Abstract
Background
The adoption of robotic surgery has increased in foregut procedures, but its comparative value to laparoscopy in Heller myotomy remains unclear. We sought to evaluate the short-term perioperative outcomes of the robotic vs laparoscopic approach for Heller myotomy.
Methods
The American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database from 2022 to 2023 was queried for patients undergoing Heller myotomy, in which the “robotic” variable was included. Outcomes were evaluated using univariable analysis and multivariable logistic and linear regression.
Results
A total of 712 patients underwent a Heller myotomy; laparoscopy accounted for 63.6% of cases (n = 453) and robotic 36.3% (n = 259). The cohorts were matched aside from higher rates of HTN in the robotic group (42.9% vs 34.7%, P < 0.05). Robotic Heller myotomy had significantly (P < 0.05) greater use of intraoperative EGD (22.8% vs 16.8%) and balloon distension studies (5.4% vs 1.5%). The robotic approach was associated with a longer median operative time (167 min vs 119.5 min) and independently associated with longer operative time on linear regression (β 35.39 min, CI 26.19-45.96 min). There were no significant differences in short-term complications including reoperation, readmission, and mortality.
Discussion
These data suggest no significant differences in the short-term outcomes between robotic and laparoscopic Heller myotomy. Robotic surgery was associated with longer operative times and utilization of more EGD and balloon distension studies. Further research is required to evaluate the clinical benefit of robotic surgery in achalasia.
Key Takeaways
• Rates of complications between laparoscopic and robotic heller myotomy are low and comparable between platforms • Patients undergoing robotic-assisted Heller myotomy had significantly longer operative times. • Robotic-assisted Heller myotomy was associated with increased utilization of intraoperative esophagogastroduodenoscopy and esophageal balloon distension studies.
Introduction
Achalasia is a rare neurodegenerative motility disorder defined as impaired relaxation of the lower esophageal sphincter and absent esophageal body peristalsis resulting in dysphagia and reduced esophageal emptying. 1 While it remains incurable, various treatment modalities ranging from pharmacotherapy to surgical myotomy have been utilized to reduce the functional outflow obstruction. Dr. Ernest Heller pioneered the surgical treatment for achalasia in 1913 through esophageal myotomy. Over the past century, the rate of Heller myotomy as a treatment modality for achalasia has steadily increased. 2 With the advent of minimally invasive approaches in the 1990s, laparoscopic Heller myotomy (LHM) has become the gold standard management with reduced morbidity, comparable functional outcomes and improved quality of life. 3 However, variations in the technical elements of Heller Myotomy (HM) have been developed and their impact remains uncertain due to the relative rarity of the disease.
Robotic platforms have been increasingly adopted in foregut procedures, driven by potential ergonomic and technical advantages such as wristed instrumentation, stable three-dimensional visualization, and enhanced precision in confined fields. These attributes may theoretically translate into more controlled dissection at the gastroesophageal junction and lower rates of mucosal injury or esophageal perforation during HM. In contemporary studies, the reported mucosal perforation rate for LHM ranges between 5% and 10%, providing a benchmark for evaluating whether the robotic platform can further enhance safety. 4 However, the clinical value of robotic Heller myotomy (RHM) relative to LHM remains uncertain. Previous literature has been dominated by single-institution retrospective series and heterogeneous observational studies, with meta-analyses generally suggesting longer operative times with robotic approaches and variable findings across secondary outcomes. 5 Pooled analyses have suggested that RHM may be associated with a reduced risk of esophageal perforation compared with LHM, though these findings remain limited by nonrandomized studies, confounding, and study heterogeneity. 6
At the same time, intraoperative adjuncts used to assess myotomy adequacy have expanded. Intraoperative esophagogastroduodenoscopy (EGD) may help confirm mucosal integrity and evaluate the extent of the myotomy, while impedance planimetry provides real-time assessment of esophagogastric junction geometry and distensibility. 7 Yet the real-world frequency of these adjuncts has not been well characterized on a national level.
Given ongoing uncertainty, large, contemporary datasets that reflect current practice patterns are useful for evaluating if the robotic platform offers measurable short-term perioperative advantages over laparoscopy. The American College of Surgeons National Surgery Quality Improvement Program (ACS-NSQIP) database has recently incorporated the use of the robotic platform as a variable. The goal of this study is to utilize the NSQIP database to evaluate the short-term safety profile of LHM compared to RHM.
Methods
Data Source
The ACS-NSQIP database is comprised of data from over 600 participating hospitals. Data from 2022 and 2023 were merged and included in this study.
Study Population
The ACS-NSQIP database was searched for all patients undergoing laparoscopic Heller myotomy (CPT 43279) inclusive of robotic cases. Robotic cases were identified using the variable “Robot_Used” within the NSQIP data set. Patients undergoing intraoperative Esophagogastroduodenoscopy (EGD) were identified by associated CPT codes including: 0652T, 3130F, 31622, 43180, and 43200-43252. Impedance planimetry was identified with CPT code 91040. Data on patient demographics included age, body mass index, race, ethnicity, diabetes mellitus, smoking status, functional status, history of severe chronic obstructive pulmonary disease, congestive heart failure, preoperative acute kidney injury, use of immunosuppressive therapy, bleeding diathesis, American Society of Anesthesiologist (ASA) classification, and preoperative albumin.
Statistical Analysis
Categorical variables were evaluated with chi-squared and Fisher's exact tests. Fischer's exact test was used when contingency table values of less than 5 represented more than 20% of cells. Quantitative variables were evaluated using Mann-Whitney U for nonparametric data, and Student's t-test for parametric data. Significant findings were further evaluated with logistic regression or linear regression. Independent variables with a significant association of P < 0.10 were included in the multivariable model. All analyses were performed with IBM Statistical Package for Social Sciences (SPSS) Version 30.0.0.0 (172).
Results
Baseline Demographics Laparoscopic vs Robotic Heller Myotomy
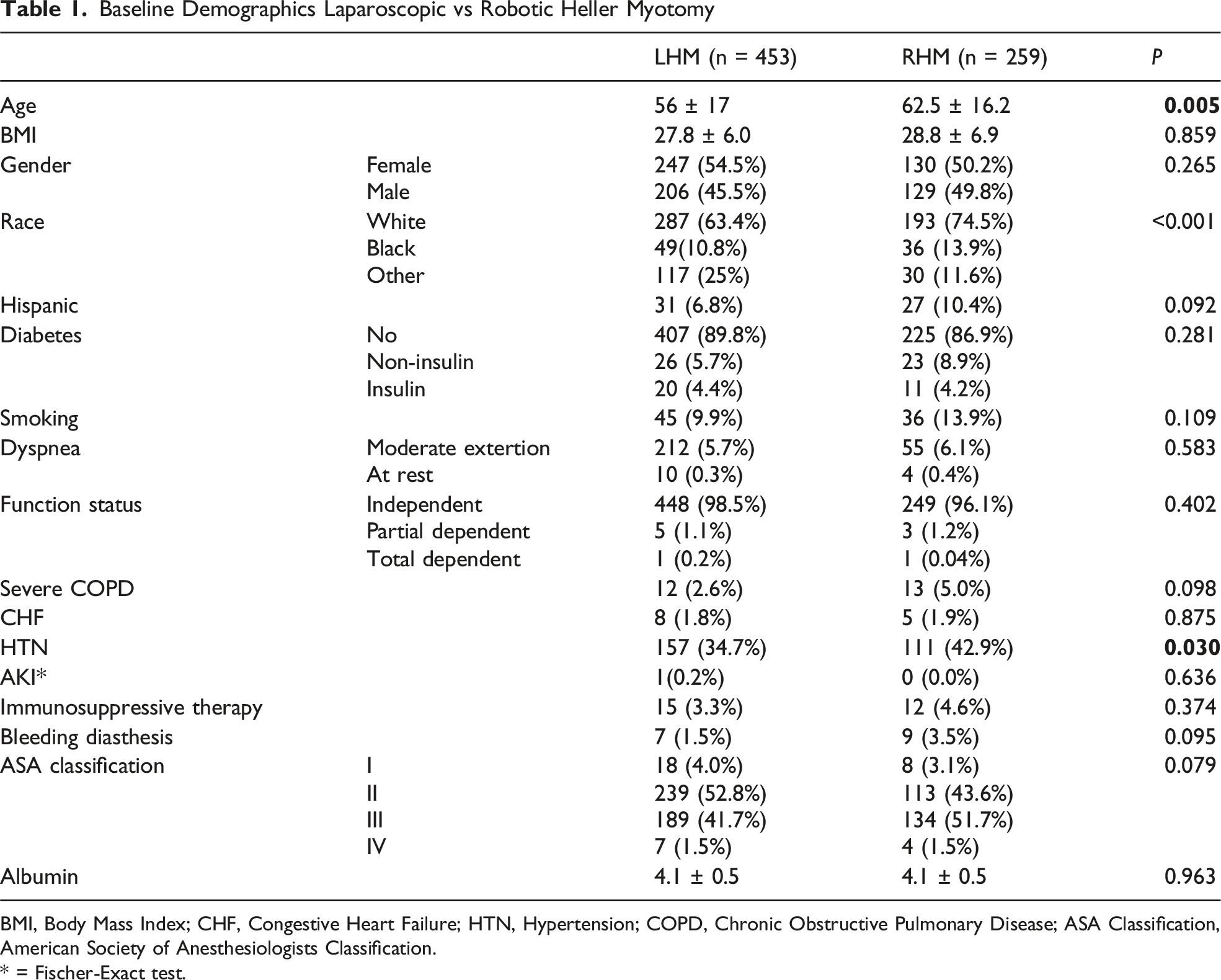
BMI, Body Mass Index; CHF, Congestive Heart Failure; HTN, Hypertension; COPD, Chronic Obstructive Pulmonary Disease; ASA Classification, American Society of Anesthesiologists Classification.
* = Fischer-Exact test.
Postoperative Complications LHR vs RHM
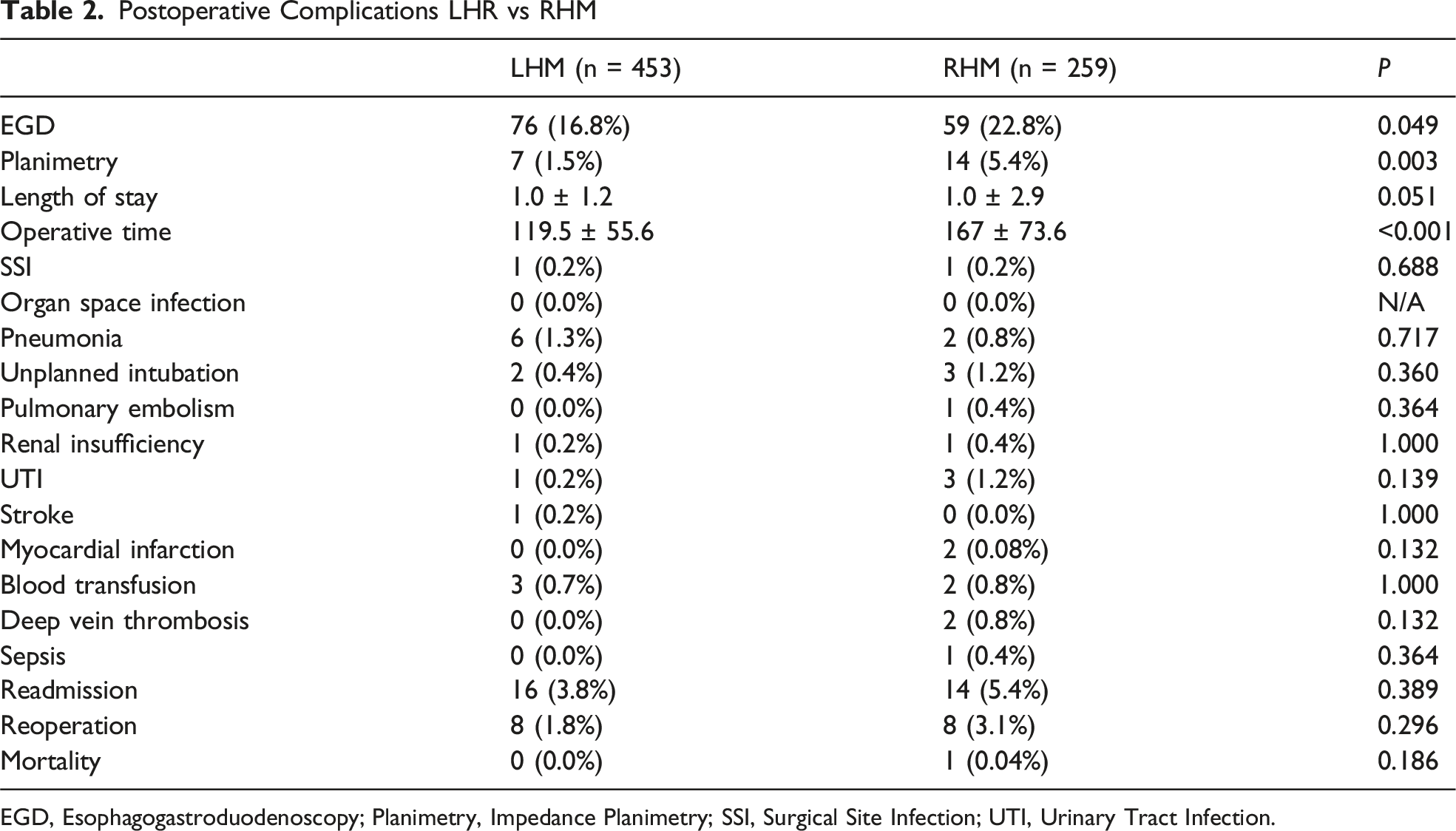
EGD, Esophagogastroduodenoscopy; Planimetry, Impedance Planimetry; SSI, Surgical Site Infection; UTI, Urinary Tract Infection.
Multivariable Linear Regression Analysis (Robotic-Assisted Surgery Baseline)

Regression analysis using the following predictor variables: Age, Gender, Race, Ethnicity, American College of Anesthesiologists Classification, Operative Time, Operative Year, Body Mass Index, EGD Use, Impedance Planimetry, and Clinical History (Diabetes, Smoking, Dyspnea, Functional Status, Severe Chronic Obstructive Pulmonary Disease, Congestive Heart Failure, Hypertension, Renal Failure, Dialysis, Wound Infection, Chronic Steroid Use, Bleeding diathesis).
There were no significant differences in rates of readmission and reoperation between the cohorts. Events were uncommon overall. When evaluating for possible esophageal perforation or leak, one patient in the laparoscopic cohort underwent an esophageal repair and one patient in the robotic cohort was readmitted with esophageal perforation.
Discussion
This study demonstrates that there is no significant difference in short-term complications between LHM and RHM. Prior studies have shown conflicting complication rates when comparing RHM to LHM.5,8–11 In a systematic review spanning seven studies from 2005 to 2020, Xie et al found a significantly lower rate of esophageal perforation with RHM as compared to LHM with other outcomes otherwise comparable. 5 A similar systematic review across 11 studies published between 2005 and 2023 found RHM with longer operative times, shorter length of stay (LOS), and lower rates of esophageal perforation with similar symptomatic relief as compared to LHR, but with significantly higher rate of reintervention. 8 Another study describes decreased overall complication rates and lower LOS associated with RHM compared to LHM, although subset analysis of only high-volume centers showed a higher perforation rate. 9 In the present analysis, possible esophageal perforation-related events were rare, with one event identified in each cohort. Taken together, these findings suggest that either minimally invasive approach can be performed safely, while any true difference in perforation risk is likely small and difficult to detect in retrospective datasets of this size. In addition, Brosnihan et al found no significant difference in mortality, LOS, or 30-day readmission. 10 Generally, in studies spanning long periods of time, familiarity with the robotic platform rapidly evolves even within a given study period due to increasing integration of this platform in training and practice, which may add heterogeneity into the interpretation of results.
More recently, Brosnihan et al also queried the 2022 NSQIP database for patients undergoing solely elective Heller myotomy and found that RHM had higher morbidity and longer operative times. 11 The current study expands upon this prior work by incorporating 2023 data and, critically, by including concurrent intraoperative adjuncts (EGD and planimetry) which were previously excluded or not prioritized. This allowed for a more comprehensive assessment of contemporary practice patterns and whether these adjuncts influence short-term complications. Interestingly, our results show that even with the inclusion of these additional procedures, which were more frequently utilized in the robotic group, there was no significant difference in morbidity.
While the NSQIP database does not provide specific cost data, this increased duration likely translates to higher hospital costs. Previous analyses of large inpatient databases have reported significantly higher overall hospital charges for RHM compared to LHM ($42,900 vs $34,300), often citing equipment costs and operative time as key drivers.4,9 The clinical benefit of the robotic platform must therefore be weighed against these economic considerations, especially in the absence of a clear short-term outcome advantage.
While the use of intraoperative esophagogastroduodenoscopy (EGD) during Heller myotomy has been shown to have no influence on outcomes during LHM, 12 planimetry during LHM has been shown to provide real-time feedback. 13 The higher rate of intraoperative planimetry use in RHM as compared to LHM may simply reflect the relative availability of these relatively more costly modalities at larger tertiary and quaternary centers or convenience of use in conjunction with the robotic platform. It also suggests that these cases may have the added benefit of a more targeted intraoperative evaluation, although this a study by Chacko et al demonstrated increased perforation rates at high-volume centers. 9
These findings should be interpreted in light of several limitations. As a retrospective database study, conclusions are limited to variables captured within ACS-NSQIP. The database lacks granular information on achalasia subtype, symptom severity, myotomy length, surgeon experience, center volume, fundoplication details, and long-term functional outcomes. In addition, esophageal perforation is challenging to infer from diagnosis and procedure codes rather than directly captured operative findings.
Our study adds to the existing literature describing differences in RHM and LHM, finding that short-term outcomes were not significantly different, but that intraoperative time and use of intraoperative adjuncts is higher with RHM. Further studies will need to be performed with larger sample sizes to better understand the impact of the increased rate of intraoperative adjunct use in RHM and LHM.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
