Abstract
Introduction
Bariatric surgery is increasingly performed in individuals of reproductive age to treat obesity and reduce associated comorbidities. However, the impact of these procedures on subsequent pregnancy outcomes remains incompletely understood. This study evaluates perinatal outcomes in patients with a history of bariatric surgery, focusing exclusively on pregnancies occurring after surgery, using a large, real-world database.
Methods
A retrospective cohort study was conducted using the TriNetX Research Network, a national, de-identified electronic health record database. We identified pregnant females aged ≥18 between June 1, 2015, and June 25, 2025. Patients with prior bariatric surgery were identified using ICD-10 and CPT codes. Propensity score matching (1:1) was performed based on age, race, ethnicity, gravida, parity, hypertension, diabetes, anemia, and delivery method, yielding 41 777 matched pairs. Perinatal outcomes including fetal growth restriction (FGR), macrosomia, gestational hypertension, gestational diabetes, preeclampsia, and preterm delivery were compared using odds ratios (ORs) and 95% confidence intervals (CIs).
Results
Bariatric surgery was associated with significantly lower risks of FGR (OR 0.831, 95% CI 0.749-0.921), macrosomia (OR 0.283, 95% CI 0.262-0.305), gestational hypertension (OR 0.391, 95% CI 0.375-0.409), preeclampsia (OR 0.313, 95% CI 0.294-0.334), gestational diabetes (OR 0.415, 95% CI 0.393-0.439), and preterm delivery (OR 0.525, 95% CI 0.493-0.560) compared to controls.
Conclusion
Patients with a history of bariatric surgery who became pregnant after the procedure experienced significantly reduced risks of several adverse perinatal outcomes. These findings support the role of bariatric surgery in improving pregnancy outcomes among individuals with obesity and underscore the importance of preconception counseling and tailored prenatal care.
Keywords
Introduction
Bariatric surgery, including procedures such as gastric bypass and sleeve gastrectomy, is increasingly utilized as an effective intervention for significant and sustained weight loss among individuals with obesity.1,2 These procedures not only help reduce excess body weight but also improve obesity-related comorbidities such as type 2 diabetes, hypertension, and polycystic ovarian syndrome, conditions that can significantly impact reproductive health and pregnancy outcomes.3-5 As more individuals of childbearing age undergo bariatric surgery, questions have emerged regarding the potential implications of these procedures on subsequent pregnancies.
Clinical guidelines often recommend delaying pregnancy for at least 12-18 months following bariatric surgery to allow for weight stabilization and adequate nutritional recovery.6,7 Despite this recommendation, unplanned pregnancies during the rapid weight loss phase are not uncommon. Literature on pregnancy after bariatric surgery suggests that these patients may face unique maternal and fetal risks due to altered gastrointestinal anatomy and nutrient absorption, including concerns about small-for-gestational-age (SGA) infants, preterm delivery, and cesarean delivery (CD).8-10 However, existing data are largely derived from small retrospective studies and case series, with mixed findings regarding the true extent of these risks.
Additionally, the type of bariatric procedure performed may influence perinatal outcomes, given differences in malabsorptive vs restrictive mechanisms and the degree of metabolic alteration. While some studies have noted improved fertility and reduced risks of gestational diabetes and hypertensive disorders of pregnancy post-surgery, others have raised concerns about increased risks of intrauterine growth restriction, nutritional deficiencies, and adverse neonatal outcomes.11-14
Our study aims to analyze the demographics and perinatal outcomes of pregnant individuals with a history of bariatric surgery using real-world data from the TriNetX Research Network (Cambridge, MA), a large multi-institutional database of electronic health records. Our study is important because the number of individuals undergoing bariatric surgery before pregnancy is steadily increasing, yet comprehensive data on their perinatal outcomes remain limited. Understanding how these surgeries impact maternal and fetal health is essential for guiding clinical decision-making, patient counseling, and risk mitigation. By leveraging a large, real-world, multi-institutional dataset, our research addresses critical knowledge gaps in obstetric outcomes following bariatric procedures. We hypothesize that perinatal outcomes will be significantly different for pregnant women who have undergone bariatric surgery vs those who do not undergo bariatric surgery.
Methods
Data Source
We utilized the TriNetX Research Network (Cambridge, MA), a large, federated, de-identified national health research database composed of real-time electronic health records from over 102 healthcare organizations (HCOs) across the United States and encompassing 154 785 537 patients. These data are contributed by academic medical centers, community hospitals, and specialty care providers and include inpatient and outpatient visits, diagnoses, procedures, medications, and laboratory values. The data underwent quality control and standardization prior to inclusion. All data used were de-identified, and the study was deemed exempt from IRB review at University of Texas Medical Branch.
Patient and Data Selection
We identified adult female patients (≥18 years old) who had at least one documented pregnancy (ICD-10: O00-O9A, Z33) between June 1, 2015 and the date of data access (June 25, 2025). From this population, we created 2 cohorts: (1) patients with a history of bariatric surgery prior to pregnancy, identified using ICD-10 procedure codes and CPT codes for common bariatric surgeries (eg, sleeve gastrectomy and gastric bypass): 1.ICD-10 codes a. b. c. d. e. f. g. h. i. j. k.
Baseline Characteristics Between Patients Who Underwent Bariatric Surgery and Controls Before and After Propensity Score Matching.
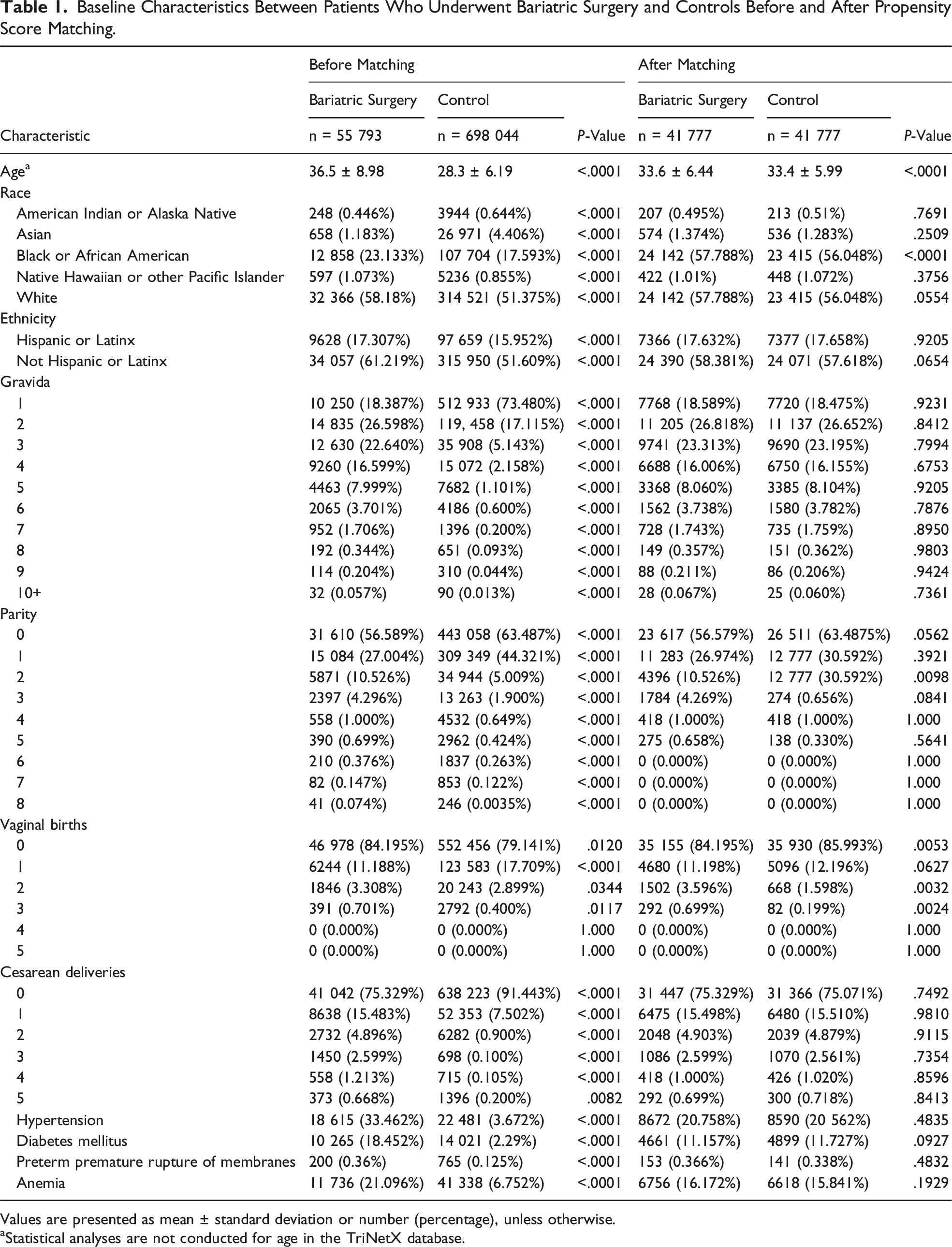
Values are presented as mean ± standard deviation or number (percentage), unless otherwise.
aStatistical analyses are not conducted for age in the TriNetX database.
Statistical Analyses
Owing to the large sample size difference between cohorts (control: n = 698 044; bariatric surgery: n = 55 793) and to account for potential confounders, we performed 1:1 propensity score matching based on race, ethnicity, age at index pregnancy, hypertension, diabetes mellitus, vaginal deliveries, C-sections, anemia, gravida, and parity. Propensity score matching was used to generate matched samples with balanced distributions of baseline characteristics between the bariatric surgery and control groups, thereby reducing the influence of confounding variables and allowing for more accurate estimation of the association between bariatric surgery and perinatal outcomes. A 1:1 matching ratio was selected to preserve statistical power while ensuring comparability between cohorts. We examined differences in baseline characteristics using independent t tests for continuous variables and chi-square or Fisher’s exact tests for categorical variables (reported as frequencies and percentages). Outcome comparisons were performed using odds ratios (ORs) with 95% confidence intervals (CIs). All perinatal outcomes in this study were assessed after bariatric surgery and during the patient’s most recent pregnancy. Patients were only included if the documented pregnancy occurred following their bariatric surgery, ensuring that all outcomes reflected the post-surgical physiologic state. Since this was a retrospective analysis using the TriNetX database, which provided a very large available cohort, no prior power calculation was performed. The large sample size and balanced propensity-matched cohorts were sufficient to detect clinically meaningful differences in outcomes with high statistical precision. All statistical tests were two-tailed with a significance threshold of P < .05.
Results
Our study identified 753 837 patients meeting inclusion criteria (bariatric surgery: n = 55 793; control: n = 698 044). At baseline, there were significant differences in demographics, comorbidities, and obstetric history between the 2 groups (Table 1). Patients who underwent bariatric surgery were older (mean age: 36.5 vs 28.3 years), had higher rates of chronic conditions such as hypertension, diabetes mellitus, and anemia, and demonstrated higher gravidity and parity compared to the control group. Among patients with a history of bariatric surgery, the mean time from surgery to delivery was 2.94 ± 1.76 years. Of the bariatric surgery group, 41 777 patients were successfully matched 1:1 with control patients based on age, race, ethnicity, and key comorbidities using propensity score matching. Following matching, the 2 cohorts were well-balanced across most baseline characteristics. Minor differences remained in variables such as parity and gravidity, which were slightly higher in the bariatric cohort. Vaginal and cesarean delivery distributions were comparable after matching. Notably, rates of chronic comorbid conditions such as hypertension, diabetes mellitus, and anemia were no longer significantly different after matching (Table 1), suggesting appropriate cohort balancing for outcome analyses.
Outcomes Between Patients Who Underwent Bariatric Surgery and Controls After Propensity Score Matching, Including ORs and 95% Cis*.
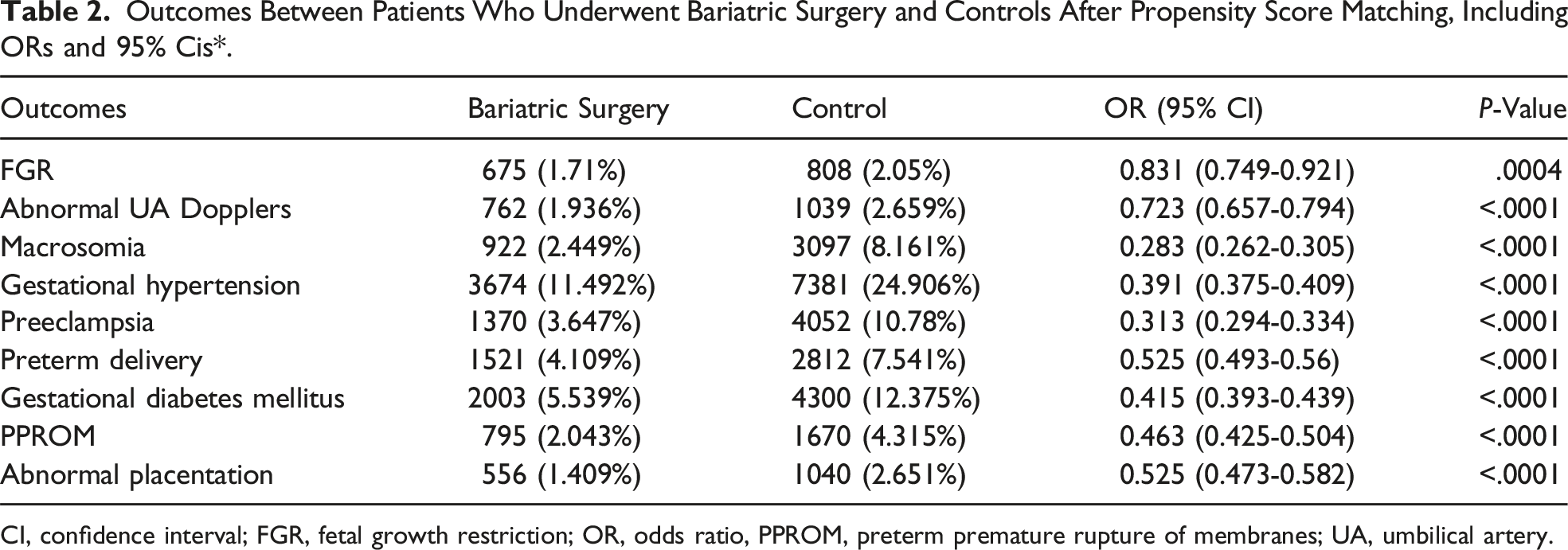
CI, confidence interval; FGR, fetal growth restriction; OR, odds ratio, PPROM, preterm premature rupture of membranes; UA, umbilical artery.
Discussion
The increasing prevalence of obesity in women of reproductive age has led to a growing number of bariatric surgeries performed to reduce obesity-related comorbidities and improve long-term health outcomes.3,15,16 Given the high risk that obesity poses to both maternal and fetal health, including gestational diabetes mellitus, hypertensive disorders, macrosomia, and preterm birth, understanding the perinatal outcomes in women with a history of bariatric surgery is critically important.10,11,14,16 Our study adds to the growing body of literature by demonstrating that bariatric surgery prior to pregnancy is associated with significantly lower risks of several adverse perinatal outcomes. Using a large, real-world, multi-institutional database (TriNetX), our analysis showed that patients with a history of bariatric surgery experienced reduced rates of fetal growth restriction (FGR), macrosomia, gestational hypertension, gestational diabetes mellitus, preeclampsia, and abnormal placentation compared to matched controls without bariatric surgery. These findings suggest that bariatric surgery may offer protective metabolic and vascular benefits during pregnancy. Weight loss and subsequent improvements in insulin sensitivity and systemic inflammation following bariatric procedures likely contribute to better placental function and reduced incidence of metabolic pregnancy complications.17-21 The reduced risk of macrosomia, for example, may reflect improved glycemic control, while the lower rates of hypertensive disorders may be due to enhanced vascular health post-surgery.13,19,21
These findings are consistent with prior studies that have shown improved metabolic profiles and pregnancy outcomes in post-bariatric patients.8,9,11,14 However, our study expands upon previous work by evaluating a broader range of outcomes, including FGR, abnormal umbilical artery Doppler, abnormal placentation, and PPROM, which have received less attention in the literature. We also observed that the bariatric cohort had lower rates of preterm delivery and PPROM. This may reflect improved maternal health status or more vigilant prenatal care. While nutritional deficiencies after bariatric surgery remain a concern, the overall benefits in reducing obesity-related complications seem to outweigh these risks in the current analysis. It is also possible that patients who undergo bariatric surgery receive more structured medical follow-up, nutritional counseling, and health education, which can positively impact pregnancy outcomes, although these factors could not be directly evaluated due to database limitations.
Strengths and Limitations
A major strength of our study is the use of a large, national dataset with robust propensity score matching to account for key demographic and clinical confounders. This approach allowed us to compare outcomes between cohorts with similar baseline characteristics, enhancing the internal validity of our findings. Additionally, we included a comprehensive array of maternal and fetal outcomes to provide a detailed characterization of risks associated with pregnancy after bariatric surgery. We acknowledge that combining different bariatric procedures may obscure procedure-specific effects on metabolic and nutritional outcomes, which could be clinically relevant for perinatal outcomes. While grouping procedures increased statistical power and minimized misclassification due to variable coding, future studies with sufficiently detailed and reliable data should evaluate each procedure individually to better characterize these distinct risks. Additionally, reliance on ICD-10 codes introduces the potential for misclassification and may incompletely capture all bariatric procedures, as some codes reflect diagnostic or status information rather than specific surgical interventions. The database also lacks information regarding patient compliance with nutritional supplements and differences in prenatal care, which may influence maternal and perinatal outcomes. Although TriNetX includes patients from both rural and urban settings, detailed socioeconomic and geographic variables are not available due to HIPAA restrictions, limiting our ability to account for these factors. These database constraints underscore the need for cautious interpretation of our findings and highlight opportunities for future studies with more granular clinical and sociodemographic data.
However, several limitations must be acknowledged. First, the retrospective nature of this study and reliance on ICD-10 coding introduces potential for misclassification and missing data. While we used multiple codes to define bariatric surgery procedures (eg, sleeve gastrectomy, gastric bypass, and others), we did not stratify outcomes by individual surgery types. This decision was made to maximize statistical power and reduce misclassification bias due to variable or incomplete procedural coding. We recognize that different procedures may have distinct nutritional and metabolic implications, and future studies should aim to compare them individually.
Due to the de-identified nature of TriNetX, important variables such as BMI at conception, nutritional status, and adherence to follow-up, our ability to determine whether the observed benefits are dependent on the timing of surgery relative to pregnancy is limited. As a result, BMI values are incompletely captured and not consistently available in TriNetX. We, as authors, elected to omit BMI from the analysis to avoid introducing incomplete or potentially biased measurements.
Third, the control group, while matched for key variables, may still differ in unmeasured ways that could influence outcomes. For instance, despite matching for age and select comorbidities, differences in genetic risk factors, social determinants of health, or baseline health behaviors may have influenced the results. Additionally, our control group consisted of pregnant patients without a history of bariatric surgery, but we did not include an “obese non-surgical” control group, which may have provided a more clinically appropriate comparator. However, such subgroup analyses introduce further challenges related to unmeasured confounding and limited data on baseline risk profiles. Importantly, we acknowledge that the absence of pre-pregnancy BMI and detailed socioeconomic variables in TriNetX may bias the observed associations, as these factors play a central role in both surgical candidacy and pregnancy outcomes. Future work incorporating these measures could help clarify whether the risks and benefits we observed are independent of these unmeasured influences. We acknowledge that women who undergo metabolic bariatric surgery (MBS) may differ from controls in ways not fully captured by the available data, such as greater motivation for health optimization or adherence to medical recommendations. Post-bariatric patients may also receive more intensive prenatal monitoring, which could influence observed outcomes. While our control group did not include an “obese non-surgical” comparator, such a group may have provided a more clinically appropriate reference. Additionally, patients with abortive outcomes were excluded because the de-identified TriNetX database does not allow reliable capture of such events using ICD-10 codes, limiting our ability to include them in the analysis. This restriction may influence the generalizability of our findings and should be considered when interpreting the results.
Finally, the lack of linked maternal-neonatal records in the database restricted our ability to explore direct neonatal outcomes or long-term infant health. Geographic information and socioeconomic status, which can influence access to care and health outcomes, were also unavailable due to HIPAA restrictions within the TriNetX platform. Moreover, a formal a priori power analysis was not performed, as the very large sample size provided sufficient power to detect clinically meaningful differences. While we believe the analysis is statistically robust, the possibility of type II error for negative findings cannot be entirely excluded, particularly for rarer outcomes or subgroup analyses. We therefore caution that null findings should be interpreted in the context of these limitations.
Clinical Implications and Future Directions
Our findings support the growing consensus that bariatric surgery before pregnancy may confer substantial benefits in reducing the risk of adverse perinatal outcomes associated with maternal obesity. These data reinforce existing clinical guidelines that encourage weight optimization prior to conception and suggest that bariatric surgery can be a valuable part of that strategy for women with severe obesity. At the same time, our study highlights the need for enhanced surveillance and individualized care planning for pregnant patients with a history of bariatric surgery. Nutritional deficiencies, surgical history, and weight trajectory should all be considered when counseling patients and managing pregnancies in this population. While our study provides important insights, prospective studies are still needed to explore the mechanisms underlying these outcomes, evaluate specific bariatric procedure types, and determine optimal timing of surgery relative to conception. This large, retrospective study demonstrates that women with a history of bariatric surgery have significantly lower risks of several maternal and fetal complications during pregnancy compared to matched controls. These findings underscore the value of bariatric surgery in reducing obesity-related pregnancy risks and support its role in preconception care for eligible patients. However, continued research is necessary to refine surgical timing recommendations, assess long-term maternal and neonatal outcomes, and optimize care pathways for this growing patient population.
Conclusion
This study highlights the significant reduction in adverse perinatal outcomes among pregnant individuals with a history of bariatric surgery compared to matched controls without such history. Using a large, multi-institutional dataset, we found lower risks of fetal growth restriction, macrosomia, gestational hypertension, gestational diabetes mellitus, and preeclampsia in the bariatric surgery group, supporting the procedure’s role in mitigating obesity-related pregnancy complications. These findings suggest that bariatric surgery may improve maternal metabolic health and placental function, positively influencing pregnancy outcomes. However, limitations such as lack of data on BMI at conception, surgical timing, and maternal-neonatal linkage must be acknowledged. While residual confounding remains possible, the protective associations observed emphasize the importance of preconception weight optimization. Clinicians should consider surgical history during prenatal care planning, and future research should explore optimal surgery-to-conception intervals and long-term infant health. Overall, our results contribute valuable evidence to guide perinatal management in women following bariatric surgery.
Footnotes
Ethics Considerations
Our institution’s Institutional Review Board (IRB) determined that this study is exempt from IRB review.
Author Contributions
Joshua E. Lewis, BS: Conceptualization, methodology, writing—original draft preparation, writing—review and editing, and data curation. Amani R. Patterson, MBS, BA: Writing—original draft preparation and writing—review and editing. Bryce Gantt, MS: Writing—original draft preparation and writing—review and editing. Raven Harding, BS: Writing—original draft preparation and writing—review and Editing. Gabriella Tavera, BS: Writing—original draft preparation and writing—review and editing. Diala Burjak, MD: Writing—original draft preparation and writing—review and editing. Nnadozie Uchegbu, MS: Writing—original draft preparation and writing—review and editing. Oyetojunbo Ibidapo-Obe: Reviewing and editing, supervision, and conceptualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Institute for Translational Sciences at the University of Texas Medical Branch, supported in part by a Clinical and Translational Science Award (UL1 TR001439) from the National Center for Advancing Translational Sciences at the National Institutes of Health (NIH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. This project was funded in part by the Department of Health and Human Services, Health Resources and Services Administration (HRSA), through its grant to the UTMB Center of Excellence for Professional Advancement and Research (grant number 1 D34HP49234‐01‐00). HRSA had no role in decisions related to the research, authorship, or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this retrospective study are available from the corresponding author on reasonable request.
