Abstract
Background
Prolonged postoperative ileus (PPOI) contributes to morbidity and prolonged hospitalization. Prucalopride, a selective 5-hydroxytryptamine receptor agonist, may enhance bowel motility. This review assesses whether the perioperative use of prucalopride compared to placebo is associated with accelerated return of bowel function post gastrointestinal (GI) surgery.
Methods
OVID, CENTRAL, and EMBASE were searched as of January 2024 to identify randomized controlled trials (RCTs) comparing prucalopride and placebo for prevention of PPOI in adult patients undergoing GI surgery. The primary outcomes were time to stool, time to flatus, and time to oral tolerance. The secondary outcomes were incidence of PPOI, length of stay (LOS), postoperative complications, adverse events, and overall costs. The Cochrane risk of bias tool for randomized trials and the Grading of Recommendations, Assessment, Development, and Evaluations framework were used. An inverse variance random effects model was used.
Results
From 174 citations, 3 RCTs with 139 patients in each treatment group were included. Patients underwent a variety of GI surgeries. Patients treated with prucalopride had a decreased time to stool (mean difference 36.82 hours, 95% CI 59.4 to 14.24 hours lower, I2 = 62%, low certainty evidence). Other outcomes were not statistically significantly different (very low certainty evidence). Postoperative complications and adverse events could not be meta-analyzed due to heterogeneity; yet individual studies suggested no significant differences (very low certainty evidence).
Discussion
Current RCT evidence suggests that prucalopride may enhance postoperative return of bowel function. Larger RCTs assessing patient important outcomes and associated costs are needed before routine use of this agent.
Introduction
Postoperative ileus (POI) refers to the impairment of coordinated motor activity of the gastrointestinal (GI) tract following surgery and manifests as obstipation and intolerance of oral intake.1,2 Approximately 10%–30% of patients undergoing abdominal surgery develop a prolonged POI (PPOI) with GI tract dysfunction extending beyond postoperative day 3.3–6 PPOI contributes to significant patient morbidity due to poor oral tolerance, abdominal distention, and frequent need for nasogastric (NG) decompression.7–9 This subsequently leads to prolonged length of hospital stay (LOS), increased risk of developing additional complications, and increased health care costs.2,5,10
Despite recent advances in perioperative care including the use of enhanced recovery after surgery (ERAS) protocols that encourage goal directed intravenous fluid administration and multimodal opioid reducing analgesia, as well as the uptake of minimally invasive surgical techniques, PPOI remains a prevalent complication.2,11–14 Consequently, there is increasing interest in investigating the pathophysiology of PPOI and the development of pharmaceutical agents that target these mechanisms.15,16 Alvimopan, a peripherally acting mu-opioid receptor antagonist has demonstrated efficacy in improving GI recovery after open bowel resection; however, there are some concerns regarding its cardiovascular complications, utility following minimally invasive surgery.17,18 Other promotility agents including metoclopramide and erythromycin have failed to show benefit in treating PPOI. 19
Prucalopride, a highly selective 5-hydroxytryptamine (5-HT4) receptor agonist, is proposed to enhance bowel motility though prokinetic and anti-inflammatory mechanisms. 20 It is currently approved for treatment of chronic idiopathic constipation in adult female patients in whom laxatives fail to provide adequate relief.21–23 In animal and pre-clinical models of PPOI, prucalopride and other 5-HT4 receptor agonists have been shown to dampen activation of macrophages residing in the intestinal muscular layers, leading to decreased surgery-induced gut inflammation and improved contractility.20,24,25 Recent randomized controlled trials (RCTs) have assessed the utility of prucalopride in treating PPOI; however, these trials individually involve smaller study populations and have varying results.25–27 This review aims to elucidate whether the use of prucalopride compared to placebo is associated with a reduction in time to return of bowel function in patients undergoing GI surgery.
Materials and Methods
Search Strategy
The following databases covering the period from database inception through initially July 2022 were searched: Medline, EMBASE and Cochrane Central Register of Controlled Trials (CENTRAL). The search was updated on January 21st 2024 to assess any additional studies. The search was designed and conducted by a medical research librarian with input from study investigators. Search terms included “prucalopride,” “5-HT4,” “postoperative ileus,” and more (complete search strategy available in Appendix). Any studies corresponding to RCT protocols, the references of published studies, and gray literature were searched manually to ensure that all relevant articles were included. Full texts were not discriminated by language. This systematic review and meta-analysis is reported in accordance with the Preferred Reporting items for Systematic Reviews and Meta-Analyses (PRISMA) and the study protocol was registered on PROSPERO before starting data collection (Prospero ID: CRD42023388053). 28
Study Selection
This review included RCTs that compared use of prucalopride vs placebo for prevention or treatment of PPOI in patients aged ≥18 years undergoing GI surgery. For the purposes of this review, a RCT was defined as a study in which participants were randomly allocated (by chance alone) to receive either prucalopride or placebo perioperatively while receiving other perioperative and operative management in keeping with the study institution’s standard of care. GI surgeries included any surgery performed on the esophagus, stomach, small intestine, large intestine, rectum, liver, gallbladder, or pancreas. Studies were not discriminated based on the timing or dosage of prucalopride administration. Cohort studies without randomized controlled design, abstracts, posters, opinions, case reports, systematic reviews, meta-analyses, letters to editors, editorials, and studies focusing on in vitro models were excluded.
Outcomes Assessed
The primary outcomes were time to defecation, time to flatus, and time to oral tolerance measured in hours. These measures have been used as surrogates for resumption of coordinated GI motility.2,3 The secondary outcomes were incidence of PPOI (defined as signs or symptoms of POI lasting ≥4 days), postoperative LOS, postoperative complications, adverse events attributed to prucalopride use, NG reinsertion, and overall costs. Postoperative complications were scored by the Clavien-Dindo classification. 29
Data Extraction
Two reviewers (SS and GT) independently evaluated the systematically searched titles and abstracts using a standardized, pilot-tested form. Discrepancies that occurred at the title and abstract screening phases were resolved by inclusion of the study. At the full-text screening stage, discrepancies were resolved by consensus between the two reviewers. If the disagreement persisted, a third reviewer (TM) was consulted. Two reviewers (SY or JK and GT) independently conducted data extraction into a data collection form designed a priori. The extracted data included study characteristics (eg, author, year of publication, and definition of outcomes), patient demographics (eg, age, gender, race, and comorbidities), operative characteristics (eg, open vs laparoscopic approach, GI organ involved, and duration of operation), treatment characteristics (eg, timing and dosing of prucalopride or placebo), and outcomes of interest.
Risk of Bias and Certainty of Recommendation Assessment
Risk of bias for each included study was assessed using the second version of the Revised Cochrane risk of bias tool (ROB 2).30,31 Two reviewers (SY or JK and GT), who previously piloted the ROB 2 on a sample of articles to familiarize themselves with the criteria, independently evaluated each study. As defined in advance, both assessors attempted to resolve any disagreements via discussion, and when that failed, a third author (TM) was consulted to adjudicate the final judgment. For the purposes of this assessment, the interventions being compared are defined as experimental (prucalopride) and comparator (placebo). Each primary and secondary outcome of interest was assessed for risk of bias, with prioritization for intention-to-treat analysis. Wherever possible, the trial protocol, non-commercial trial registry record (eg, ClinicalTrials.gov record), and any obtainable gray literature for each study were obtained to help inform the final ROB 2 decision. Additionally, these sources were used to identify any trials that are not published but aimed to address the study question. Outcomes to be reported a priori were compared between the trial protocols and publications to evaluate for reporting bias.
A priori, the authors collectively agreed that the seven most important outcomes were as follows: time to stool, time to flatus, time of oral tolerance, incidence of PPOI, LOS, NG insertion, and postoperative adverse events and complications. For these outcomes, three reviewers (SS, GT, and TM) independently evaluated the certainty of evidence using Cochrane’s Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. 32 The online GRADEpro tool was used to develop the summary of findings table.
Optimal Information Size
The optimal information size (OIS) for each of the seven most important outcomes was calculated to assist with GRADE assessment for the domain “imprecision.” A P value <.05 and power of 80% was used for all calculations. Using a baseline time to return of bowel function including time to flatus, time to stool and time to oral tolerance of mean 96 hours +/− SD 36 hours, and to detect a difference of 24 hours, an OIS of 35 patients per group is required. 33 Using a baseline rate of postoperative NG insertion of 12%, and to detect a decreased rate of 9%, the OIS is 1638 patients per group.34,35 The OIS for incidence of PPOI is 686 patients per group, using a baseline incidence of 15% and to detect a decreased rate of 10%.4–6 For postoperative LOS, the calculated OIS is 251 patients per group using a baseline LOS of mean 7 days +/− SD 4 days and to detect a decreased rate by 1 day.36–38
Statistical Analysis
All statistical analysis and meta-analysis were performed on Cochrane Review Manager 5.4 (London, United Kingdom). The threshold for statistical significance was set a priori at a p of <.05. A pairwise meta-analysis was performed using an inverse variance random effects model for all meta-analyzed outcomes. Intention-to-treat analysis for each outcome of interest was prioritized over modified intention-to-treat or per-protocol analysis. Pooled effect estimates were obtained by calculating the mean difference (MD) in outcomes for continuous variables and risk ratios (RRs) for dichotomous variables along with their respective 95% confidence intervals (CIs) to confirm the effect size estimation. In addition, mean and standard deviation (SD) was estimated for studies that only reported median and interquartile range using the method described by Wan et al. 39 Assessment of heterogeneity was completed using the inconsistency (I2) statistic. An I2 greater than 30% was considered to represent moderate heterogeneity and I2 greater than 60% was considered to represent substantial heterogeneity. 40 In addition, a sensitivity analysis adjusting for gastrointestinal organ system involved, and laparoscopic vs open surgery was attempted. For outcomes that were reported in less than two studies, a systematic narrative summary was provided.
Results
Study and Patient Characteristics
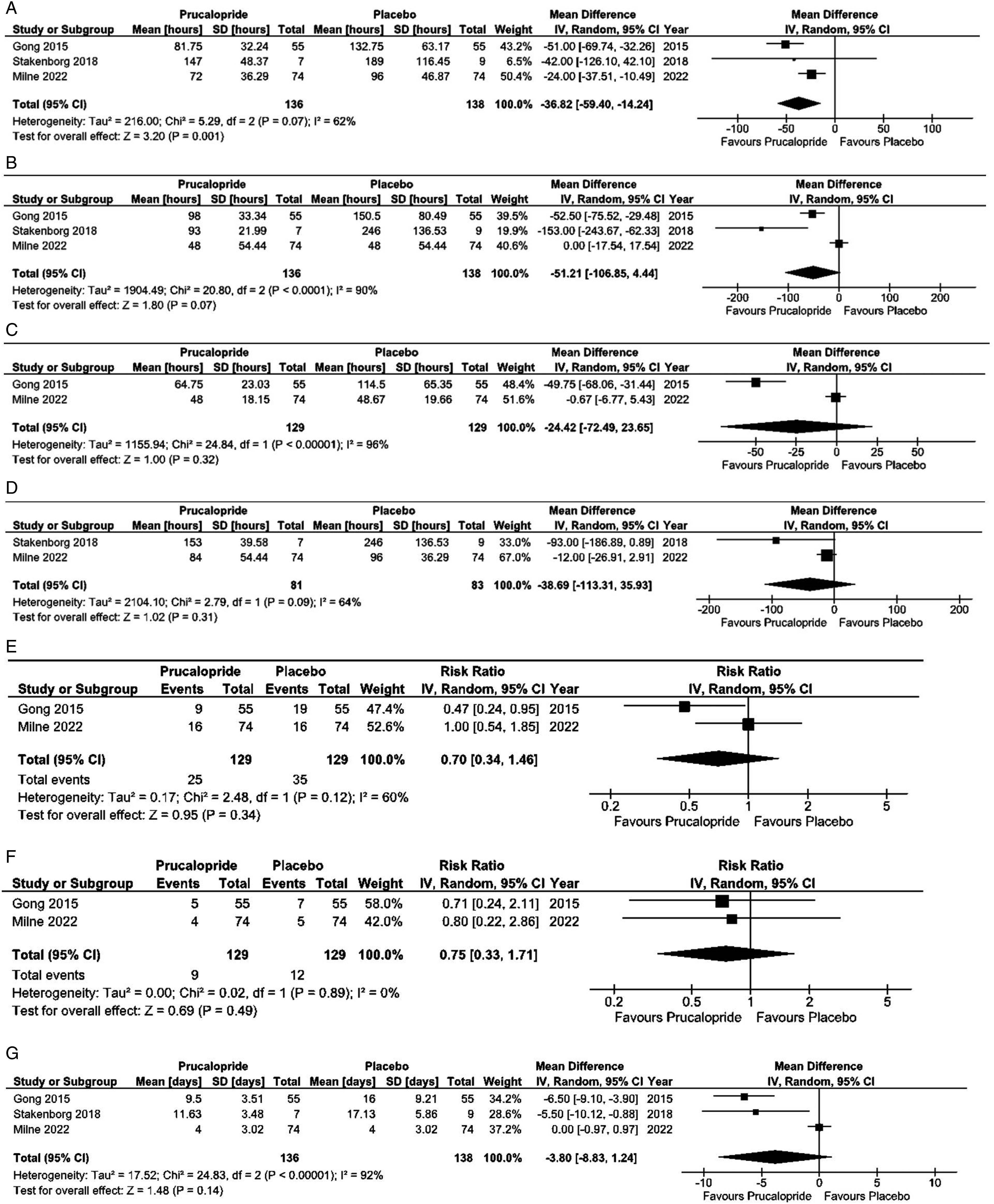
The initial search in July 2022 identified 150 relevant citations, from which 3 RCTs published between 2015 and 2022 met inclusion criteria.25–27 The search was updated in January 2024 and identified 24 additional citations, of which none met inclusion criteria. A PRISMA flow diagram of the updated study selection process is illustrated in Figure 1 and 2. PRISMA flowchart for included and excluded studies. Forrest plots for meta-analyzed outcomes. (A) Time to stool. (B) Time to oral tolerance. (C) Time to flatus. (D) Time to GI-2 (passage of stool and tolerance of oral intake). (E) Incidence of prolonged postoperative ileus. (F) Incidence of nasogastric tube insertion. (G) Length of stay.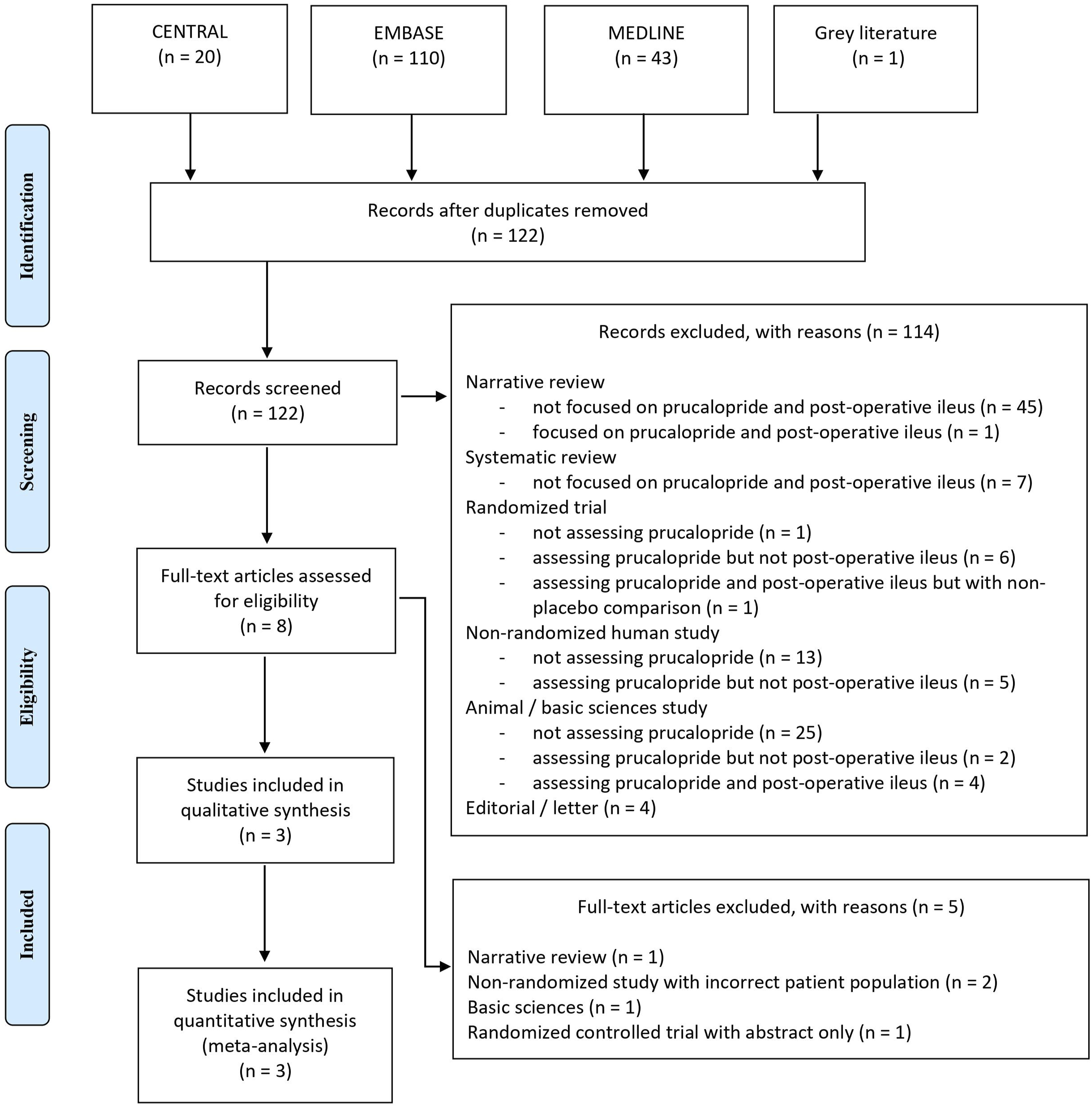
In total, 139 patients were included in each study arm. Patients who received prucalopride had a mean age of 59.4 years (SD 17.9 years) with 33% being female. Patients who received placebo had a mean age of 56.6 years (SD 17.1 years) with 40% being female. Stakenborg et al performed a per-protocol analysis, with results presented for 7 out of 10 patients receiving prucalopride and 9 out of 10 patients receiving placebo; as a result, up to 136 patients in the prucalopride group and 138 patients in the placebo group had available outcome data and were included in data syntheses. 25
Study, patient, and Operative Characteristics. ASA: American Society of Anesthesiologists Classification; Min: Minutes; RHC: Right Hemicolectomy; LAR: Low Anterior Resection; APR: Abdominoperineal Resection; Sub Col: Subtotal Colectomy; Lap: Laparoscopic; ERAS: Enhance Recovery After Surgery; NG: Nasogastric; TPN: Total Parenteral Nutrition; ICR: Ileocecal Resection; LHC: Left Hemicolectomy; SR: Sigmoid Resection; SBR: Small Bowel Resection; GDS: Gastroduodenal Surgery; Stoma R: Stoma Reversal.
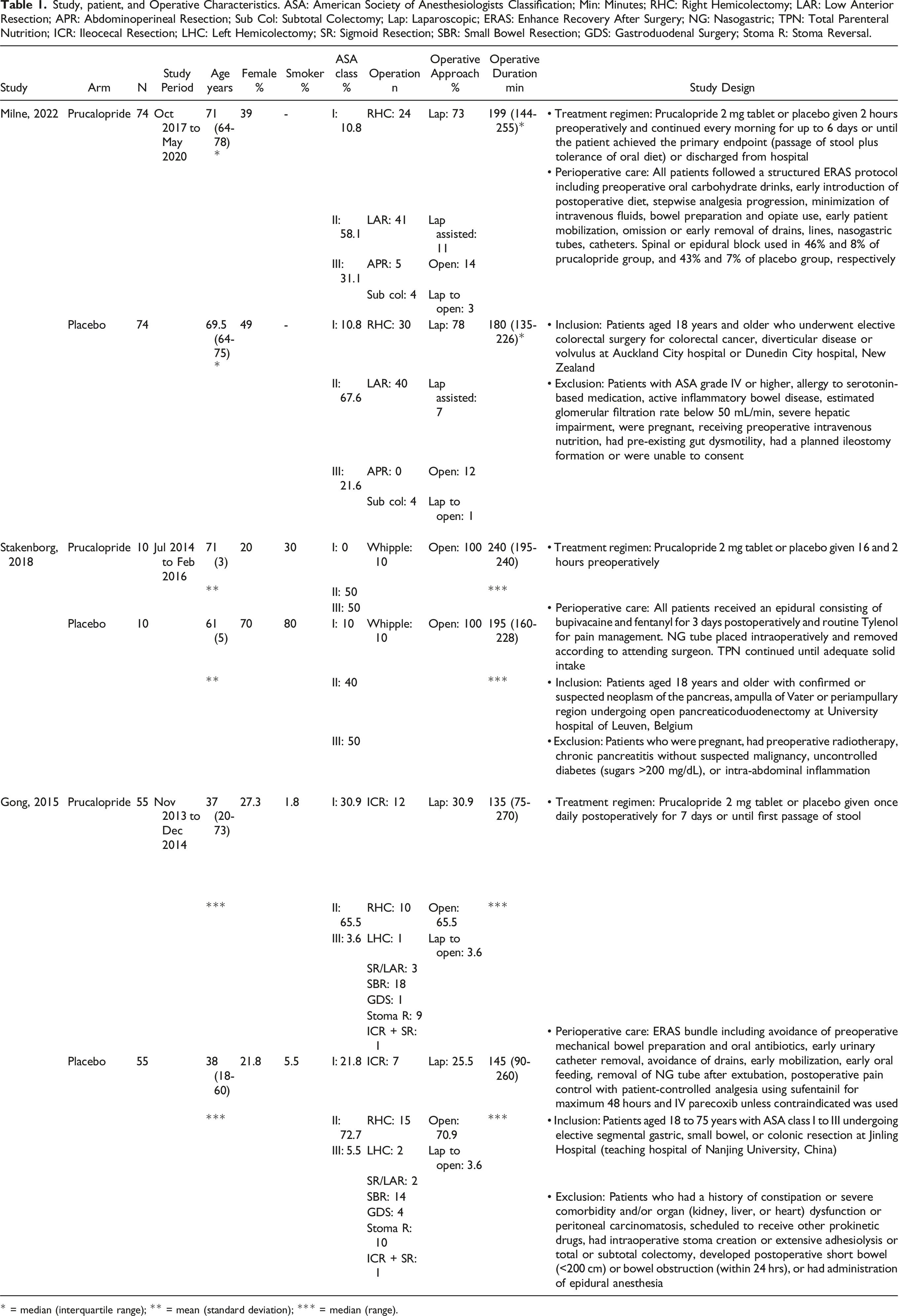
* = median (interquartile range); ** = mean (standard deviation); *** = median (range).
Risk of Bias
Risk of Bias assessment using the Cochrane Risk of Bias Tool for Randomized Trials Version 2.

Return of Bowel Function
Postoperative outcomes Related to Return of Bowel Function, Length of Stay, and overall Costs. NG: Nasogastric.
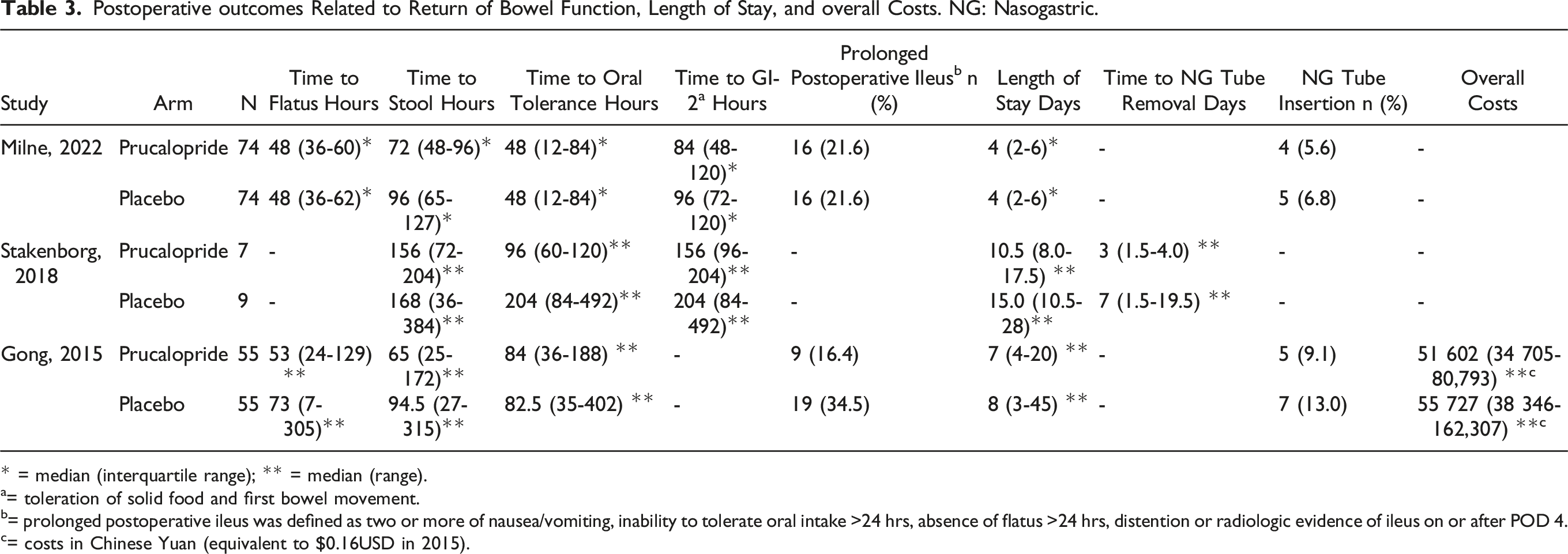
* = median (interquartile range); ** = median (range).
a= toleration of solid food and first bowel movement.
b= prolonged postoperative ileus was defined as two or more of nausea/vomiting, inability to tolerate oral intake >24 hrs, absence of flatus >24 hrs, distention or radiologic evidence of ileus on or after POD 4.
c= costs in Chinese Yuan (equivalent to $0.16USD in 2015).
Length of Stay and Hospital Costs
All 3 studies reported postoperative LOS. Gong et al and Stakenborg et al found a statistically significant decrease in postoperative LOS favoring prucalopride use. The mean LOS for patients in Milne et al was 4 days, and this was identical between the prucalopride and control groups. Upon pooling of data, LOS was decreased by 3.8 days in the prucalopride group but wide 95% CIs (95% CI 8.83 days lower to 1.24 days higher) resulted in a non-statistically significant difference (P = .14, I2 = 92%, very low certainty evidence) (Figure 1(G)). Hospital costs were only evaluated by Gong et al who found a signal towards decreased costs in the prucalopride group, but this did not reach statistical significance (median 51 602, range 34 705-80 792 Chinese Yuan in prucalopride group vs median 55 727, range 38 346-162.307 Chinese Yuan, P = .059).
Postoperative Complications and Adverse Events
Postoperative Complications and Treatment Related Adverse Events. CD: Clavien-Dindo Classification; CTCAE: Common Terminology Criteria for Adverse Events; SBO: Small Bowel Obstruction.
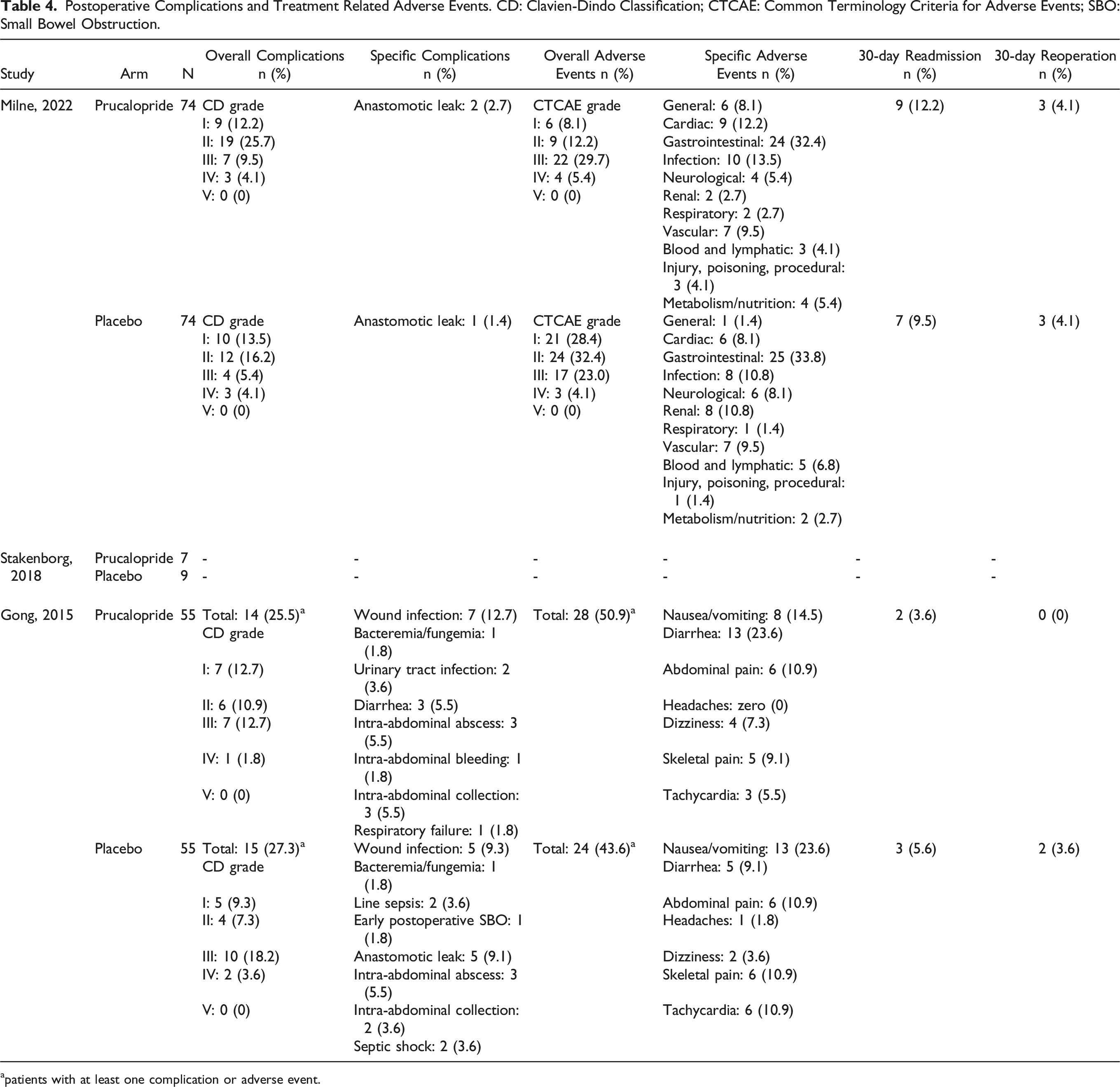
apatients with at least one complication or adverse event.
Certainty of Evidence
Summary of Recommendations for the use of prucalopride Versus Placebo to Promote Return of Bowel Function Postoperatively; CI: Confidence Interval; MD: Mean Difference; RR: Risk Ratio.
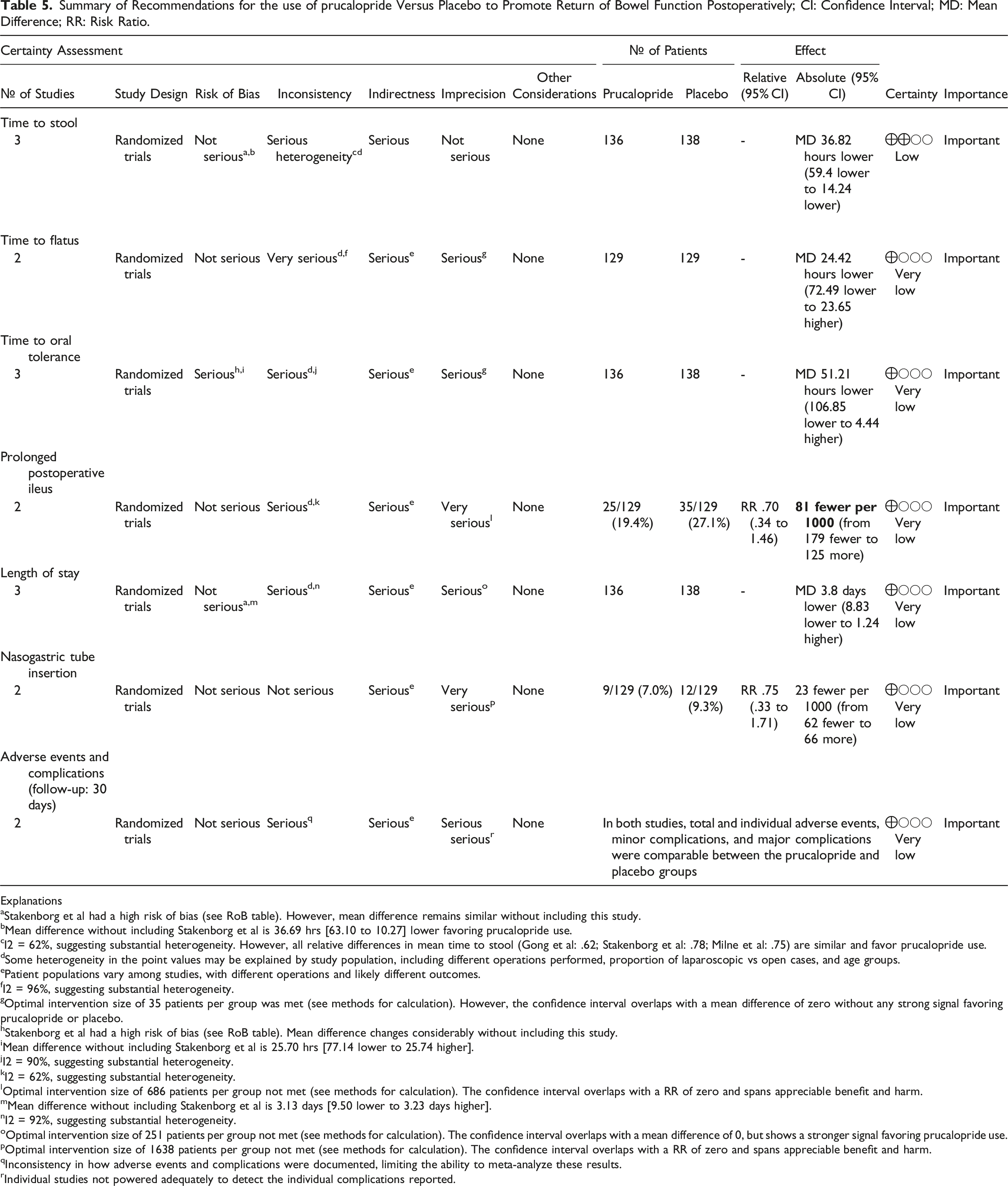
Explanations
aStakenborg et al had a high risk of bias (see RoB table). However, mean difference remains similar without including this study.
bMean difference without including Stakenborg et al is 36.69 hrs [63.10 to 10.27] lower favoring prucalopride use.
I2 = 62%, suggesting substantial heterogeneity. However, all relative differences in mean time to stool (Gong et al: .62; Stakenborg et al: .78; Milne et al: .75) are similar and favor prucalopride use.
dSome heterogeneity in the point values may be explained by study population, including different operations performed, proportion of laparoscopic vs open cases, and age groups.
ePatient populations vary among studies, with different operations and likely different outcomes.
fI2 = 96%, suggesting substantial heterogeneity.
gOptimal intervention size of 35 patients per group was met (see methods for calculation). However, the confidence interval overlaps with a mean difference of zero without any strong signal favoring prucalopride or placebo.
hStakenborg et al had a high risk of bias (see RoB table). Mean difference changes considerably without including this study.
iMean difference without including Stakenborg et al is 25.70 hrs [77.14 lower to 25.74 higher].
jI2 = 90%, suggesting substantial heterogeneity.
kI2 = 62%, suggesting substantial heterogeneity.
lOptimal intervention size of 686 patients per group not met (see methods for calculation). The confidence interval overlaps with a RR of zero and spans appreciable benefit and harm.
mMean difference without including Stakenborg et al is 3.13 days [9.50 lower to 3.23 days higher].
nI2 = 92%, suggesting substantial heterogeneity.
oOptimal intervention size of 251 patients per group not met (see methods for calculation). The confidence interval overlaps with a mean difference of 0, but shows a stronger signal favoring prucalopride use.
pOptimal intervention size of 1638 patients per group not met (see methods for calculation). The confidence interval overlaps with a RR of zero and spans appreciable benefit and harm.
qInconsistency in how adverse events and complications were documented, limiting the ability to meta-analyze these results.
Individual studies not powered adequately to detect the individual complications reported.
Discussion
PPOI is a major complication of GI surgery that increases patient morbidity and places a financial burden on the health care system.2–5 Unfortunately, therapies to prevent or treat PPOI are limited.11–18 This systematic review and meta-analysis pooled data from RCTs assessing the use of prucalopride vs placebo to promote return of bowel function in patients undergoing GI surgery. Patients treated with prucalopride were found to have a decreased time to stool of 1.5 days. Other outcomes for return of bowel function were not statistically significantly different. Postoperative complications and adverse events could not be meta-analyzed due to heterogeneity, yet data from individual studies suggested no significant differences between the two treatment groups.
The ideal regimen, dosing, and route of prucalopride administration for PPOI prevention is unclear. Although 2 mg pills of prucalopride were used across the three included RCTs, the timing of administration varied; Stakenborg et al provided 2 preoperative doses, Milne et al provided 1 preoperative dose and daily postoperative doses, and Gong et al provided daily postoperative doses only.25–27 Stakenborg et al performed animal studies showing that prucalopride administered before, but not after abdominal surgery, reduced intestinal inflammation and prevented PPOI. 25 Hence, preoperative dosing may be warranted to obtain maximal anti-inflammatory effects. For treatment of chronic constipation, 1 mg to 4 mg daily doses have been investigated, with 2 mg and 4 mg daily doses being most efficacious.21–23 Both 2 mg and 4 mg doses have similar benefits; hence, 2 mg doses have been approved for chronic constipation treatment for most patients.21–23 However, in patients with POI, absorption of oral medications may be affected due to gastroparesis and gut edema 40 ; hence, alternative routes or larger doses may be required. 41 Moreover, patients with POI may not tolerate oral intake. This was evident in the included studies as administration of prucalopride or placebo was discontinued in approximately 8% of patients in each study arm due to NG insertion or development of PPOI.26,27 Patients with severe PPOI are in most need of treatment, and it may be important to consider subcutaneous or intravenous forms of medication for more reliable delivery. Of note, Galandiuk et al performed a RCT assessing .5 mg, 2 mg and 4 mg doses of subcutaneous prucalopride vs placebo provided postoperatively in patients undergoing elective colectomies. 42 The authors found that 4 mg daily subcutaneous dosing resulted in the fastest time to flatus or stool. 42 Unfortunately, no manuscript for this study was found, preventing analysis of its methodology and incorporation into this meta-analysis. 42 Ultimately, additional RCTs focused on subcutaneous or intravenous prucalopride with varying doses and regimens may be warranted.
The first generation 5-HT4 receptor agonists enhanced GI function but were nonselective and associated with increased cardiovascular adverse events including development of prolonged QT interval and arrhythmias. 43 In contrast, prucalopride is a highly selective 5-HT4 receptor agonist which has not been associated with increased cardiovascular adverse events both at therapeutic and supratherapeutic doses in vivo and early-phase human trials. 43 The most common side effects attributed to prucalopride include headache and GI upset with nausea, abdominal pain, or diarrhea. 44 These findings are in keeping with the studies by Milne et al and Gong et al who identified no statistically significantl difference in postoperative complications or adverse events except for increased incidence of diarrhea in the prucalopride group.26,27 Of note, both studies excluded patients with American Association of Anesthesia (ASA) grade IV or severe hepatic or renal impairment. However, in prior studies assessing more comorbid patient populations, and patients with impaired renal function receiving reduced dose, prucalopride was well tolerated.45,46 Overall, these findings suggest that prucalopride is likely well tolerated by most patients.
PPOI has a complex pathophysiology, with multiple proposed mechanisms including inflammatory, hormonal, neurogenic, and pharmacologic pathways. 2 Moreover, after abdominal surgery, different segments of the GI tract are expected to regain coordinated function in different time intervals, including 0-24 hours for the small intestine, 24-48 hours for the stomach, and 48-72 hours for the colon.1,47 The mechanism of action for prucalopride is likely multifaceted, including mimicking vagal stimulation to release acetylcholine from enteric neurons along the GI tract, with subsequent anti-inflammatory effects on resident macrophages in the muscularis layer of the bowel as well as promotility effects on gastric emptying and small as well as large bowel transit.45,48 Additional pathways involving opioid receptor antagonists, ghrelin agonists, sympathetic nervous system blockade, and specific interleukin receptor modulation have and continue to be investigated. 47 Unfortunately, many studies assessing the impact of a single pharmaceutical on POI have failed to show any significant benefit.19,49,50 It is possible that to maximize the effect of pharmaceutical agents, a combination of two or more drugs used together rather than a single drug may need to be investigated as targeting multiple mechanisms and segments of the GI tract may more effectively accelerate PPOI recovery.
This study has several limitations. First, only 3 RCTs met the inclusion criteria with relatively small sample sizes. Although the OIS was achieved for the primary outcomes of time to stool, time to flatus, and time to oral tolerance, the OIS was not achieved for the secondary outcomes. As such, despite pooling the data, the study remained underpowered for the secondary outcomes of interest, and it is possible that there were additional differences in postoperative outcomes that were not detected. Nonetheless, prucalopride demonstrated decreased time to stool, and had a signal towards decreased time to flatus and decreased time to oral tolerance suggesting that it may provide benefit for return of bowel function. Second, the studies were heterogeneous with respect to patient age groups, operations performed, and proportion of laparoscopic vs open surgeries. Older age, longer and more complex operations involving greater anesthetic time and resuscitation, and open surgeries involving more manipulation of bowel are risk factors for PPOI.6,11,13–15 Moreover, different parts of the GI tract regain return of function at different times postoperatively.2,17 These clinical differences likely explain some of the heterogeneity in outcomes found between each study. Ideally, sensitivity analyses or meta-regressions incorporating these known risk factors for PPOI would be performed to better delineate the role of prucalopride for PPOI. Using the GRADE framework to provide a certainty of evidence assessment, we aimed to transparently highlight these limitations for cautious interpretation of the results.
In summary, this systematic review and meta-analysis demonstrated a 1.5 day improvement in time to stool post GI surgery with the use of prucalopride vs placebo; however, other outcomes addressing return of bowel function and LOS were not statistically significantly different. Prucalopride provided minimal additional side effects as per 2 of the RCTs and thus is likely safe to prescribe in these patient populations. Based on the limited data available, prucalopride may be an appropriate pharmacologic adjunct to enhance postoperative return of bowel function. Larger RCTs assessing clinically relevant outcomes and associated costs are needed before routine use of this agent.
Footnotes
Author Contributions
Conception and design of the study: All authors.
Acquisition of data: Talwar, Sharma, McKechnie, Yang, and Khamar.
Analysis and interpretation of data: All authors.
Drafting and revision of the manuscript: All authors.
Approval of the final version of the manuscript: All authors.
Agreement to be accountable for all aspects of the work: All authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Prospero ID
CRD42023388053
Data Availability Statement
Template data collection forms, data extracted from included studies, data used for all analyses, analytic codes can be requested for access by contacting the corresponding author.
