Abstract
Background
Lymph node ratio is the number of lymph nodes with evidence of metastases on pathological review compared to the total number of lymph nodes harvested during oncologic resection. Lymph node ratio is a proven predictor of long-term survival. These data have not been meta-analyzed to determine the prognosis associated with different lymph node ratio cut-offs in colon cancer.
Methods
Medline, Embase, and CENTRAL were systematically searched. Articles were included if they compared 5-year overall survival (OS) or disease-free survival (DFS) between different lymph node ratios for patients undergoing oncologic resection for stages I-III colon cancer. Pairwise meta-analyses using inverse variance random effects were performed.
Results
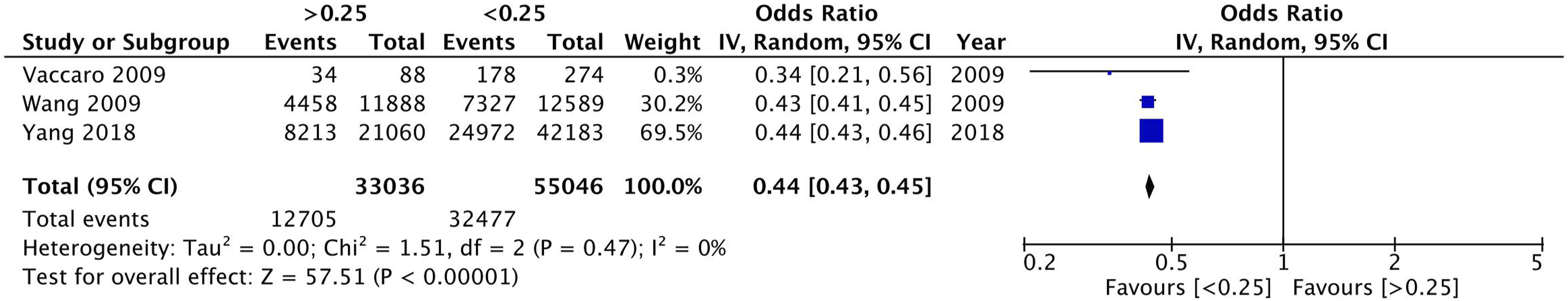
From 2587 citations, nine studies with 97,631 patients (female: 51.9%, median age: 61.65 years) were included. A lymph node ratio above .1 resulted in a 49% decrease in the odds of 5-year OS (2 studies; OR: 0.51, 95% CI: 0.49-.53, P < .00001). A lymph node ratio above .25 resulted in a 56% decrease in the odds of 5-year OS (3 studies; OR: 0.44, 95% CI: 0.43-.45, P < .00001). A lymph node ratio above .5 resulted in a 65% decrease in the odds of 5-year OS (3 studies; OR: 0.35, 95% CI: 0.33-.37, P < .00001).
Conclusions
Lymph node ratios from .1 to .5 are effective predictors of 5-year OS for colon cancer. There appears to be an inverse dose-response relationship between lymph node ratio and 5-year OS. Further study is required to determine whether there is an optimal lymph node ratio cut-off for prognostication and whether it can inform which patients may benefit from more aggressive adjuvant therapy and follow-up protocols.
Key Takeaways
• Lymph node ratios from .1 to .5 are effective predictors of 5-year overall survival for colon cancer. • There is an inverse dose-response relationship between lymph node ratio and 5-year overall survival. • Further study is required to determine whether there is an optimal lymph node ratio cut-off for prognostication.
Introduction
Colon cancer commonly metastasizes to other solid organs via hematogenous and lymphatic spread. 1 Lymphatic spread can be considered a proxy of cancer metastasis and can therefore be used to prognosticate outcomes. 2 Current National Comprehensive Cancer Network (NCCN) guidelines consider total number of positive lymph nodes in staging patients. 3 Cancer stage subsequently informs treatment decisions such as the need for adjuvant therapy and surveillance investigations. 4
Lymph node ratio is a prognostic indicator, defined by the ratio between the number of lymph nodes harboring metastatic disease on pathologic review compared to the total number of lymph nodes harvested. 2 Therefore, a higher ratio indicates increased number of positive lymph nodes relative to the total number of lymph nodes in the surgical specimen. Theoretically, the greater the lymph node ratio, the worse the prognosis. 5 A number of observational studies have demonstrated that lymph node ratio to be a prognostic factor for other cancers such as head-and-neck, 6 esophageal, 7 and breast. 8 These findings are also reflected in numerous colorectal cancer observational studies examining lymph node ratio and long-term survival.9–11 However, there is no consensus regarding the extent to which different lymph node ratio cut-off values predict long-term survival and how to use this value in conjunction with overall lymph node harvest. Nor is there a standard reference lymph node ratio value to predict prognosis. 2
A systemic review and meta-analysis in 2010 by Ceelen et al supported the use of lymph node ratio in colorectal cancer prognostication. 5 This study conducted a meta-analysis of the hazard ratios associated with lymph node ratios for overall survival (OS) or disease-free survival (DFS) and did not examine the use of different lymph node ratio cut-offs. 5 A systemic review and meta-analysis in 2020 by Karjol et al considered the prognostic effect of lymph node ratio on rectal cancer. 12 This study excluded colon cancer and meta-analyzed extracted hazard ratios for OS and DFS. There has yet to be a systematic review and meta-analysis aimed at determining the long-term prognosis associated with different lymph node ratio cut-offs in colon cancer. Moreover, numerous observational studies have been conducted since the most recent meta-analysis examining lymph node ratio in colon cancer patients. Therefore, the aim of this study is to evaluate the association of various lymph node ratio cut-offs on long-term survival for patients undergoing curative oncologic colonic resection for non-metastatic colon cancer.
Material and Methods
Search Strategy
The following databases covering the period from database inception through to November 2022 were searched: Medline, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL). Search terms included “colorectal cancer,” “lymphadenectomy,” “survival,” and more (complete search strategy available in Appendix 1). References of published studies and gray literature were manually searched to ensure that all relevant articles were included. This systematic review and meta-analysis is reported in accordance with Preferred Reporting items for Systematic Reviews and Meta-Analyses (PRISMA) 13 and the Meta-Analysis of Observational Studies in Epidemiology (MOOSE). The study protocol was registered on the PROSPERO International Prospective Register for Systematic Reviews a priori (ID: CRD42022338568).
Study Selection
Articles were eligible for inclusion if they compared 5-year OS or DFS between groups solely differentiated by the lymph node ratio following surgical resection for colon cancer. Exclusion criteria for this study are as follows: • Studies that solely evaluated patients with rectal cancer were excluded. A study including patients with both colon and rectal cancer was excluded if more than 50% of the included patients had rectal primaries in the given lymph node harvest cut-off analysis. • Studies including less than 10 patients were excluded. • Data from large, population level databases were carefully inspected. Significant chronologic overlap in data between studies was an indication for exclusion of one or more of the studies evaluating the same database. • Studies including stage IV patients were excluded. • Studies including patients undergoing emergent resection were excluded. • Non-English studies were excluded. • Abstracts, posters, opinions, case reports, reviews, meta-analyses, letters to editors, and editorials were excluded.
Outcomes Assessed
The primary outcome was long-term survival, reported as 5-year OS. 5-year OS was specified as the primary outcome as it the most common reported survival interval following colon cancer treatment. 14 Additionally, 5-year OS serves as the benchmark for long-term survival in colon cancer. 14
Secondary outcomes included 5-year DFS and the number of lymph node ratio cut-offs reported in the literature.
Data Extraction
Three reviewers independently screened the systematically searched titles and abstracts using a standardized, pilot-tested form. Discrepancies that occurred at the title and abstract screening phases were resolved by inclusion of the study. At the full-text screening stage, discrepancies were resolved by consensus between the three reviewers. If the disagreement persisted, a fourth reviewer was consulted. The same reviewers extracted data into a data collection form designed a priori. The extracted data included study characteristics (eg, author, year of publication, and study design), patient demographics (eg, age, gender, operation type, cancer histology, cancer location, and cancer stage), lymph node harvesting data (eg, lymph node ratio cut-offs), and oncologic outcome (ie, OS and DFS).
Risk of Bias Assessment
Risk of bias for each of the included observational study was assessed using the Methodological Index for Non-Randomized Studies (MINORS) tool. 15 Two reviewers assessed study quality for each study. Discrepancies were discussed amongst the reviewers until consensus was reached.
Statistical Analysis
All statistical analyses and meta-analyses were performed on STATA version 14 (StataCorp, College, TX) and Cochrane Review Manager 5.3 (London, United Kingdom). The threshold for statistical significance was set a priori at a P of <.05. A pairwise meta-analysis was performed using an inverse variance random effects model for all meta-analyzed outcomes. Studies were meta-analyzed according to their reported lymph node ratio cut-offs. For example, all studies reporting 5-year OS or DFS for patients above and below a lymph node ratio cut-off of .1 were included in the same meta-analysis, all studies reporting 5-year OS or DFS for patients above and below a lymph node ratio cut-off of .2 were included in the same meta-analysis, and so on. Pooled effect estimates were obtained by calculating the odds ratios (ORs) along with their respective 95% confidence intervals (CIs) to confirm the effect size estimation. Mean and standard deviation (SD) was estimated for studies that only reported median and interquartile range using the method described by Wan et al. 16 For studies that did not report a measure of central tendency, authors were contacted for missing data. Data were presumed to be unreported if no response was received from study authors within 2 weeks from the index point of contact. Missing SD data were then calculated according to the prognostic method. 17 Assessment of heterogeneity was completed using the inconsistency (I2) statistic. 18 An I2 value was subdivided into <25% (low), 25%–50% (medium), 50%-75% (high), >75% (substantial) for assessment of heterogeneity. 19 Bias in meta-analyzed outcomes was assessed with funnel plots when data from more than 10 studies were included in the analysis. 20 Meta-analyses were performed for all lymph node ratios cut-offs that were reported in 2 or more studies. 21
Results
Study Characteristics
From 2587 relevant citations, one prospective cohort study and 8 retrospective cohort studies met inclusion criteria. Four of the included studies were included in the quantitative synthesis. A PRISMA flow diagram of the study selection process is illustrated in Figure 1. In total, the included studies had 97,631 patients (51.9% female, median age: 61.65). The most commonly studied lymph node ratio was .25 (four studies), followed by a lymph node ratio of .1 (three studies), .5 (three studies), and .2 (two studies). There were three studies using large, population level databases (ie, SEER), and six independent, single-center studies. Detailed study characteristics are reported in Table 1. PRISMA diagram – Transparent reporting of systematic reviews and meta-analysis flow diagram outlining the search strategy results from initial search to included studies. Study Characteristics of Included Studies. N, number of participants; SD, standard deviation; LNR, lymph node ratio; SEER, Surveillance Epidemiology and End Results; USA, United States of America.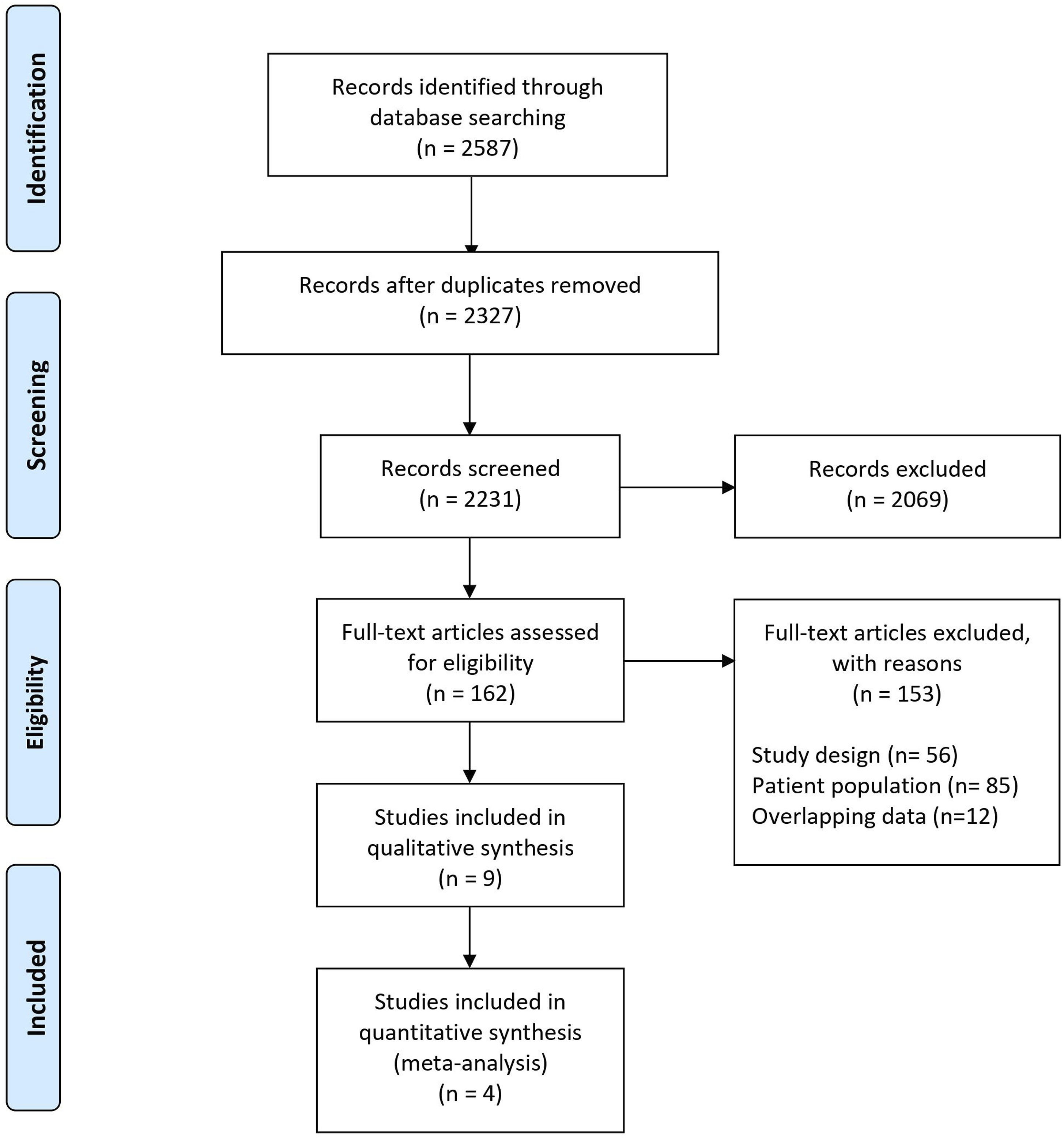
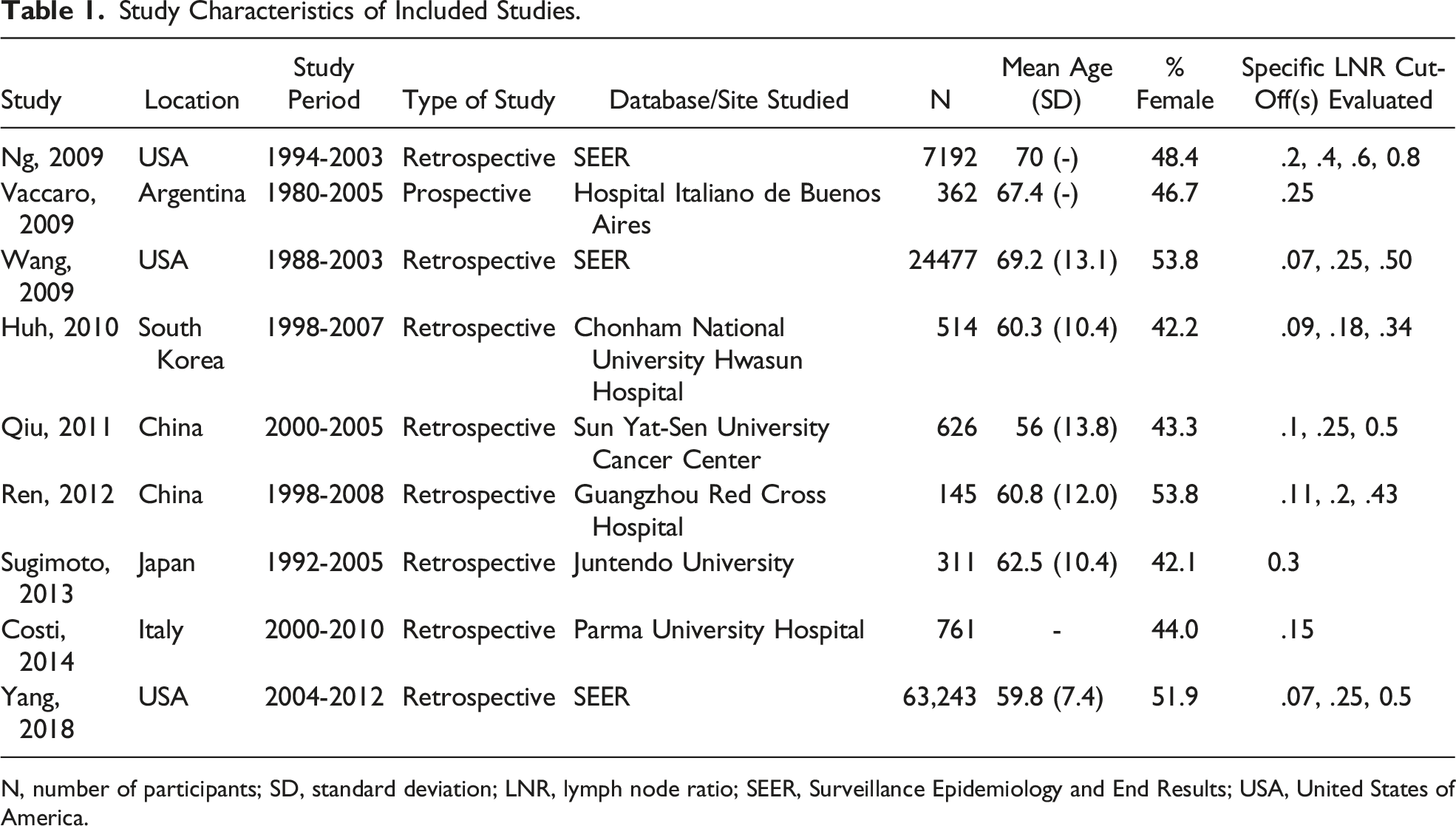
Disease Characteristics
Most included patients had right-sided colon cancers (n = 54,716, 56.94%). Of the remaining patients, 27,574 (28.69%) had left-sided colon cancer, and917 (.93%) had rectal cancers. Tumor location was not reported in 13.5% of patients. Of all studies reporting cancer stage, all included patients were stage III.
Tumor- and Treatment-Related Data for Included Studies.
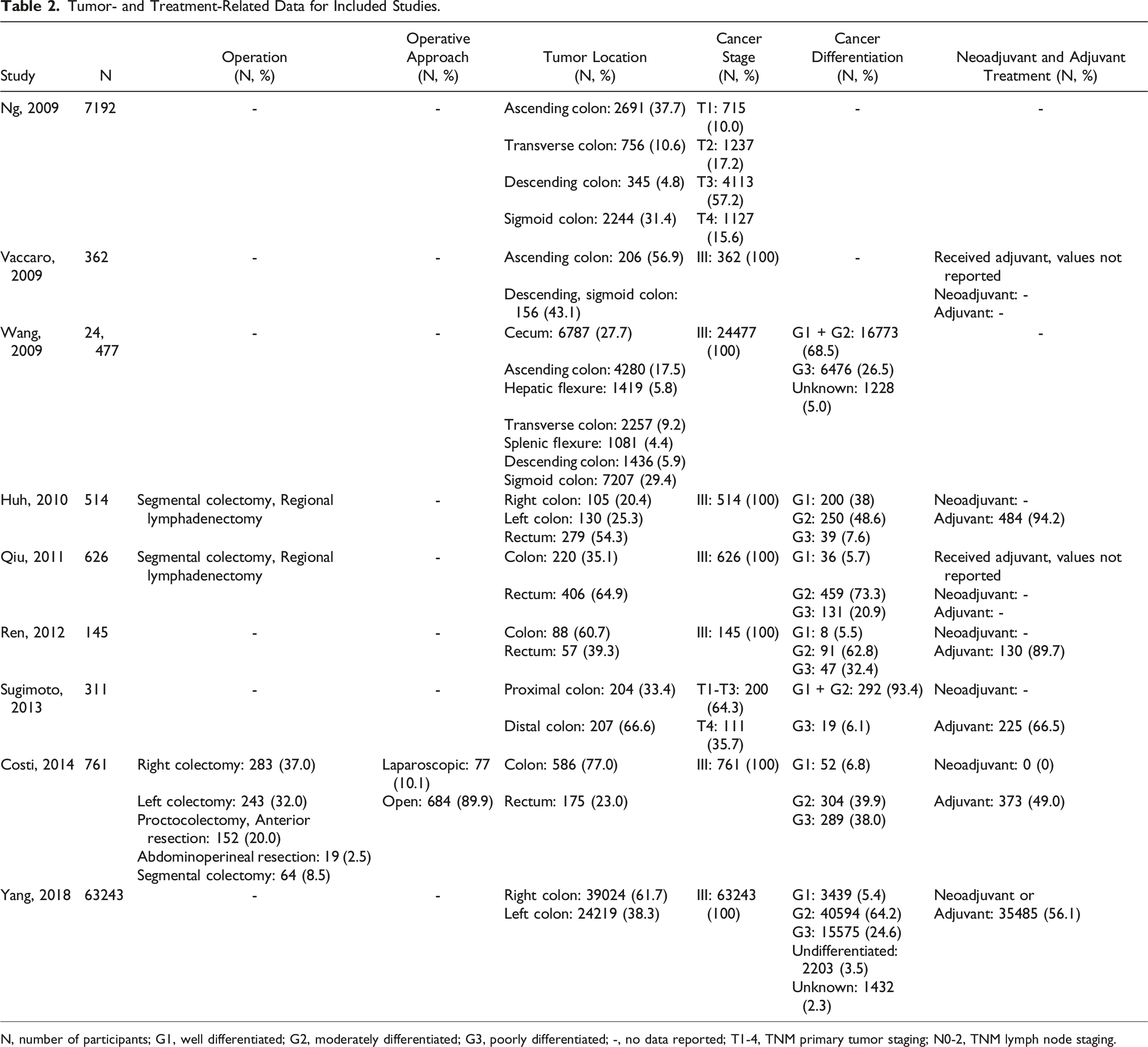
N, number of participants; G1, well differentiated; G2, moderately differentiated; G3, poorly differentiated; -, no data reported; T1-4, TNM primary tumor staging; N0-2, TNM lymph node staging.
Five-Year Survival Outcomes for Included Studies.
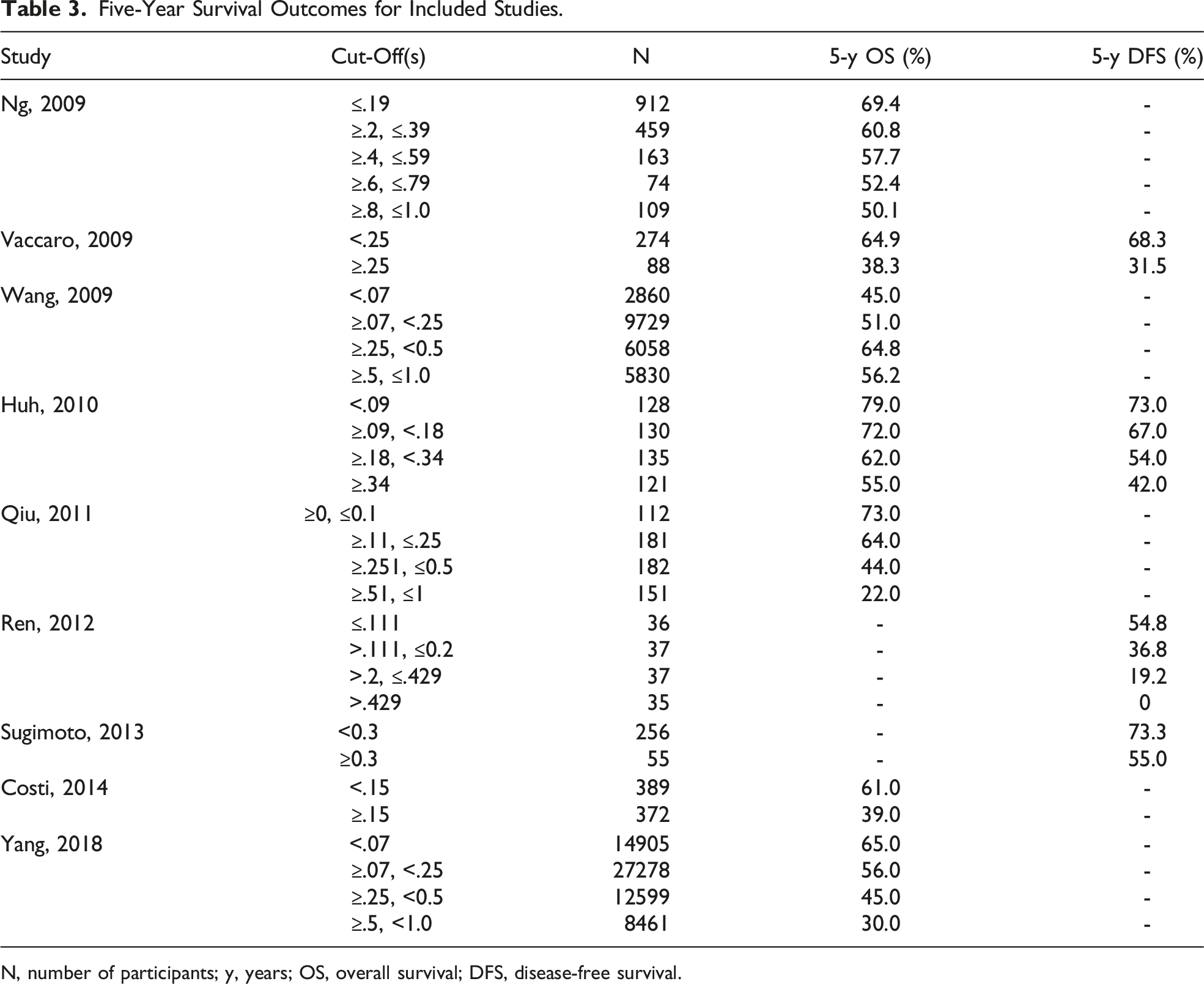
N, number of participants; y, years; OS, overall survival; DFS, disease-free survival.
Long-Term Oncologic Outcomes
Meta-analyses were possible for 5-year OS reported according to cut-offs at lymph node ratios of .1, .25, and .5. A lymph node ratio above .1 demonstrated a 49% decrease in odds of 5-year OS (2 studies; OR: .51, 95% CI: .49-.53, P < .00001, I2 = 0). A lymph node ratio above .25 demonstrated a 56% decrease in odds of 5-year OS (3 studies; OR: .44, 95% CI: .43-.45, P < .00001, I2 = 0). A lymph node ratio above .5 demonstrated a 65% decrease in odds of 5-year OS (2 studies; OR: .35, 95% CI: .33-.37, P < .00001, I2 = 56). The results of all pair-wide meta-analyses performed for the various lymph node ratio cut-offs (Figures 2–4).
Risk of Bias
Risk of bias analysis was completed according to the MINORS tool for each of the included studies. The mean MINORS score was 16.75 (±2.19). All 9 of the included studies stated the aim of their study appropriately in the manuscript, included consecutive patients, reported appropriate end points, had adequate follow-up periods, had adequate statistical models (ie, Kaplan-Meier curves), and had adequate control groups. One of the included studies evaluated prospectively collected data. None of the included studies calculated an a priori sample size, nor did any of the included studies report having blinded data outcome accrual and statistical analysis.
Discussion
Lymph node ratio is a prognostic indicator based on the percent of positive lymph nodes examined during oncologic resection for colon cancer. 2 This systematic review and meta-analysis analyzed the impact of various lymph node ratio cut-offs on long-term survival for patients undergoing curative oncologic colonic resection for non-metastatic colon cancer. Data was included from 9 studies evaluating various lymph node ratio cut-offs in terms of 5-year OS in colon cancer patients. Patients with lymph node ratios greater than .1 (OR: .51, 95% CI: .49-.53, P < .00001), .25 (OR: .44, 95% CI: .43-.45, P < .00001), and .5 (OR: .35, 95% CI: .33-.37, P < .00001) had a significant decrease in the odds of 5-year OS when compared to patients with lymph node ratios below the respective cut-offs. Altogether, any ratio greater than .1 can effectively predict long-term survival in patients undergoing oncologic resection for colon cancer. However, there is potentially an inverse dose-response relationship, with increasing lymph node ratio conferring a worsened 5-year OS.
Existing literature supports the use of lymph node ratio for colon cancer prognostication. The systematic review and meta-analysis by Ceelen et al in 2010 considered how lymph node ratios prognosticate colorectal cancer through analysis of hazard ratios. Ceelen et al examined 16 studies evaluating stage III colorectal cancer patients. 5 They pooled hazard ratios of six studies, with various lymph node ratio cut-offs, and demonstrated that increased lymph node ratio was associated with decreased OS (4 studies, HR: 2.36, 95% CI: 2.14-2.61, I2 = 79%) and DFS (4 studies, HR: 3.71, 95% CI: 2.56-5.38, I2 = 40%). 5 Similar findings were reported in a 2016 systematic review and meta-analysis by Zhang et al for OS (24 studies, HR: 1.91, 95% CI: 1.71-2.14, I2 = 60.5), and DFS (19 studies, HR: 2.75, 95% CI: 2.14-3.53, I2 = 71.7). 26 However, in both Ceelen et al and Zhang et al, different lymph node ratios were pooled rather than analyzed seperately.5,26 Individual lymph node ratios have been pooled and meta-analyzed for rectal cancer however. A 2020 systematic review and meta-analysis by Karjol et al pooled hazard ratios from OS and DFS for lymph node ratios of .1, .2, .3, and .6. They demonstrated that patients above each of the respective lymph node ratio cut-off experienced decreased OS and DFS; however, there was not a clear inverse dose-response relationship. 12 A similar analysis of individual lymph node ratios for colon cancer had yet to be completed until the present study.
Increased lymph node ratio has been repeatedly shown to predict long-term survival in various cancers.6–8,12 Selecting patients for adjuvant therapy based on lymph node ratio, or evaluating impacts of lymph node ratio on adjuvant therapy effectiveness, has been studied for others cancers like gastric, 27 pancreatic, 28 and esophageal. 29 As for colon cancer, Moug et al considered the value in lymph node ratio for staging colon cancer, and its implications on determining which patients would benefit from adjuvant chemotherapy. 30 Their data suggested that only patients with lymph node ratios between .05 and .19 had improved 5-year OS when receiving adjuvant chemotherapy (HR: .513, 95% CI: .343-.769, P < .001), 30 thereby exhibiting potential uses of lymph node ratios in adjuvant chemotherapy decisions for colon cancer.
The strengths of this systematic review and meta-analysis include rigorous methodology, a comprehensive risk of bias assessment, selective analysis of 5-year OS and DFS, inclusion of all identified lymph node ratio cut-offs, meta-analysis based upon specific lymph node ratio cut-offs, and a large number of patients (n = 97,631) in the included studies. The study limitations include a small number of studies per lymph node ratio cut-off, inclusion of only a single prospective study, and heterogeneity in methodologies between included studies. The few studies evaluating similar lymph node ratio cut-offs limited the meta-analysis, reduced statistical power, and lowered the confidence in the effect estimates. It is possible that the quantitative findings of the present review may significantly change as further data become available. Furthermore, the statistical heterogeneity I2 value was >50% for a lymph node ratio .5, thus highlighting potential inconsistencies in methodologies between studies. 18 Lastly, given that all the included data were observational, there are possible confounders impacting these findings such as cancer stage, tumor grade, tumor location, and individual patient factors (eg, smoking status, body mass index, and age) that were not accounted for. Unfortunately, we were unable to control for these data through subgroup analyses due to heterogeneity.
Conclusion
This systematic review and meta-analysis demonstrated that lymph node ratios greater than .1 can prognosticate 5-year OS for colon cancer, while providing survival data for specific lymph node ratios. Specifically, for patients with a lymph node ratio greater than the studied cut-offs (ie, .1, .25, and .5) 5-year OS was decreased by at least 50% relative to patients below that same cut-off. Moreover, long-term survival likely worsens with greater lymph node ratios. However, these findings are significantly limited by statistical power and heterogeneity. Further prospective study is warranted to determine optimal lymph node ratio cut-offs for prognostic use and if lymph node ratios may inform adjuvant therapy use in colon cancer. 0.1 lymph node ratio overall survival – Random effect meta-analysis comparing between a <0.1 and ≥0.1 lymph node ratio. 0.25 lymph node ratio overall survival – Random effect meta-analysis comparing between a <0.25 and ≥0.25 lymph node ratio. 0.5 lymph node ratio overall survival – Random effect meta-analysis comparing between a <0.5 and ≥0.5 lymph node ratio.

Footnotes
Authors’ Contributions
Conception and design of the study – all authors. Acquisition of data – Ichhpuniani, McKechnie, Lee, and Biro. Analysis and interpretation of data – all authors. Drafting and revision of the manuscript – all authors. Approval of the final version of the manuscript – all authors. Agreement to be accountable for all aspects of the work – all authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
