Abstract
Background
Postoperative gastrointestinal dysfunction (POGD) commonly occurs following gastrointestinal (GI) surgery and is associated with specific anesthetic agents. Cholinesterase inhibitors employed for reversing neuromuscular blockade have been implicated in development of POGD. Sugammadex, a novel reversal agent, is linked with reduced POGD. However, there is a lack of comprehensive comparative review between these agents regarding their impact on POGD following GI surgery. This study aims to systematically review the effects of sugammadex on POGD compared to cholinesterase inhibitors following GI surgery.
Methods
MEDLINE, EMBASE, and CENTRAL were searched as of July 2022 to identify articles comparing sugammadex with cholinesterase inhibitors in patients undergoing gastrointestinal surgery, specifically in relation to POGD. Secondary endpoints included length of hospital stay, readmission rates, pulmonary complications, and postoperative morbidity.
Results
From 198 citations, 2 randomized controlled trials (RCTs) and 3 retrospective cohorts with 717 patients receiving sugammadex and 812 patients receiving cholinesterase inhibitors were included. Significantly lower rates of prolonged postoperative ileus (OR .44, 95% CI .25-.77, P < .05, I2 = 56%, low certainty evidence) was observed with sugammadex. No significant difference in any other outcome was observed. Narrative review of readmission data demonstrated no significant difference.
Conclusion
The use of sugammadex following gastrointestinal surgery is associated with significantly lower rates of prolonged postoperative ileus compared to cholinesterase inhibitors. However, these do not translate into a significant reduction in length of stay, morbidity, or postoperative nausea and vomiting. Results are limited by the numer of studies included and missing data, more robust RCTs are needed before recommendations can be made.
Introduction
Postoperative gastrointestinal dysfunction (POGD) is characterized by transient impairment in gastrointestinal (GI) function following surgery and encompasses symptoms including prolonged postoperative ileus (PPOI), and postoperative nausea and vomiting (PONV). 1 POGD is common after GI surgery due to increased prevalence of surgical risk factors such as manual manipulation of intestine and prolonged anesthetic duration.1-3
Anesthetic reversal agents used to reverse the effects of deep neuromuscular blockade (NMB) may also play a key role in POGD. 4 NMB reversal traditionally involves a combination of cholinesterase inhibitors (CI) such as neostigmine and pyridostigmine, co-administered with anticholinergics (AC) like atropine and glycopyrrolate. Coadministration of AC avoids the potential lethal side effects of unopposed muscarinic activity. 5 However, due to the longer half-life of AC relative to CI, AC effects predominate in the immediate postoperative period. These effects can include reduction in intestinal peristalsis, gastric secretion, and an increase in bronchodilation.6-9
Sugammadex is a novel reversal agent that forms tight inhibitory complexes with steroidal non-depolarizing muscle relaxants. It lacks muscarinic activity, thereby obviating the need for anticholinergic coadministration. 10 This property forms the basis of the hypothesis that sugammadex may lead to less POGD.
Several studies recently have recently examined the effect of sugammadex on POGD following abdominal surgery.11-13 However, study populations have been heterogenous with mixed results. Furthermore, no systematic review or meta-analysis comparing CI agents and sugammadex following GI surgery currently exists. Therefore, the primary aim of this study was to systematically review the effects of sugammadex on POGD compared to CI following GI surgery. Secondary endpoints included length of stay (LOS), readmission within 30 days, and postoperative morbidity.
Materials and Methods
Search Strategy
The following databases encompassing a period of studies from database inception to July 2022 were searched: Medline, EMBASE, and Cochrane Register of Controlled Trials (CENTRAL). Search strategy included terms such as “Sugammadex,” “Ileus,” “Bridion,” “Neostigmine,” and more. Relevant grey literature and published studies were manually surveyed for completion of search. This systematic review and meta-analysis is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and the Meta-Analysis of Observational Studies in Epidemiology. 14 The review was registered with PROSPERO (CRD42023389880).
We included both prospective and retrospective studies if they compared the use of sugammadex with CI in patients undergoing GI surgery and reported on POGD. Studies from all languages were included. Authors of studies without full-text available were contacted for the full-text, if no response in 30 days, the studies were excluded. Commentaries, opinion articles, case reports, pediatric population, animal studies, studies with less than 10 patients, and studies in non-GI surgery patients were excluded.
Outcomes Assessed
Primary study outcome included incidence of PPOI (defined as signs or symptoms of POI lasting ≥4 days) and PONV (defined as nausea or vomiting within 24 hours of surgery postoperatively). Secondary outcomes included postoperative LOS, readmission rates within 30 days, pulmonary complications, and postoperative morbidity. LOS was defined as the time from arrival to the post-anesthesia recovery unit (PACU) to time of discharge. Readmission was classified as readmission for any cause within 30 days of discharge from hospital.
Data Extraction
The search strategy was created from input by all members of the team and carried out by author SS. Two study authors independently searched the titles and abstracts using a standardized, pilot tested form. Any discrepancies at this stage were resolved by inclusion of the study. At the subsequent full-text screening stage, discrepancies were resolved by bringing the text in question to the whole team for consensus. Subsequent data abstraction was done independently by 2 reviewers with the use of a data collection excel sheet that was designed a priori. Abstracted data included study level data (eg, author, publication year, study design, and surgical procedure details), patient level data (eg, gender, body mass index [BMI], and ASA status) and postoperative data (eg, PPOI, PONV, LOS, and readmission).
Risk of Bias Assessment and Certainty of Evidence
The risk of bias in each study was evaluated with either the Cochrane Risk of Bias Tool for Randomized Controlled Trials 2.0 (RoB 2.0) or Risk of Bias in Non-randomized Studies – of Interventions (ROBINS-I).15,16 Estimates derived from meta-analysis were assessed by Grading of Recommendations, Assessment, Development and Evaluation (GRADE). 17 Two reviewers assessed the risk of bias and certainty of evidence independently. Discrepancies with assessment were discussed with all study authors until consensus was achieved.
Statistical Analysis
Statistical analyses and meta-analyses were performed on STATA version 14 (StataCord, College, TX0 and Cochrane Review Manager 5.3 (London, UK)). Threshold for statistical significance was set a priori at P < .05. Pairwise meta-analysis was performed using an inverse variance, random effects model for all outcomes that were meta-analyzed. Mean difference (MD) was calculated in outcomes for continuous variables and odds ratio (OR) for dichotomous variables along with their respective 95% confidence intervals (CI) to confirm effect size estimation. Lastly, mean and standard deviation (SD) when not reported was calculated from the median and range or interquartile range using methods described by Wan et al. 18 Heterogeneity was assessed using the inconsistency (I2) statistic. Inconsistency greater than 50% was considered to represent considerable heterogeneity. Publication bias was assessed with a funnel plot if > 10 studies were included in the meta-analysis. In addition, a sensitivity analysis adjusting for gastrointestinal organ system involved, and laparoscopic vs open surgery was attempted. A systematic narrative summary was provided for all outcomes in which less than 3 studies had reported the outcome.
Results
Study and Patient Characteristics
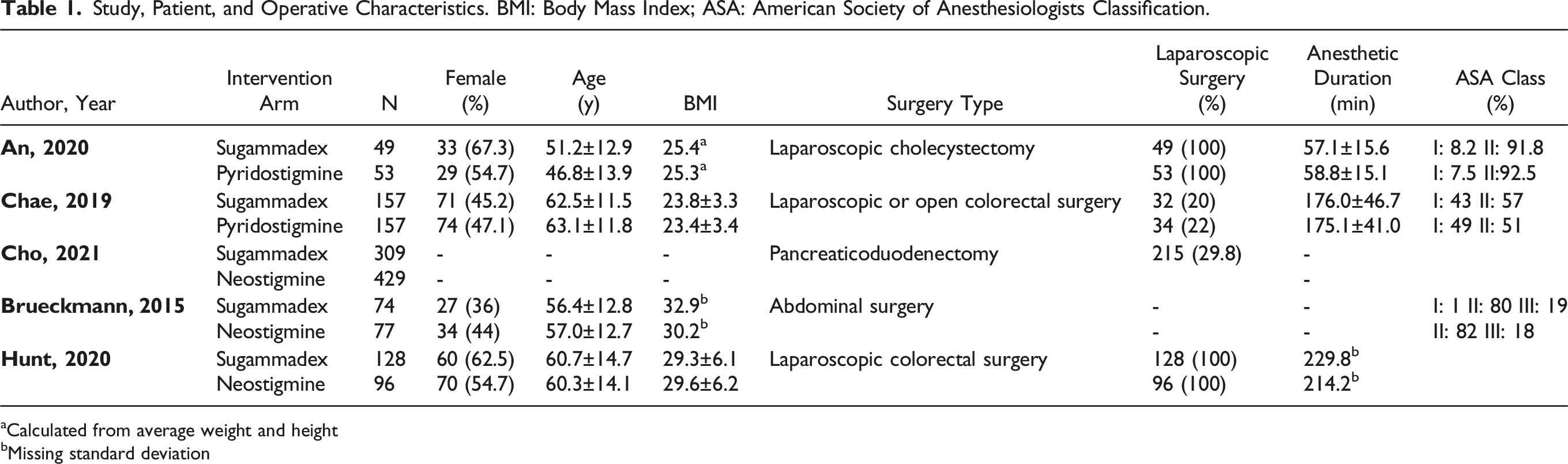
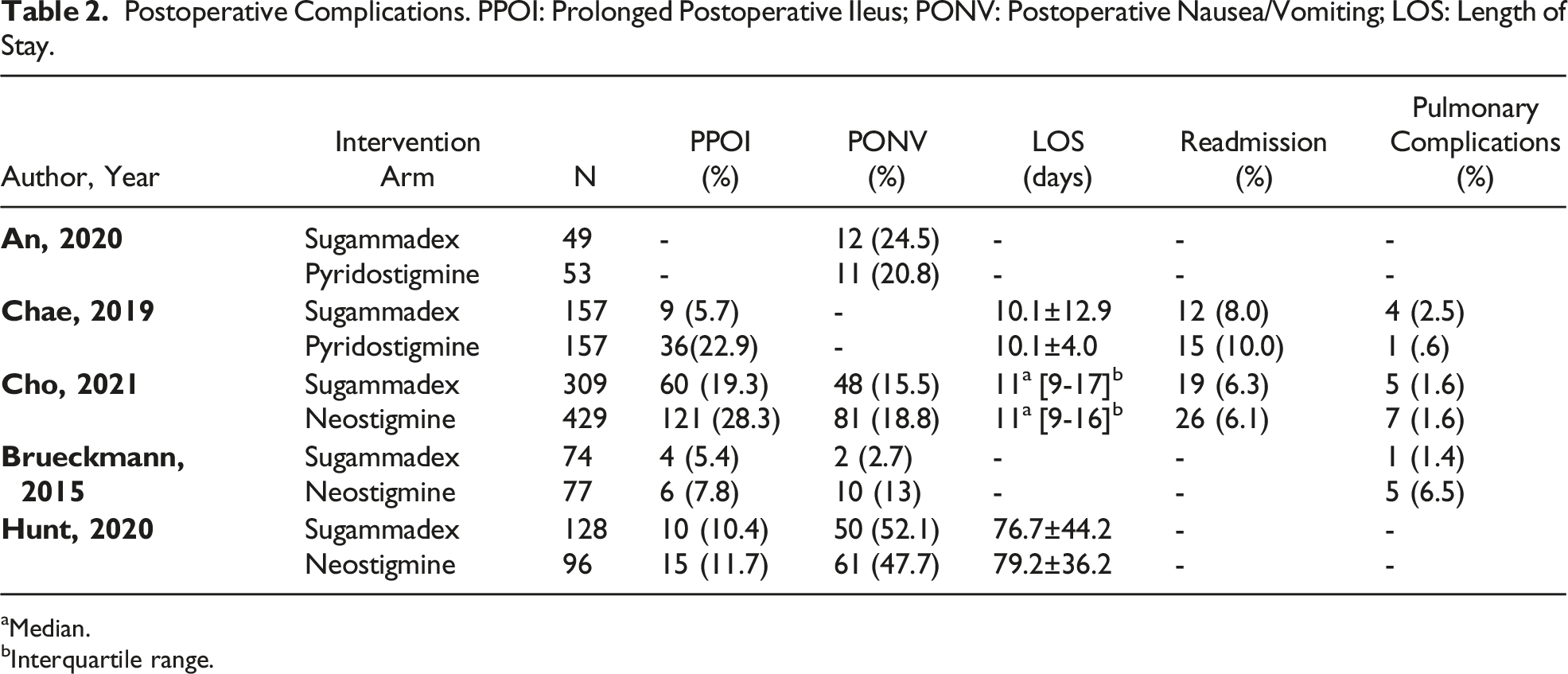
From of 198 relevant citations, 2 randomized controlled trials (RCT) and 3 retrospective cohorts met inclusion criteria.19-22 A PRISMA flow diagram for the study selection is illustrated in Figure 1. Overall, 717 patients were in the sugammadex group (mean age 59 +/- 13 years, 53.4% female) and 812 patients were in the CI group (mean age 59 +/- 14 years, 50% female). Detailed Study, Patient, and Operative characteristics are presented in Table 1. Detailed postoperative complications are presented in Table 2. PRISMA flowchart for included and excluded studies. Study, Patient, and Operative Characteristics. BMI: Body Mass Index; ASA: American Society of Anesthesiologists Classification. aCalculated from average weight and height bMissing standard deviation Postoperative Complications. PPOI: Prolonged Postoperative Ileus; PONV: Postoperative Nausea/Vomiting; LOS: Length of Stay. aMedian. bInterquartile range.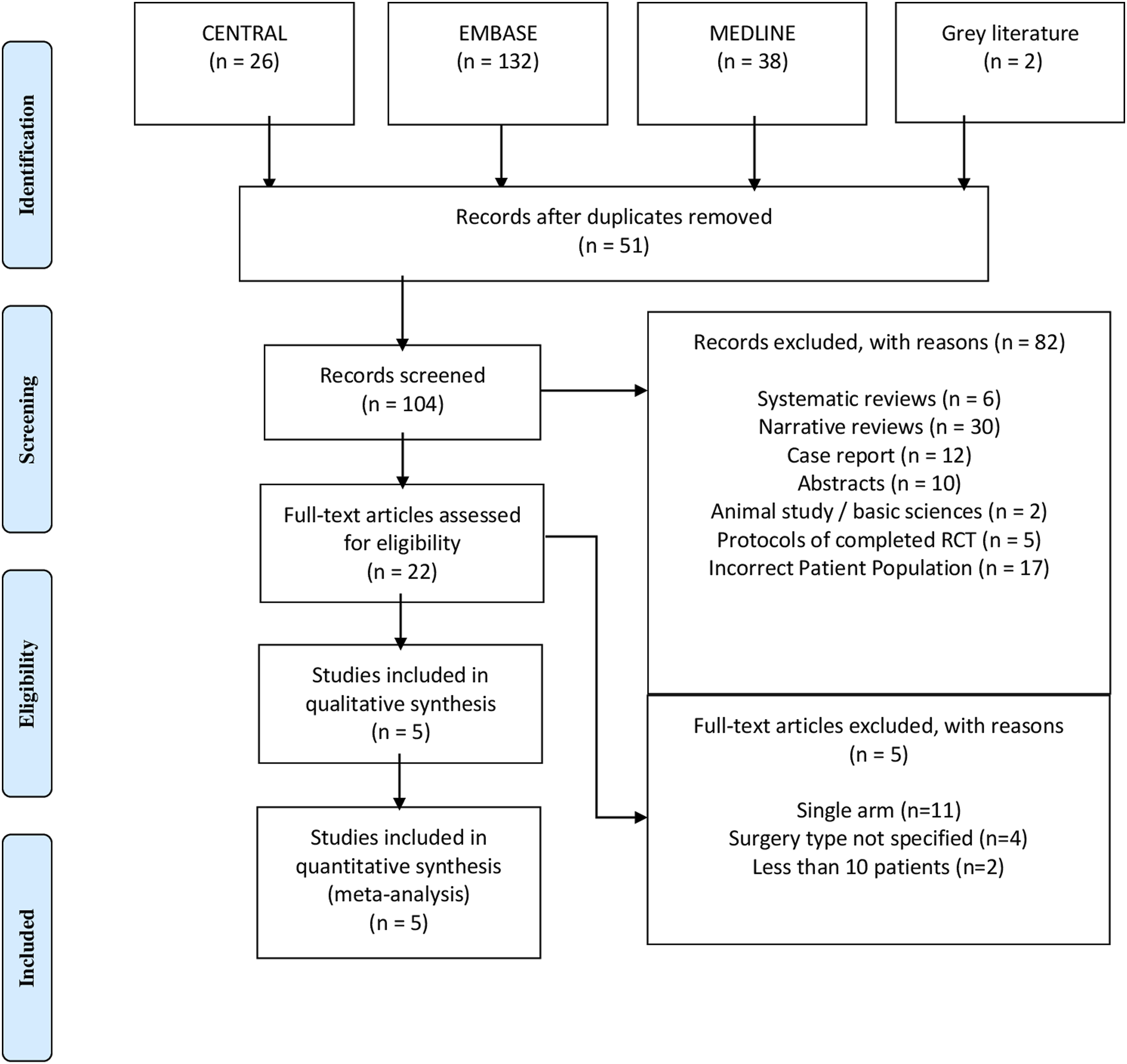
Gastrointestinal Symptoms
Four studies were included in the meta-analysis for PONV with 560 patients receiving sugammadex and 655 patients receiving CI (Figure 2).20-23 PONV data was extracted by review of PACU data for documentation of emesis or nausea in the PACU or administration of antiemetics within 24 hours of NMB reversal in one study.
21
The other 3 studies did not specifically mention how PONV data was recorded. There was no significant difference in the rates of PONV (OR .58, 95% CI .31-1.09, P = .09, I2 = 68%, very low certainty evidence) between the 2 groups. Overall odds ratio for development of postoperative nausea or vomiting in patients undergoing anesthetic reversal with Sugammadex vs Cholinesterase inhibitors.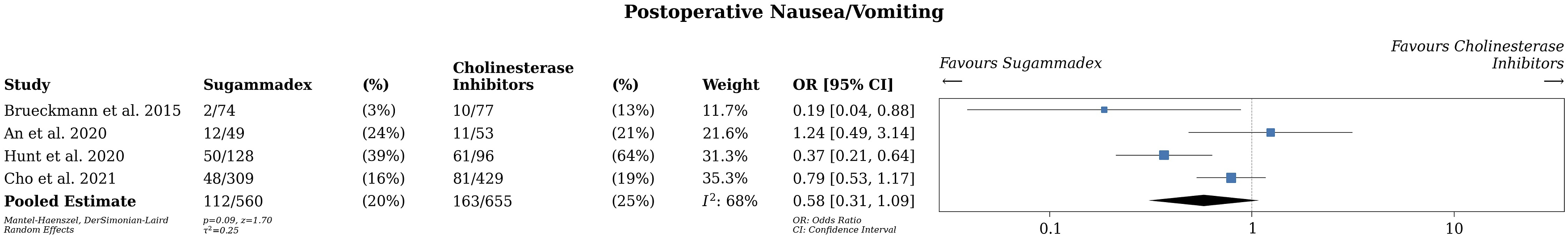
Similarly, 4 studies reported PPOI data with 668 patients receiving sugammadex and 759 receiving CI included in the analysis (Figure 3).19-21,23 Hunt et al extracted ileus data from discharge summaries or documentation of ICD-10 code for ileus.
21
Lastly, Cho et al defined ileus as postoperative time to first flatus or oral intake tolerance greater than the 75th percentile of that observed in our cohort.
20
There was a significantly lower rate of PPOI for patients receiving sugammadex (OR .44, 95% CI .25-.77, P < .05, I2 = 56%, low certainty evidence) compared to CI. Overall odds ratio for development of prolonged postoperative ileus in patients undergoing anesthetic reversal with Sugammadex vs Cholinesterase inhibitors.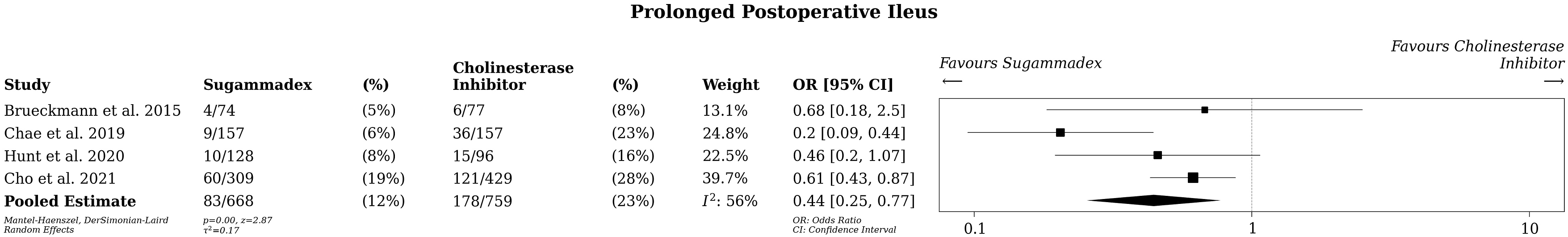
An et al did not specifically mention ileus in their results but did document time to first passage of flatus as their primary outcome. They found that patients receiving sugammadex (15.03h, 6.36-20.24h) compared to pyridostigmine (20.85h, 16.34-25.86h) had a statistically significant shorter time to passage of flatus (P = .001). Similarly, time to passage of stool was also shorter for the sugammadex group, although not statistically significant (P = .087). 22
Length of Stay
Three studies were included in the meta-analysis for LOS with 594 receiving sugammadex and 562 patients receiving CI (Figure 4).19-21 There was no difference in LOS between groups (MD -.01, 95% CI -.39-.37, P = .97, I2 = 0, low certainty evidence). Two studies reported readmission data with 466 patients receiving sugammadex, of which 31 (6.7%) were readmitted within 30 days whereas 41 (7.0%) patients of the 586 receiving CI required readmission. In both studies there was no significant difference in readmission between the 2 groups. Overall mean difference in length of stay in patients undergoing anesthetic reversal with Sugammadex vs Cholinesterase inhibitors.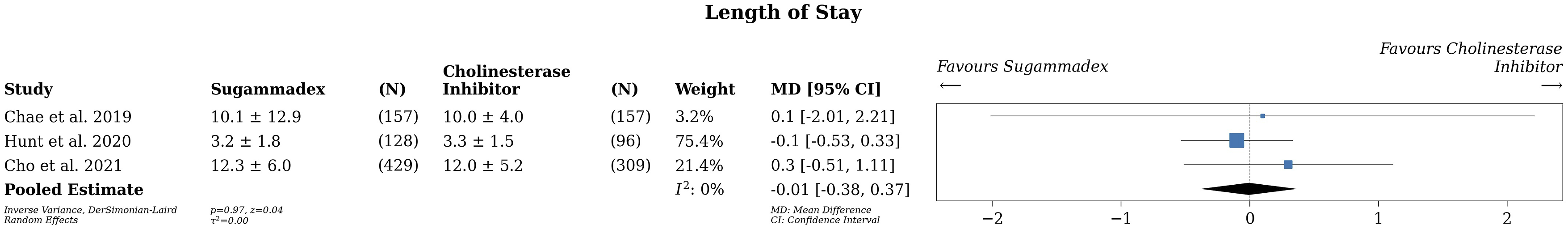
Postoperative Morbidity
Four studies reported postoperative morbidity, in which 668 patients received sugammadex and 759 patients received CI for reversal (Figure 5).19-21,23 There was no significant difference between the 2 groups for postoperative morbidity (OR .6, 95% CI .3-1.21, P = .15, I2 = 86, very low certainty evidence). Postoperative morbidity was calculated in all studies as documentation of at least one adverse event postoperatively. Overall odds ratio of postoperative morbidity in patients undergoing anesthetic reversal with Sugammadex vs Cholinesterase inhibitors.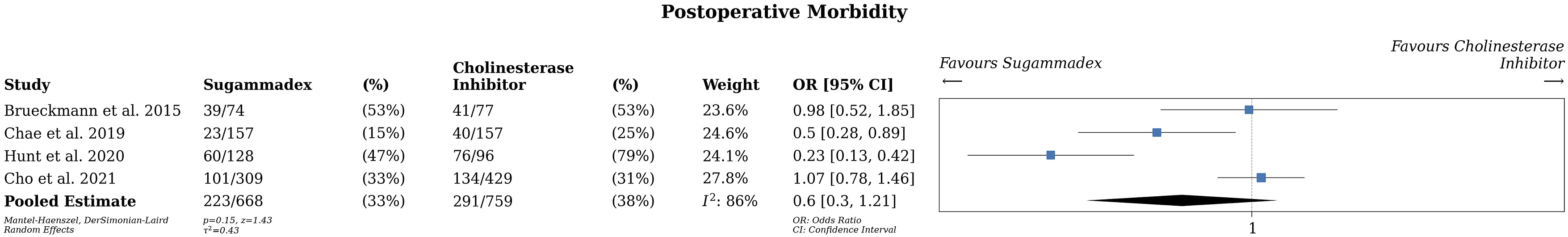
Pulmonary Complications
Pulmonary complications were reported in 3 studies with 540 patients receiving sugammadex, and 663 patients receiving CI (Figure 6).19,20,23 There was no significant difference in pulmonary complications between 2 groups postoperatively (OR .93, 95% CI .23-3.81, P = .92, I2 = 46, low certainty evidence). Two studies did not define what specific diagnosis were included as “pulmonary complications,” and Chae et al documented incidence of pneumonia postoperatively and these were included by study authors as pulmonary complications. Overall odds ratio for development of pulmonary complications in patients undergoing anesthetic reversal with Sugammadex vs Cholinesterase inhibitors.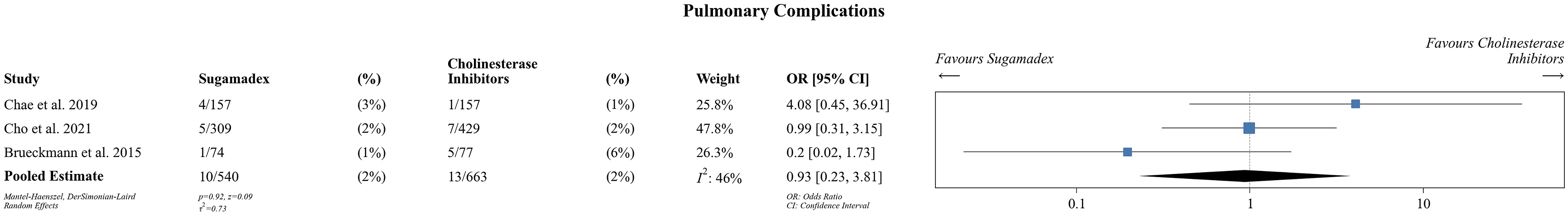
Risk of Bias
Risk of bias assessment included for full text articles was analyzed using Rob 2 and ROBINS-1 tools, presented in Figures 7 and 8, respectively.15,16 Both randomized controlled trials had overall low level of bias. In both, patients were blinded to the treatment, and in one study the safety assessors were also blinded to the treatment group. The intervention protocol was developed and described a priori. The number of patients excluded from analysis and reasons for exclusion were outlined in both studies. There was some concern with missing outcome data in one study due to 2 patients that were lost to follow-up for safety data.
23
Among the 3 retrospective studies, all three were classified as having overall low level of bias. Risk of bias tool for randomized controlled trials 2.0 (RoB 2.0). Risk of bias in non-randomized studies—of interventions (ROBINS-I).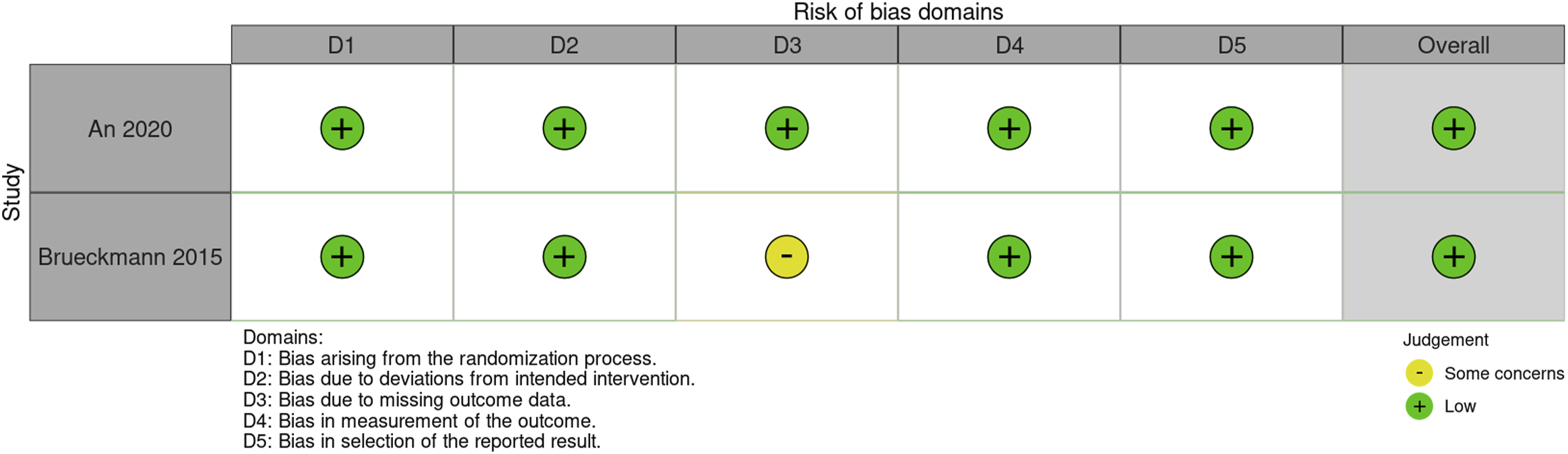

Certainty of Evidence
Summary of Recommendations for the Use of Sugammadex Versus Cholinesterase Inhibitor for Postoperative Gastrointestinal Dysfunction; RCT
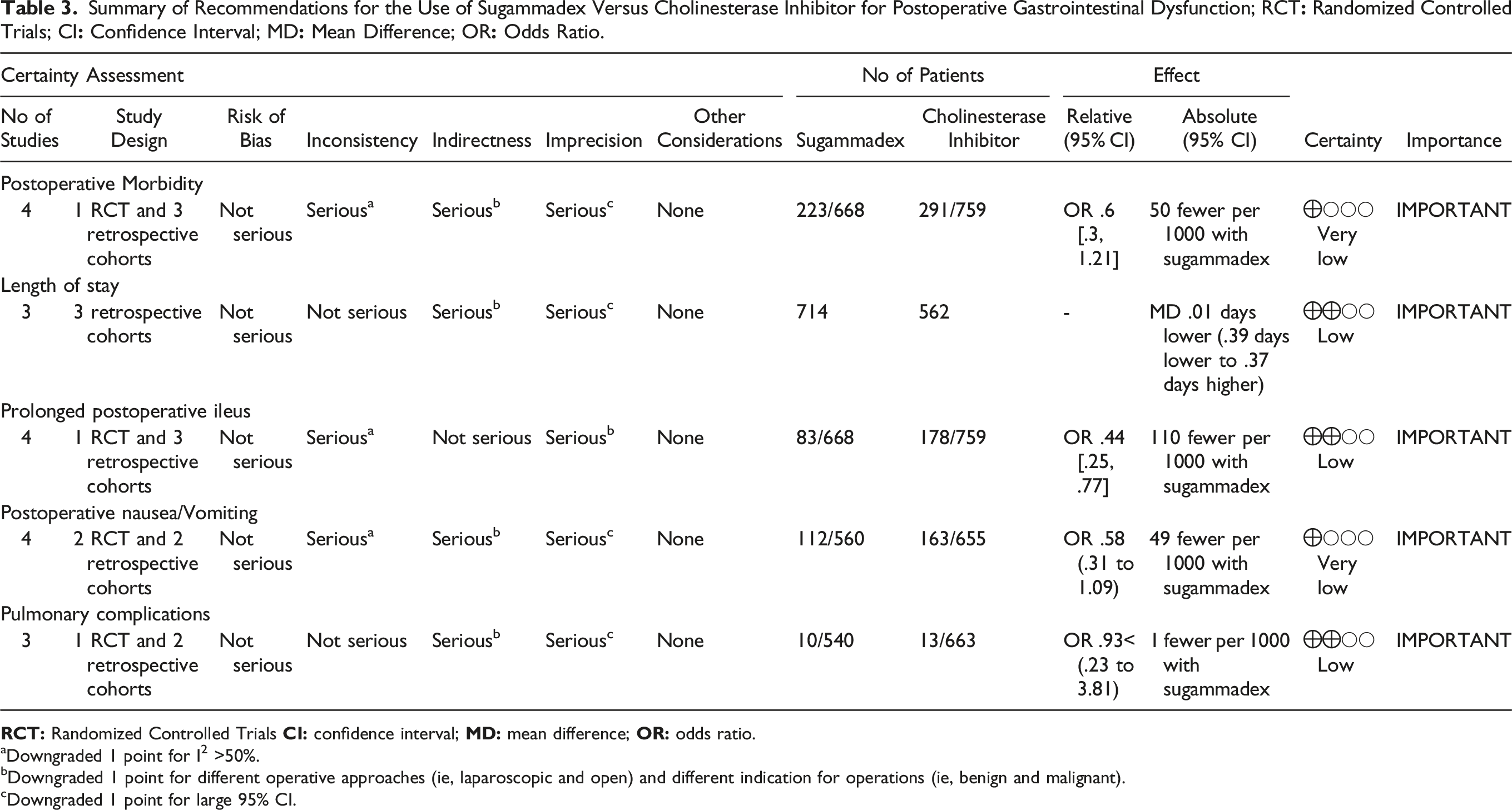
aDowngraded 1 point for I2 >50%.
Downgraded 1 point for different operative approaches (ie, laparoscopic and open) and different indication for operations (ie, benign and malignant).
cDowngraded 1 point for large 95% CI.
Discussion
POGD is prevalent among patients undergoing GI surgery, leading to increased morbidity, prolonged hospitalization, and higher health care costs. Pathophysiology of POGD is multifactorial and strategies for its prevention remain limited. 1 Traditional NMB reversal agents such as CIs, have been implicated as causes of POGD due to their influence on acetylcholine, a key neurotransmitter in GI motility.24-26 Sugammadex, a novel reversal agent that does not exert its effects via an acetylcholine pathway, is suggested to reduce POGD.10,12 There remains equipoise regarding the influence of sugammadex on POGD compared to CI in patients undergoing GI surgery.13,27 Our systematic review found a statistically significant lower incidence of PPOI (OR .44, 95% CI .25-.77, P < .05, I2 = 56%, low certainty evidence) in patients undergoing GI surgery receiving sugammadex compared to CI. However, this did not translate into a statistically significant difference in PONV, LOS, or morbidity.
Postoperative GI motility is influenced by both anesthetic and surgical factors. 28 Surgical risk factors for PPOI such as larger incision size and bowel handling during surgery are common during GI surgery. 28 Therefore, minimizing anesthetic risk factors remains critical. The initial studies comparing CI with sugammadex for PPOI were done in thyroid surgery with mixed results.13,27 Subsequent studies looking at intraperitoneal surgeries found consistent evidence supporting sugammadex over a broad range of procedures.12,29 Our systematic review looking at comparative studies in patients undergoing GI surgery is in keeping with these results. Our cohort included a wide variation in surgical modality (laparoscopic, open, robotic), anesthetic duration, malignant, and benign disease with all studies demonstrating a lower rate of PPOI in patients undergoing NMB reversal with sugammadex.
Pre-clinical studies suggest that development of PPOI after abdominal surgery is biphasic. The second phase, also known as the prolonged phase, begins 3 hours after intestinal manipulation with its duration dependent on the nature of surgery. This prolonged phase of PPOI is mediated through a cholinergic anti-inflammatory pathway. 30 Although CI independently are known pro-motility agents, it is unlikely that they influence this pathway due to their rapid onset of action and relatively short half-life. 31 Sugammadex also has little influence on this pathway due to its acetylcholine independent mechanism of action. 10 Therefore, the observed difference in PPOI is likely related to the coadministration of CI with AC, as AC are associated with delayed gut peristalsis and have a longer half-life than CI, leading to their effects predominating in the postoperative period.9,32
PONV occurs in up to 30%-70% of high risk patients with many patients considering it the most undesirable outcome of surgery.33,34 CI are hypothesized to lead to development of PONV due to their muscarinic effects on the GI tract and central receptors, with previous studies demonstrating an association with high dose neostigmine and postoperative PONV.35,36 Sugammadex, lacking this muscarinic activity is expected to lead to less PONV. No significant difference in PONV between the 2 reversal agents was found in our review (OR .58, 95% CI .31-1.09, P = .09, I2 = 68%, very low certainty evidence). This is consistent with a previous meta-analysis of 17 randomized clinical trials including various gastrointestinal and non-gastrointestinal surgeries with 1552 patients which was unable to conclusively demonstrate any difference in PONV between sugammadex and neostigmine. 37 However, subsequent small studies have demonstrated reduction in PONV with sugammadex within the first 6 hours postoperatively compared to CI.38,39 In our review, no study recorded PONV at different time points to elucidate temporal differences. This could suggest that differences in PONV are likely a function of CI avoidance rather than a benefit of sugammadex, with muscarinic effects of CI diminishing overtime leading to no difference in PONV after 6 hours. Further, PONV is influenced by several perioperative factors such as opioid use, intraoperative inhalant anesthetic, abdominal surgery, and emergent surgery. 34 Our review of patients undergoing GI surgery with varying indications for surgery and opiate use did not demonstrate any significant difference based on reversal agent used in PONV.
Meta-analysis of LOS and narrative review of readmission data did not demonstrate any difference between the 2 reversal agents in our review. Existing literature on the influence of sugammadex vs CI on LOS is inconclusive, owing to the multitude of factors which influence LOS other than the surgery itself.40-42 Tan et al, retrospectively reviewed 1614 patients who underwent abdominal surgery, of which 517 received sugammadex and 645 spontaneously recovered and found no difference in LOS. They found that open surgery and HPB surgery often led to increased hospital and PACU stays. 29 Conversely, TK Oh et al found a trend towards shorter hospital stay and lower 30-day readmissions in those who received sugammadex for NMB reversal following major abdominal surgery and Watts et al found sugammadex significantly reduced the LOS among all surgeries.43,44 In our cohort of 3 studies which reported LOS data, none found a significant difference in LOS between the 2 groups. However, Chae et al, did find a significantly lower incidence of delayed discharge in the sugammadex group, defined as an LOS greater than 11 days.19-21 This highlights the complexity of patient LOS and readmission with many surgical factors that can be controlled for in future studies, and many patient factors that are difficult to standardize. PACU stay may be a better variable for assessing efficacy of NMB reversal and less influenced by patient related factors. Previous data reporting PACU discharge times with sugammadex have been mixed. 45,46 Studies reporting reduction in PACU stay with sugammadex highlight its ability to reduce postoperative pulmonary complications which can often prolong PACU stay. 47 Our review found no significant difference in pulmonary complications following reversal between the 2 groups (OR .93, 95% CI .23-3.81, P = .92, I2 = 46, low certainty evidence). Only one study in our review reported PACU stay and found no difference. 19
The strengths of our study were derived from a focus on GI surgeries and inclusion of studies with a direct control group with CI, a rigorous risk of bis analysis, and a broad systematic search strategy. There are several limitations that would require future studies to address. First, of the five studies included, 3 were retrospective studies which can introduce an element of bias into our analysis. This was addressed by our risk of bias analysis which found low to moderate bias in all 3 studies. Due to the nature of observational studies, residual confounding factors that weren’t measured such as nutritional status and steroid use could be imbalanced between groups contributing to prognostic imbalance and thus biasing results. Second, our review had heterogeneity in the presentation and modality of surgeries included. Open and emergent surgeries are known to independently influence postoperative outcomes such as LOS, pulmonary complications, and morbidity.48-51 Third, the definition of these variables such as pulmonary complication and ileus was left to the discretion of the respective authors and varied greatly among studies. Fourth, the reversal dose used in each study, and inclusions of patients who required sugammadex as a rescue agent was not standardized. This likely reflects the different levels of NMB required for each study, which could be accounted for by including factors such as train of four analysis and postoperative residual curarization but was not done so in our analysis.
In summary, this systematic review and meta-analysis demonstrated a potential benefit in terms of PPOI for patients undergoing GI surgery receiving sugammadex compared to CI. However, there was no impact in terms of PONV, LOS, morbidity, or pulmonary complications. Large RCTs with standardization in measurement for clinically relevant outcomes, in addition to studies assessing cost effectiveness are required before routine use of sugammadex can be recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
