Abstract
Background
Perforated marginal ulcers (PMUs) are a rare but known complication of bariatric surgery. Management typically involves prompt surgical intervention, but limited data exists on non-operative approaches. This study reviews published data on non-operative management of PMUs and presents a case series of patients who were managed non-operatively. Our hypothesis is that certain patients with signs of perforation can be successfully managed non-operatively with close observation.
Methods
We completed a systematic review searching PubMed, Embase, Web of Science, Cochrane, and clinicaltrials.gov. Ultimately 3 studies described the presentation and non-operative management of 5 patients. Additionally, we prospectively collected data from our institution on all patients who presented between Dec. 2022 and Dec. 2023 with PMUs confirmed on imaging and managed non-operatively.
Results
In our literature review, three patients had Roux-en-Y gastric bypass (RYGB), while two had one anastomosis gastric bypass. One patient required surgery two days after admission. Another underwent elective conversion surgery weeks later for a non-healing ulcer. Two received endoscopic interventions. One patient recovered with nil-per-os (NPO) status, and intravenous proton pump inhibitor (PPI) treatment. The patients in our case series presented with normal vital signs, an average of 30 months after RYGB, and with CT scan signs of perforation. None of these patients required surgical or endoscopic intervention.
Conclusion
In conclusion, while perforated marginal ulcers have traditionally been considered a surgical emergency, some patients can be successfully treated with non-operative management. More research is needed to identify the clinical presentation features, comorbidities, and imaging findings of this group.
Key Takeaways
• There is not a large amount of literature describing the non-operative management of perforated marginal ulcers, but some reviews describe successful methods of management without surgery. • Perforated marginal ulcers are a serious complication of bariatric surgery that in certain cases may be amenable to conservative management. • With advances in CT imaging modalities, some findings of perforation need to be confirmed with fluoroscopic prior to proceeding to the operating room.
Introduction
A perforated marginal ulcer (PMU) is a rare but serious complication of Roux-en-Y gastric bypass (RYGB). PMUs are full-thickness defects in the bowel wall at the gastrojejunostomy (GJ) between the Roux limb and the gastric pouch. The incidence of marginal ulcers has been reported to be as high as 25%, with an incidence of PMUs around 1%. 1 Despite their low incidence, PMUs are frequently associated with high morbidity and mortality. Reported mortality rates of perforated peptic and marginal ulcers range from 2% to 20%, depending on comorbidities and timing of presentation.2,3
Management of PMUs is typically prompt surgical intervention, with the goal of addressing the defect and any associated complications, using techniques ranging from omental patching to full revision of the GJ anastomosis.4,5 While some studies have shown that non-surgical approaches, such as endoscopic suturing, may be effective in managing small bleeding marginal ulcers without the need for surgery, there is limited data on managing PMUs non-operatively.5,6 In the setting of advanced imaging modalities and thin-slice computed tomography (CT) scanners, there is a subset of patients with “microperforations” that can potentially be successfully managed with non-operative therapy. However, no studies to date have evaluated or defined the criteria for successful non-operative management. With the rising rates of bariatric operations, providers must be prepared to manage several cases of PMUs each year.
This study reviews published data on non-operative management of PMUs and presents a case series with a proposed treatment algorithm of patients who were successfully managed non-operatively. We hypothesize that patients who present with signs of perforation on initial imaging, but stable vital signs and minor laboratory derangements, can be managed with medical therapy and close observation.
Methods
Information Sources and Search Strategy
A comprehensive search for English language studies was conducted in PubMed (NLM), Embase (Ovid), Web of Science (Clarivate), Cochrane Library (Wiley), and clinicaltrials.gov by a health sciences research librarian. The search strategy used subject headings and keywords to find literature on the presentation of marginal ulcers in patients following bariatric surgery. All the searches were conducted on September 2, 2022. The study protocol can be accessed at PROSPERO (CRD42022354519), and the full database search strategies are in Appendix 1.
Study Selection
All screening and data extraction of included studies was done independently and in duplicate using the Covidence systematic review software.
7
See Figure 1 for the study flowchart showing the stages from identification to final inclusion, and Appendix 2 for the full list of data extracted from all the included studies. Disagreements were resolved by discussion until consensus was met on all exclusions and inclusions. Articles were excluded if there was absence of imaging within 12 hours of admission, absence of prior bariatric surgery, prior hospitalization or operative intervention for marginal ulcer, perforation secondary to iatrogenic injury, patients under the age of eighteen, or the study was not in English. Study selection.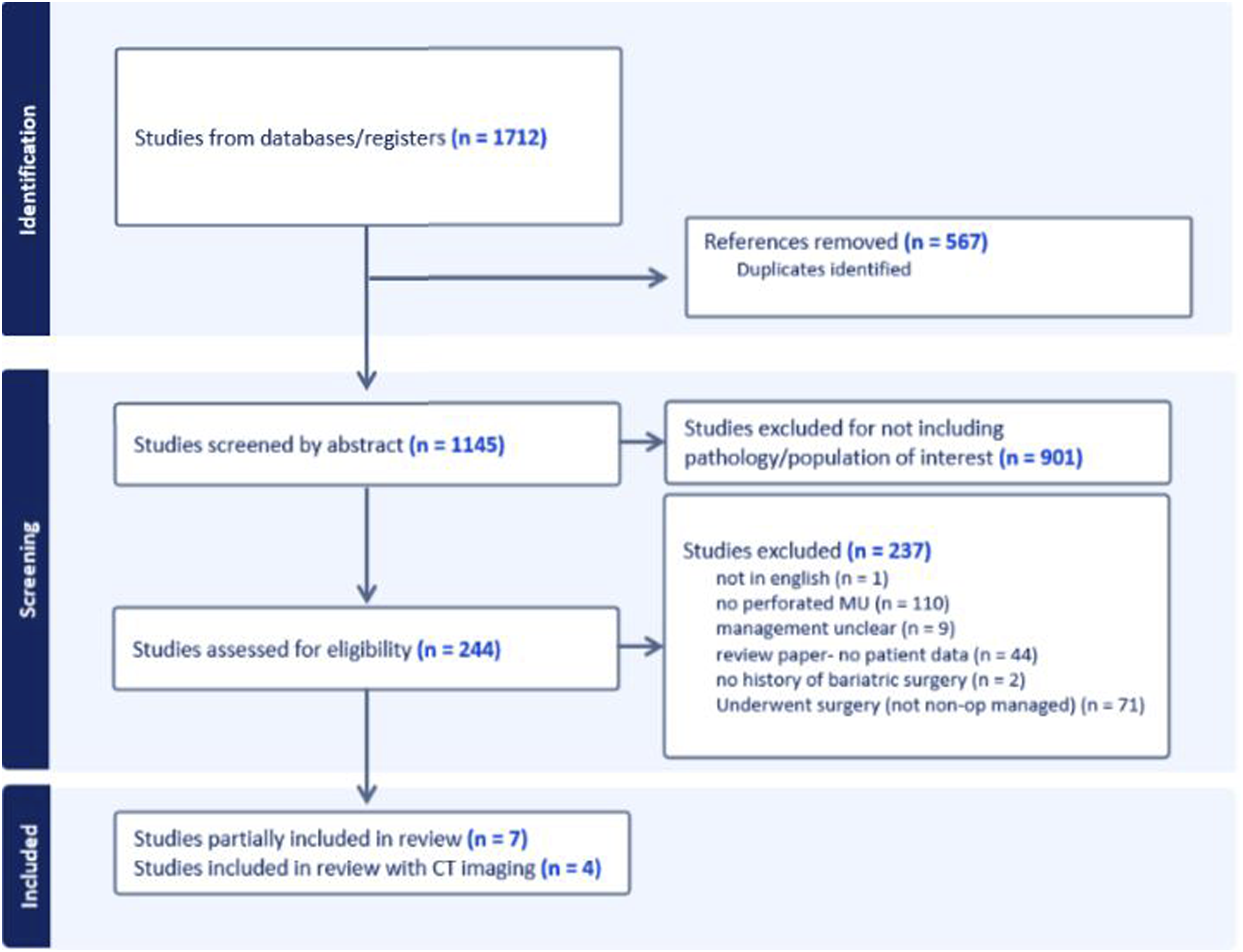
Primary and Secondary Outcomes
The primary outcome of interest was the incidence of successful non-operative management of PMUs, defined as a discharge to home without morbidity after a hospitalization that did not require any surgical intervention. Secondary outcomes of interest included number of ICU days, hospital length of stay, and need for non-operative intervention (eg, image-guided drain placement or endoscopy), and discharge destination.
Case Series
Patients were prospectively identified patients who presented with CT signs of PMUs between December 2022 and December 2023 and were managed with non-operative therapy. Data were obtained via manual chart review of the electronic medical record.
Results
Description of Studies
Of the seven studies included in our initial literature review, three included only a single patient who was managed non-operatively and did not provide any additional specific patient data, and one did not include any imaging. The systematic review by Martino et al included 102 patients with PMUs managed non-operatively, but 101 of these patients were from the study by Altieri et al, which had no specific patient data. However, notably these studies reported a 0% 30-day mortality, likely representing a more stable presentation.1,4 The one remaining patient in the Martino et al study was identified in the paper by Aviran et al, 8 which was included in this systematic review. Coblijn et al noted a patient in 2015 who developed a PMU adjacent to a nasojejunal tube 3 weeks after conversion of adjustable gastric banding to RYGB. Similarly in 2020, Vedantam et al described a single patient who developed a PMU shortly after endoscopic stent placement.9,10 It is unclear whether these perforations were iatrogenic or due to the ulcer. Although no imaging used for initial diagnosis was described, Wei et al 11 described a strategy using two overlapping partially covered stents placed endoscopically. This patient developed a jejunal leak 25 days later and became dependent on total parenteral nutrition.
Demographic Features and Imaging Findings of Patients With PMU on Presentation.
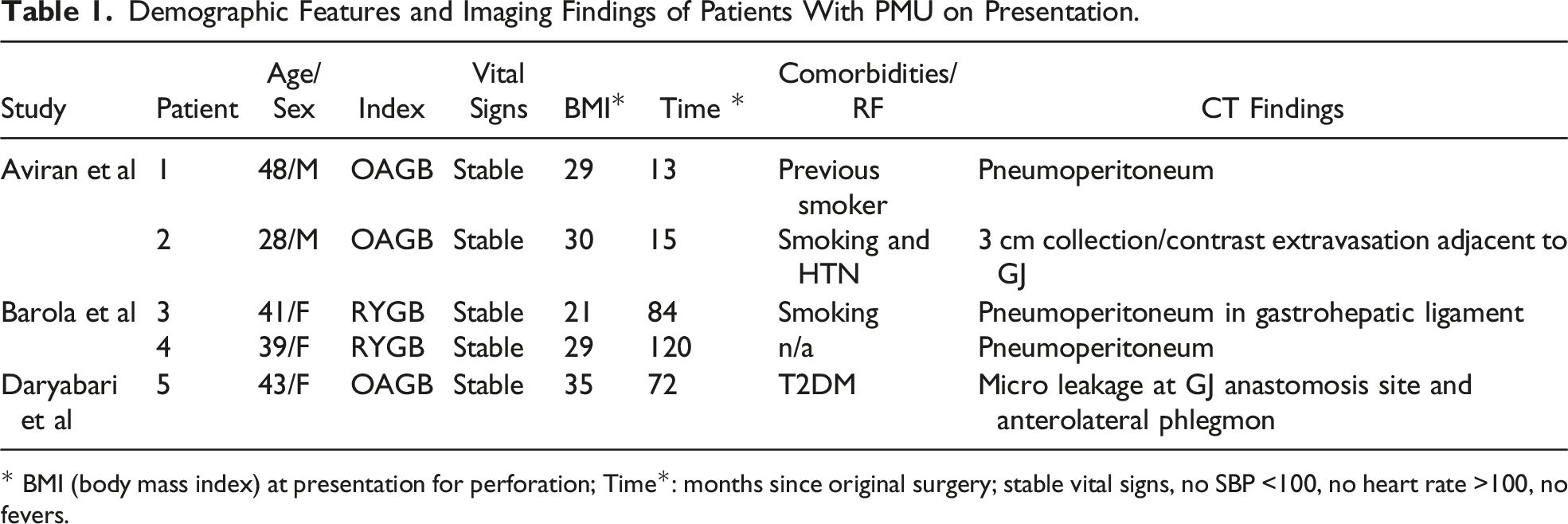
* BMI (body mass index) at presentation for perforation; Time*: months since original surgery; stable vital signs, no SBP <100, no heart rate >100, no fevers.
Patient Demographics
Of the five patients, three had RYGB, and two had a one anastomosis gastric bypass (OAGB). The median age of the cohort was 41, with a roughly even distribution of men and women. The median body mass index (BMI) of the patients at the time of presentation with perforation was 29 kg/m2. The median time from surgery to presentation was 72 months. Two patients had data on comorbidities, with type 2 diabetes in one and hypertension in the other. Two patients were current smokers, and one patient was a prior smoker. Only one patient (#2) had a history of a prior marginal ulcer, and no patients were using non-steroid anti-inflammatory drugs (NSAIDs). None of the patients initially presented with unstable vital signs or fevers in the emergency department (ED).
Patient Data
Initial Non-Operative Management Strategies and if Surgery was Ultimately Needed for These Patients.
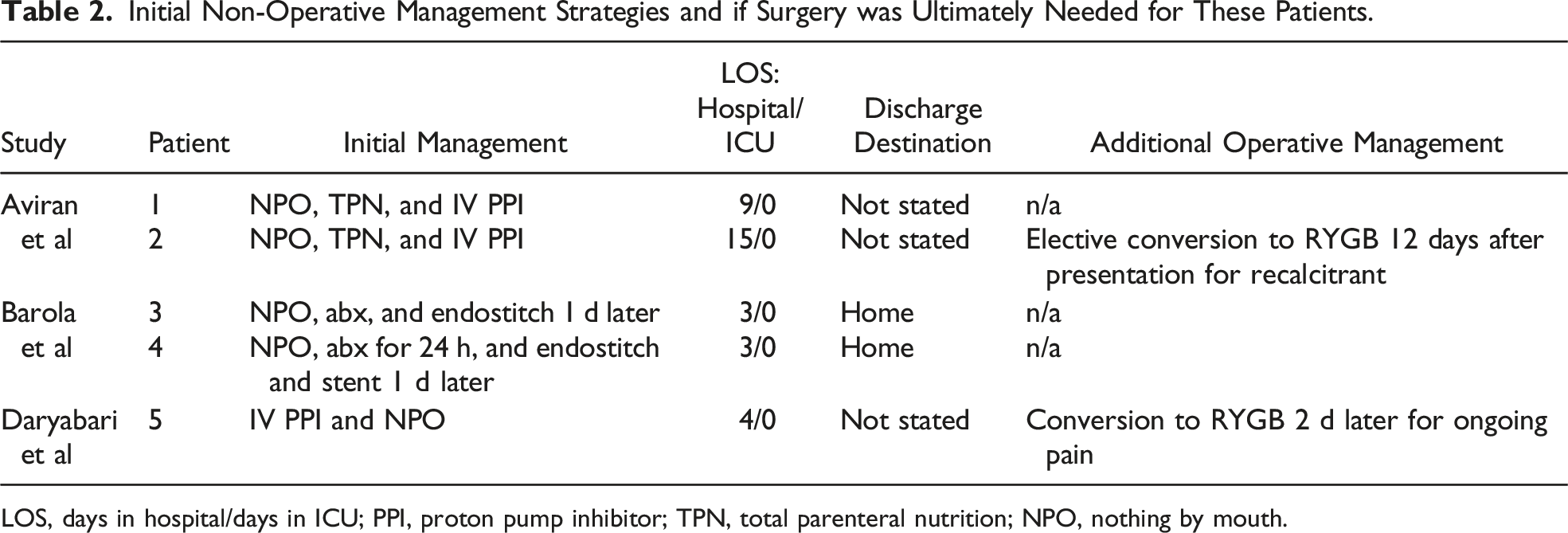
LOS, days in hospital/days in ICU; PPI, proton pump inhibitor; TPN, total parenteral nutrition; NPO, nothing by mouth.
Case Series – Non-Operative Management of Perforated Marginal Ulcers
Patient #1
The first patient is a 65-year-old female who had laparoscopic RYGB two years prior to presentation at an outside facility. She had a history of endoscopy-proven marginal ulcer identified three months prior to presentation. She developed two days of acute epigastric pain and limited oral intake. A CT scan demonstrated small areas of extraluminal air without extraluminal enteric contrast (Figure 2A). A small subdiaphragmatic fluid collection was also seen. The patient was afebrile with a normal heart rate and blood pressure, and a benign abdominal exam. She had a leukocytosis with a white blood cell count of 20.9 (x103/mcl). Case series CT scans – (A) Patient #1, (B) Patient #2, (C) Patient #3, and (D) Patient #4.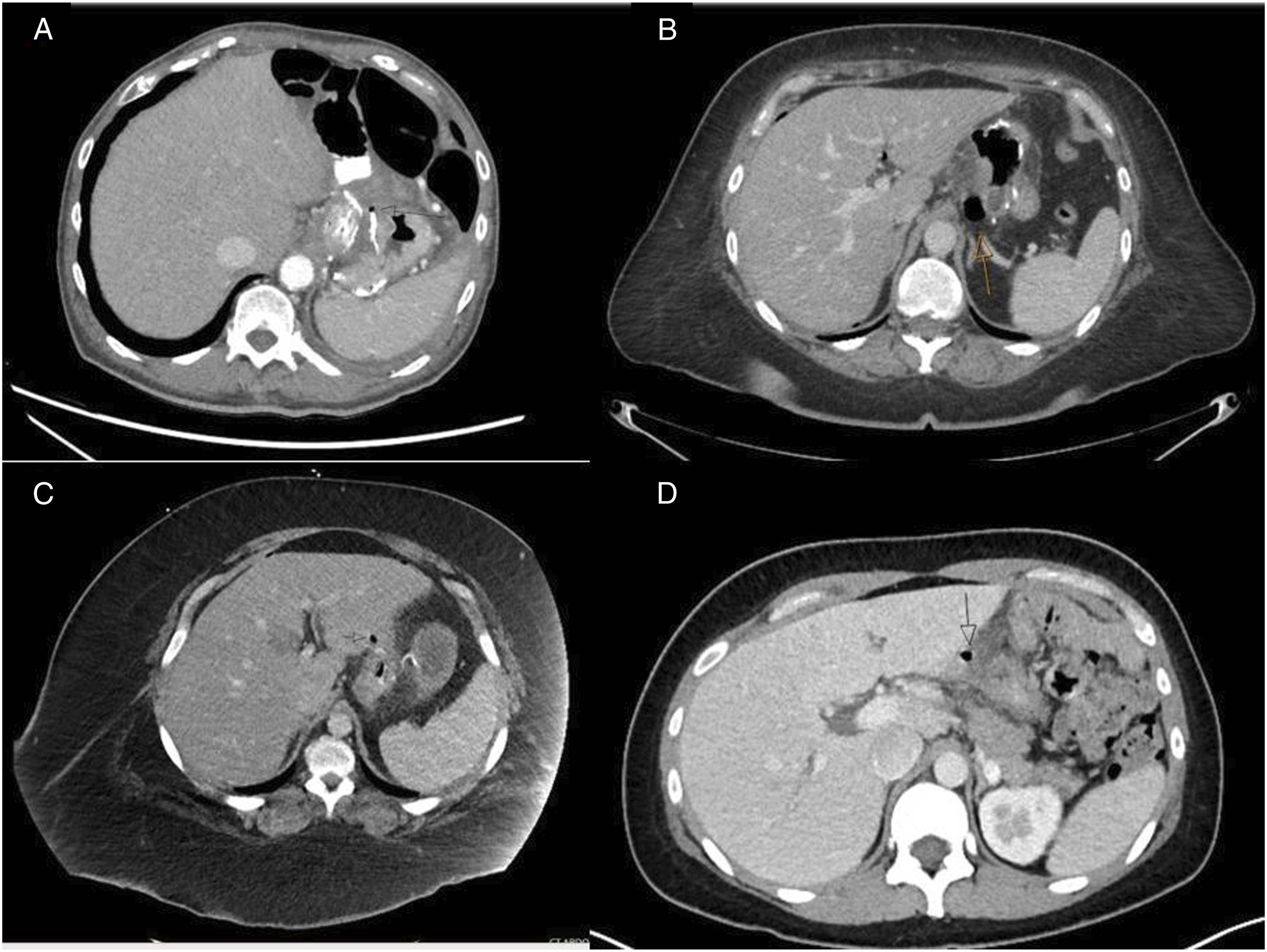
Given her clinical stability and benign exam, she was admitted to the surgical floor for serial exams and telemetry. She was started on empiric antibiotic treatment for contained perforation with piperacillin/tazobactam and vancomycin. Given plans for seven days of strict NPO, a peripherally inserted central catheter (PICC) was placed and total parenteral nutrition (TPN) was initiated. High dose proton pump inhibitor (PPI) therapy was initiated with sucralfate. The subdiaphragmatic fluid collection was managed with image-guided aspiration. On hospital day 7, an upper gastrointestinal series was completed, with no leak demonstrated. Her diet was advanced without complication, and she was discharged on a full liquid diet. She completed a 2-week course of intravenous antibiotics. A repeat outpatient CT scan did not demonstrate any persistent or new fluid collections and showed resolution of inflammation surrounding the GJ anastomosis. At 3-week follow-up, she was doing well and tolerating a bariatric regular diet.
Patient #2
The following patient is a 40-year-old woman four months after laparoscopic RYGB who presented to the ED with acute onset epigastric pain with radiation to the right shoulder. She was hemodynamically stable on arrival with a normal white count and normal lactate. A CT scan demonstrated several foci of pneumoperitoneum adjacent to the GJ anastomosis, which was thickened and inflamed (Figure 2B). Given her clinical stability and reassuring vitals and labs, the patient was admitted for close observation, serial exams, with strict NPO status, intravenous (IV) antibiotics, and TPN for nutrition. High dose PPI and sucralfate were also initiated.
During her hospitalization, the patient remained clinically well. On hospital day 6, an upper gastrointestinal series was obtained which did not demonstrate any ongoing leak. The patient was thus started on a bariatric clear liquid diet with continued PPI and sucralfate therapy. TPN was discontinued with toleration of an oral diet, and she was discharged home on a bariatric full liquid diet. As an outpatient, her diet was further successfully advanced without issue to a bariatric regular diet, and at a 3-month post-hospitalization follow-up, she was doing well without complication.
Patient #3
The third patient is a 31-year-old woman who presented to the ED four weeks following laparoscopic RYGB with acute onset, severe burning epigastric pain. Prior to this, she had convalesced well from her bariatric operation without issue. She had discontinued her prescribed PPI one week prior to presentation. On arrival, she was hemodynamically stable, with no leukocytosis, acidosis, or anemia. A CT scan on arrival demonstrated a locule of extraluminal gas adjacent to the GJ anastomosis, with several other foci of pneumoperitoneum and inflammatory changes surrounding the GJ anastomosis, concerning for PMU (Figure 2C). Oral contrast had been administered without identification of extraluminal contrast, suggesting a contained perforation. Given these findings, she was admitted for serial exams, strict NPO status, IV antibiotics, and parenteral nutrition.
The patient did not demonstrate any signs of sepsis or uncontained perforation during her hospitalization. On hospital day 6, she underwent an upper gastrointestinal series which did not demonstrate any leak. She was given a diet and transitioned to oral antibiotics, both of which were well tolerated, and she was subsequently discharged. At follow-up two weeks later, the patient was doing well without complaints and tolerating a bariatric regular diet.
Patient #4
The final patient is a 34-year-old woman who presented to the ED eight years after laparoscopic RYGB with a 24-hour history of sharp abdominal pain. She previously had mild epigastric pain a year prior, and upper endoscopy at the time showed no identifiable issues. On this presentation, she had nausea, but no emesis, fever, or other symptoms. Her abdomen was tender to palpation, but she had no signs of peritonitis. She had no history of NSAID use but had taken a 5-day course of steroids for sinus infection. She presented with stable vitals and a slight leukocytosis to 13.3 (x103/mcl). In the ED, a CT scan demonstrated punctate pneumoperitoneum near the gastrohepatic ligament and surrounding stranding (Figure 2D), concerning for PMU. Given her overall stability, she was admitted and made NPO, given high dose IV PPIs, sucralfate, and broad-spectrum antibiotics.
The next morning, she underwent an upper gastrointestinal series with oral contrast that did not demonstrate any ongoing leak or perforation. On hospital day 2, her pain had improved, and she was transitioned to an oral PPI, ciprofloxacin, flagyl, and a clear liquid diet. With continued improvement, she was discharged on hospital day 3, tolerating a bariatric full liquid diet. She remained on sucralfate and a PPI, and continued a planned 14-day course of antibiotics. On follow-up a week later, she was not having ongoing pain issues and was tolerating a soft diet.
Discussion
This study demonstrates that a cohort of patients with PMUs exists which can be successfully managed non-operatively. Patients likely to be successfully managed with a non-operative approach are those who present with stable vital signs, no signs of peritonitis, and only mild lab derangements. However, a paucity of data exists comparing an observation-first strategy to the current more common approach of initial surgical intervention.
The clinical presentation of PMUs is often dramatic, with patients presenting with acute abdominal pain, nausea, vomiting, and signs of peritonitis. The diagnosis is based on clinical findings, with radiologic or endoscopic confirmation. The traditional treatment of PMUs involves surgical intervention to address the defect and any associated complications like intra-abdominal abscess or sepsis. Multiple techniques have been developed for the management of PMUs, including laparoscopic repair, endoscopic closure, and omental patching.5,6 However, the use of observation alone, akin to the management of perforated diverticulitis, remains largely unstudied.
While no studies have directly compared operative and non-operative management of PMUs, studies have compared these approaches in other gastrointestinal perforations. Udelsman et al
12
compared both approaches in management of spontaneous pneumoperitoneum and similarly found no difference in 30-day mortality in either group when comparing patients without peritonitis. Cao et al
13
obtained similar findings in their study of patients with perforated gastroduodenal ulcers, with no difference in morbidity or hospital stay between those managed with or without an operation. These studies, like ours, found that a majority of patients successfully managed non-operatively present with stable vitals, mild lab derangements, and non-peritonitic exam. Based on these studies and our own experience, we recommend a conservative approach to stable appearing patients as outlined in Figure 3. Proposed management algorithm of PMU.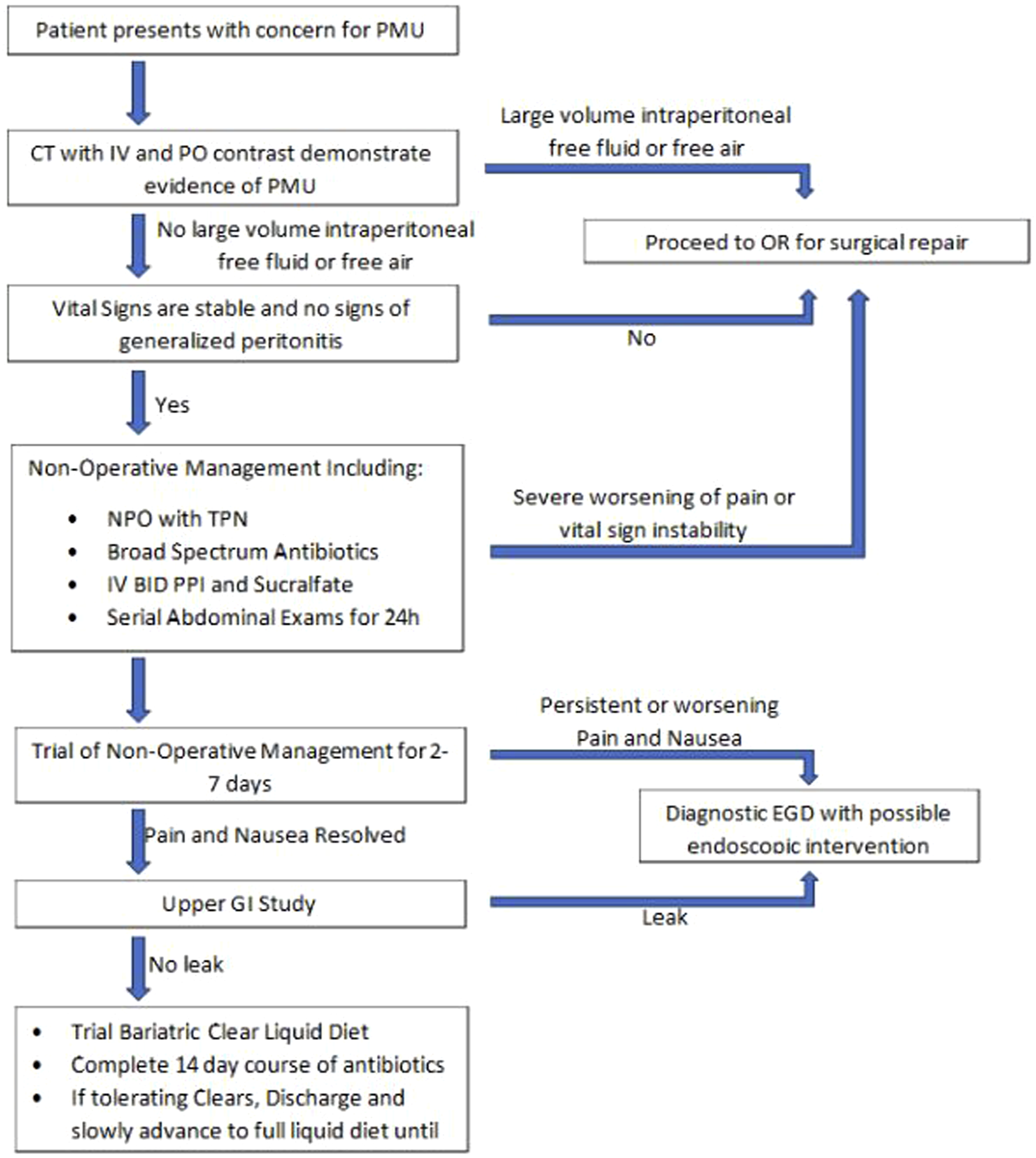
Advancements in imaging may explain an evolution in the management of PMUs, as thin-slice CT may be able to better detect microperforations compared to older modalities. 14 However, extraluminal air can occur without true perforation, and a diagnosis of PMU should be confirmed with a fluoroscopic gastrointestinal series demonstrating extravasation of contrast. To the authors’ knowledge, the specificity of pneumoperitoneum in diagnosing PMUs is unstudied. Data presented in our study raise the need for determining true PMUs, and the criteria for deciding which patients do not need surgical management. Ultimately, patients should have a holistic evaluation of their clinical presentation, including abdominal exam, labs, and close observation for those managed initially with a non-operative approach.
This study has several limitations beyond the retrospective nature and small sample size. First, the sample sizes are too small with the current literature to identify risk factors for failure of non-operative management of PMUs or for addressing any selection bias in patients described as successfully managed in this systematic review. Additionally, given the long median time periods between index surgery and presentation with PMUs, there is not enough data to know what the rates of recurrence of PMUs between these groups managed non-operatively and operatively.
Conclusion
This systematic review of PMUs identified a cohort of patients who can successfully be managed with a non-operative approach. Patients with small volume pneumoperitoneum, stable initial vitals, and minimal laboratory derangements may benefit from close observation and further evaluation instead of immediately proceeding to the operating room. More research is needed to determine the specificity of CT scans for PMUs, and the indications for non-operative management need further clarification.
Footnotes
Appendix 1:
Appendix 2:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
