Abstract
Background
Cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) is well established for mucinous cancers with peritoneal dissemination. Its role for non-mucinous tumors is less defined. This study compares outcomes between mucinous and non-mucinous cancer patients undergoing CRS-HIPEC to better understand therapeutic impact.
Methods
A prospectively maintained database of CRS-HIPEC patients at an academic tertiary referral center from 2011-2023 was analyzed, including patients with appendiceal, colorectal, gastric, ovarian tumors, and soft tissue sarcomas. Survival outcomes were assessed using Kaplan Meier curves and multivariate Cox-proportional hazards models.
Results
Among 195 patients, 55 (28%) had non-mucinous cancers and 140 (72%) mucinous tumors. The non-mucinous group had lower PCI (median 9 vs 14,
Discussion
Despite differential loco-regional features, non-mucinous cancer patients had similar survival after CRS-HIPEC. Differences in progression were primarily seen in those with appendiceal cancers, not colorectal tumors. These findings support the use of CRS-HIPEC across histologic subtypes, contributing to prognostication and risk-stratification for patients with differing cancer histopathology.
Keywords
Key Takeaways
• After CRS-HIPEC, there were no significant differences in overall or cancer-specific survival for patients with non-mucinous tumor histology vs mucinous tumors. • While poorer progression-free survival is seen in non-mucinous cancers after CRS-HIPEC, this difference is primarily driven by patients with appendiceal cancers. There was no difference in risk of progression for patients with colorectal tumors. • CRS-HIPEC should be considered as an appropriate treatment option for select patients with non-mucinous cancers with peritoneal dissemination, as histologic differences in mucin production alone should not be a contraindication.
Introduction
Mucinous carcinoma is a rare histologic subtype of epithelial tumor characterized by abnormal mucus production. While prognostic factors such as tumor differentiation, depth of invasion, nodal involvement, and lymphovascular invasion are well understood in cancer outcomes, mucin production is less well-defined. Secreted and transmembrane mucins form a protective barrier for epithelial tissues to naturally protect against harmful conditions and regulate inflammatory responses. However, many different types of malignancies have been found to exploit mucin overexpression to promote tumor cell signaling, growth, and survival. 1 As a unique disease process and potential poor prognostic indicator, mucin production has necessitated the development of alternative oncologic treatment strategies such as cytoreductive surgery with the additional of hyperthermic intraperitoneal chemotherapy (CRS-HIPEC). 1 Mucinous adenocarcinomas are frequently of colorectal and appendiceal origin, though they may arise from any glandular organ including the breast, stomach, ovary, and lung. 2
Although CRS-HIPEC was initially developed as a treatment for pseudomyxoma peritonei (a rare peritoneal cancer characterized by accumulation of mucin in the abdomen), favorable outcomes have been observed when applied to patients with peritoneal carcinomatosis from a variety of origins. 3 CRS focuses on safely removing visible macroscopic disease, while HIPEC targets residual microscopic and low-volume disease. While CRS-HIPEC been well-studied and utilized in treating peritoneal dissemination for patients with mucinous cancers, its application in non-mucinous tumors remains less extensively explored.
While mucin-secreting and non-mucinous tumors exhibit distinct histopathologic features, prognostic data remains varied. In some studies, mucinous tumors of various locations have been associated with poor differentiation, more advanced T stage and worse survival.2,4,5 Other studies have found no significant survival differences for patients with tumors with and without mucin production. 6 Particularly in the setting of peritoneal dissemination, differences in biological behavior and therapeutic response of mucinous and non-mucinous cancers remains poorly understood. Detailing the differential response of mucinous and non-mucinous cancer histopathology to CRS-HIPEC is valuable for guiding treatment and prognostication for patients and providers. In this study, we aim to compare oncologic outcomes among patients with mucinous vs non-mucinous cancers with peritoneal carcinomatosis after CRS-HIPEC to better understand its therapeutic role in these 2 distinct patient populations.
Materials and Methods
This study was approved by our Institutional Review Board (IRB #200638) with a waiver of informed consent. Data was retrospectively obtained from a prospectively maintained database of all patients who underwent CRS-HIPEC from 2011-2023 at our institution. Cases were excluded if they were repeat CRS-HIPEC procedures performed for the same patient. This database included patients with appendiceal, colorectal, gastric, ovarian, soft tissue sarcoma, and peritoneal mesothelioma tumors. For the analytic cohort, patients with peritoneal mesothelioma were excluded due to lack of validated tumor grading system for histologic subtypes other than epithelioid mesothelioma, as tumor-grade was included as a covariate in our multivariable models. Patients were divided into 2 groups: those who had non-mucinous vs mucinous tumors based on identification of mucin in the final surgical pathology report from CRS-HIPEC. Baseline patient demographics, medical history, and operative data were collected from the electronic medical record. Charlson comorbidity index was used as a composite score of patient’s medical comorbidities, predictive of 10 year survival. 7 Continuous variables are reported as median and quartiles, and categorical variables as number and percent.
Outcomes included overall survival (OS), cancer-specific survival (CSS) and progression free survival (PFS) from the date of CRS-HIPEC. Within the survival analyses, patients were censored at the time of outcome of interest or at loss to follow up. We also compared short-term postoperative outcomes between groups, including postoperative complications (severity classified by Clavien Dindo score 8 ), length of postoperative hospitalization, readmissions, and mortality within 30 days.
Categorical variables were compared between groups using the Chi-squared test, while continuous variables were compared using the Mann-Whitey U test for non-parametric data. A
Results
Patients and Operative Characteristics
Study Population Demographic and Operative Characteristics
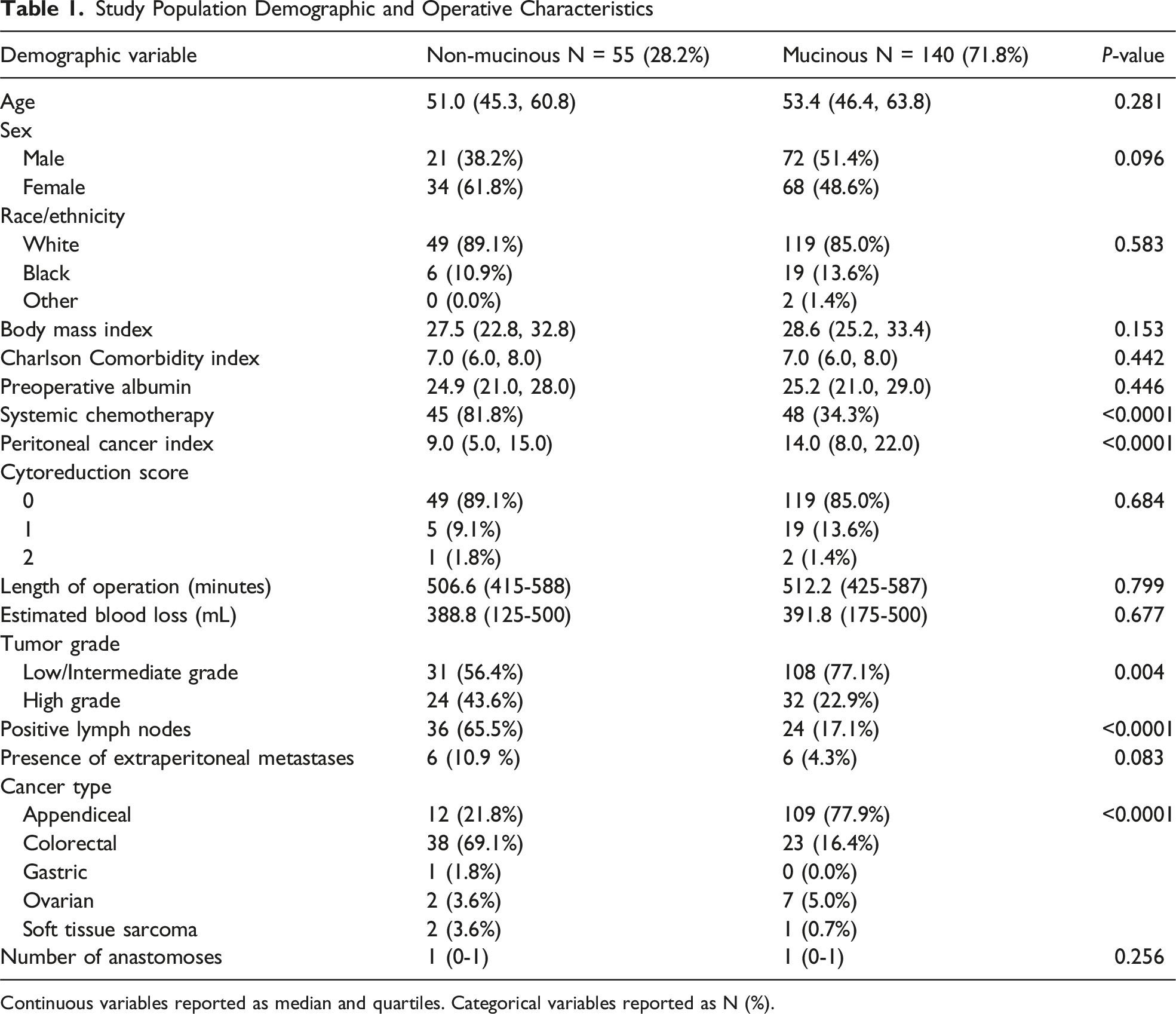
Continuous variables reported as median and quartiles. Categorical variables reported as N (%).
Postoperative Outcomes
Short Term Post-operative Outcomes
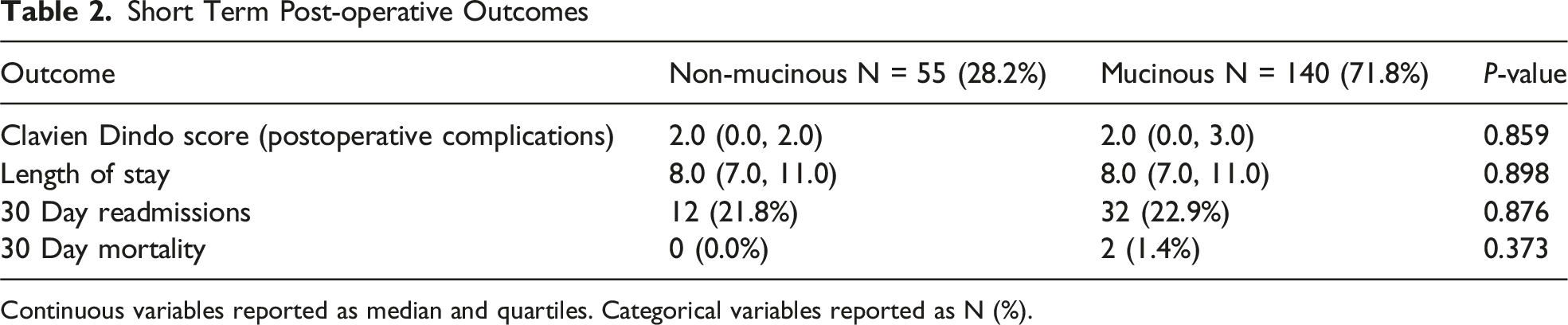
Continuous variables reported as median and quartiles. Categorical variables reported as N (%).
Survival Outcomes
Among all CRS-HIPEC patients, mortality at 5 years was 23.6% vs 11.4% ( Kaplan Meier Curves Comparing Mucinous and Non-Mucinous Tumors. Survival Outcomes including A) Overall Survival (OS), B) Cancer-Specific Survival (CSS) and C) Progression Free Survival (PFS) Over 150 months after Diagnosis are Demonstrated for Patients With Non-mucinous (Blue) and Mucinous Tumors (Red). On a Univariate Comparison With Log-Rank Tests, Patients With Non-mucinous Cancers Exhibit Poorer OS, CSS, and PFS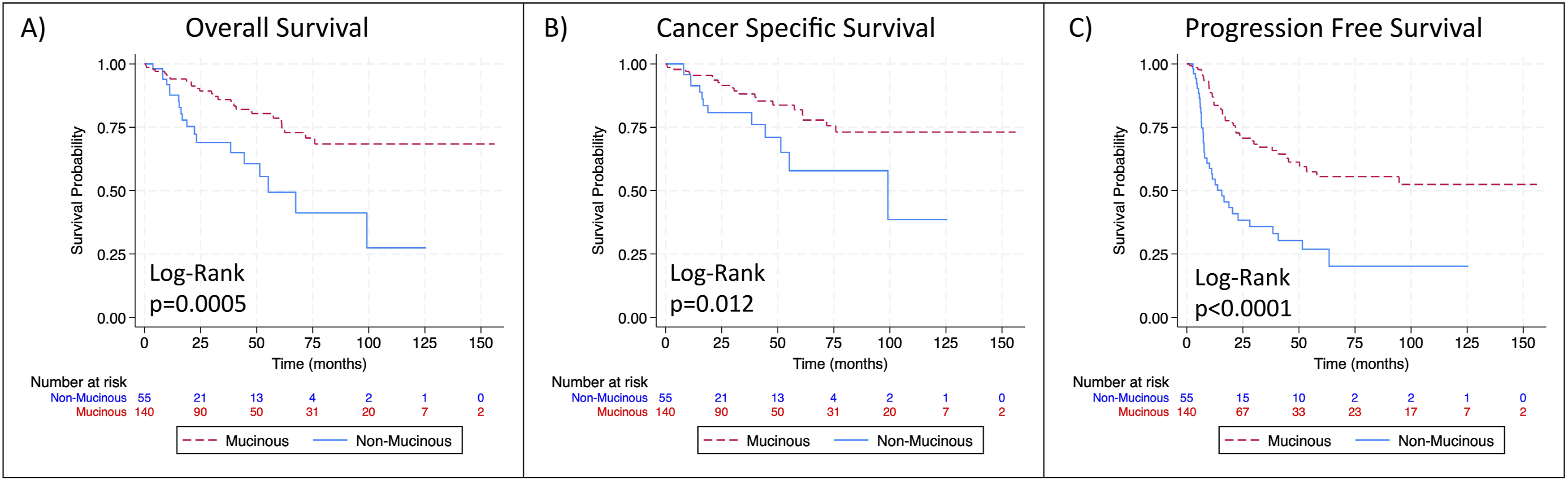
Multivariable Analysis of Factors Associated With Survival
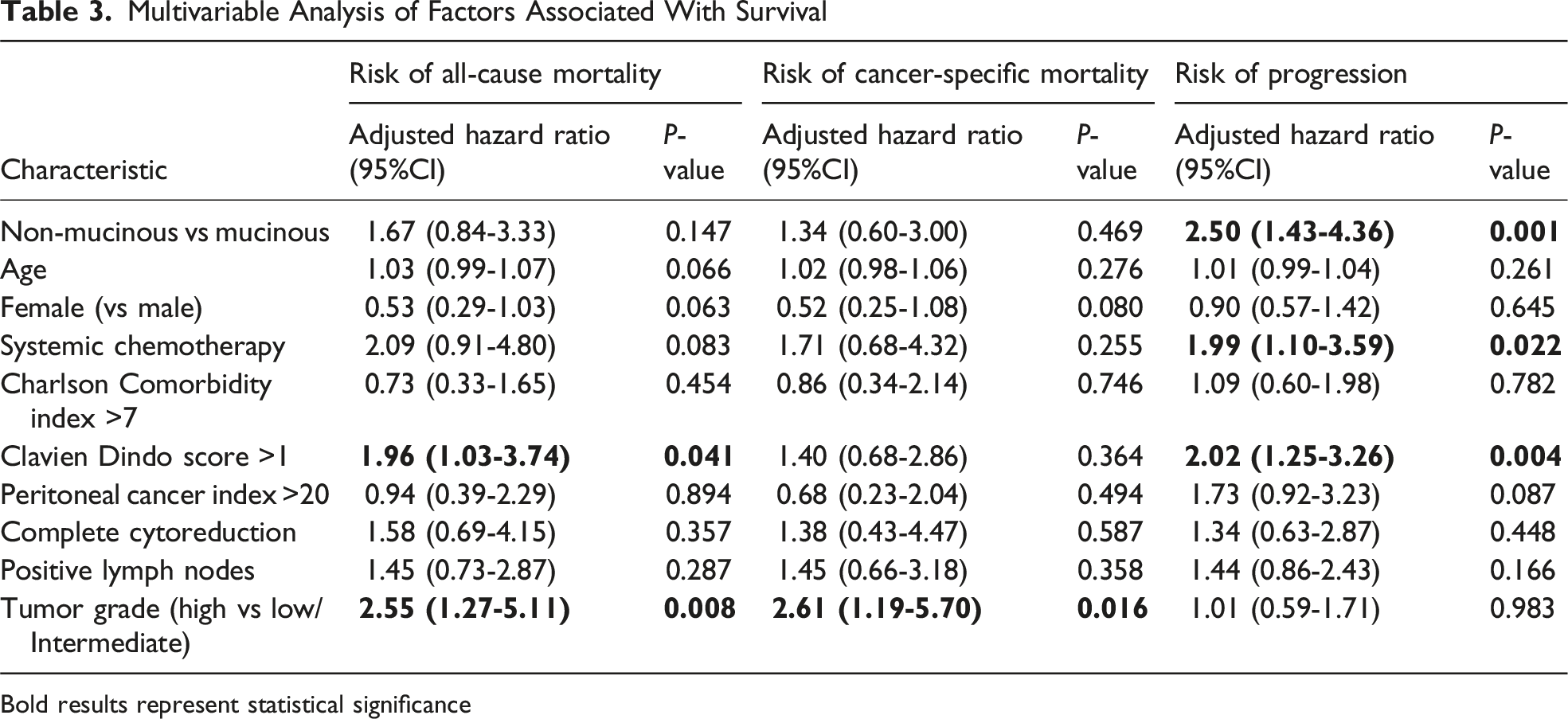
Bold results represent statistical significance
Subgroup Analyses
Out of 121 patients with appendiceal tumors, there were 12 patients with non-mucinous tumors, 74 with appendiceal mucinous neoplasms (AMN) (including 67 low-grade (LAMN) and 7 high-grade (HAMN)) and 35 with mucinous adenocarcinomas (MA). By subtype, 3-year mortality rates were 16.7% for CRS-HIPEC patients with non-mucinous tumors, 3.0% for those with LAMN, 28.6% for HAMN, and 11.4% for MA (
Among the 61 patients with colorectal cancer, there were 38 patients (62.3%) with non-mucinous tumors and 23 with mucinous tumors (37.7%). Three and 5 year mortality rates were similar between those with non-mucinous and mucinous colorectal cancers (3 y: 26.3% vs 34.8%,
Discussion
In this study, we found that patients with non-mucinous cancers who underwent CRS-HIPEC did not experience significantly different overall or cancer-free survival outcomes when compared to patients with mucinous tumors, after adjusting for relevant clinical and oncologic factors. Furthermore, there were no differences in short-term postoperative outcomes between groups. Although progression free survival was statistically poorer in patients with non-mucinous tumors undergoing CRS-HIPEC, this was predominantly driven by differences for patients with appendiceal cancers, not colorectal tumors. These findings suggest that while mucinous histology in appendiceal cancer may be more responsive to CRS-HIPEC from a progression standpoint, overall oncologic survival remains comparable for patients with non-mucinous cancers.
Our findings are consistent with prior studies demonstrating the prognostic importance of histology in appendiceal tumors. In a large SEER database study including 5655 appendiceal cancer patients, 5-year survival rates were lowest for patients with signet ring, colonic-type and mucinous adenocarcinoma, while those with goblet cell carcinoid and malignant carcinoid tumors had better outcomes. 9 When matched by stage, survival rates between patients with mucinous and non-mucinous appendiceal tumors have been shown to be similar, although this was without attention to treatment regimen. 10 Our findings align with a prior evidence suggesting poorer outcomes for those with non-mucinous appendiceal histologies, 11 as well as the well-established oncologic benefits of CRS-HIPEC for appendiceal mucinous neoplasms, particularly LAMNs.12,13 Given that nearly half of the patients in our mucinous histology group had LAMNs (48.5%), the superior PFS observed in this group is consistent with favorable outcomes reported following CRS-HIPEC for this subtype. Nevertheless, patients with non-mucinous appendiceal tumors continued to demonstrate significantly poorer PFS when compared to mucinous adenocarcinomas.
Among the colorectal cancer subgroup undergoing CRS-HIPEC, there were no significant differences in OS or PFS between those with mucinous vs non-mucinous cancers. Associations between histologic subtype in colorectal cancer and prognosis have been varied in the literature. Although mucinous colorectal cancers tend to present with more advanced disease in various retrospective studies, a large meta-analysis demonstrated that the prevalence of stage IV disease was similar for patients with mucinous and non-mucinous cancers.4,6,14 Among stage I-III colorectal cancer patients, mucinous histology has been associated with poorer prognosis. 5 For patients with stage IV disease some studies have found no differences in OS by histologic subtype, while others have demonstrated worse oncologic outcomes and treatment responses for those with mucinous histologies.6,14,15 Our findings are consistent with a retrospective analysis from Australia, 16 demonstrating similar survival outcomes for patients with mucinous vs non-mucinous colorectal tumors after CRS-HIPEC, suggesting that this treatment may offer similar oncologic benefit for both subtypes of colorectal cancers with peritoneal spread.
Data on the impact of histologic subtype on outcomes after CRS-HIPEC for other tumors, such as ovarian and gastric cancer, is limited. Advanced-stage mucinous ovarian cancers with peritoneal spread are associated with worse prognosis compared to serous cancers of the same stage. 17 Mucinous gastric carcinoma, while rare, also tends to present at more advanced stages, although survival differences in general between patients with mucinous and non-mucinous gastric cancers are less consistent across studies.18,19 To the best of our knowledge, histologic subtype has not been extensively studied in the context of CRS-HIPEC for these malignancies, representing an area for future investigation.
Limitations of this study include its retrospective, single institution nature which may affect generalizability. Sample size (particularly for less common histologies such as HAMN, and cancer types other than appendiceal/colorectal) may limit power in detecting differences between groups. Although reflective of the rarity of certain cancer subtypes in general, it restricts direct comparisons. Additionally, heterogeneity across different primary tumor types and within histologic categories—such as the presence of signet ring cells or neuroendocrine differentiation—may confound prognostic interpretation. While the vast majority of patients (98.5%) received mitomycin-based HIPEC according to the same protocol, unmeasured variability in types of neoadjuvant and adjuvant therapies may also influence outcomes. We did control for the use of systemic chemotherapy in general within our multivariable models to account for these differences. Future areas of investigation include exploring specific patient and tumor characteristics that predict responsiveness to CRS-HIPEC among non-mucinous cancers.
In conclusion, despite differences in PFS primarily seen in appendiceal cancer patients, overall and cancer-specific survival following CRS-HIPEC did not significantly differ across histologic subtypes after adjustment for key clinical and oncologic factors. This suggests that CRS-HIPEC may offer a meaningful and comparable survival benefit even for patients with non-mucinous cancers with peritoneal spread. Prior studies have provided evidence to support benefits with CRS-HIPEC compared to other treatments for patients with malignancies with peritoneal dissemination—including in appendiceal, colorectal, ovarian, gastric, and mesothelioma cancers. 20 Although further investigation is needed to clarify histology-specific outcomes, the comparable OS and CSS seen in our cohort supports the continued use of CRS-HIPEC across histologic subtypes. Based on these findings, non-mucinous histology should not be considered a contraindication to CRS-HIPEC. In all, these findings are valuable in contributing to the larger data pool regarding differential patient responsiveness to CRS-HIPEC, contributing to treatment decision-making, risk stratification, and underscoring the need for further research into optimizing outcomes after CRS-HIPEC for different cancer histologies.
Supplemental Material
Supplemental Material - Adaptive and Maladaptive Regulation of Positive and Negative Affect: Specific Comparison of Outcomes after Cytoreductive Surgery with Hyperthermic Intraperitoneal Chemotherapy Among Patients with Non-Mucinous vs Mucinous Tumors
Supplemental Material for Comparison of Outcomes after Cytoreductive Surgery with Hyperthermic Intraperitoneal Chemotherapy Among Patients with Non-Mucinous vs Mucinous Tumors by Emma M. Bradley, Julia Brickey, Marissa Kuo, Jean Mok, Emily Breeding, and Deepa Magge in The American Surgeon™
Footnotes
Ethical Considerations
This study was approved by our Institutional Review Board (IRB #200638) with a waiver of informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The lead PI of this study works as a consultant for GE Healthcare for a new PET scan under development for peritoneal disease. The corresponding author is supported by the Veterans Affairs Quality Scholars Fellowship. The authors have no additional conflicts of interest to disclose regarding this work.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
