings to other populations. Finally, we did not evaluate other important indicators of inflammation such as high sensitivity C-reactive protein,53,54 which limits our capacity of detailed evaluation for all potential inflammation pathways.
Conclusion
The findings of this study suggested that blood inflammatory markers may affect the development of atherosclerosis as indicated by the increase in the presence of plaques and stenosis. The observed associations may be stronger for the femoral artery. The findings of our cross-sectional study should be treated with caution. It is necessary to conduct prospective studies to further investigate the observed associations.
Supplemental Material
Supplemental Material - Leukocyte and Platelet Related Inflammatory Indicators and Risk of Carotid and Femoral Plaques: A Population-Based Cross-Sectional Study in Southeast China
Supplemental Material for Leukocyte and Platelet Related Inflammatory Indicators and Risk of Carotid and Femoral Plaques: A Population-Based Cross-Sectional Study in Southeast China by Ruimei Feng, PhD, Yiquan Dai, MM, Shanshan Du, PhD, Wenbin Liang, PhD, Hongyu Chen, MM, Cheng Chen, MM, Tianmin He, MM, Tao Tao, PhD, Zhijian Hu, PhD, Pingfan Guo, MD, and Weimin Ye, PhD, MD in Angiology
Footnotes
Acknowledgments
The research team would like to thank the participants, interviewers, nurses of the Fuqing cohort study and local government officials, doctors and other healthcare professionals, who have been providing generous support for the day-to-day research field work operation.
Author Contributions
All authors contributed to: (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology of the People’s Republic of China (grant number: 2017YFC0907102), Government of Fuqing city (grant number: 2019B003), Department of Science and Technology of Fujian, China (grant number: 2019L3006), and High-level Talents Research Start-up Project of Fujian Medical University (No. XRCZX2017035 and No. XRCZX2020034).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
