Abstract
Background
This study aimed to investigate the relationship between the ratio of negative lymph nodes (NLN) number to the number of metastatic lymph nodes (MLN) and the harvested lymph nodes (HLN) number ratio survival rate and compare its prognostic value.
Methods
This retrospective cohort study included 207 stage III colon cancer patients between 2010 and 2018 at a single center. NLN/MLN and NLN/HLN cut-off values were determined with the receiver operating characteristic (ROC) curve according to 5-year survival. The patients were divided into high-risk and low-risk groups according to the cut-off value. These 2 groups were evaluated according to the clinicopathological data of the patients.
Result
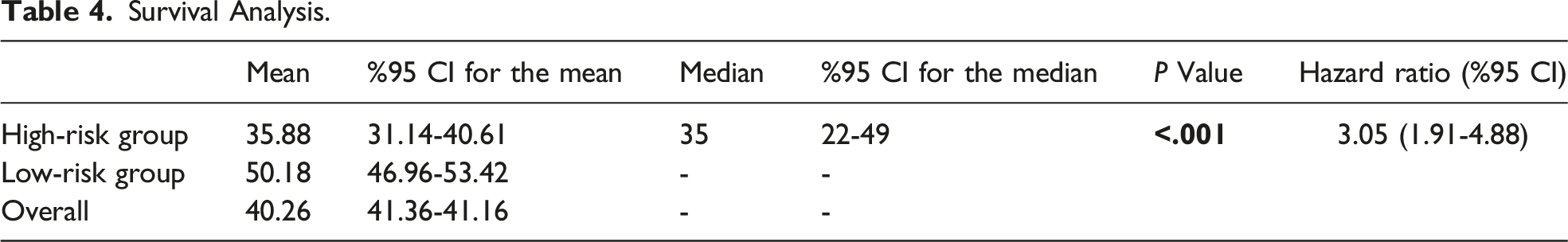
The time-dependent ROC curve showed the optimal cut-off values of NLN as 3.86 and .79, respectively. These values show 83 patients in the high-risk group and 124 in the low-risk group. There was no difference between the groups in tumor localization and T stage. According to Kaplan-Meier survival analysis, mean survival was 35.88 months in the high-risk group and 50.18 months in the low-risk group. The risk of death in the high-risk group was 305% compared to the low-risk group (Hazard Ratio: 3.05, 95% 1.91 – 4.88) (P < .001).
Conclusion
NLNs are among the critical prognostic factors in colon cancer. Although NLNs have a positive correlation with the survival rate of the patients, there is no statistical difference in tumor T stage and localization.
Introduction
According to the GLOBOCAN 2020 reports, colorectal cancers are the third most common cancer among cancers and second in cancer-related deaths, and 1.93 million people are diagnosed with colorectal cancer annually, and 930 000 people die from colorectal cancers. 1 However, in non-metastatic colorectal cancers, complete resection of the tumor with lymphatic tissues is the most effective treatment for early-stage colon cancer. 2 In addition, it provides essential information about the prognostic and predictive values of the disease with +mesocolic excision. 3
The tumor-node-metastasis (TNM) staging system recommended by the American Joint Committee on Cancer (AJCC) constitutes the most crucial step in patient management after appropriate surgery, and according to this staging system, metastatic lymph node (MLN), after dissection of at least 12 lymph nodes with pN stage determined by the number. 4 However, the determination of the number of lymph nodes varies according to the methods of the pathologist in obtaining the lymph nodes, primarily depending on the surgical technique.5,6
In today's medicine, more is needed to evaluate the lymph node stage only according to the number of MLN, mainly due to molecular pathology and genetic developments. For this reason, new staging systems have been proposed, including the harvested lymph nodes (HLN) dissected in pN evaluation. 7 These include the lymph node ratio (LNR) 8 and log odds of positive LNs (LODDS). 9 Furthermore, it has been reported that LNR and LODDS are independent prognostic factors in colorectal cancers.10-13
While it does not provide information about the prognosis in LNR node-negative patients, In LODDS, only positive and negative lymph nodes (NLN) were evaluated. However, there is no system to evaluate the prognostic significance of isolated NLN. A more significant number of NLN may indicate a lower probability of occult lesions and is associated with a better prognosis. 14 However, there are not enough studies on the prognostic importance of NLN in colon cancer. Therefore, our study aims to examine the prognostic significance of NLN in stage III colon cancers in line with the patients' clinicopathological data.
Material and Method
Patient selection
Patients who were operated on with the diagnosis of colon cancer in Mersin University Faculty of Medicine, Department of General Surgery between January 2010 and January 2018 were retrospectively analyzed. Patients with a colon cancer diagnosis, who underwent curative surgery, had a stage III pathology report, and had complete data were included in the study. Patients with rectum cancer, palliative surgery, non-stage III, other malignancy, recurrent disease, harvested lymph nodes dissection of less than 12 nodes, and missing data were excluded from the study. The study was designed for colon tumors only; rectal tumors were excluded. Rectal cancer was excluded from the study because preoperative systemic (chemotherapy/radiotherapy) treatment can be applied, and the treatment may affect the lymph nodes.
Data collecting
Demographic data (age, gender), comorbid disease, American Society of Anesthesiologists (ASA) score, tumor location, operation timing, operation time, length of hospital stay, postoperative complication, pathology result, tumor stage, and overall survival (OS) were recorded. In addition, the presence of postoperative complications was recorded. Complications were classified according to the severity of the complications. Complication severity was classified using the Clavien-Dindo scale.
The histopathology of the tumor, TNM staging, HLN, MLN, and NLN count, tumor stage, degree of differentiation, and perineural and lymphovascular invasion were recorded in the pathology report. In addition, the K-ras mutation was examined in immunohistochemical examination. Finally, tumor staging was done according to AJCC.
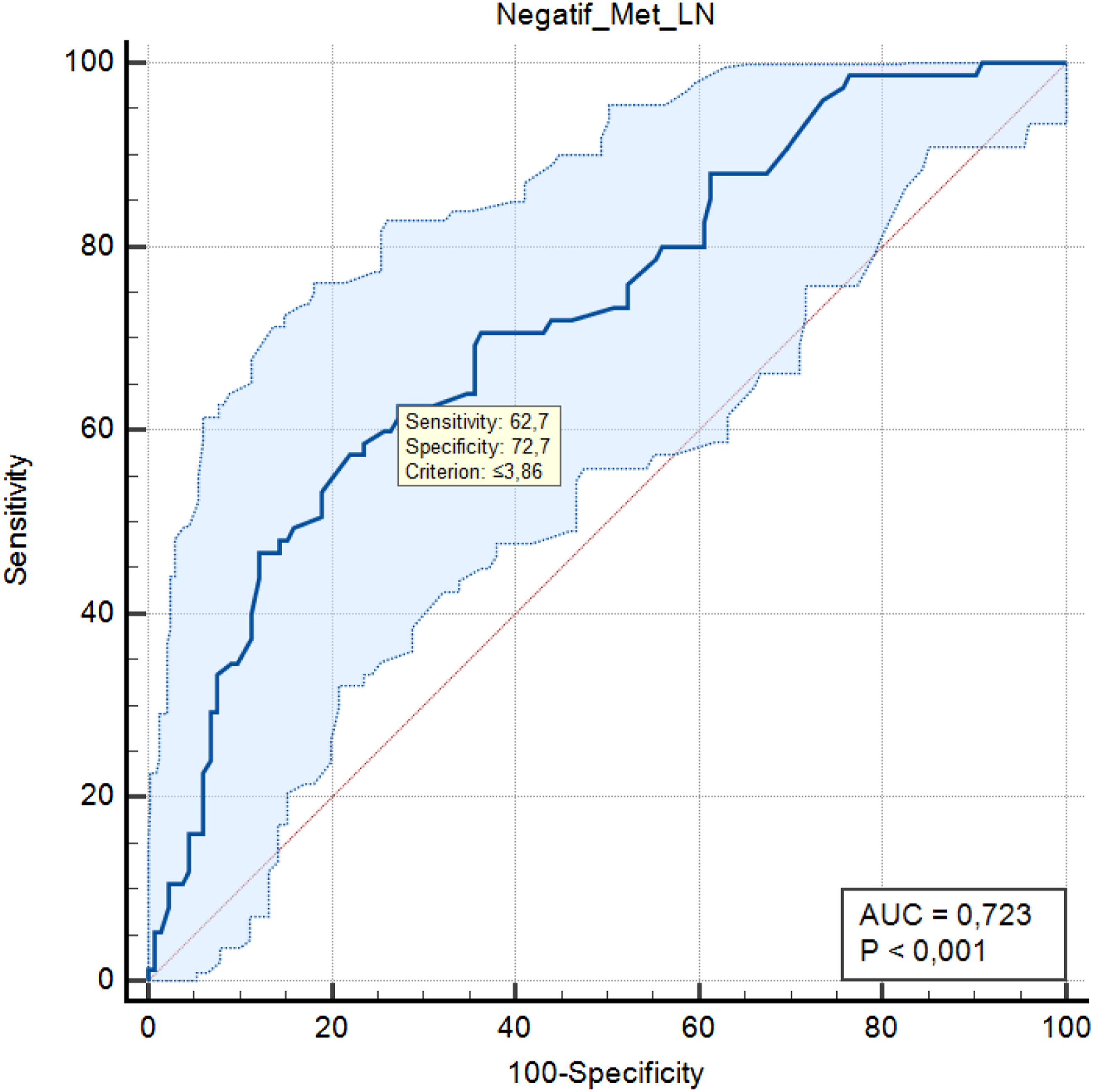
Study design
HLN, MLN, and NLN counts were recorded from the pathology report. NLN/MLN and NLN/HLN ratios were calculated for each patient. In order to calculate the 5-year OS, the study was limited to the 2018 patient recruitment period. Five-year OS of the patients was calculated. ROC analysis and ROC curve were created to establish the cut-off value in lymph node ratios. The cut-off value of NLN/MLN and NLN/HLN was determined according to the 5-year disease-free survival (DFS). NLN/MLN and NLN/HLN cut-off values were parallel. Cut-off values were ≤3.86 for NLN/MLN and ≤.79 for NLN/HLN, with 62.7% sensitivity and 72.7% specificity (Figures 1 and 2). The patients were divided into 2 groups as lymph node cut-off values were lower (≤3.86 or ≤.79) (high-risk group) and higher (>3.86 or >.79) (low-risk group) and compared. Negative lymph node cut-off values by 5-year survival are shown in Table 1 The time-dependent receiver operating characteristic (ROC) curve of NLNs/Total LNs for 5-year survival. Area under the ROC curve in NLNs/Total LNs: .724, P < .001. The time-dependent receiver operating characteristic (ROC) curve of NLNs/Metastatic LNs for 5-year survival. Area under the ROC curve in NLNs/Total LNs: .723, P < .001. Negative Lymph Node Cut-Off Values By 5-Year Survival. AUC: area under curve, NLN: negative lymph nodes, MLN: metastatic lymph nodes, HLN: harvested lymph nodes.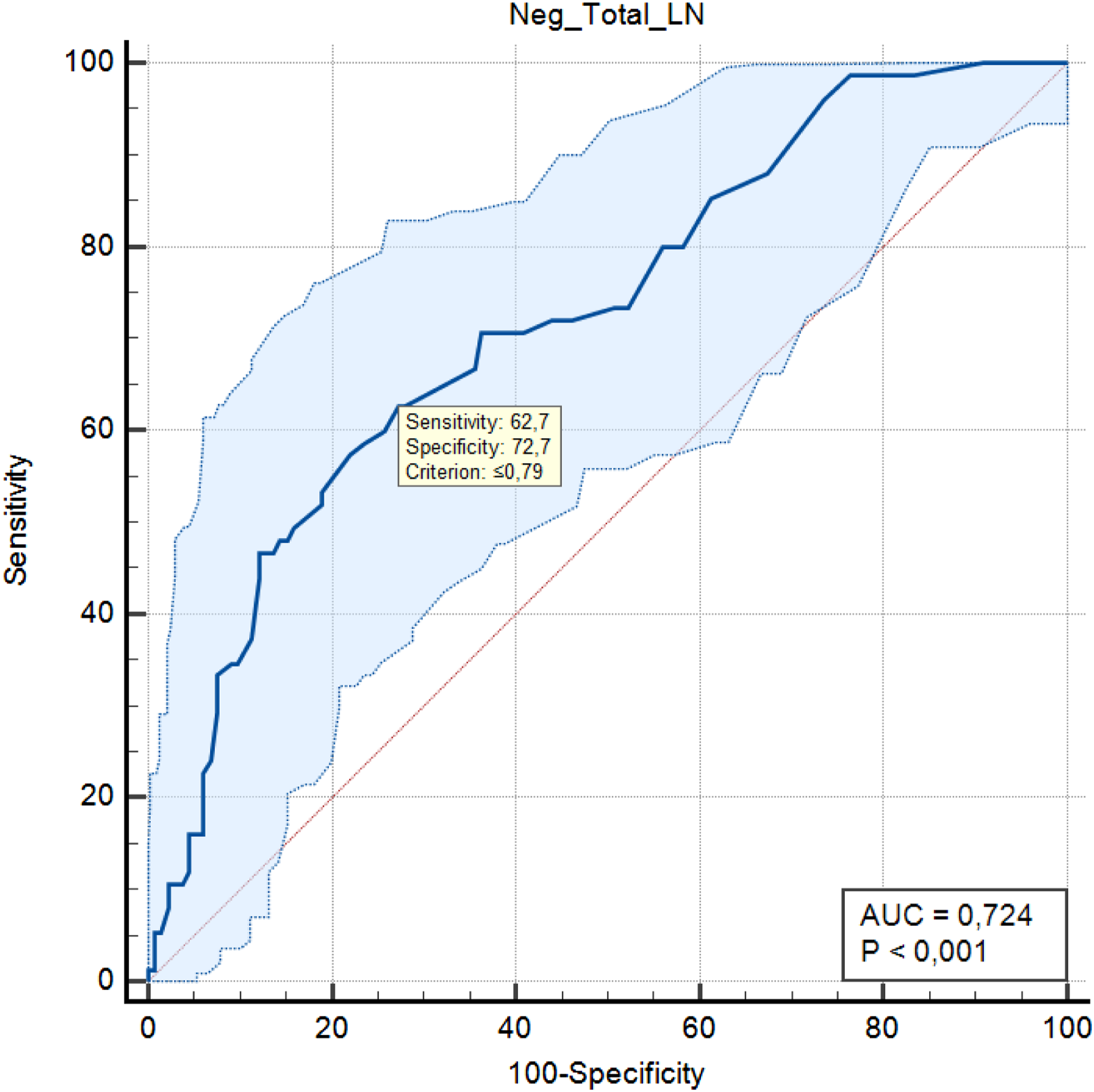

Statistical Analysis
The data analysis used mean, median, standard deviation, minimum and maximum values while performing the continuous data statistics on the scales. Frequency and percentage values were used to define categorical variables. ROC was determined by curve analysis to determine the cut-off in the lymph node ratio measurements in predicting 5-year OS. Significance in ROC analysis was determined by sensitivity, specificity, likelihood ratio (+), and likelihood ratio (-) statistics. Studentst test was used to compare the means of 2 groups in continuous measurements. The Mann-Whitney U test compared the 2 groups for non-normally distributed and ordinal variables. The chi-square test was used to evaluate the relationship between categorical variables. OS curves were estimated by the Kaplan-Meier method. Differences according to the group were determined by the Log-Rank test. The statistical significance level of the data was taken as P < .05. In the evaluation of the data, www.e-picos.com and MedCalc statistical package programs were used. Survival curves of stage III patients according to the Negative/Total LNs and Negative/Metastatic LNs.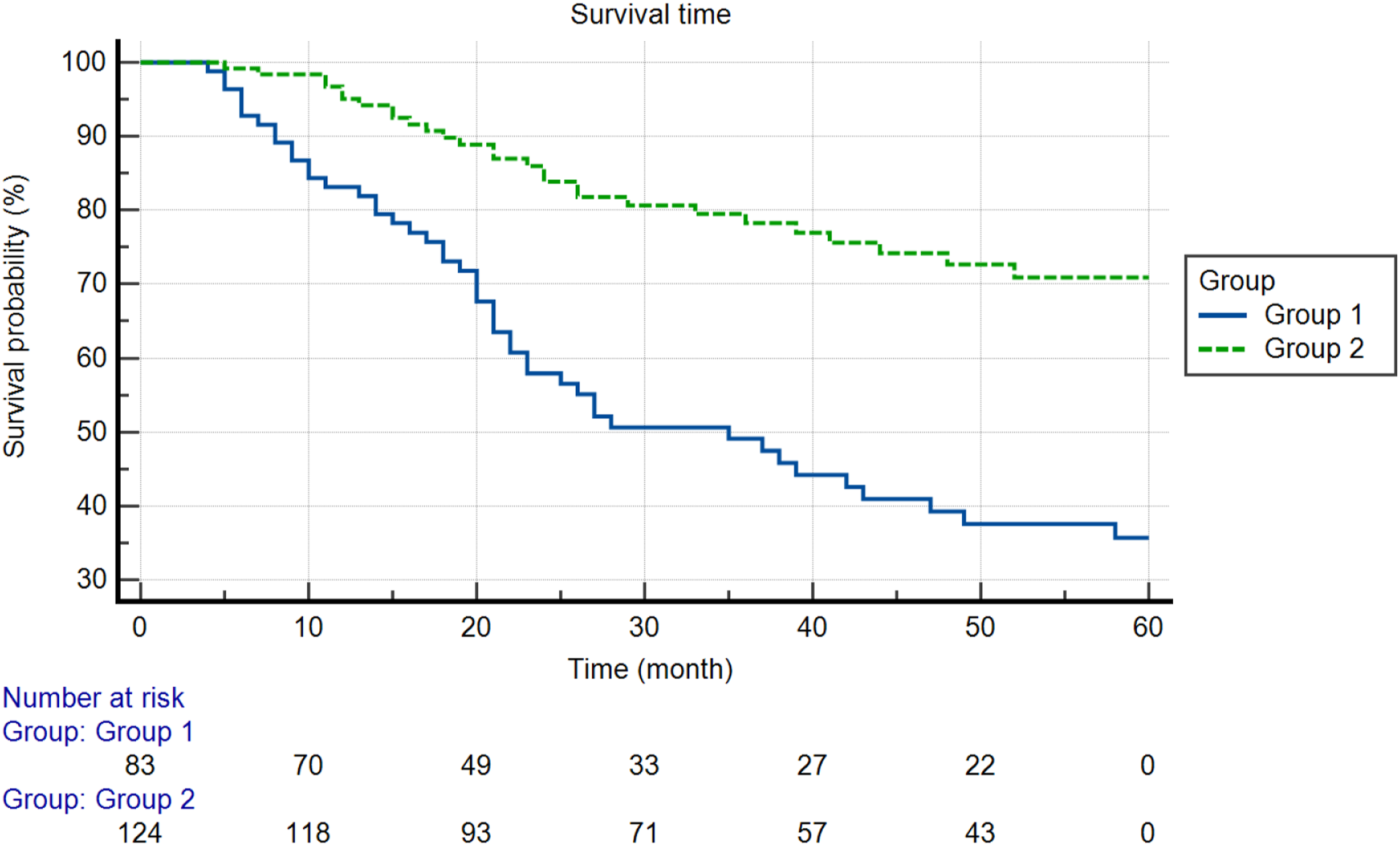
Results
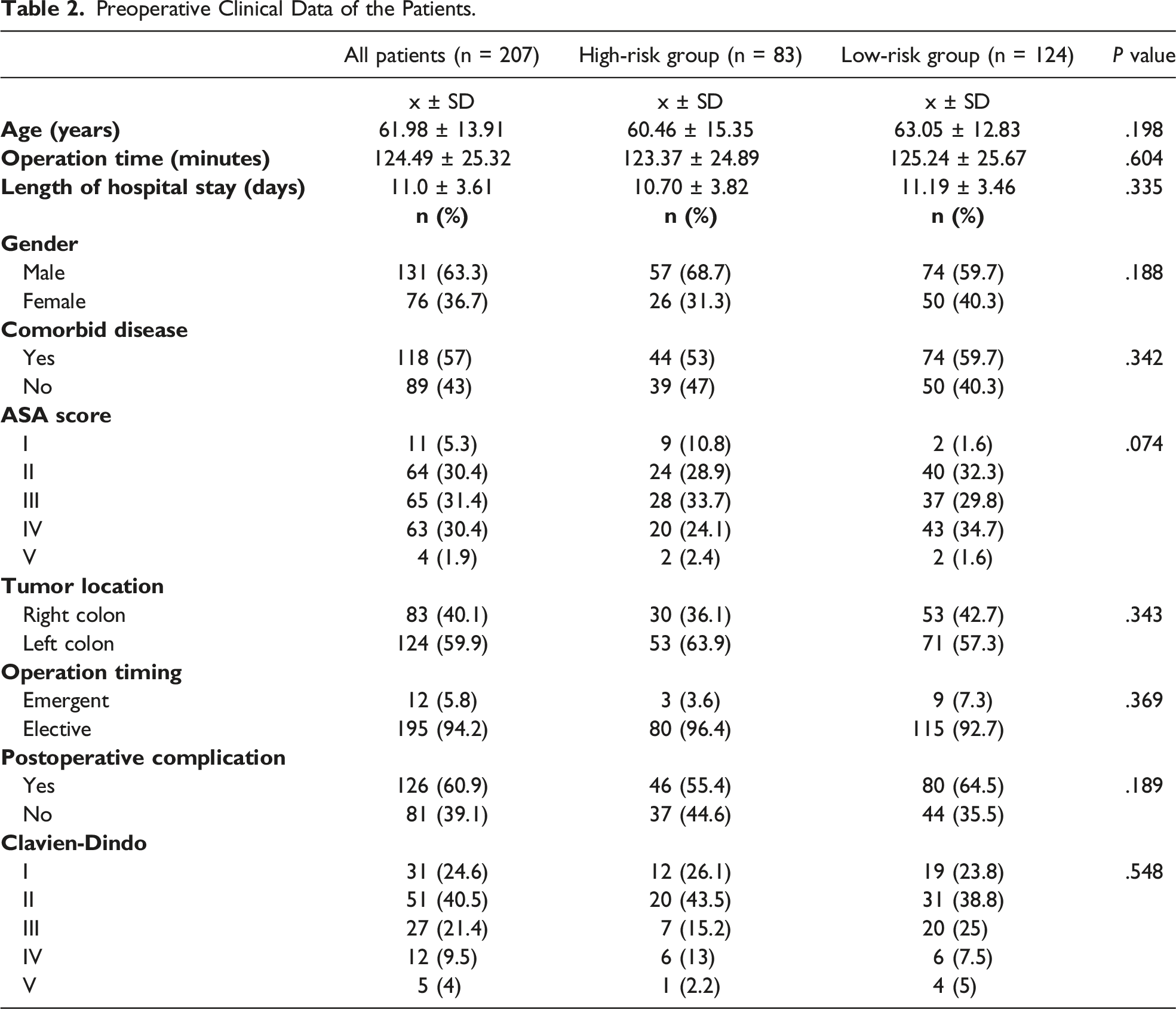
Preoperative Clinical Data of the Patients.
Histopathological Data of the Patients.
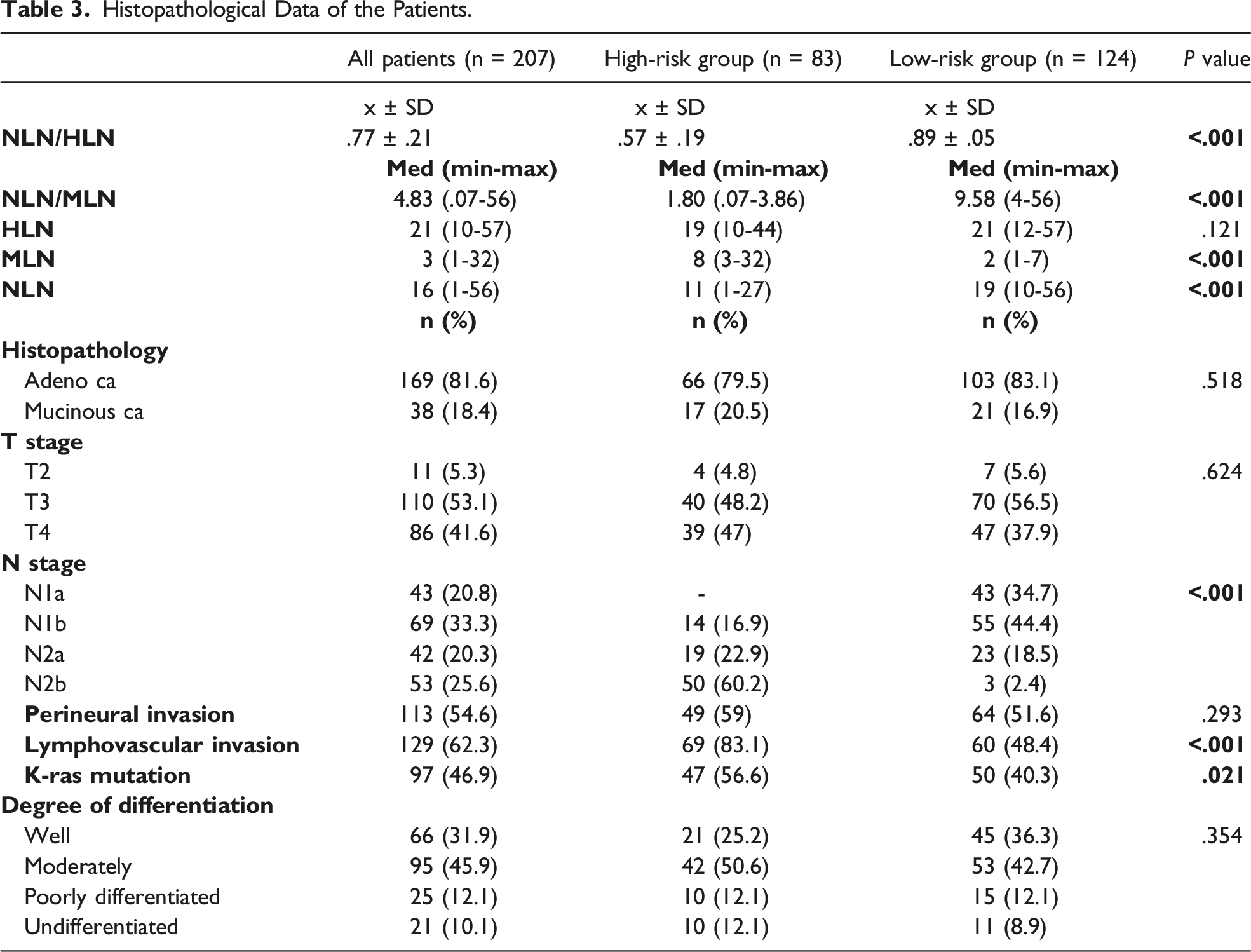
The median HLN was 19 (10-44) in the high-risk group and 21 (12-57) in the low-risk group. The median value of MLN was 8 (3-32) in the high-risk group and 2 (1-7) in the low-risk group. The median value of NLN was 11 (1-27) in the high-risk group and 19 (10-56) in the low-risk group. The median NLN/MLN value was 1.80 (.07-3.86) in the high-risk group and 9.58 (4-56) in the low-risk group. The mean of NLN/HLN was .57 ± .19 in the high-risk group and .89 ± .05 in the low-risk group. There was no statistically significant difference between the groups regarding HLN count (P = .121). NLN/HLN mean, NLN/MLN ratio, and the median value of the MLN - NLN were statistically lower in the high-risk group than in the low-risk group (P < .001). (Table 3).
Perineural invasion was present in 49 (59%) patients in the high-risk group and 64 (51.6%) in the low-risk group. Lymphovascular invasion was present in 69 (83.1%) patients in the high-risk group and 60 (48.4%) patients in the low-risk group. K-ras mutation was present in 47 (56.6%) patients in the high-risk group and 50 (40.3%) in the low-risk group. There was no significant difference between the groups regarding the degree of perineural invasion and differentiation (P = .293, P = .354, respectively). However, the frequency of lymphovascular invasion and K-ras mutation was statistically significantly higher in a high-risk group than in the low-risk group (P < .001, P = .021, respectively) (Table 3).
Survival Analysis.
Discussion
In the current study, it has been shown that the number of NLN is in direct relationship with the survival rate in stage III colon cancer patients. However, there was no correlation between the number of NLN and tumor T stage and tumor localization.
Lymph node status is among the most important prognostic markers in colon cancer and provides critical information about prognosis. 15 However, considering the inadequacy of the current lymph node staging system, alternative methods have emerged.16-18 Studies conducted in recent years have revealed the importance of the number of NLNs in various cancers.19,20 In a study in which clinical data of 20 702 patients with stage III colorectal cancers were analyzed, the effect of NLN on prognosis was shown. 21
The relationship between increased NLN and more prolonged survival has yet to be fully understood. In addition, HLN may vary depending on the patient's age, the tumor grade, the molecular characteristics of the tumor, and the location.22-24 This situation directly affects the number of NLNs. According to a hypothesis, it is thought that in patients with high anticancer immunity, a robust reactive response to the tumor, increased NLN number, and a stronger tumor immunity is thought to affect survival positively. 25
In recent years, studies have shown the effect of NLN number on prognosis in various malignancies. However, there is no accepted classification system for the NLN optimal ratio and cut-off values, and different statistical methods and threshold values have been determined in different studies.26,27 Therefore, most studies on the prognostic significance of NLN were evaluated only according to the number of NLN. For example, in a study, the NLN number was taken as the cut-off value of 9; right colon and left colon tumors were compared, and as a result, the NLN count had a positive prognostic effect. 28 Similarly, in a study on stage III gastric cancer patients, patients were grouped by taking the cut-off value of NLN count of 9. Survival analyses showed that NLN count was significantly associated with OS (P = .001) and was an independent predictor of prognosis in stage III patients. (P < .01). 29
A study on right colon cancers found no relationship between increased lymph node number and recurrence-free survival, regardless of MLN status. In addition, in this study, the relationship between the increased number of lymph nodes and OS could not be demonstrated in the case of nodal metastasis. 30 However, in Le Voyer et al’s study, the positive effect of an increasing number of NLNs on DFS and OS was shown in multivariate analysis. 31 In our study, when low-risk and high-risk groups were evaluated, no correlation was found between tumor localization and the T stage of the tumor.
In our current study, not only the number of NLN but also the number of NLN, MLN, and HLN were evaluated, their ratios were evaluated by survival analysis, and cut-off values were created. NLN/MLN cut-off value of .79; NLN/HLN cut-off value was determined as 3.86, and low and high-risk groups were formed according to these values. There is a significant difference in survival in the high-risk group compared to the low-risk group (P < .001, 95% CI = 3.05 (1.91-4.88)).
Our study could not elucidate the mechanism underlying the relationship between NLN and survival. However, it reveals the prognostic importance of the NLN. Therefore, interactions between tumor and host are thought to increase lymph node proliferation, increasing both metastatic and negative lymph node counts. However, the number of negative LNs can be considered indicative of host defense based on the indicator of the host's immune response to the tumor as a marker of underlying tumor-host interactions, which may have an independent effect on survival. 32 In addition, however, NLN count may be an indicator of the adequacy of lymph node dissection. It may give an idea about the competence of the surgeon and pathologist concerning this.
There are several limitations to our study. The first is that the study was designed as a single center and retrospective. In addition, the small number of patients included in the study is another limitation. Another limitation is the inclusion of genetic factors that affect the prognosis of colon cancer is among the crucial limitations. Finally, another limitation is that DFS needed to be evaluated. However, the study's major strength is that it is a rare study in which the effect of the ratio of NLN to MLN and HLN dissected was evaluated together.
Conclusion
Our study has shown that; the number of NLN is among the critical prognostic factors in stage III colon cancer, and there is a positive correlation with the survival rate. However, there was no correlation between the number of NLN and tumor T stage and tumor localization. However, the positive correlation with the survival needs to be clearly explained. For this, prospective studies in which genetic and immune parameters are included in patients with more patient populations are needed. These mechanisms will lead to significant changes in tumor staging systems when thoroughly explained.
Since the effect of NLN number is associated with lower metastatic foci as the number of NLN increases, it has prognostic significance. Therefore, when used with the current TNM classification, the number and ratio of NLN can be helpful in predicting prognosis, especially in stage III colon cancer patients.
Footnotes
Acknowledgments
This article has been accepted as an oral presentation to the 2023 International Congress of Turkish Colon and Rectum Surgery.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
