Abstract
Burn injuries over previously grafted tissue present a formidable challenge for excision and debridement, particularly when there are critical underlying structures such as bowel. Enzymatic debridement with the recently approved anacaulase-bcdb, a bromelain-based enzymatic debridement gel (Nexobrid®), presents an additional method of burn excision that may be useful in such a situation. This brief report presents the management of a complex third-degree burn over a remotely skin-grafted bowel mass using anacaulase-bcdb gel. This report is written with documented patient consent and approval by the Human Research Protection Program office in compliance with institutional policy. A 52-year-old man presented to our level I burn center with a third-degree 3% total body surface area contact burn to a remotely skin-grafted bowel mass. The patient was admitted with the decision to proceed with anacaulase-bcdb debridement of his wound to minimize the risk of compromising his underlying bowel. The patient underwent the debridement without any sign of succus emanating from the wound. Post-debridement, he was transitioned to a negative pressure wound dressing and discharged home. He continued receiving wound care at clinic follow-ups and eventually underwent complex open ventral hernia repair. This brief report provides a safe alternative to operative excision of wounds with underlying critical structures.
Keywords
Key Takeaways
• Non-operative, enzymatic excision is a viable management strategy for a complex third-degree burn to a skin-grafted bowel mass. • Bromelain-based enzymatic debridement may be appropriate in excision of burn wounds with underlying visceral structures.
Prompt excision of burn eschar is a crucial step in the healing process of deep partial and full-thickness burn wounds. 1 Since the early 1970s, Dr Janzekovic’s technique of tangential excision to remove all eschar and damaged dermis by layers has become a standard of care. 2 However, surgical excision is associated with complications such as bleeding, heat loss, the removal of viable tissue with the eschar, and the need for operating room (OR) availability and trained surgeons. The removal of viable tissue in particular is an important risk to weigh in patients with burns overlying critical structures such as in the case of a previous skin graft over exposed bowel without intervening fascia. Although it is uncommon, such a complex case presents a challenge in surgical management of the burn wound as operative excision may result in the development of an enterocutaneous fistula. An alternative approach with enzymatic debridement may be a potential solution. Since its approval by the US Food and Drug Administration (FDA) in 2022, 3 burn surgeons have sought to delineate the appropriate role of the recently approved anacaulase-bcdb, a bromelain-based enzymatic debridement gel (Nexobrid®), in clinical practice. Here, we present the management of a complex third-degree burn to a remotely skin-grafted bowel mass using anacaulase-bcdb gel. This report is written with documented patient consent and approval to be exempt from the internal research board review by the Human Research Protection Program office in compliance with institutional policy.
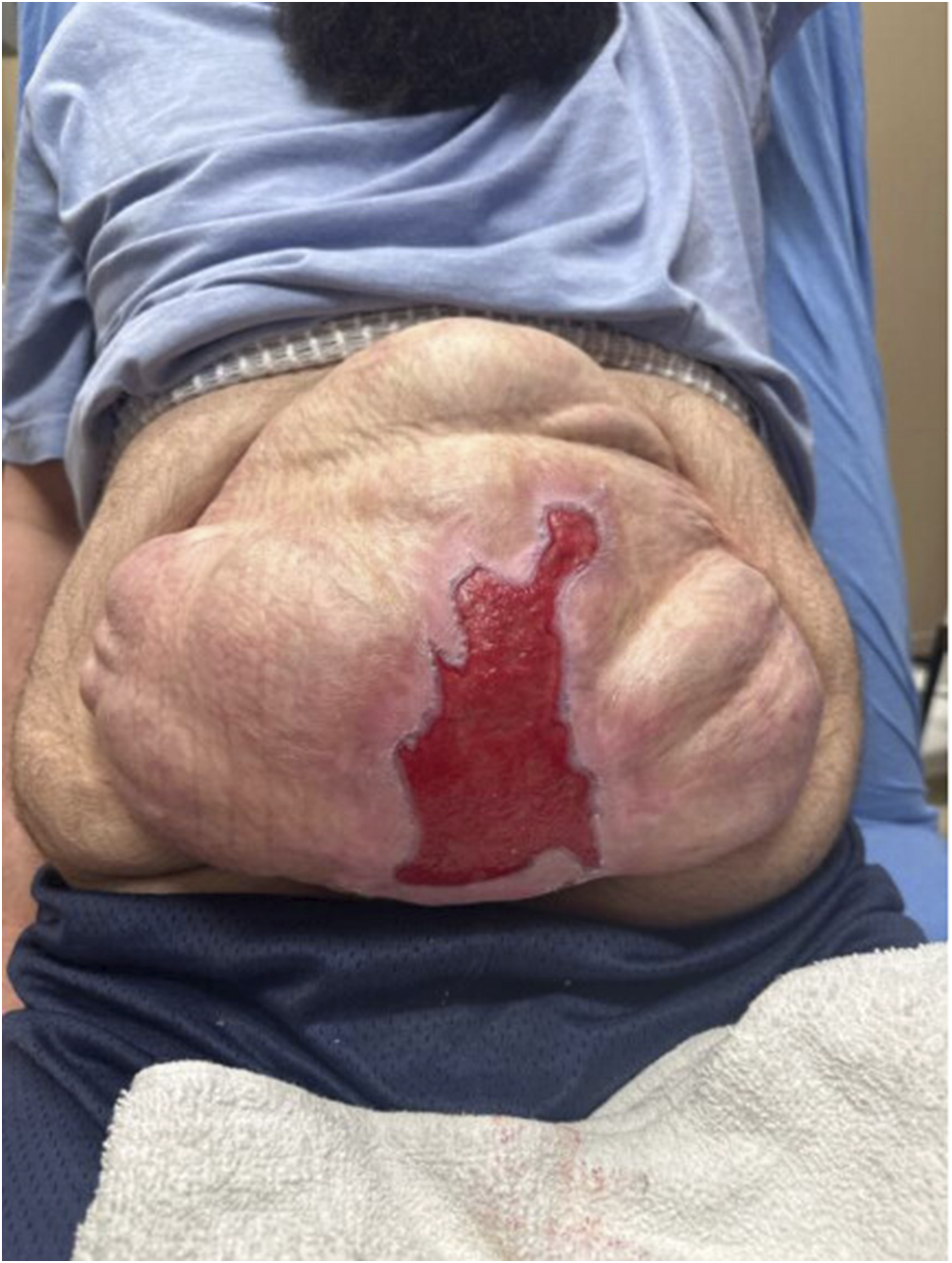
A 52-year-old man presented to our Level I Burn Center in February of 2024 with a 2-day history of a 3% total body surface area (TBSA) full-thickness contact burn to a skin-grafted bowel mass. The skin-grafted bowel mass was a result of a traumatic injury to the abdomen that occurred in 2020 treated with a laparotomy that resulted in a large ventral hernia and loss of abdominal domain. Other past medical history was notable for essential hypertension, type II diabetes mellitus (A1C 6.9), and hyperlipidemia. Reportedly, the patient was cooking on a stove and rested his insensate bowel mass on the stove to relieve discomfort. This resulted in a contact burn to the previously skin-grafted area. He presented in a delayed fashion due to reluctance to return to the hospital given previous history of long admission. Axial CT of Abdomen With Ventral Hernia and Subcutaneous Inflammatory Changes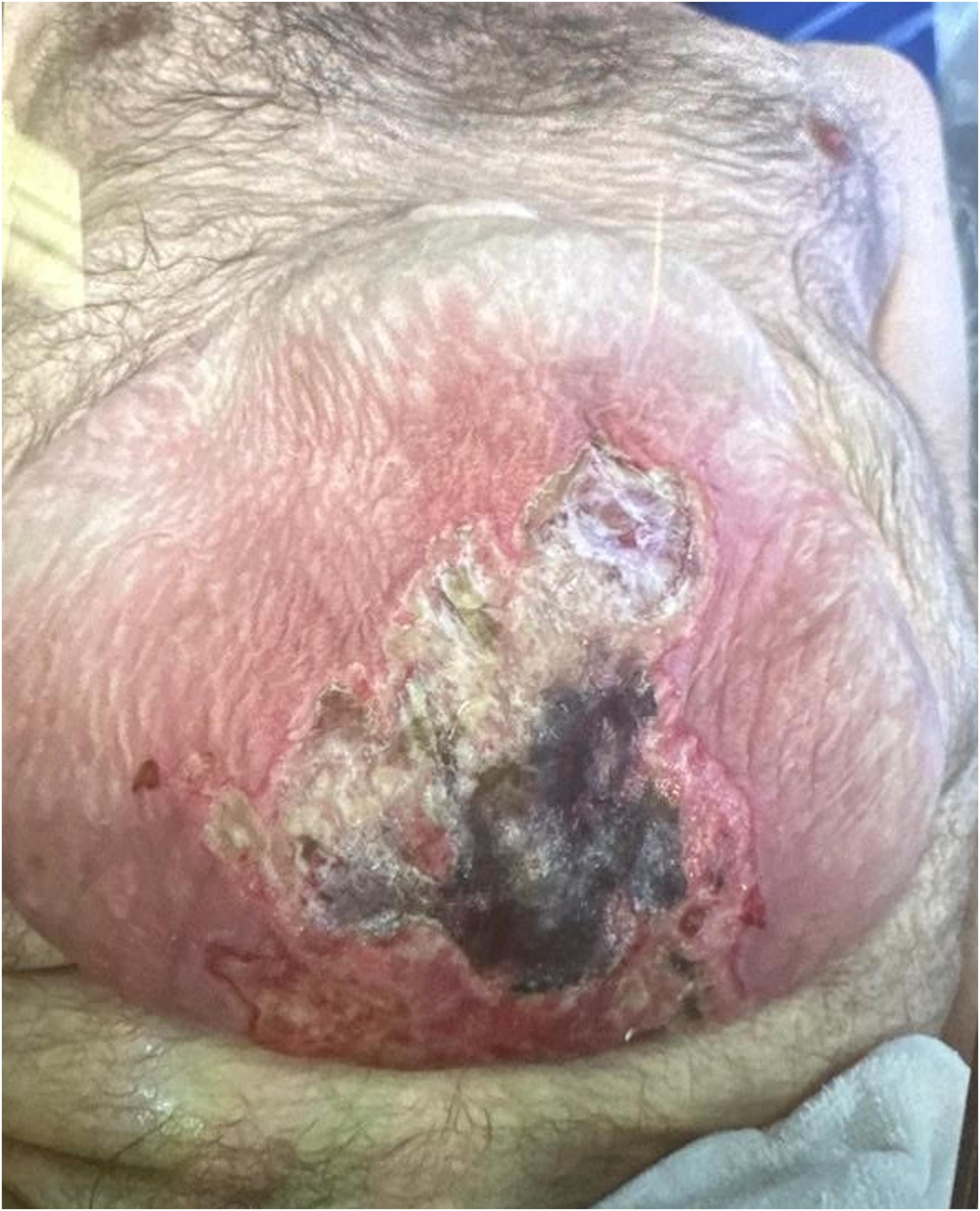
On arrival, the patient was hemodynamically stable and afebrile. Physical exam revealed erythema due to suspected cellulitis surrounding the third-degree burn (Image 1). Labs were unremarkable except for leukocytosis at 28 × 103 cells/uL. A computed tomography (CT) scan of the abdomen was performed which did not show any visceral damage to the bowel although subcutaneous inflammatory changes and focal fluid was present in the cutaneous tissue overlying loops of bowel (Image 2). The patient was admitted to the burn intermediate care unit and cefazolin was initiated secondary to cellulitis surrounding his burn wound. Initially, the wound was managed with daily bacitracin and Xeroform before transitioning to silver sulfadiazine (Silvadene®) cream on hospital day 2. After much discussion regarding the risk that surgical excision would pose to his closely underlying bowel, the decision was made to proceed with enzymatic debridement on hospital day 5. The delay in anacaulase-bcdb gel application was to accommodate for 48 hours of hypochlorous acid solution (Vashe®) washes to ensure no silver sulfadiazine remained in the wound bed due to its inhibitory effects on enzymatic debridement. Pre-debridement (Hospital Day 3)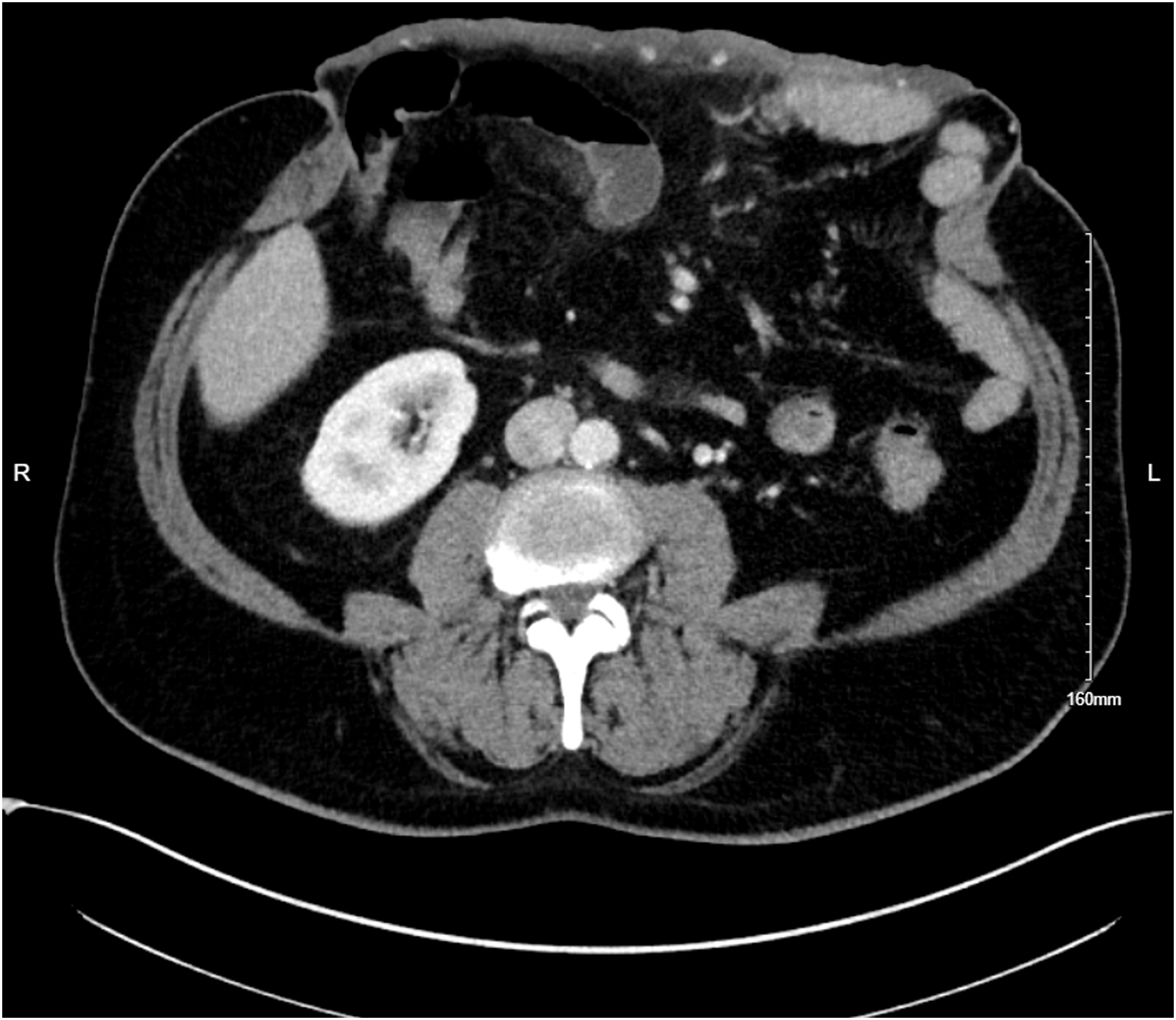
For the anacaulase-bcdb application, the patient underwent sedation with boluses of fentanyl (100 mcg total) and propofol (80 mg total) and local analgesia with lidocaine injection (10 mg total). Patient-controlled fentanyl analgesia was available to the patient for pain control during the enzyme application, the 4-hour debridement process, and enzyme removal procedures. The burn wound was cleaned and an antibacterial solution was applied 2 hours prior to the enzymatic debridement agent. Once in the wound care tank, the wound was cleaned again to ensure no residual silver sulfadiazine remained. A petrolatum skin barrier was applied around with the outside of the burn wound bed. The 5 g lyophilized anacaulase-bcdb gel was mixed with the 50 g gel vehicle according to the manufacturer’s instruction. Once mixed, a 3 millimeter thick layer of the gel was immediately applied to the burn wound. A sterile occlusive dressing was applied and then a layer of loose fluff gauze was applied and secured for the 4-hour dwell time. The dressings were taken down in an aseptic manner, removing the protective ointment and any dissolved eschar with sterile saline soaked gauze. An antibacterial soak solution was applied for an additional 2 hours. The patient was monitored closely throughout the 4-hour application. There was no sign of succus emanating from the wound. Physical exam showed that dermis in most areas of the wound after debridement appeared viable (Image 3). Post-debridement Day 0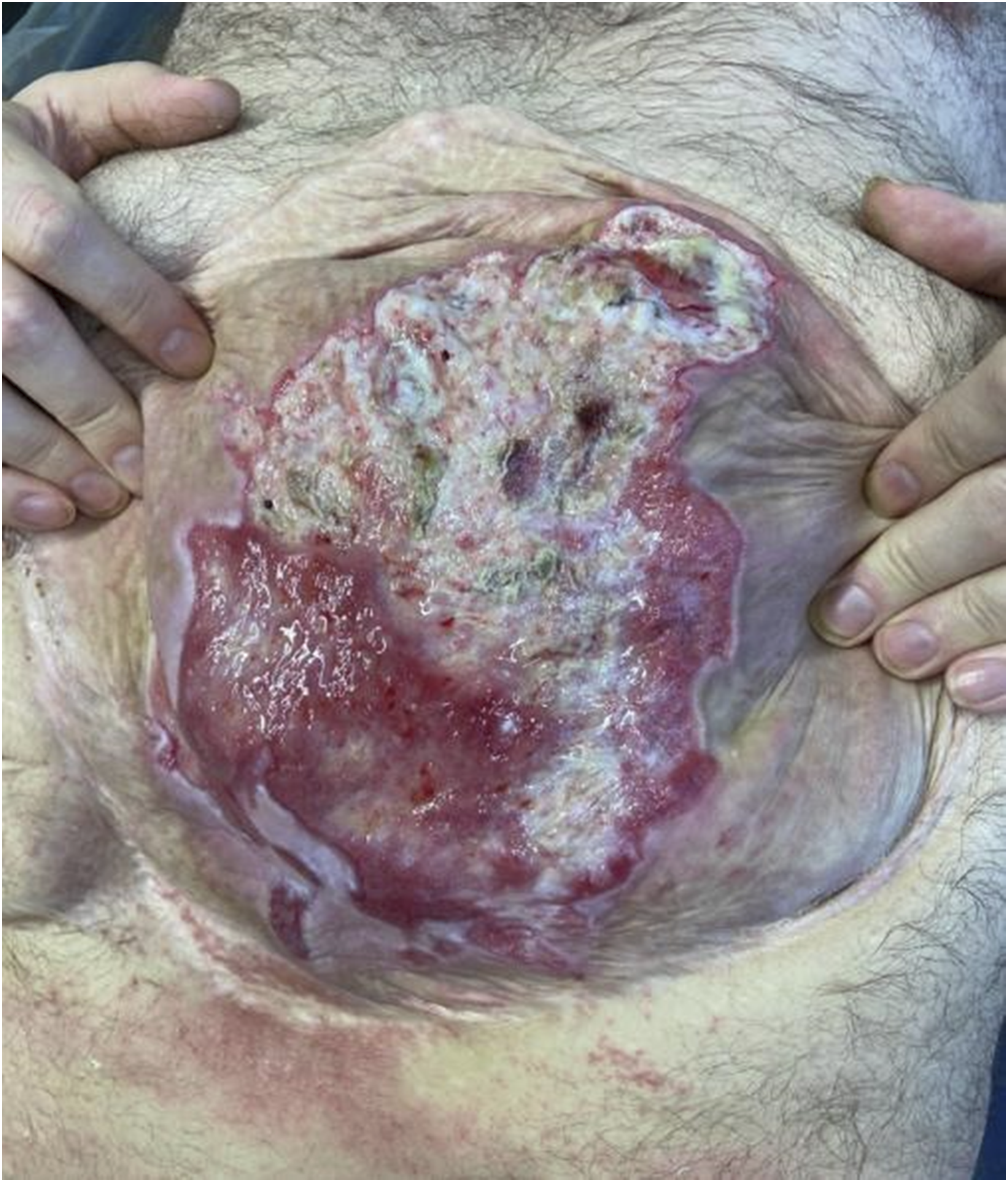
Post-debridement, the patient was initially managed with hypochlorous acid solution applied to the wound every 4 hours and dressed with non-adherent gauze. During this time, the patient also completed his 7-day course of cefazolin on post-debridement day 1. He was transitioned to a negative pressure wound dressing on post-debridement day 2 (Image 4), which was fashioned with non-adherent gauze (Adaptic®) and a black sponge and set to −125 mmHg suction. The negative pressure wound dressing was changed inpatient on post-debridement day 5. He was discharged home on post-debridement day 5 with a plan for definitive outpatient split thickness skin grafting. The patient has since had 5 months of follow-up with our burn clinic. He continued the negative pressure wound dressing for 1 week outpatient before transitioning to topical wound care with bacitracin and Xeroform for an additional week. The patient declined skin grafting and continued with wound care with an antibacterial foam dressing containing methylene blue and gentian violet (Hydrofera Blue READY®) changed every 2 to 3 days. During his last follow-up with our burn clinic in July of 2024, the wound remained open but had decreased in size (Image 5). The patient was evaluated by the general surgery team and deemed a suitable candidate for complex open ventral hernia repair which he underwent 5 months post-burn with excision of the remaining burn wound. First Negative Pressure Wound Dressing Change (Post-Debridement Day 2) Wound at Last Follow-Up (5 Months Post-burn)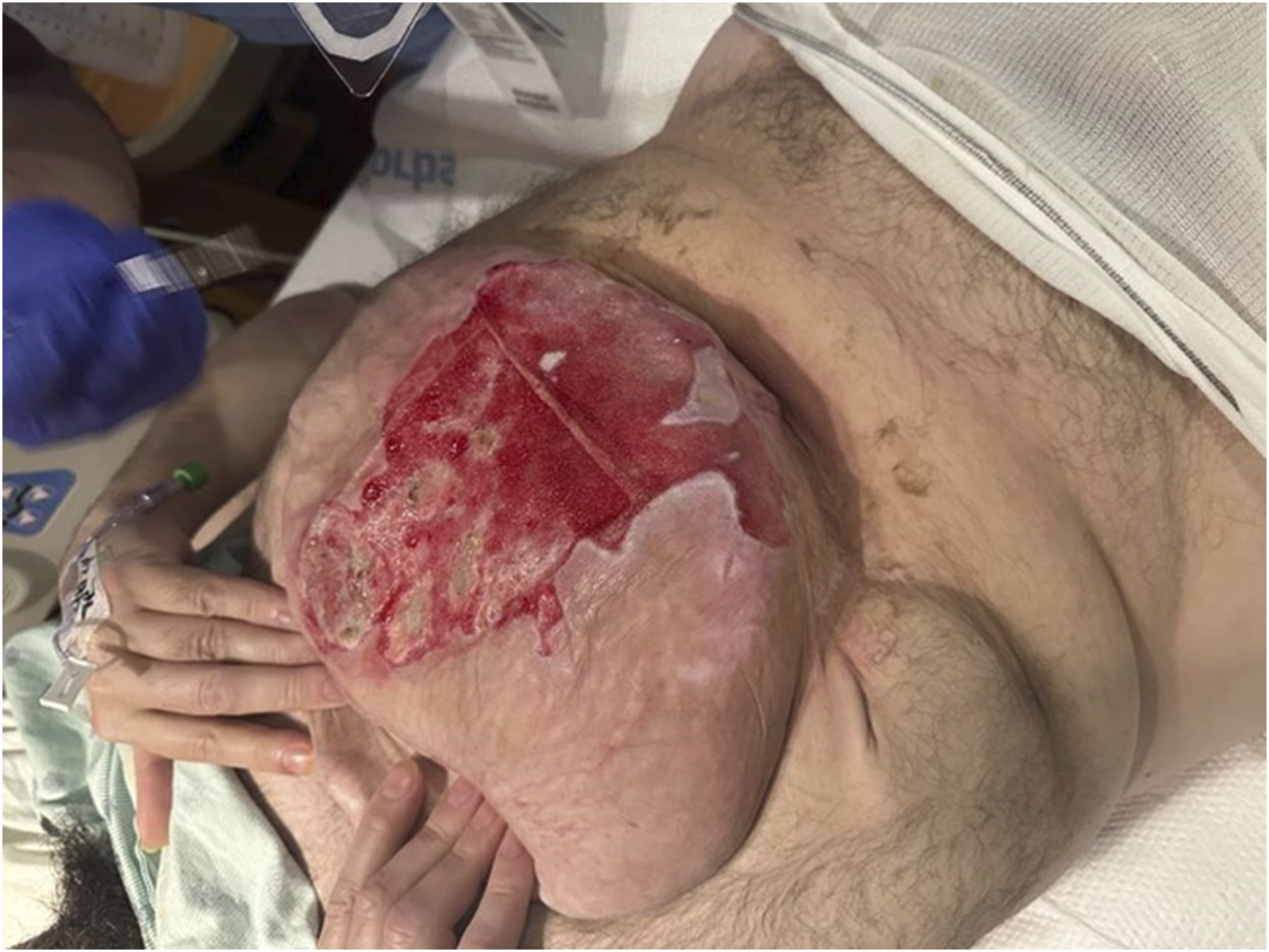
Although rare, third-degree burns over a previously skin-grafted bowel mass present a unique challenge when considering surgical excision. In addition to the association of sharp excision with complications such as bleeding, heat loss, excision of healthy tissue, and need for operating room (OR) resources, 4 there is also the added risk of formation of an enterocutaneous or enteroatmospheric fistula due to the proximity of the wound to the underlying bowel. In this case, enzymatic debridement was presented as an alternative approach to operative excision with an excellent outcome for the patient. This allowed the patient to bridge to a definitive ventral hernia repair.
Current literature, including multiple randomized clinical trials, support the use of enzymatic debridement for faster debridement times, reduced need for operations, reduced blood loss, and reduced need for autografting when compared to the standard of care when used for appropriate wounds.5-9 Most studies have focused on the utilization of enzymatic debridement in burns isolated to a single extremity that may be operatively prohibitive.10-12 However, there has also been some evidence in the efficacy of off-label use of enzymatic debridement such as in burns of the face and genitalia, especially in patients who are otherwise not reasonable operative candidates.4,5 Our brief report offers an example of the potential utilization of anacaulase-bcdb gel debridement in third-degree burns that directly overlay critical structures and to prevent the removal of essential, healthy tissue. Given that this patient did not require operative excision, it also demonstrates that successful treatment with NexoBrid® may reduce the total number of surgical procedures for excision and grafting.
It is important to note that this procedure was performed at our facility which has surgeons and staff that have been trained in, and are experienced in, the use of anacaulase-bcdb gel. One of the difficulties of utilizing an enzymatic debridement agent is the learning curve for mastering the application technique and the assessment of the post-debridement wound bed.5,13 In particular, pain control during the administration is important to the success of the procedure and the lack of an appropriate pain management protocol has been a barrier to the widespread feasibility of this procedure.5,14 Different anesthesia techniques have shown to be effective during enzymatic treatment, including conscious sedation such as was used in this case. 4 Other logistical alterations to consider include the avoidance of iodine and silver sulfadiazine products or, in the case of this patient, including the appropriate wash out period if such products have been used. Potential erosion of anacaulase-bcdb into nearby critical structures such as vessels or visceral organs was also considered, and the patient was monitored closely during the application process for the erosion of anacaulase-bcdb into the bowel.
There are no similar cases of burn management of a skin-grafted bowel mass in the literature. This case demonstrates that non-operative, bromelain-based enzymatic excision is a viable management tool for a complex third-degree burn to a skin-grafted bowel mass.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
