Abstract
Low-grade inflammation and arterial stiffness are key factors in the development of vascular aging. However, the interplay between arterial stiffness and inflammation for cardiovascular (CV) disease is unclear. Aortic pulse wave velocity (aPWV) and the inflammatory markers, high-sensitivity C-reactive protein (CRP) and orosomucoid, were measured in 2710 participants (median age: 72 years). These participants were followed up for a mean of 7.6 years for a composite CV disease end point. Per 1 interquartile range increment of CRP and orosomucoid, respectively, aPWV increased by 0.19 m/s (95% CI: 0.07-0.32) and 0.19 m/s (0.11-0.27), after multifactorial adjustment. Mediation analysis showed that aPWV, after multifactorial adjustment, mediated 8% (−4, 20) of the CV disease risk associated with CRP and 8% (−4, 18) of orosomucoid risk. The associated risk increased with combinations of high aPWV and high CRP or orosomucoid. We found no evidence that arterial PWV acted as an important mediator of the relationship between systemic inflammation and CV disease risk in this elderly population. The results instead indicate an additive effect. Our study supports the view that arterial stiffness and chronic inflammation affects CV risk mainly through separate causal pathways.
Keywords
Introduction
Chronic inflammation is an important component of cardiovascular (CV) risk, but hard to measure in a precise way, as the inflammatory process may be local and not fully mirrored by inflammatory biomarkers in the general circulation. Previous studies have shown that a number of disease conditions characterized by increased systemic inflammation (rheumatoid arthritis, inflammatory bowel disease, etc) are associated with higher levels of arterial (aortic) pulse wave velocity (aPWV),1–5 a marker of vascular aging (arterial stiffness), and with increased risk of CV disease (CVD) events.6,7 Inflammatory biomarkers in blood, which are often increased in these disease conditions, have also been shown to be associated with increased aPWV. 8 Furthermore, decreases in aPWV have been observed after inhibition of components of the inflammatory cascade (interleukin-12 [IL], IL-6, and tumor necrosis factor-α) in smaller clinical studies (35-50 patients) with inflammatory diseases, including psoriasis, rheumatoid arthritis, or ankylosing spondylitis.9–11 These findings were highlighted in a consensus document of the European Society of Cardiology Working Group on Aorta and Peripheral Vascular Diseases, European Association of Cardiovascular Imaging, and Heart Failure Association. 12
The consequences of inflammation for arterial structure and function are especially relevant in the current COVID-19 (coronavirus disease of 2019) era and have recently been highlighted in a position statement from the European Society of Hypertension (ESH) and the Artery society. 13 Herein, it was proposed that aPWV could be used to identify high risk of CVD in individuals with inflammatory conditions. However, as also stated, a direct link between arterial stiffness and the increased CVD risk associated specifically with systemic inflammation has not yet been proven.
Hypothetically, systemic inflammation could be a confounding factor for the relationship between arterial aging and CVD. Arterial aging could also be a consequence of long-standing low-grade systemic inflammation, which partly mediates the increased CV risk. Alternatively, arterial stiffness and inflammation could represent 2 different pathways of CV risk. Although systemic inflammation is strongly associated with atherosclerosis, the relationship with arteriosclerosis (arterial stiffness) is not equally well documented in humans from the population. Using the Malmö Diet and Cancer Study cohort, we aimed to further elucidate the role that systemic inflammation—as measured by inflammatory biomarkers—plays for the increased prospective CV risk associated with arterial stiffness.
Participants and Methods
The aim was investigated in 3 steps. First, we examined the relationships between aPWV and 2 plasma markers of systemic inflammation, C-reactive protein (CRP), and orosomucoid (ORM). Both biomarkers have been previously associated with arterial stiffness and incidence of CVD.8,14,15 Secondly, we investigated whether the CV risk associated with the 2 inflammatory biomarkers could be mediated by elevated aPWV. Finally, we investigated the combined effects of elevated aPWV and elevated CRP and ORM, respectively, on the incidence of CV events. The analyses were performed using the reexamination of the population based Malmö Diet and Cancer study.
Data are only available upon reasonable request because of confidentiality of human research subjects (legal restrictions). For transparency of the results, a file containing the data underlying this study is available after application to the Malmö Diet and Cancer study Board (https://www.malmo-kohorter.lu.se/english).
Study Population
In this population-based, observational study, we used data from the CV branch of the Malmö Diet and Cancer Cohort (MDC-CV), 16 a prospective cohort study based on the population of the city Malmö in southern Sweden (230 000 inhabitants at the time). All men and women 45 to 73 years of age were invited, 30 446 individuals attended with a participation rate of approximately 40% in the MDC baseline study between 1991 and 1996. 17 Of the invited, 19% were immigrants, mainly from other European countries. Only those with sufficient understanding of Swedish were included in the cohort. The MDC study was primarily designed to explore the associations between dietary habits and health outcomes, for example, cancer or CVD. 17
The CV branch, MDC-CV included 6103 men and women with extended CV examinations,18–20 who were randomly selected from the total MDC cohort between 1991 and 1994. A reexamination of MDC-CV was performed between 2007 and 2012. Of 4924 eligible participants (still alive and not emigrated), 3734 participated in the reexamination (76% attendance rate). 18
The inclusion and exclusion criteria are presented as a flowchart (Figure 1) and in a previous paper. 21 The final study population included 2710 participants, 62% women, with a mean age of 72 years (standard deviation: 5.5 years) at the time of aPWV measurement.
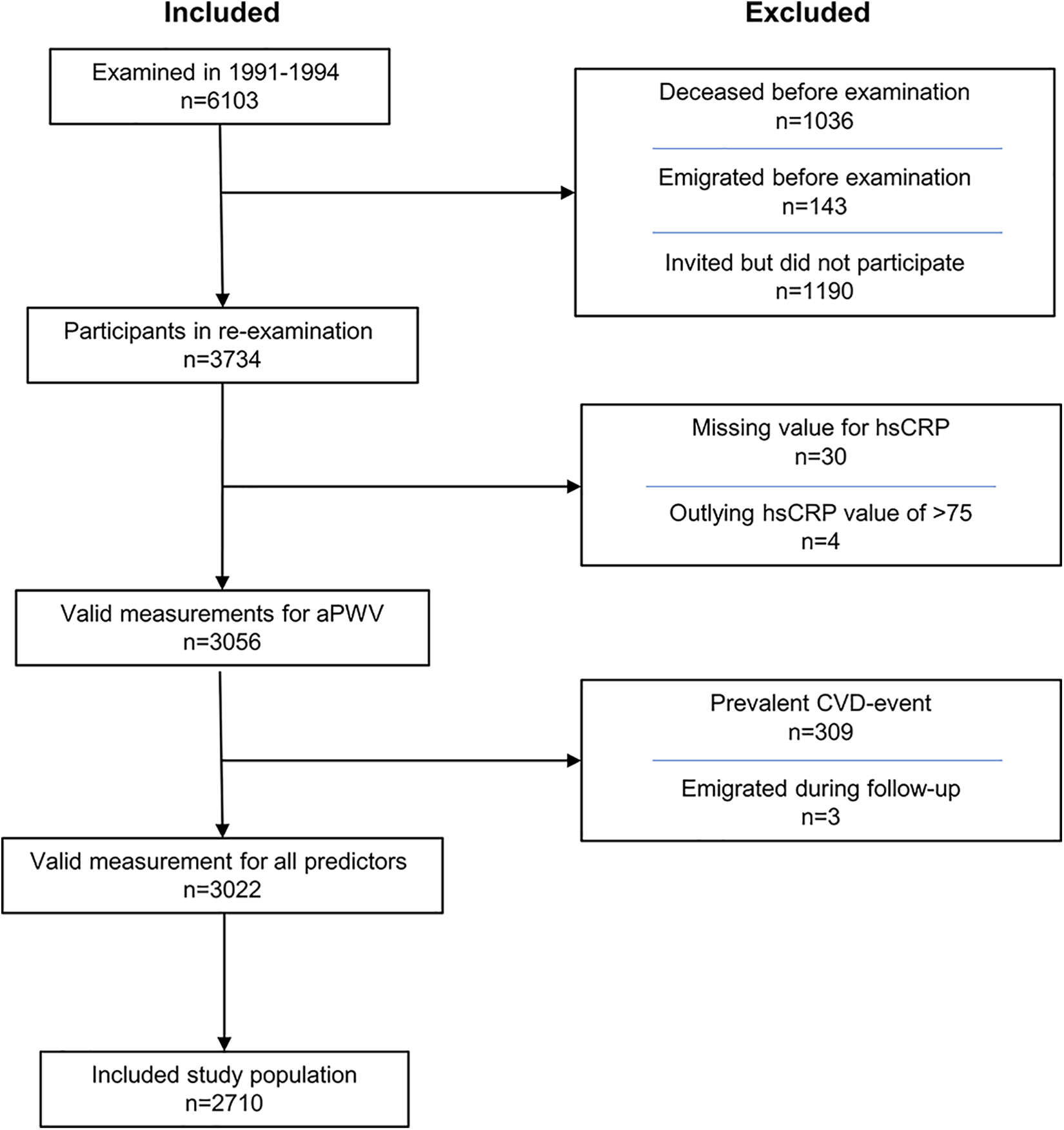
Inclusion/exclusion flowchart for the Malmö Diet Cancer Study—cardiovascular arm. aPWV indicates arterial pulse wave velocity; CVD, cardiovascular disease; hsCRP, high-sensitivity C-reactive protein; n, number of participants.
The study was approved by the Regional Ethical Review Board at Lund University, Lund, Sweden [MDC LU 51–90 and 532-2006]. All participants provided written informed consent.
Baseline Examinations
The MDC-CV reexamination from 2007 to 2012 consisted of 2 to 3 separate visits, which included a self-administered questionnaire, anthropometric data, fasting blood sampling, and carotid-femoral aPWV measurements, as described elsewhere. 18
The aPWV measurements were performed during the final study visit of each participant. There was some time delay for these visits, due to logistical reasons and shortage of technical staff, why the mean time difference between baseline and the aPWV measurements was 267 days.
Previous CVD events, history of hypertension or diabetes mellitus (DM), and use of blood pressure- or lipid-lowering drugs were assessed using the self-administered questionnaires.
Details for assessment of body mass index (BMI), mean blood pressure, history of hypertension, and DM measurements and diagnostics have been described previously. 21
Laboratory Methods
The plasma levels of high-sensitivity CRP and ORM were analyzed using immunoturbidimetric methods on Cobas 6000 systems (Roche Diagnostics GmbH) at the Department of Clinical Chemistry, Skåne University Hospital, Malmö, Sweden.
Technical Investigations
The aPWV measurements were carried out using applanation tonometry (SphygmoCor, AtCOR), measuring the PWV (m/s) between the carotid and the femoral artery (c-f aPWV). This is the gold standard for measurement of arterial stiffness, according to a European consensus document from 2012. 22 The distance calculated was (suprasternal notch to umbilicus) − (umbilicus to measuring point at the femoral artery) − (suprasternal notch to measuring point at the carotid artery), in accordance with the consensus at the time. The measurements were adapted using a factor to correspond to that measured using the 80% of direct distance, which is the currently recommended method. The details of the aPWV measurements and correction have been described elsewhere.21,23
End Point Data Retrieval
The participants were followed up until emigration, death or the first CVD event, up until December 31, 2018. The cause of death register of the Swedish National Board of Health and Welfare provided data on mortality and cause of death. The Swedish National Inpatient Register was the source for data on nonfatal myocardial infarction and stroke. This register has been externally validated and shown to have a high positive predictive value and sensitivity for myocardial infarction. 24
Percutaneous coronary intervention (PCI) procedures were identified from a nationwide register, which includes all consecutive patients at all 29 centers that perform coronary angiography or PCI in Sweden. 25 Coronary artery bypass graft surgical procedures were identified from the National Inpatient Register based on national Swedish classification systems of surgical procedures.
A composite CVD end point was used for the risk analyses.21,26 The end point included fatal or nonfatal myocardial infarction (ICD-10 code I21), fatal IHD (I21-I25), PCI, coronary artery bypass grafting (CABG), and stroke (I60, I61, I63, I64). The composite CVD end point consisted of the first CVD event for each patient during follow-up.
Statistical Analysis
Baseline Characteristics
Baseline characteristics of participants with and without future CVD were compared using t test for continuous variables and χ2 test for categorical (binary) variables.
Cross-Sectional Associations
Total covariate missing data were less than 1%, this was imputed using a single imputation, estimated through linear regression with shrinkage.27,28
A restricted cubic spline transformation with 4 knots placed at the quintile intervals was applied for CRP, which had significant nonlinear relationship (P < .001) with aPWV. An identical transformation of ORM did not show any nonlinear relationship with aPWV (P = .9).
Using multiple regression, associations between CRP and aPWV, and between ORM and aPWV, were investigated. Results were presented as the difference in aPWV comparing the 25th and the 75th percentile of the distribution of CRP and ORM, respectively.
Multiple regressions were carried out using 2 different adjustment models, Model 1 was adjusted for age, sex, mean arterial pressure, and heart rate. Model 2 was additionally adjusted for time between baseline and aPWV measurement, BMI, current smoking, low-density lipoprotein cholesterol, diabetes, and use of lipid-lowering or blood pressure–lowering medication.
Cardiovascular Disease Event Prediction
Cumulative incidence curves were plotted to visualize incidence of CVD events over time, for groups of CRP and aPWV, or ORM and aPWV, split at their respective median values. Death from non-CV causes and emigration were treated as competing risks.
Cox regressions were carried out for the composite CVD end point for the same groups. Using the same 2 adjustment models as in the multiple regression analyses, hazard ratios (HRs) with 95% CIs were calculated for groups with different combinations of low and high aPWV and CRP or ORM, respectively, using the low/low group as reference.
Mediation analysis is a useful tool when exploring potential causal pathways, according to a hypothesis presented in a directed acyclic graph (Supplemental Figure S1). A natural effects model based on Cox regression was used, as presented by Lange et al, 29 to determine the proportion of CVD risk associated with inflammatory biomarkers that could be explained by increased aPWV. The association between aPWV and risk of CVD has been described previously in the MDC-CV cohort. 21 Results were also yielded for the CVD risk associated with systolic blood pressure, which is known to be highly correlated with aPWV, and smoking status, which has been found to be only weakly, or perhaps not at all correlated with aPWV,20,30 for comparison. The CIs for the Cox models were yielded from percentile bootstrap using 3000 replicates.
The Cox regression covariate modeling included investigation of nonlinear relationships, as previously described. 21 All statistical analyses were carried out using R version 5.1-2 (Lucent Technologies).
Results
Baseline Characteristics
As presented in Table 1, 12.6% (n = 342) of the participants experienced an incident CVD event during follow-up. In unadjusted analysis, participants with events had significantly higher age, aPWV, plasma levels of CRP and ORM, systolic and diastolic blood pressure, and plasma levels of fasting glucose and triglycerides, but lower levels of HDL cholesterol and LDL cholesterol. Participants with CVD events were also significantly more likely to be men or to have a medical history of diabetes, hypertension, or use of blood pressure–lowering drugs.
Baseline Characteristics of the Study Population.
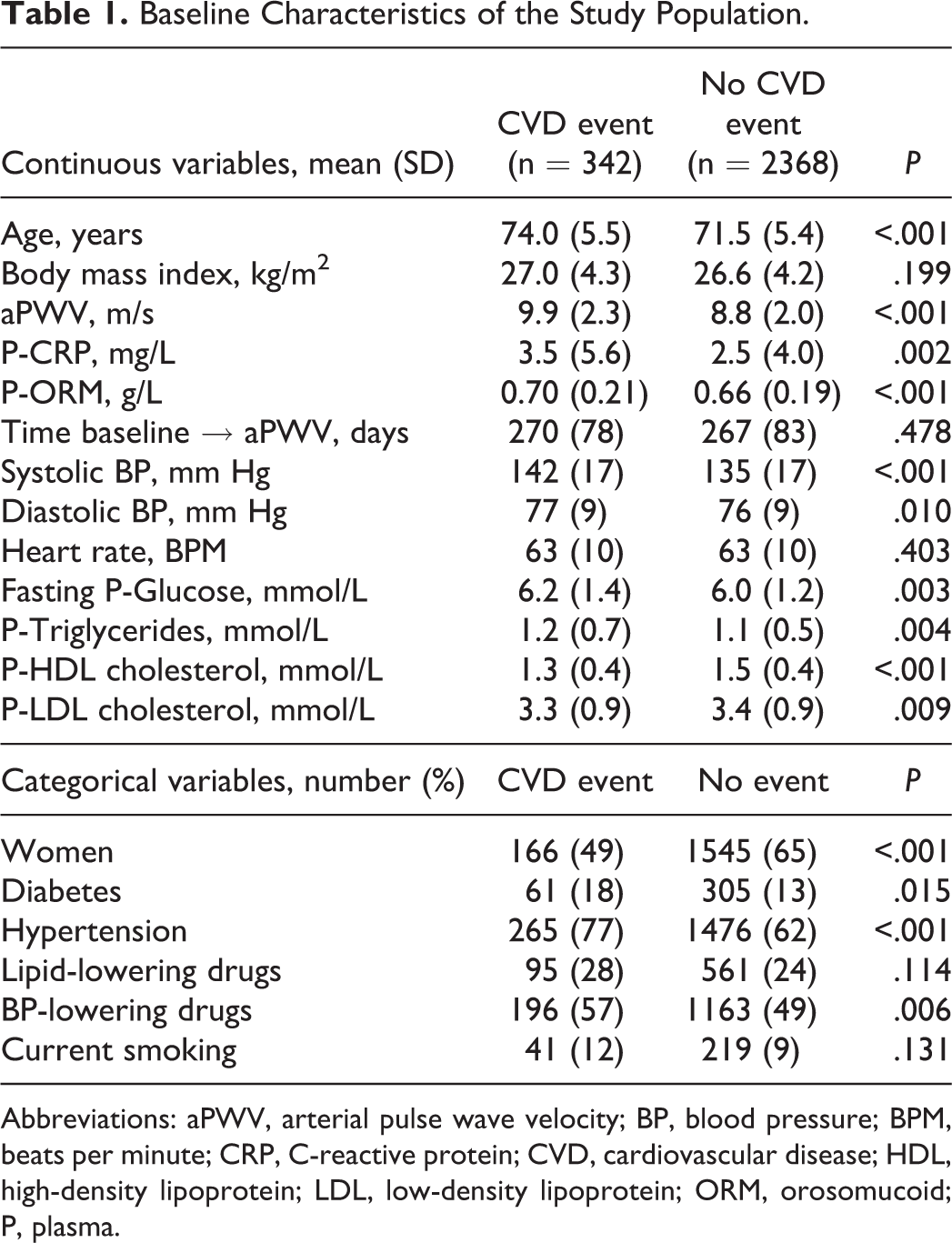
Abbreviations: aPWV, arterial pulse wave velocity; BP, blood pressure; BPM, beats per minute; CRP, C-reactive protein; CVD, cardiovascular disease; HDL, high-density lipoprotein; LDL, low-density lipoprotein; ORM, orosomucoid; P, plasma.
Cross-Sectional Associations
Both CRP and ORM were significantly positively correlated with aPWV. The effect size decreased but remained significant after adjustment for known CVD risk markers (Figure 2). The associated increase in aPWV was similar for CRP and ORM when comparing interquartile ranges.
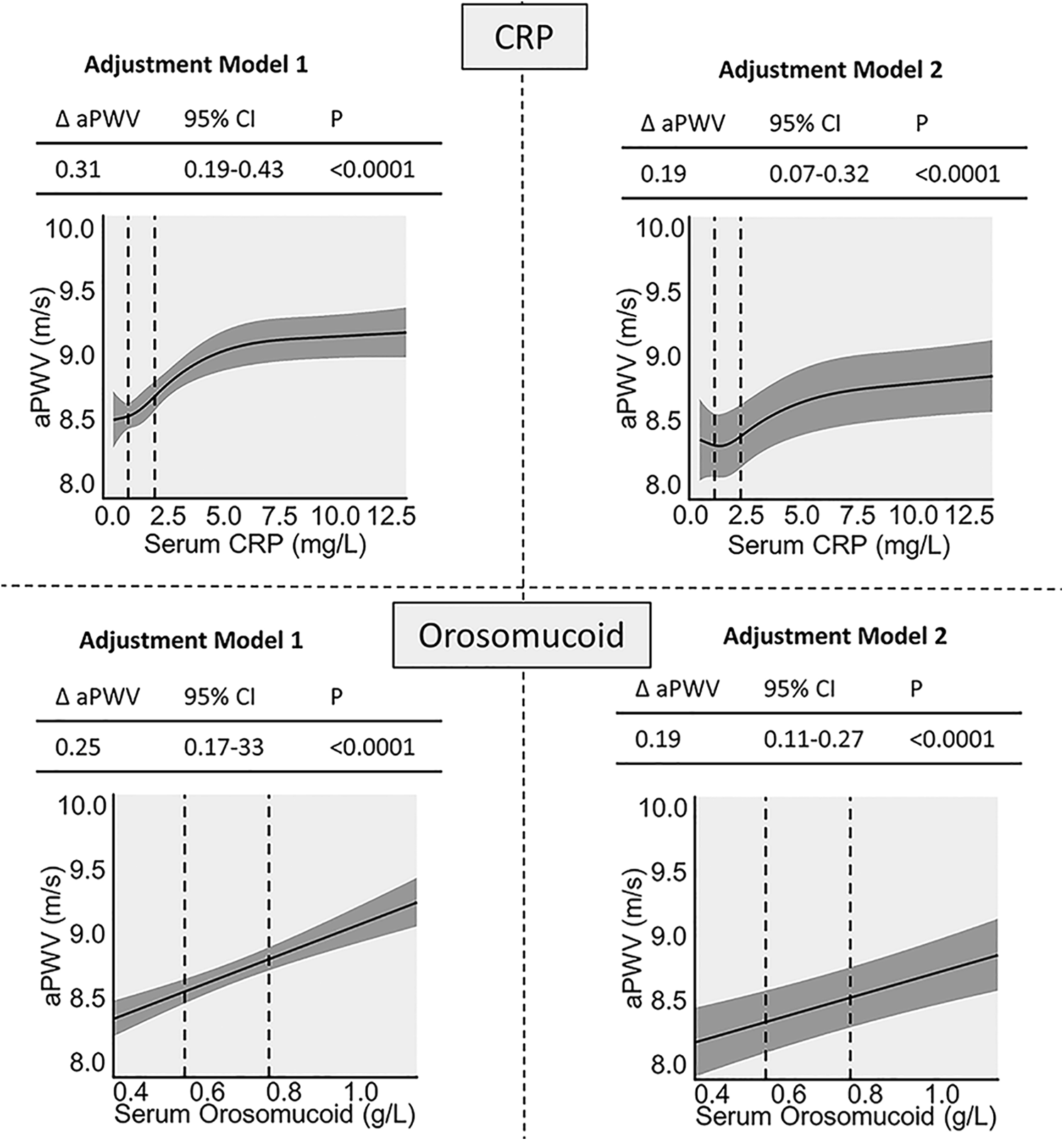
Associations between aPWV and inflammatory biomarkers in multiple linear regression. Δ = Difference in aPWV between the 25th and the 75th percentile of CRP or ORM Adjustment Model 1 is adjusted for age, sex, mean arterial pressure, and heart rate. Model 2 is additionally adjusted for time between baseline and aPWV measurement, body mass index, current smoking, low-density lipoprotein cholesterol, diabetes, and use of lipid-lowering or blood pressure–lowering medication. aPWV indicates arterial pulse wave velocity; CRP, C-reactive protein; ORM, orosomucoid.
Cardiovascular Disease Event Prediction
Cumulative incidence curves showed that above median aPWV was associated with a higher incidence of CVD. Both for CRP and ORM, the highest incidence rates were observed for those with both aPWV and the inflammatory marker above median (Figure 3).
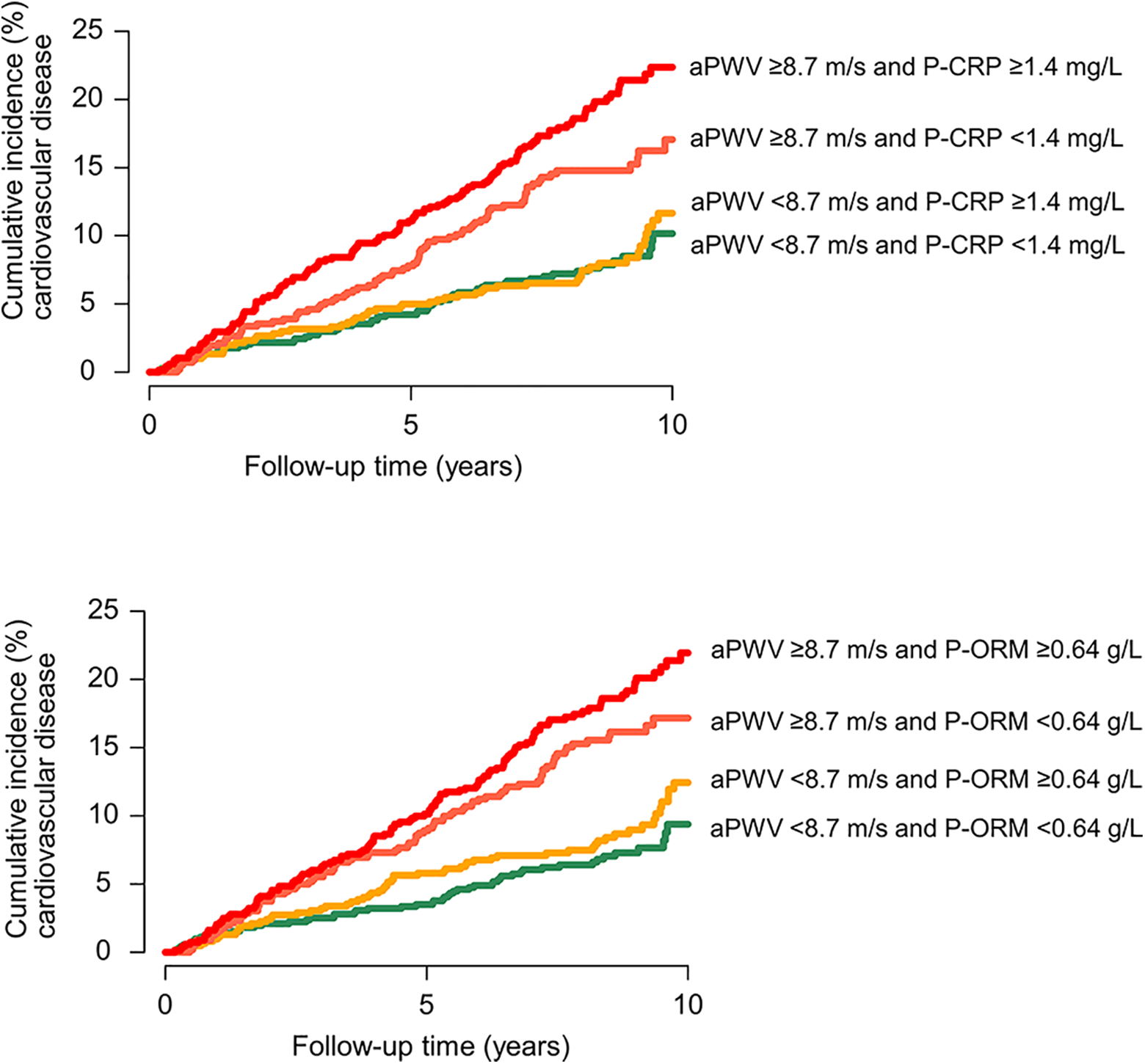
Cumulative incidence curves for cardiovascular disease. With death from noncardiovascular causes and emigration as competing risks. Groups are defined by combined high or low levels of aPWV and CRP and aPWV and ORM. aPWV indicates arterial pulse wave velocity; CRP, C-reactive protein; ORM, orosomucoid.
The HRs of a 1 standard deviation higher level of aPWV were similar in groups with high and low CRP, respectively. The HRs were higher in the group with below-median levels of ORM (HR 2.3) than in the above median group (HR 1.6) in an unadjusted model (Supplement Table).
Combinations of above and below median aPWV and CRP or ORM trended toward an increased risk for combinations of high values. The combination high aPWV + high CRP had an HR of 1.57 and the combination high aPWV + high ORM had an HR of 1.71 after extensive adjustment (Figure 4).
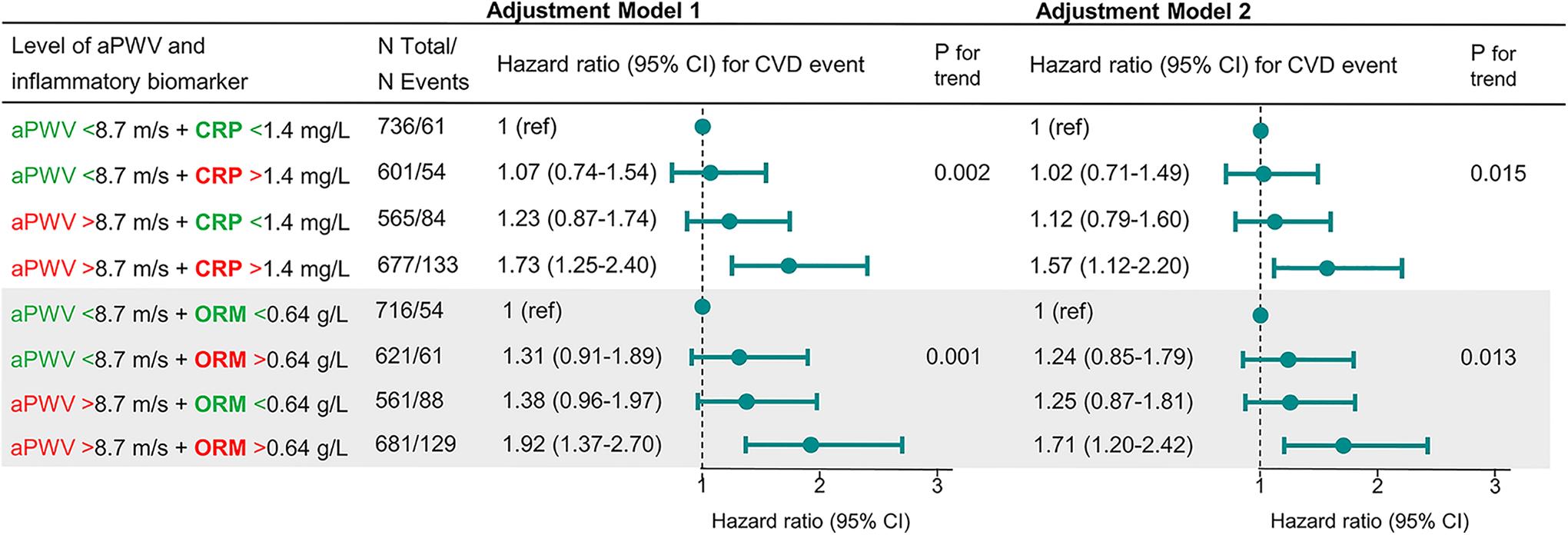
Cox regressions results for categories of high or flow values of aPWV and inflammatory biomarkers. Showing HRs in subgroups of combinations of above and below median levels of aPWV (8.7 m/s) and CRP (1.4 mg/L) or ORM (0.64 g/L), respectively. Adjustment Model 1 is adjusted for age, sex, mean arterial pressure, and heart rate. Model 2 is additionally adjusted for time between baseline and aPWV measurement, body mass index, current smoking, low-density lipoprotein cholesterol, diabetes, and use of lipid-lowering or blood pressure–lowering medication. aPWV indicates arterial pulse wave velocity; CRP, C-reactive protein; CVD, cardiovascular disease; HR, hazard ratio; ORM, orosomucoid.
The mediation analysis using the Cox natural model showed that aPWV mediated 11% (95% CI: 1-21) of the relationship between CRP and incidence of CVD, in adjustment Model 1. After adjustments for risk factors in Model 2, 8% (−4 to 20) of the CRP-CVD relationship was mediated by aPWV. The results for ORM were almost identical in both survival models (Table 2). These results were similar to those for current smoking, at −1% (−0.28 to 0.18) for adjustment Model 1 and 8% (−1 to 25) for adjustment Model 2. In comparison, a larger proportion of the risk associated with systolic blood pressure, 26% (10-38) and 22% (5-36), respectively, was statistically mediated by aPWV in the 2 adjustment models.
Mediating Effect of aPWV for the Prediction of Incident CVD Events, by Inflammatory Biomarkers and Other Risk Factors in a Natural Effects Cox Regression Model.a
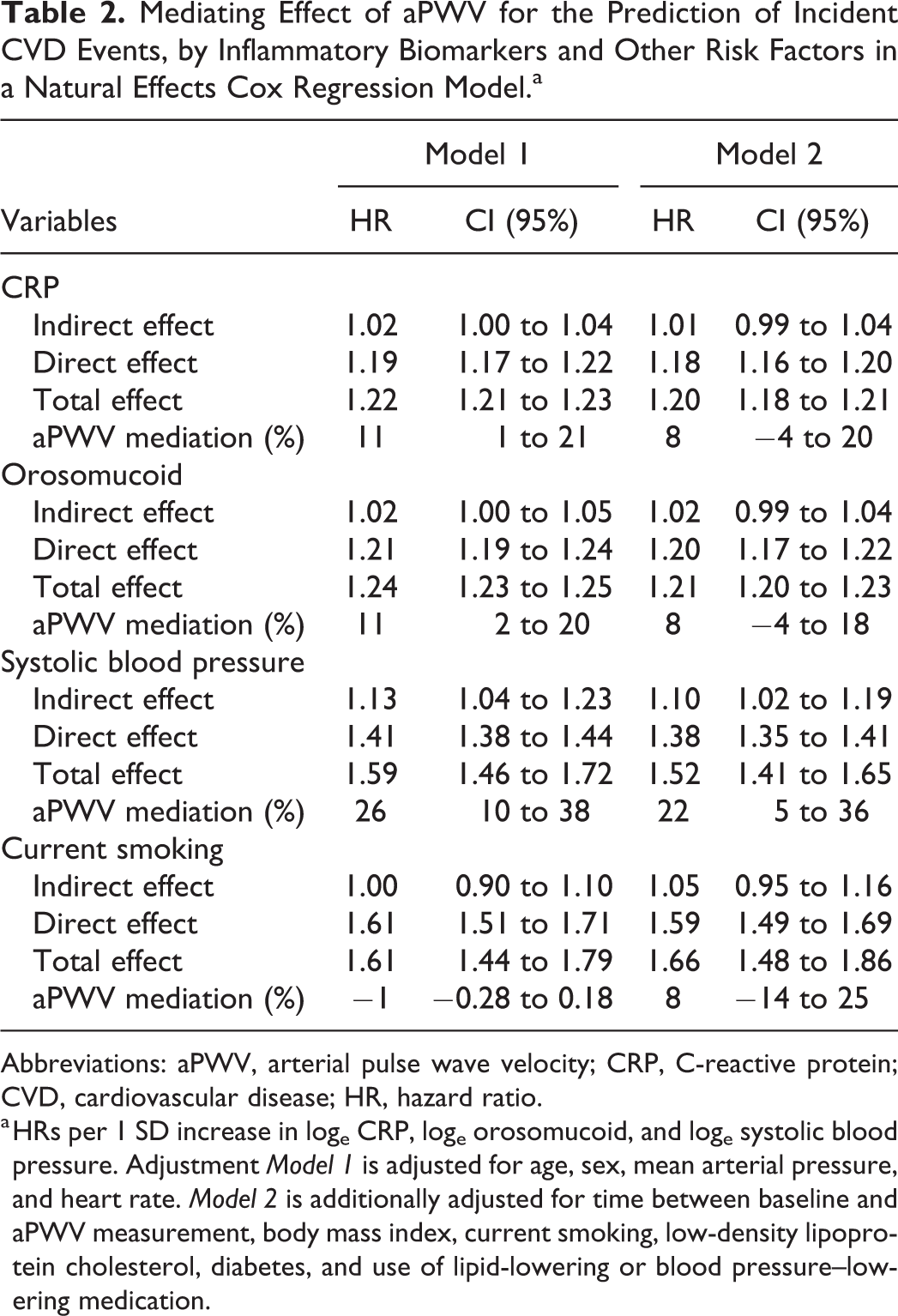
Abbreviations: aPWV, arterial pulse wave velocity; CRP, C-reactive protein; CVD, cardiovascular disease; HR, hazard ratio.
a HRs per 1 SD increase in loge CRP, loge orosomucoid, and loge systolic blood pressure. Adjustment Model 1 is adjusted for age, sex, mean arterial pressure, and heart rate. Model 2 is additionally adjusted for time between baseline and aPWV measurement, body mass index, current smoking, low-density lipoprotein cholesterol, diabetes, and use of lipid-lowering or blood pressure–lowering medication.
Discussion
This population-based cohort study analyzed possible relationships between 2 well-known circulating biomarkers of systemic inflammation and aPWV, and their relationship with incident CVD events. After full adjustment for known CVD risk markers, both CRP and ORM were found to be weakly but significantly positively correlated with aPWV. The incidence of CVD events was highest for those with both aPWV and an inflammatory marker above median in combination. We did not find that arterial stiffness has the potential to be an important mediator of the relationship between inflammation and CVD, and the results therefore support the view that chronic low-grade inflammation and arterial stiffness (aPWV) represent 2 different pathways of CV risk (Supplement Figure S2).
Few previous studies have examined the interrelationships between aPWV, inflammatory biomarkers, and incidence of CVD events. A previous study reported that the Augmentation Index (Aix), an indirect and partial marker of arterial (aortic) stiffness, is independently associated with CV mortality in a small cohort of patients with advanced chronic kidney disease-5, even after adjustment for high-sensitive CRP. 31 These associations have been further discussed in a review by Maloberti et al. 32
As shown in clinical trials, anti-inflammatory therapy targeting molecules upstream from CRP in the inflammatory cascade decreases aPWV in patients with inflammatory diseases.9–11 In our epidemiological study, we studied the general population, in which there mainly is low-grade inflammation due to nonclinical causes. It is possible that the intense inflammation in inflammatory diseases more sharply increases aPWV. Furthermore, clinical trials have not determined the importance of the decrease in aPWV for lowering of CVD risk, relative to the other effects of anti-inflammatory therapy, including direct effects on atherosclerosis and thrombosis. 33 In our study, we provide an estimation of the importance of aPWV as a mediator in the pathway between inflammation and CVD in the general population.
Strengths and Limitations
The strengths of this observational, follow-up study include that it is population based with data collected from a relatively large cohort with well-validated baseline measurements, including those of aPWV, and data from validated national registers for follow-up of CVD events. The follow-up time and the number of events are sufficient for relatively robust results. Some limitations should, however, also be acknowledged. This was a rather homogenous cohort (almost exclusively Caucasian of European descent, mostly northern European ethnicity) with participants of a relatively advanced age, which limits the generalizability of the results. It has been previously reported that the association between aPWV and risk of CVD events is stronger for middle-aged participants than in the elderly. 34
The method used to calculate the carotid-femoral distance for the aPWV measurements was different from the one suggested by the current guidelines of the ESH (not available when the MDC-CV Re-exam Study started in 2007), which advise using 80% of the direct carotid-femoral distance. 22 The method used in this study on average underestimates the distance and to account for this the measurements were subsequently corrected using a constant factor, as previously described. 23 This factor may not be correct for all individuals. There was also a time-lag (mean 268 days) between the baseline study visits when most of the data were collected, and the later visits for aPWV measurements. By adjusting for the time interval between cIMT and aPWV measurements, this bias was minimized.
Even though the cohort included 2710 individuals followed over 7.6 years, the CIs were wide for some of the analyses. It is still possible that aPWV mediates up to 20% of the CVD risk (upper limit of 95% CI) associated with CRP. The mediation analysis in this study was based on observational data, and as such the results are subject to confounding and reverse causation.
Other kinds of studies, for example, Mendelian randomization (causal inference) based on genetics or randomized clinical trials, would be required to finally determine any causal relationships between systemic inflammation and arterial stiffness. Furthermore, the role of local (perivascular) inflammation has not been possible to analyze in this or in other large studies. We instead measured ORM and CRP, the latter an extensively used clinical biomarker, even if not causal, produced downstream of the systemic inflammatory cascade which causes CVD. 33 Finally, the CVD end point included PCI and CABG, which are invasive procedures dependent on local clinical guidelines and clinical judgment, and as such they may provide slightly biased measures of actual CVD morbidity.
In the future, similar studies are needed in different populations across wider age ranges, also adding other inflammatory biomarkers such as ILs. In addition, intervention studies are needed to reverse inflammation and risk, as was evident in the CANTOS (The Canakinumab Antiinflammatory Thrombosis Outcome Study) trial in post-MI patients treated with a monoclonal antibody against IL-1 β with a 15% relative risk reduction versus placebo in spite of a high use of drugs for secondary prevention. 35 Recently also the anti-inflammatory drug colchicine was shown to significantly reduce CV events in patients with stable coronary disease versus placebo. 36 Measurement of aPWV in interventions like these could determine whether the decrease in risk is likely to be mediated by a decrease in arterial stiffness, or not.
Conclusions
Arterial stiffness is associated with 2 markers of systemic inflammation (CRP and ORM) in a population-based study involving elderly participants. The association between aPWV, a marker of arterial stiffness, and future CV events was confirmed. We did not find evidence that aPWV mediates any major part of the increased CVD risk associated with systemic inflammation in the general population. There was a trend toward an additive effect on risk with combinations of high CRP or ORM and high aPWV. Taken together, these findings support the view that arterial stiffness and systemic inflammation represent 2 different pathways of CV risk.
Perspectives
Chronic low-grade inflammation in the elderly general population is likely to affect CV risk mainly through other mechanisms and not by increasing arterial stiffness. The treatment effect of anti-inflammatory medications for lowering of CVD risk could most likely not be monitored with measurement of aPWV. Furthermore, to decrease arterial stiffness and the associated CVD risk, it is possible that selective anti-inflammatory approaches will not be effective. Other types of studies are needed to determine a lack of a causal effect of systemic inflammation on aPWV and associated risks, or if local (perivascular) inflammation is more relevant.
Supplemental Material
Supplemental Material, sj-docx-1-ang-10.1177_00033197211017406 - Aortic Stiffness, Inflammation, and Incidence of Cardiovascular Events in Elderly Participants From the General Population
Supplemental Material, sj-docx-1-ang-10.1177_00033197211017406 for Aortic Stiffness, Inflammation, and Incidence of Cardiovascular Events in Elderly Participants From the General Population by Benjamin Nilsson Wadström, Margaretha Persson, Gunnar Engström and Peter M. Nilsson in Angiology
Footnotes
Acknowledgments
The authors thank Gerd Östling, PhD, Cecilia Kennbäck, and ultrasound technicians at the clinical sciences department, responsible for aPWV and cIMT data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by the Research Council of Sweden [grant number K2011-65X-20752-04-6]; the Region Skane County Council; the Heart- and Lung Foundation to PM Nilsson, and the Ernhold Lundstrom Foundation to M Persson.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
