Abstract
Background: Morel-Lavallee lesions (MLLs) are rare closed soft tissue degloving injuries caused by shearing forces that separate skin and subcutaneous tissue from underlying fascia. These injuries may be underrecognized in polytrauma patients, increasing the risk of delayed diagnosis, infection, prolonged healing, and long-term morbidity. This scoping review aims to provide a comprehensive summary of clinical presentation, imaging features, and evolving management for MLLs, with the goal of identifying gaps in the literature and guiding clinical decision making. Methods: We systematically searched PubMed, Scopus, Web of Science, Cochrane CENTRAL, and ClinicalTrials.gov between 1992 and 2025 for studies pertaining to adult patients with MLLs. Four independent reviewers screened full texts for inclusion. Data were extracted and summarized using narrative synthesis. Of 633 unique articles, 102 underwent full-text review, and 69 studies met inclusion/exclusion criteria. Results: MLLs commonly present with fluctuance, ecchymosis, swelling, and skin hypermobility. Diagnosis is clinical, with imaging playing a supportive role. Computed tomography and ultrasound can aid diagnosis in acute trauma settings, while magnetic resonance imaging is best for evaluating composition, chronicity, and extent. Management strategies vary based on size, chronicity, and anatomic locations. Acute lesions may be managed with compression or aspiration. Chronic or recurrent MLLs often require sclerodesis, minimally invasive debridement, or open surgical excision, often with subsequent soft tissue reconstruction. Conclusions: MLLs remain a diagnostic and therapeutic challenge in trauma. Given the heterogeneity of existing evidence, high-quality prospective studies are needed to establish evidence-based optimal management guidelines and improve long-term outcomes.
Keywords
Introduction
Morel-Lavallee lesions (MLLs) are closed soft tissue degloving injuries that occur following trauma. These lesions were first described in 1853 by the French physician Maurice Morel-Lavallee. 1 Morel-Lavallee lesions result from shearing forces that separate skin and subcutaneous tissue from the underlying fascia, which disrupts local microvascular and lymphatic networks. This results in the accumulation of hemolymphatic fluid, necrotic fat, and/or debris. 2 If untreated, chronic inflammation and hemosiderin deposition can lead to fibrous capsule formation, which impairs fluid reabsorption and results in persistent collections of blood products, necrotic tissue, and fibrinous exudate.3,4 The true incidence of MLLs remains unknown due to underdiagnosis and delayed presentations; however, prior literature estimates that up to 8% of patients with acetabular fractures have concomitant MLLs.2,4
Morel-Lavallee lesions can be underrecognized in polytrauma patients, where the initial clinical focus is on life-threatening injuries. In the acute setting, MLLs typically present as soft fluctuant masses and are often mistaken for simple hematomas/seromas. Although initially sterile, MLLs are prone to wound complications such as secondary infection if not promptly managed, which can result in significant functional impairment and poor cosmetic outcomes.5,6 Delayed recognition has also been linked to longer hospital stays, higher resource utilization, and occasional need for complex soft-tissue reconstruction.6,7
Prior reviews on MLLs have been limited by narrow search methodologies and insufficient study inclusion, with most being specialized to specific areas of MLL occurrence (eg, orthopedic injuries).1,2,4,6,8 This has resulted in a lack of comprehensive understanding of available evidence. Given these gaps, this scoping review aimed to provide a more comprehensive and updated summary of existing literature for all MLLs (including non-orthopedic injuries), with a focus on physical examination findings, imaging characteristics, and evolving treatment paradigms.
Methods
This scoping review was preregistered, and the protocol was published on Open Science Framework (https://osf.io/8khfw). The review was conducted in adherence to the Joanna Briggs Institute Methods Manual for Scoping Reviews and reported according to the EQUATOR (Enhancing the QUAlity and Transparency Of health Research) Network Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) checklist (Supplemental Table 1).9,10
Eligibility Criteria
Eligible studies included randomized control trials, case-control, case reports (with ≥2 patients), case series, cohort, and observational studies. Studies involved adult patients (≥18 years old) with closed, internal degloving injuries consistent with MLL.
We excluded editorials, letters, narrative reviews, systematic reviews, and single-patient case reports. Studies were also excluded if they involved penetrating trauma, open wounds, open degloving injuries, or patients <18 years old.
Database Searches
A comprehensive search was conducted across five databases: PubMed, Scopus, Web of Science, Cochrane CENTRAL, and ClinicalTrials.gov, for studies on the diagnosis and management of MLLs from 1992 to 2025, with no language restrictions. A health sciences librarian (LM) developed the PubMed search strategy using relevant keywords, synonyms, and MeSH terms, and adapted it for the other databases. Manual reference checks of all included studies were performed to identify additional eligible literature. All records were imported and managed in Covidence (Veritas Health Innovation, Melbourne, Australia). The complete search strategy is available in Appendix A (last updated on June 7, 2025).
Study Selection
Following de-duplication, four reviewers (PDN, AB, MJ, and NT) independently screened titles and abstracts against predefined eligibility criteria. Full texts of potentially eligible studies were subsequently retrieved and independently reviewed by two of three physician reviewers (PDN, MJ, and AB). Inter-reviewer agreement for full-text inclusion was excellent (Cohen κ = 0.80). A total of 102 full-text studies assessed, with 69 meeting inclusion criteria. Of the 11 non-English (Hebrew, Spanish, German, Mandarin, and Hungarian) articles, 8 were included following translation using ChatGPT 4o (OpenAI, San Francisco, CA), accessed in May 2025. The study selection process is illustrated in the PRISMA flow diagram (Figure 1). PRISMA Flow Diagram With Identification, Screening, Eligibility, and Article Inclusion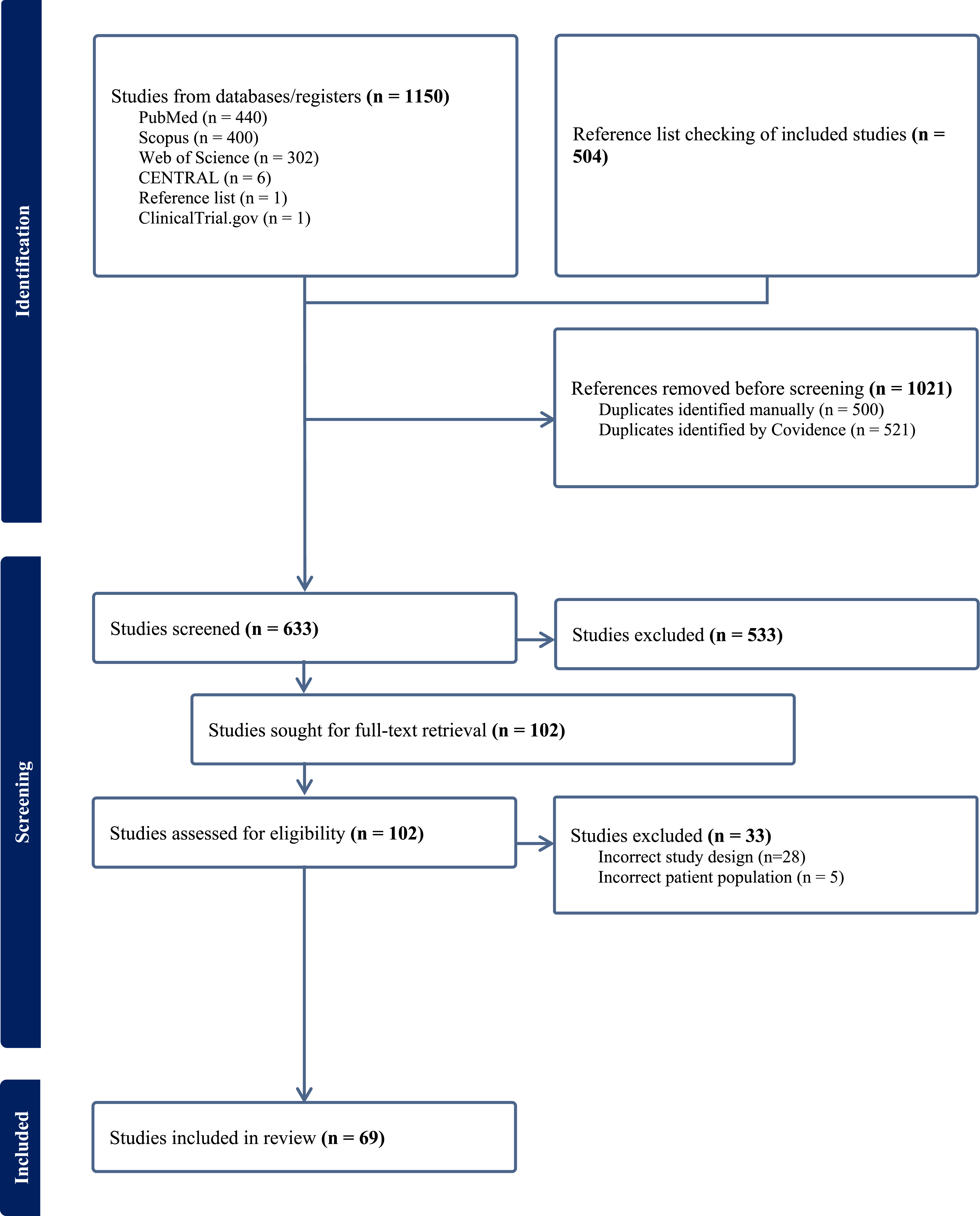
Data Extraction
A standardized data extraction template was developed and pilot-tested within Covidence using two included studies. Extraction was performed independently by two of the three reviewers (PDN, MJ, and AB) for each article evaluated, with conflicts resolved via discussion with the senior author (JN). Extracted data included demographics (age, sex), injury profile (mechanism of injury, Injury Severity Score (ISS), and MLL anatomic location), physical exam findings, imaging (ultrasound (US), computed tomography (CT), magnetic resonance imaging (MRI)), management strategies (non-operative, percutaneous, and operative), reconstructive techniques, and clinical outcomes. A complete list of data elements is provided in Appendix B.
In alignment with scoping review methodology, we did not perform formal quality assessment or risk of bias appraisal of included studies. All data were synthesized descriptively and organized into thematic categories related to MLL diagnosis, management strategies, and predictors of treatment failure.
Results
Search Results
The initial search yielded 1654 records. Following de-duplication, 633 unique studies were screened. Of these, 102 full-text studies were assessed for eligibility, and 69 met inclusion criteria (Supplemental Table 2). A total of 33 studies were excluded for incorrect study design (n = 28) or patient population (n = 5). The included studies were published between 1992 and 2025, with most (98.6%) being non-comparative case series or observational studies (Supplemental Table 3).
Clinical Presentation
Morel-Lavallee lesions may present within hours after trauma; however, many are initially overlooked and time to diagnosis can range from index hospitalization to several years post-injury. The clinical presentation of MLLs varies and is influenced by rate of hemolymphatic fluid accumulation and patient body habitus. 5 Early physical exam findings include localized ecchymosis, fluctuance, palpable mass, tenderness, and skin hypermobility (Supplemental Table 4). These external signs may be subtle, delayed, or absent entirely. In severe cases, the accumulation of fluid exerts pressure on overlying skin, which compromises perfusion and results in secondary skin necrosis.1,2,11-14
Although MLLs can occur anywhere shear forces are transmitted, certain anatomic regions are predisposed due to greater superficial tissue mobility relative to underlying fascia. When the Trauma Quality Improvement Project (TQIP) database study of abdominal MLLs is excluded, 15 the majority of MLLs occurred in the lower extremity (n = 890, 52.3%) with the most common locations being the thigh (n = 410, 24.1%), greater trochanter (n = 302, 17.8%), and knee (n = 140, 8.2%).7,14,16,17 Other reported sites of MLLs include lumbar (n = 356, 20.9%), gluteal (n = 176, 10.3%), pelvis (n = 139, 8.2%), flank (n = 79, 4.6%), and abdomen (n = 48, 2.8%) regions (Supplemental Table 5).7,16,18
Morel-Lavallee lesions often follow high-energy trauma and are frequently associated with concomitant injuries. Approximately 50% of MLL cases have associated fractures, most often involving the pelvis, acetabulum, or proximal femur, which anatomically overlap with high-risk areas for MLL development.16,19,20 In cases of abdominal MLLs, up to 40% of patients have concomitant hollow viscus injuries. 15
Chronic MLLs may manifest as persistent pain, skin tightness, or a palpable mass. Secondary infection may occur and present as cellulitis or abscess. Chronic lesions are unlikely to resolve without procedural or operative intervention due to fibrous capsule development and can impair activities of daily living.21,22 These findings reinforce the importance of early clinical recognition and timely management to limit complications and long-term morbidity.
Imaging Features
Morel-Lavallee Lesion Computed Tomography Findings

Morel-Lavallee Lesion Ultrasound Findings

Morel-Lavallee Lesion Magnetic Resonance Imaging Findings

STIR = Short-TI Inversion Recovery’ PDFS = Proton Density Fat Fraction; SPIR = Spectral Presaturation with Inversion Recovery; SPAIR = Spectral Attenuated Inversion Recovery.
Computed Tomography
Computed tomography scans are often the initial imaging modality utilized in the acute trauma setting. Despite their frequent use in clinical practice, CT findings of MLLs were reported in only 15 studies (21.7%). This limited representation may be due to publication bias, as CT offers poor specificity for MLLs. However, CT imaging can reveal nonspecific early findings (ie, fluid, fat stranding, and/or hematoma), 18 with most MLLs appearing as fluid collections between subcutaneous fat and underlying fascia (Table 1).17,23-26 When intravenous contrast is used, CT can help detect active extravasation, which may prompt treatment (ie, compression, angioembolization, and/or surgery). Morel-Lavallee lesions usually exhibit lower attenuation values (<30 Hounsfield units) than hematomas, which often measure around 75 Hounsfield units, although this may depend on timing of CT imaging in relation to injury. 27
Ultrasound
Ultrasound serves as an easily accessible adjunct in evaluation of MLLs and was reported in 16 studies (23.2%). Ultrasound can rapidly confirm fluid collections, determine lesion size and depth, assess compressibility, and exclude vascular flow through Doppler imaging; however, it lacks diagnostic specificity. 3 The sonographic appearance of MLLs depends on the chronicity of the lesion (Table 2).12,23-25,28-30 In acute and subacute phases (<1 month), MLLs typically present as heterogenous fluid collections with irregular, lobulated margins.17,26,31-35 Chronic lesions (>18 months) tend to appear more homogenous, with smooth, flat, or fusiform contours. 32 Color Doppler may demonstrate mild peripheral hyperemia in inflamed lesions without internal vascularity, a feature that helps distinguish MLLs from high-flow vascular malformations. 1 Although, US cannot reliably distinguish MLLs from other soft tissue pathologies, such as hematomas, abscesses, fat necrosis, or neoplastic processes. As a result, the role of US is largely adjunctive, often requiring additional imaging for definitive diagnosis and treatment planning, despite some reports of its assistance with percutaneous drainage for diagnostic/therapeutic purposes.7,36,37
Magnetic Resonance Imaging
Magnetic resonance imaging is the preferred modality to radiographically evaluate MLLs, with findings reported in 20 studies (29.0%). Magnetic resonance imaging offers superior characterization of lesion morphology, internal composition, and stage of evolution. The MRI appearance of MLL is highly influenced by anatomic location, chronicity, and internal contents (Table 3).29,34,37 Acute lesions typically appear heterogenous with irregular borders, while chronic lesions are more homogenous with smooth margins.3,38 Chronic MLLs often form a fibrous capsule, which appears as a hypointense ring due to hemosiderin deposition.3,39 Signal characteristics on MRI evolve over time, with acute and subacute lesions showing high signal intensity on T2-weighted images and low signal on T1-weighted images, consistent with serolymphatic fluid accumulation.12,17,23,24,26,33,35,39,40 As blood products within the lesion degrade, the T1-weighted images become progressively hyperintense.1,3
Mellado and Bercandino developed a six-type MRI classification system to categorize MLLs based on lesion shape, signal characteristics, and presence of a capsule. 38 However, this system lacks guidance on prognosis and treatment. To address this gap, Shen et al 8 proposed a simplified classification that dichotomizes MLLs into acute or chronic based on capsular formation, and offers recommendations for treatment strategies and prognostic outcomes, although with limited supporting evidence.
Management
Morel-Lavallee Lesion Management Strategies
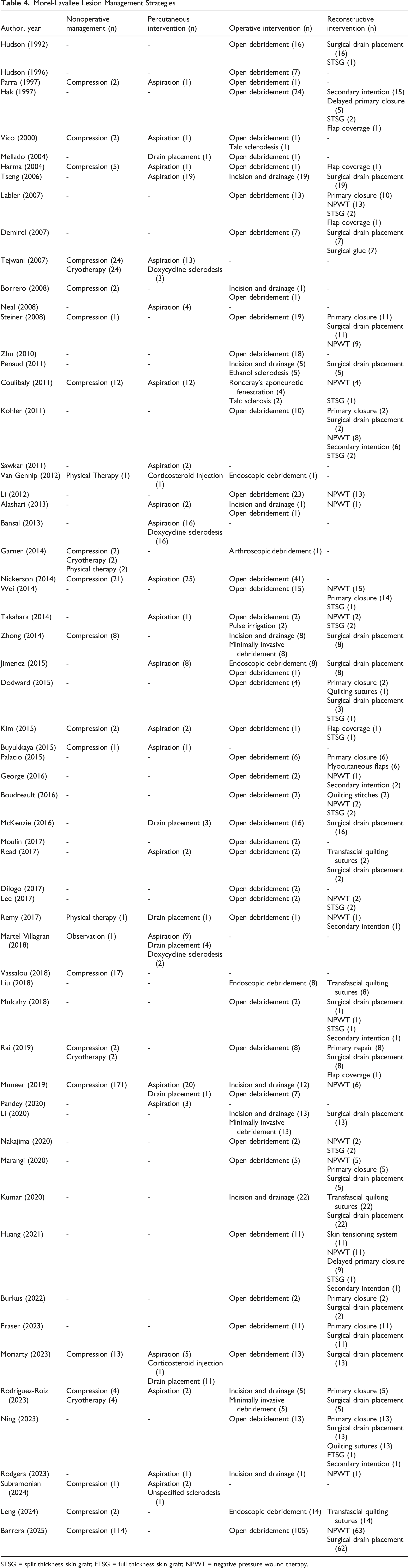
STSG = split thickness skin graft; FTSG = full thickness skin graft; NPWT = negative pressure wound therapy.
Nonoperative Management
Small acute MLLs without capsular formation may be managed nonoperatively with compression bandages.7,8,37,40 However, this approach carries a high risk of progression, recurrence, delayed healing, and subsequent procedural intervention.8,41 Lesions located in regions difficult to compress (eg, greater trochanter) or in obese patients are less likely to respond to conservative measures. 7
Percutaneous Management
Percutaneous aspiration, sometimes performed under US or CT guidance, aims to definitively diagnose MLL and reduce dead space. It is typically combined with compression therapy. 32 Catheters may be inserted for continuous fluid evacuation and can be coupled with negative pressure systems or delivery of sclerosing agents. 8 Nickerson et al 16 put forth the largest study investigating the utility of percutaneous management and demonstrated that aspiration alone is associated with a high recurrence rate, particularly for lesions >50 mL. Accordingly, many authors now advocate for early operative drainage/debridement for high-volume cavities.1,16
Sclerodesis
In chronic MLLs with well-formed capsules, sclerodesis can eliminate dead space by disrupting the capsule lining and promoting inflammation, fibrosis, and adhesion of tissue planes to prevent fluid reaccumulation. Multiple sclerosing agents have been reported, including doxycycline, erythromycin, bleomycin, vancomycin, ethanol, tetracycline, talc, and surgical glue.3,4,8,21,26,31,34,36,37,41 Percutaneous sclerodesis is typically reserved for lesions refractory to aspiration/compression or for those <400 mL in volume.1,16 In operative settings, sclerodesis with talc or ethanol has been used adjunctively to promote fibrosis.21,31,41,42 Reported success rates of sclerodesis reach as high as 95%, although these findings may be influenced by selection bias and a lack of high-quality comparative studies.8,36
Minimally Invasive Techniques
Minimally invasive approaches are increasingly favored for patients with viable overlying skin and appropriately sized lesions. Traditional techniques include small incisions for irrigation and drainage. Liposuction cannula debridement has been described to disrupt and collapse the lesion cavity.22,28 Tseng et al described a long plastic brush debridement technique followed by pulse lavage and drain placement. 11 Other described techniques include video-assisted endoscopic debridement.12,43,44 These minimally invasive approaches highlight the evolving landscape of treatment options for MLL and given the potential for decreased wound morbidity, these techniques merit future comparative research.
Open Operative Management
Open surgery is generally indicated for large, chronic, recurrent lesions or those in anatomic areas not amenable to compression (eg, greater trochanter, pelvis, and perineum), especially when associated with overlying tissue compromise.8,14,19,22,45 Alternatively, open operative management may occur in conjunction with other planned operations (eg, laparotomy for hollow viscus injury). Acute lesions with skin necrosis or significant soft tissue devitalization require open excisional debridement. Surgical intervention involves curettage or capsular excision to enable soft tissue apposition and healing. 46 Capsule fenestration has also been described in the literature. 41 Early surgical management is important to mitigate secondary infection by removing necrotic debris and fluid collections. However, open surgery also risks disruption of the remaining subdermal vascular plexus, potentially resulting in further tissue necrosis. 1
Reconstructive Techniques
The management of large MLLs may necessitate reconstructive procedures to address tissue loss. 11 Quilting sutures are used in both open and minimally invasive procedures to tack soft tissue or skin to underlying fascia and eliminate dead space.30,44,47 Primary closure is feasible for smaller lesions with minimal skin loss. Larger defects may require negative pressure wound therapy (NPWT) to reduce dead space and stimulate granulation tissue.13,47 Skin grafting or flap reconstruction may follow NPWT to restore soft tissue coverage. 13 Other adjuncts that have been attempted include progressive tension sutures, fibrin sealants, and synthetic glues to promote tissue adherence.1,42
Complications
Morel-Lavallee Lesion Outcomes and Follow-up
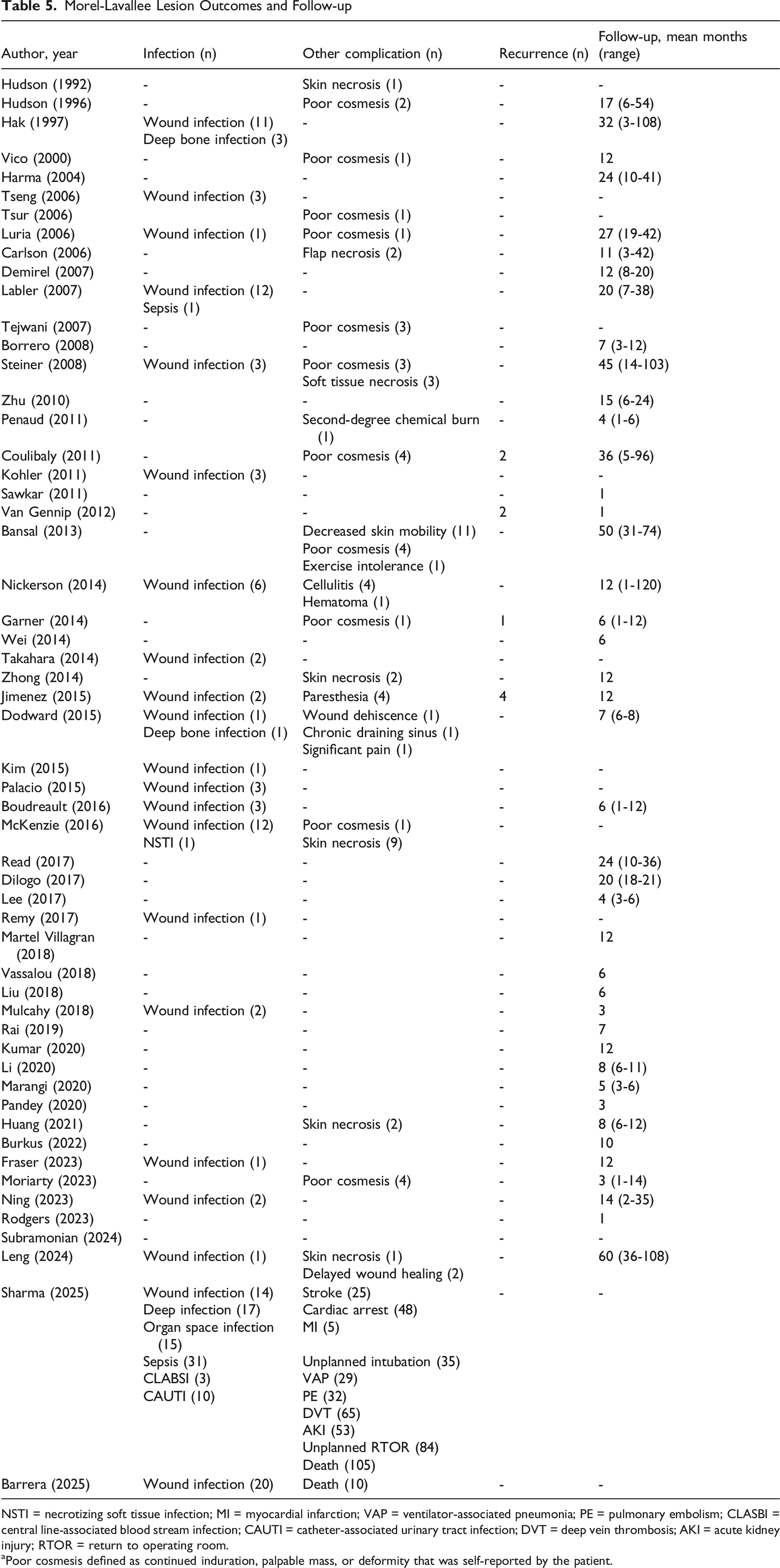
NSTI = necrotizing soft tissue infection; MI = myocardial infarction; VAP = ventilator-associated pneumonia; PE = pulmonary embolism; CLASBI = central line-associated blood stream infection; CAUTI = catheter-associated urinary tract infection; DVT = deep vein thrombosis; AKI = acute kidney injury; RTOR = return to operating room.
aPoor cosmesis defined as continued induration, palpable mass, or deformity that was self-reported by the patient.
Pressure-induced necrosis of the overlying skin can develop, particularly in large or chronic lesions, potentially exposing underlying fractures or orthopedic hardware. 8 Additionally, disruption of the subdermal vascular plexus during aggressive debridement or flap reconstruction may further compromise skin viability.1,18,20,46 Therefore, successful management requires a delicate balance between adequate debridement, effective dead space elimination, and preservation of viable skin and/or soft tissue for closure.
Infection is another significant concern in the management of MLL, which may result from direct inoculation through traumatic injury, hematogenous spread, or manipulation during treatment.4,8,19,45 The literature reports inconsistent findings on bacterial colonization, with some identifying high rates of positive cultures, while others note negative cultures following aspiration.1,8,19,36 Regardless, surgical procedures that traverse or disrupt an MLL cavity are generally regarded as high-risk for infection, reinforcing the importance of intraoperative cultures.
Recurrence is a common complication, with rates in the literature ranging from 0% to 75%, reflecting heterogeneity in study populations, definitions, and follow-up protocols.1,8,16,36 This underscores the need for standardized treatment algorithms and post-treatment surveillance.
Suboptimal healing and poor cosmesis are also a source of morbidity. Primary closure under tension can dehisce or reduce skin mobility, while inadequate soft tissue apposition can lead to delayed healing or chronic draining sinuses.36,44,45 Disruption of superficial cutaneous nerves may result in localized parasthesias. 43 Sclerosing agents, while effective, carry the risk for extravasation beyond the lesion cavity, leading to chemical burns of adjacent skin. 21 Additionally, sclerodesis has been associated with contour deformities, due to uneven collapse of the lesion cavity and subsequent poorly apposed fibrotic remodeling. 36
Limitations
This scoping review provides a summary of the clinical presentation, diagnostic modalities, and evolving treatment strategies for MLLs to aid clinical decision making. However, several limitations must be acknowledged. This review includes predominantly lower-quality retrospective studies with small sample sizes, heterogeneity in study populations, and inconsistent outcomes reported. Variability in lesion size measurements, recurrence definitions, and follow-up durations limit the findings of this scoping review. However, this highlights MLLs as another area of trauma research that may benefit from the development of a core outcome set.48-50 To maximize comprehensiveness and generalizability, we included case series irrespective of study quality. The predominance of observational studies introduces inherent biases and limits the ability to form casual inferences. Additionally, this review does not offer a comparative analysis of treatment efficacy, as no large high-quality comparative studies, let alone randomized trials currently exist for MLL management.
Future Research
Future research should prioritize large prospective cohort comparative studies to evaluate optimal timing of procedural interventions and study surgical and reconstructive techniques. The role of imaging, particularly CT scans, in guiding operative decision making for MLLs also remains unclear. Studies exploring imaging findings and/or volumetric analyses of MLLs could help refine treatment selection. Future randomized control trials are warranted to compare outcomes between treatment modalities, particularly stratified by lesion size and chronicity.
Conclusion
Morel-Lavallee lesions are frequently associated with concomitant orthopedic injuries but can also occur in other anatomical regions (eg, abdomen). Clinically, MLLs present with soft tissue swelling, ecchymosis, and fluctuance. Diagnosis is primarily based on a thorough history and physical examination, although imaging modalities such as US, CT, and MRI may be helpful adjuncts.
Management should be individualized, considering lesion size, chronicity, location, presence of wound complications, and associated injuries. The primary therapeutic goal is to obliterate the dead space and reduce ongoing hemolymphatic disruption. Small acute lesions may be managed nonoperatively with observation, compression therapy, and/or aspiration with or without drain placement. Chronic, large, and/or recurrent lesions often necessitate operative interventions, ranging from minimally invasive approaches to open surgery. Given the paucity of high-quality comparative data, a multidisciplinary approach involving trauma, orthopedics, and/or plastic surgery teams is essential to achieving optimal outcomes. Future efforts to develop standardized guidelines, core outcome sets, and large prospective randomized trials appear warranted to inform evidence-based management of MLLs.
Supplemental Material
Supplemental Material - Clinical Presentation, Imaging Features, and Management of Morel-Lavallee Lesions: A Scoping Review
Supplemental Material for Clinical Presentation, Imaging Features, and Management of Morel-Lavallee Lesions: A Scoping Review by Peter D. Nguyen, Nicholas Truong, Mallory Jebbia, Anushka Bagga, Linda S. Murphy, Jiabao Nie, Areg Grigorian, and Jeffry T. Nahmias in The American Surgeon™.
Footnotes
Author Contributions
Conception and study design: PDN, NT, MJ, AB, LSM, JN, AG, and JTN. Literature review: PDN, LSM, and NT. Data acquisition: LSM, NT, and JN. Data analysis and interpretation: PDN, MJ, and AB. Drafting of the manuscript: PDN, LSM, and NT. Critical revision: PDN, MJ, AG, and JTN
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
