Abstract
Purpose
Severe obesity is a barrier to listing for kidney transplantation due to concern for poor outcomes. This study aims to compare bariatric surgery with medical weight loss as a means of achieving weight loss and subsequent listing for renal transplant. We hypothesize that bariatric surgery will induce greater frequency of listing for transplant within 18 months of study initiation.
Materials and Methods
We performed a randomized study of metabolic bariatric surgery (MBS) vs medical weight loss (MM) in patients on dialysis with a body mass index (BMI) of 40-55 kg/m2. The primary outcome was suitability for renal transplant within 18 months of initiating treatment. Secondary outcomes included weight loss, mortality, and complications.
Results
Twenty patients enrolled, only 9 (5 MBS, 4 MM) received treatment. Treated groups did not differ in age, gender, or race (P ≥ .44). There was no statistically significant difference in the primary endpoint: 2 MBS (40%) and 1 MM (25%) listed for transplant ≤18 months (P = 1.00). With additional time, 100% MBS and 25% MM patients achieved listing status (P = .048); 100% of MBS and 0 MM received kidney transplants to date (P = .008). Weight, weight loss, and BMI trajectories differed between the groups (P ≤ .002). One death from COVID-19 occurred in the MM group, and 1 MBS patient had a myocardial infarction 3.75 years after baseline evaluation.
Conclusion
These results suggest MBS is superior to MM in achieving weight loss prior to listing for kidney transplantation. Larger studies are needed to ensure the safety profile is acceptable in patients with ESRD undergoing bariatric surgery.
Key Points
• In patients with ESRD, bariatric surgery achieves greater weight loss than medical management alone. • In this small randomized trial, bariatric surgery appears to lead to greater frequency of listing for transplant in patients with ESRD and severe obesity. • Bariatric surgery appears well tolerated in patients with ESRD, but larger studies are needed to fully evaluate the risk-to-benefit profile.
Introduction
Severe obesity (Body Mass Index (BMI) > 40 kg/m2) remains a major barrier for patients with end stage renal disease (ESRD) to become listed for kidney transplant. Most centers exclude patients with severe obesity from transplant. 1 Several societies specifically recommend patients achieve a BMI <30 kg/m2 before being considered for renal transplant. 2 With over 25% of patients with ESRD experiencing concomitant obesity, therapies to achieve weight loss are needed to improve access to transplantation in this population. 3 This is particularly critical given the far superior outcomes with renal transplant vs dialysis in the ESRD population. Prior studies have found medical weight loss leads to active transplant listing in only a quarter of patients with severe obesity. 4 This likely reflects the limited efficacy of current medical weight loss therapies in achieving significant, durable weight loss.
While bariatric surgery has been shown to be safe and effective in a broad population of patients with severe obesity, whether bariatric surgery should be recommended in ESRD patients remains undetermined. It is possible that bariatric surgery achieves significant and durable weight loss in patients with concurrent severe obesity and ESRD, allowing for transplant listing. On the other hand, surgical risk and the need for strict dietary compliance may subject this population to poor outcomes after bariatric surgery.
To date, data evaluating the use of bariatric surgery to achieve weight loss and transplant listing are limited to retrospective case-control studies or case series, with the exception of one prospective single arm study.5-7 Importantly, no study to date has prospectively compared bariatric surgery to medical weight loss with regard to their achieving weight loss with subsequent listing for kidney transplantation. Thus, the true safety and benefit of bariatric surgery in a population of ESRD patients seeking weight loss for successful transplant listing is poorly defined. The aim of this study is to compare the rate of successful kidney transplant listing between patients receiving medical weight loss therapy and those receiving bariatric surgery. We hypothesize that patients undergoing bariatric surgery will be more likely to achieve listing for kidney transplantation compared to those managed with medical weight loss.
Methods
Study Design
This is a prospective, randomized, non-blinded, single center study enrolling patients with class III obesity and concurrent ESRD requiring hemodialysis. This study was approved by the Institutional Review Board and was listed on clinicaltrials.gov.
Study participants
Patients were enrolled if they had a BMI of 40-55 kg/m2, with age ≤62 years, and had insurance coverage for bariatric surgery. Patients were excluded if they had any of the following: cirrhosis, prior foregut or bariatric surgery, inability to walk 200 feet, significant heart or lung disease, HIV, Crohn’s disease, psychiatric disorder, alcohol or drug addiction, active tobacco use, non-compliance with dialysis regimen, previous renal transplant, non-English speaking, or currently receiving peritoneal dialysis. Patients were followed from date of enrollment (beginning August 2014) to November 2022.
Randomization and interventions
Participants underwent an initial screening visit which included a visit with a transplant nephrologist and member of the weight loss clinic team. Patients were enrolled after completion of the screening visit and were then randomized 2:1 to metabolic bariatric surgery (MBS) with laparoscopic Roux-en-Y gastric bypass or medical management (MM). Given an intended enrollment of 75 consecutive participants, an allocation sequence that was not known in advance to investigators or study personnel was developed using a restricted randomization for unbalanced allocation approach. A participant’s treatment condition was conveyed to study personnel shortly after consent was obtained, and the patient was subsequently notified. Patients with MM received standard of care at the medical weight loss center. The baseline study visit occurred after the screening visit, prior to, and in close proximity to initiation of treatment.
Laparoscopic Roux-en-Y Gastric Bypass was carried out by a single surgeon to achieve a standardized surgical approach. Briefly, surgery was conducted with the use of 6 laparoscopic port sites in the same configuration. A 50 mL volume gastric pouch and Roux-en-Y anatomy was achieved with a 75 cm Roux limb and 50 cm BP limb. Both mesenteric defects were closed in all cases. Patients were initiated on a bariatric clear liquid diet evening of surgery and advanced along a standardized bariatric diet protocol in the ensuing weeks under the guidance of bariatric-specific dietitians.
Medical weight management was conducted per the VUMC Medical Weight Loss Clinic and at the discretion of the obesity medicine physician and consisted of routine visits with the physician and dietitians. Patients first undergo a standard of care visit which includes an initial evaluation with physician or nurse practitioner. Following this, patients attend clinic visits every 4-6 weeks with a physician or nurse practitioner, dietician, psychologist (as needed), or exercise physiologist per standard of care for the medical weight loss program, and weights were obtained at each visit. Use of anti-obesity medications in the MM group were at the discretion of the obesity medicine physician.
Suitability for transplantation was determined by the local Transplant review committee. No predetermined criteria were specified by the study protocol and study personnel were not involved with this determination. A review committee consists of a nephrologist, transplant surgeon, dietitian, social worker, and patient liaison. A BMI >40 is considered one of several absolute contraindications to renal transplant.
Outcomes
The primary outcome was successful listing for renal transplant within 18 months of surgery or initiating MM (BMI of 40-55 kg/m2). Secondary outcomes were total body weight loss (TBWL) defined as percent weight change from baseline, BMI, their trajectories, transplantation rates, morbidity, including metabolic or electrolyte abnormalities, wound infections, organ space infections, reoperations, reinterventions, and mortality.
Data Management and Statistical Analysis
Data were collected and managed using REDCap electronic data capture.8,9 Person-level data were analyzed using summary statistics, comparisons of proportions including Fisher’s exact test for 2 by 2 tables, and analysis of variance methods. Mixed effects models evaluated the longitudinal trajectories of BMI, weight, and percent change in weight (in relation to pre-treatment values), with a time by treatment group interaction effect testing whether trajectories differed between MBS and MM. Statistical power for the primary endpoint in the originally-intended treatment groups (50 MBS and 25 MM) was 81% to detect a 40-point rate difference (non-directional α = 0.01) and 84% to detect a 35-point rate difference (non-directional α = 0.05). Analyses were performed using IBM SPSS Statistics (Armonk, NY).
Results
Patient Characteristics
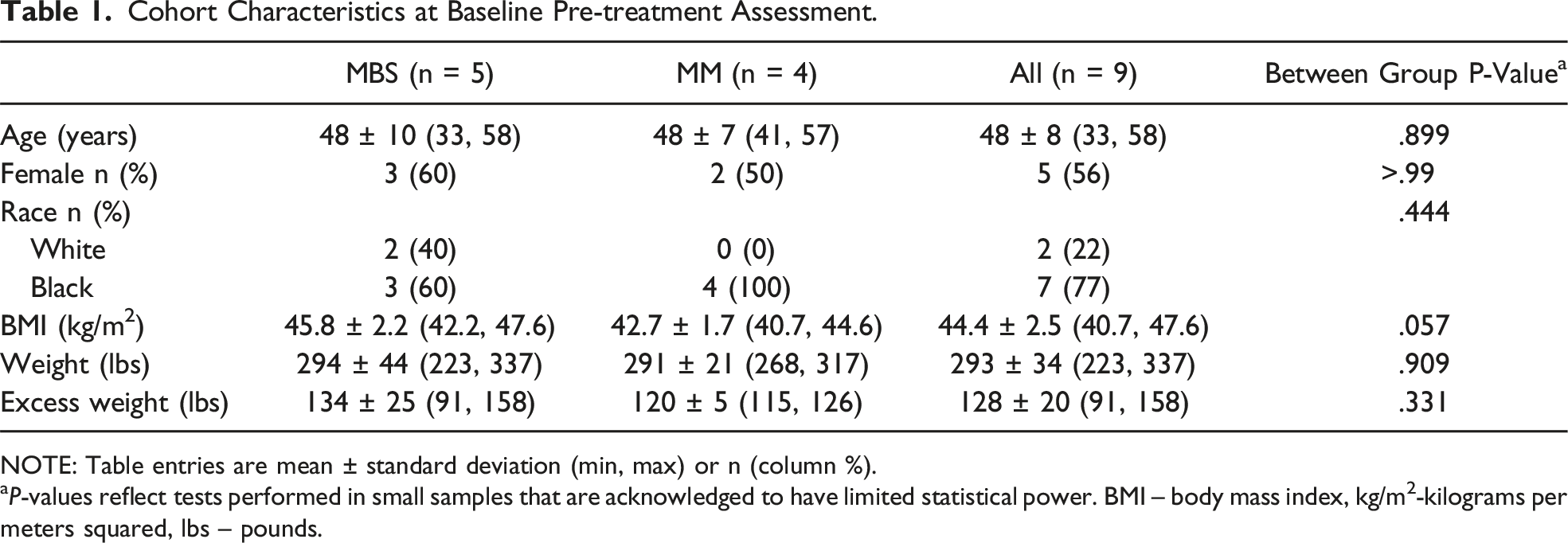
Twenty patients were enrolled (ie,, were deemed eligible, consented to participate, and were randomized (Figure 1). These 20 patients (12 MBS, 8 MM) comprised the intention to treat sample. Nine patients (5 MBS, 4 MM), who completed the comprehensive baseline study visit in the Clinical Research Center and subsequently initiated treatment, comprised the treated cohort that was followed longitudinally. Eleven patients in the intention to treat sample (7 MBS, 4 MM) did not complete the baseline study visit and did not receive treatment (Figure 1). There was no difference in age, gender, BMI, weight, or excess weight between patients randomized to MBS and MM in the intention to treat sample (all P > 0.148). Additionally, there were no differences in these measures between those who were treated (n = 9) and those who were not (n = 11, all P ≥ 0.10), and there were no significant randomization conditions by whether treated interaction effects (all interaction P ≥ 0.25, Supplemental Table 1). Characteristics of the treated cohort are summarized in Table 1. With the caveat that the comparisons are insufficiently powered due to the limited sample, no differences in baseline characteristics were observed between the MBS and MM groups in the treated cohort. All 4 participants in the MM group received dietary education for weight loss including recommendation for a higher protein diet (40-50 grams per day) with minimal starches and simple sugars as well as increased physical activity and strength training. Only one subject in the MM group received an obesity treatment medication (semaglutide 0.25-0.5 mg). Consort diagram. Cohort Characteristics at Baseline Pre-treatment Assessment. NOTE: Table entries are mean ± standard deviation (min, max) or n (column %). aP-values reflect tests performed in small samples that are acknowledged to have limited statistical power. BMI – body mass index, kg/m2-kilograms per meters squared, lbs – pounds.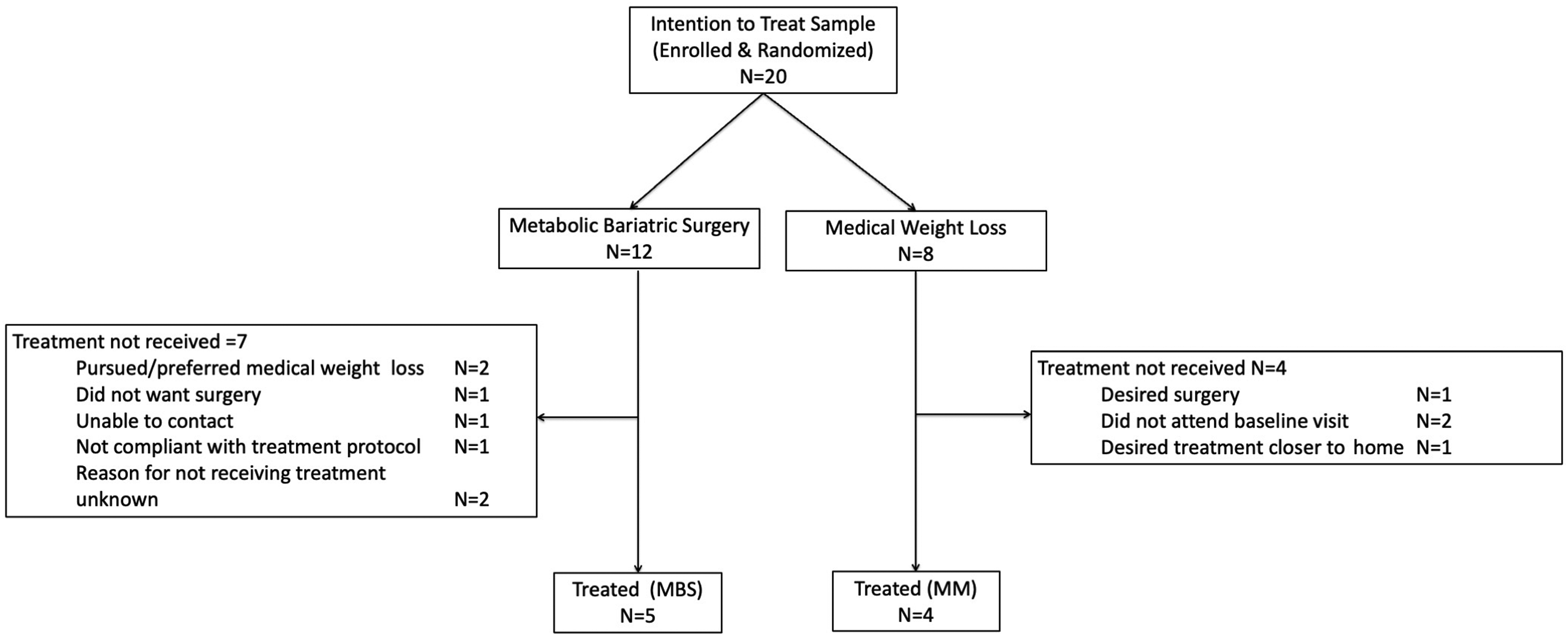
Listing for Kidney Transplantation and Transplantation
The primary endpoint analysis was not statistically significant in this limited sample, with 2 MBS (40%) and 1 (25%) MM patient being listed for kidney transplantation within 18 months (P > 0.99). Total surveillance time to whether the primary endpoint was achieved averaged 19 ± 9 (median = 22, min = 8, max = 30) months in the MBS and 60 ± 30 (median = 71, min = 16, max = 81) in the MM group (P = 0.021). At the most recent analysis, 5 (100%) of MBS and 1 MM patient (25%) were listed for transplant (P = 0.048). Partly due to the higher rate of listing among MBS patients, the proportion of persons transplanted among the entire treated cohort (regardless of whether listed) was significantly higher (5 of 5) in MBS patients compared to zero of four MM patients (P = 0.008).
Adverse Events
Two patients in the MBS group required emergency department care within the first 12 months following baseline visit: 1 for dehydration and syncope, and the other for a missed dialysis appointment necessitating semi-urgent hemodialysis. One death occurred in the MM group at 4.5 years due to complications from COVID-19. One MBS patient was treated for myocardial infarction 3.75 years after the baseline visit.
Weight Loss and the Trajectories of Weight, Weight Loss, and BMI
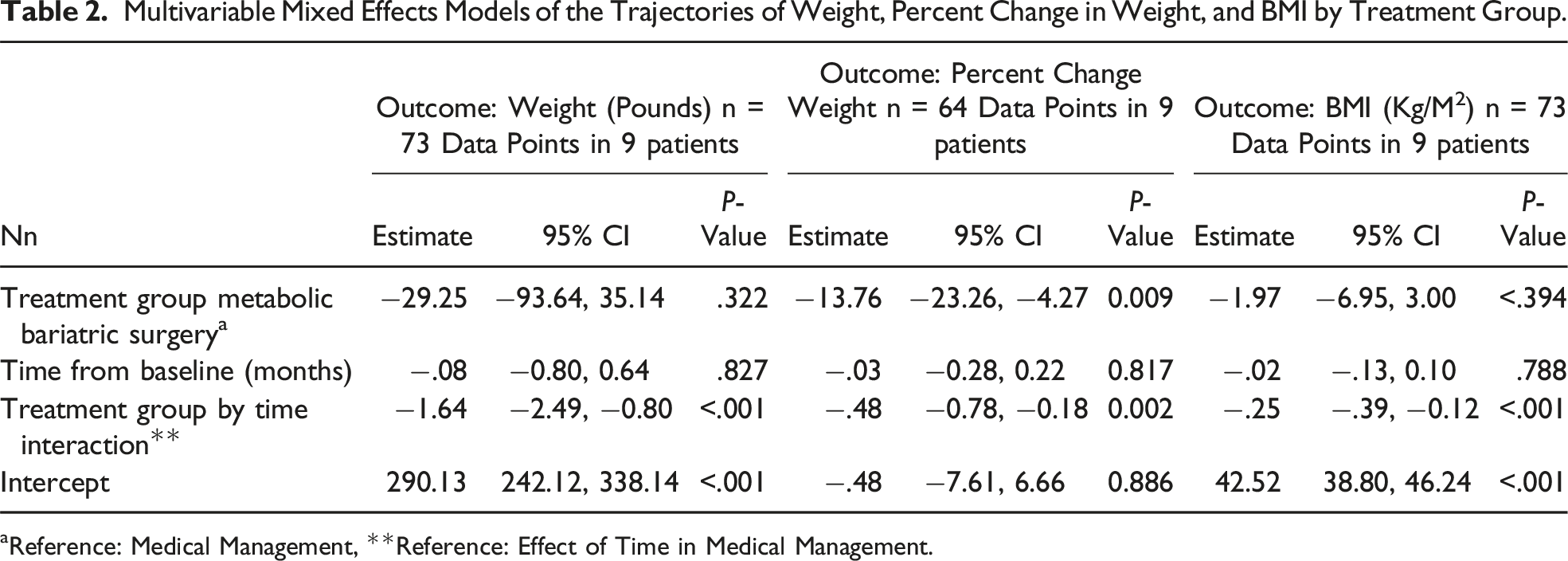
Patient follow-up through the most recent weight assessment data point averaged 45 ± 5 (median = 47, min = 37, max = 49) months in the MBS and 30 ± 8 (median = 28, min = 12, max = 50) months in the MM group (P = 0.075). Two persons in the MM group were considered ineligible to continue to participate in the study after post-baseline months 12 and 14 due to non-compliance with protocol visits. After adjusting for follow-up time, overall weight loss was greater, averaging 31 ± 13% in the MBS patients compared to 3 ± 7% in MM patients (P = 0.046). Longitudinal mixed effects models, that comprised all available data points, demonstrated that the trajectories of BMI (Figure 2(a)) and weight over pre- and post-treatment data points and of post-treatment percent change in weight (Figure 2(b)) differed significantly between MM and MBS (all time by group interaction effects P ≤ 0.002, Table 2). Specifically, these interaction effects demonstrated that the temporal trajectories of BMI, weight, and percent change in weight, were significantly more negative in the MBS group compared to those of the MM group. From an effect size perspective, the proportions of variance in BMI and percent change in weight that were associated with follow-up time were negligible (R2 ≤ 0.0007) in the MM group and large (R2 ≥ 0.399) in the MBS group (Figure 2). Analyses indicated that BMI declined to <40 kg/m2 by month 12 in MBS patients and remained >40 kg/m2 in MM patients through the most recent data point. Trajectories of (A) BMI over all observation points and (B) percent change in weight after treatment. Multivariable Mixed Effects Models of the Trajectories of Weight, Percent Change in Weight, and BMI by Treatment Group. aReference: Medical Management, **Reference: Effect of Time in Medical Management.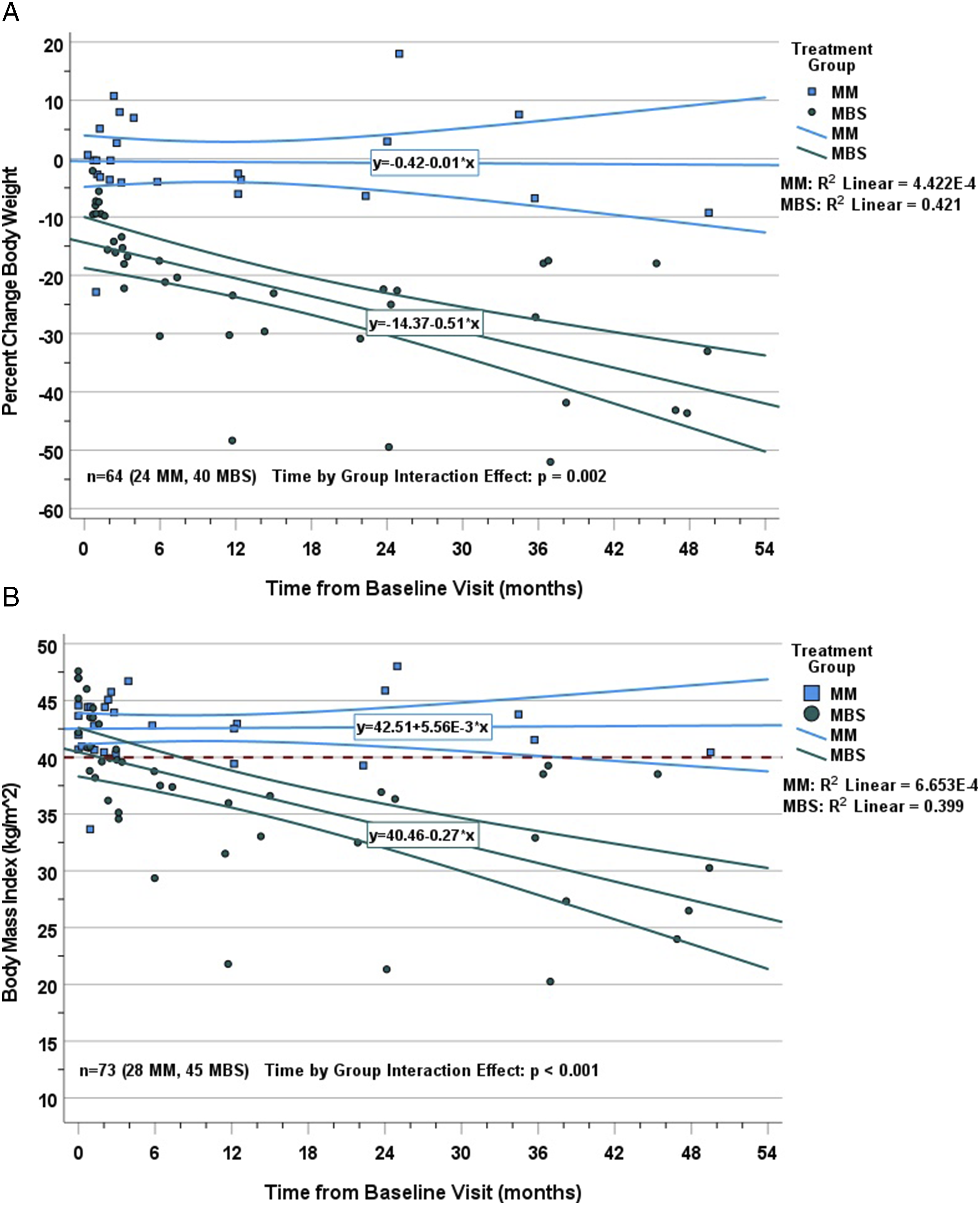
Discussion
In this post-hoc analysis of a small, randomized trial having limited participant enrollment, the primary endpoint of listing for kidney transplant within 18 months after therapy initiation was not different between the treatment groups. On the other hand, bariatric surgery was superior to medical therapy for achieving weight loss in patients receiving dialysis and ultimately listing for renal transplant over a more extended period. Analysis of the data is complicated by the high dropout rate prior to treatment initiation in the surgical group and after treatment initiation in the MM group. Bariatric surgery appeared safe in this small sample, consistent with other reports.7,10-12
Determining the optimal treatment for patients with obesity and ESRD is critical given the major individual health risks of both obesity and ESRD. Obesity is an impediment to listing for kidney transplant and is associated with a longer time to transplant.1,13 Medical weight loss allows listing in only 5 to 45% of patients.4,14,15 In contrast, bariatric surgery achieves adequate weight loss and subsequent listing for transplant. In the systematic review by Orandi et al 7 evaluating bariatric surgery in patients seeking kidney transplantation, of the 288 patients who underwent bariatric surgery, 50% ultimately achieved transplantation. In a recent retrospective review of 38 patients with ESRD undergoing bariatric surgery, Soliman et al 11 found 47% of patients achieved listing for transplant following surgery. As the only randomized trial to date, our results that 100% of MBS patients were able to be listed for transplant vs only 25% in the MM group argue strongly that MBS should be considered in appropriate patients.
Although the weight loss achieved by the MBS group is concordant with large systematic reviews of surgical weight loss, the MM group experienced weight loss below what has been found in prior studies of lifestyle and behavioral interventions. A meta-analysis of primary care-driven lifestyle weight loss interventions found patients experienced up to 9 kg of weight loss. 16 In contrast, a Cochrane review found that patients with ESRD achieved on average only 3.6 kg of weight loss with such lifestyle interventions, in line with what was found in our study. 17 These differences reflect the challenges with medically-managed weight loss in patients with ESRD, as interventions such as very low calorie diets and aerobic exercise are resource intensive and carry risks in these patients.18-20
Limited long-term data exist evaluating the impact on overall patient and graft survival in bariatric patients who subsequently undergo kidney transplantation. Short term follow-up studies suggest that bariatric surgery does not impact graft or patient survival. For example, in a matched case-control study, Kim et al 21 found no impact from sleeve gastrectomy on graft or patient survival. However, compared to bariatric patients with normal renal function, ESRD is associated with higher rates of complications and post-operative interventions. 22 One strength of this study is the extended duration of follow-up, with a median of 45 months (28 months in the MM and 47 months in the MBS), which exceeds the less than 2-year median follow-up in the systematic review by Orandi et al. Thus, while our study had a limited sample, there were no post-operative complications in the MBS group, suggesting acceptable outcomes at a high-volume bariatric center of excellence.
There are two possible explanations as to why our study failed to detect a difference between the treatment groups with respect to the primary endpoint. The first is that the two therapies are equivalent. The second is that our final treatment cohort of 5 MBS and 4 MM patients had insufficient statistical power to address that specific endpoint in light of the observed rate difference in this small sample. Given our additional data, including the long-term superiority of MBS and the longitudinal analyses of weight loss trajectories, we favor the second explanation. We did not meet our enrollment target of 75 patients, and our failure to detect a difference in the primary endpoint highlights the challenges with randomization in trials comparing surgical to non-surgical interventions.
This study faced challenges with identifying potential study subjects seeking weight loss, who were agnostic to whether this would be accomplished with a medical or surgical approach. As a result, relatively few patients who were screened consented to participate. Additionally, 11 of the 20 patients who consented and were randomized were ultimately excluded due to their not attending subsequent visits. We presume this is due to patients entering the trial that had a predetermined desire for one of the treatment arms and subsequently leaving the study if they did not get the desired randomization. Previous studies have reported instances of patients seeking care for non-life threatening issues may not be seeking a surgical intervention, or may preferentially want surgery. 23 Likewise, in our study, a minority of patients elected to undergo treatment opposite to their randomization condition. COVID-19 likely played a significant role in challenges with follow-up in our study as well, as enrollment and follow-up challenges increased shortly after the beginning of the pandemic. Other studies have experienced this phenomenon as well. For example, oncology studies reported a significant drop in enrollment willingness by patients, with one-fifth of patients reporting COVID-19 having a negative influence on their willingness to enroll. 24 Although no interim analysis was planned within the initial study design, given the slow enrollment and challenges during the COVID pandemic, the study seemed unlikely to demonstrate equipoise and was unlikely to achieve the intended sample size. Enrollment was thus closed early, but patients who had received treatment underwent ongoing observation.
Notably, this study did not compare sleeve gastrectomy and medical management, or with Roux-en-Y Gastric bypass. Sleeve gastrectomy is now the most common bariatric surgery conducted across the United States. 25 However, at the time of the design of this study, a significantly greater volume of literature existed showing the safety of Roux-en-Y gastric bypass in patients with ESRD, and this aligned with the experience of the surgeons involved with this study. One concern with gastric bypass in patients with ESRD is the development of secondary oxalosis. A meta-analysis of twelve observational studies found that gastric bypass had a nearly 80% higher relative risk of nephrolithiasis. 26 To date however, data evaluating the impact of nephrolithiasis impact renal allografts in prior gastric bypass patients is limited to case series.27,28 No episodes of allograft nephrolithiasis was found in our small cohort of patients who underwent transplant following gastric bypass. Additionally, several studies have shown greater weight loss and comorbid disease remission with gastric bypass compared to sleeve gastrectomy. 29 Importantly, no guidelines exist at the time of writing that specify when a particular patient should undergo a particular bariatric procedure, including patients needing renal transplant. Future studies should specifically compare the safety and effectiveness of Roux-en-Y Gastric Bypass and Sleeve Gastrectomy in achieving durable weight loss in patients with ESRD seeking renal transplant.
Despite these limitations, this study provides further evidence yet that bariatric surgery should be considered in patients with concurrent ESRD and obesity seeking kidney transplant. Future studies should include longer follow-up to ensure durable weight loss and appropriate graft survival following kidney transplantation. Additionally, larger studies can confirm that short and long-term adverse events are not significantly elevated above that of the general population of patients undergoing bariatric surgery.
Conclusion
This small, non-blinded randomized trial found that, compared to medical management, bariatric surgery achieves greater weight loss in a population of patients with obesity and ESRD without a significantly higher rate of complications or worsening of underlying renal disease. Importantly, this resultant weight loss leads to a higher rate of successful listing for kidney transplantation.
Supplemental Material
Supplemental Material - Medical and Surgical Weight Loss as a Pathway to Renal Transplant Listing
Supplemental Material for Medical and Surgical Weight Loss as a Pathway to Renal Transplant Listing by Jason M. Samuels, Wayne English, Kelly A. Birdwell, Irene D. Feurer, David Shaffer, Sunil K. Geevarghese, and Seth J. Karp in The American Surgeon
Footnotes
Acknowledgments
The authors would like to acknowledge Alysse Sephel, MPH, BSN, RN and Scott A. Rega MS, MMHC for their assistance with this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CTSA award from the National Center for Advancing Translational Sciences. Its contents are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
