Abstract
Background
With a demonstrated association between adiposity and parathyroid hormone (PTH) levels, we hypothesized that patients with a higher body mass index (BMI) would have lower rates of postoperative hypoparathyroidism following total thyroidectomy.
Methods
retrospective review of patients undergoing total thyroidectomy from 2015 to 2021. Demographics, BMI, surgical indications, and laboratory data including pre- and postoperative PTH values were examined.
Results
Of the 352 patients with complete clinicopathologic data, most were female (n = 272, 77.3%) with an average age of 42.7 (SD+/−19.4). Obese (BMI 30-39.99) was most common BMI group (n = 108, 30.8%), with 11.7% (n = 41) morbidly obese (BMI > 40). Morbidly obese patients had significantly higher postoperative PTH levels than BMI < 18.5 (46.0 vs 19.3 pg/mL, P = .004). Patient race was significantly associated with pre- and postoperative PTH (P = .03, P = .004.) On multivariable analysis, preoperative PTH, race, and BMI were independent predictors of higher postoperative PTH (P < .05 for all).
Discussion
Patients with higher BMI and non-white race have relative protection from postoperative hypoparathyroidism.
Key Takeaways
• Patients with higher BMIs have a relative protection from postoperative hypoparathyroidism independent of other factors including surgical indication, number of parathyroid glands visualized during surgery, or vitamin D levels. • Race has a statistically significant relationship with the development of postoperative hypoparathyroidism following thyroid surgery. • Preoperative PTH values, BMI, and race were all independent predictors of postoperative PTH levels on multivariable analysis.
Introduction
With a prevalence in the United States of 37 per 100,000, hypoparathyroidism is a rare diagnosis 1 but most commonly occurs following anterior neck surgery (75% of all cases) with the incidence after surgery being as high as 8% in one study.2,3 Postoperative etiologies of hypoparathyroidism include inadvertent trauma, devascularization, or unplanned removal of parathyroid glands during surgery with risk factors including extensive neck dissection, bilateral neck exploration, re-operative surgery, pediatric or hyperthyroid patients, and patients with malabsorptive conditions such as a history of bariatric surgery.4,5 Most postoperative hypoparathyroidism is transient with a range of 2-7.3% being persistent past 6 months across several studies.1,6,7
Parathyroid hormone (PTH) regulates calcium and phosphate levels through multiple processes in the body. Parathyroid hormone stimulates bone resorption, increases calcium reabsorption while decreasing phosphate reabsorption in the kidneys, and increases both calcium and phosphate absorption in the intestines. A PTH deficiency, consequently, results in hypocalcemia, hyperphosphatemia, and hypercalciuria. Hypoparathyroidism has multiple deleterious effects on the body including basal ganglia calcifications, cataracts, potential arrhythmias, tetany, and multiple neuropsychiatric symptoms ranging from vague complaints of fatigue and brain fog to seizures. 8
An association has been demonstrated between adiposity and parathyroid hormone with PTH levels increasing as adiposity increases. 9 A number of reasons may explain this association, including PTH inhibition of lipolysis and promotion of insulin resistance, as well as adiposity lowering vitamin D levels through sequestration through fat tissue.9-11 This relationship between lower vitamin D with higher PTH has also been noted to be more prevalent in African American and Hispanic patients.12,13 Given these relationships, we hypothesized that patients with a higher body mass index (BMI) would have lower rates of postoperative hypoparathyroidism following total thyroidectomy. As a secondary goal, we wanted to examine the relationship of race and postoperative PTH values.
Methods
Our Institutional Review Board reviewed and approved this study protocol. Using a prospectively maintained database, we performed a retrospective review of adult and pediatric patients undergoing total thyroidectomy by five high-volume endocrine surgeons from 2015 to 2021. Patients were excluded for missing BMI or postoperative PTH data points, undergoing completion thyroidectomy, or concurrent parathyroid surgery. Patient age, gender, race, BMI, surgical indication, and laboratory values for vitamin D, preoperative PTH (pre-PTH), and postoperative PTH (post-PTH) were examined. Surgical indications included an enlarging gland, nodule, diagnosis of malignancy, and Graves’ disease. The number of parathyroid glands seen during the operation, the number of parathyroid glands autotransplanted, and surgeon performing the operation were also examined. Parathyroid hormone reference range was 12-88 pg/mL with hypoparathyroidism defined as PTH < 12 pg/mL. Calcium reference range was 8.4-10.4 mg/dL with hypocalcemia defined as Ca < 8.4 mg/dL.
At our institution, a PTH value is routinely obtained while the patient is in the post-anesthesia care unit (PACU) and is used to determine the discharge calcium supplementation plan. Graves’ disease patients and all patients with PTH values between 10 and 19 pg/mL are instructed to take 1000 mg calcium carbonate three times daily. For PTH values less than 10 pg/mL, the patient is discharged with calcium carbonate and calcitriol. Calcium citrate is used instead of calcium carbonate in patients taking acid-reducing medication and in those with a history of gastric bypass surgery. All supplementation is weaned after the patient is seen in clinic 2 weeks postoperatively if laboratory studies are normal.
Our primary outcome of interest was the relationship between BMI and postoperative PTH, with below reference range postoperative PTH used to define postoperative hypoparathyroidism. Calcium lab values were not included in this definition. Secondary outcomes included the relationship of postoperative PTH with race, surgeon, indication for surgery, and number of glands identified or autotransplanted during the operation. Bivariate analysis was conducted for ordinal and categorical variables with Kruskal-Wallis and Chi-square tests, respectively. A multivariable linear regression model was also constructed to identify independent predictors of postoperative PTH. Analysis was performed using Stata 17 (StataCorp, College Station, TX).
Results
Patient Demographics.
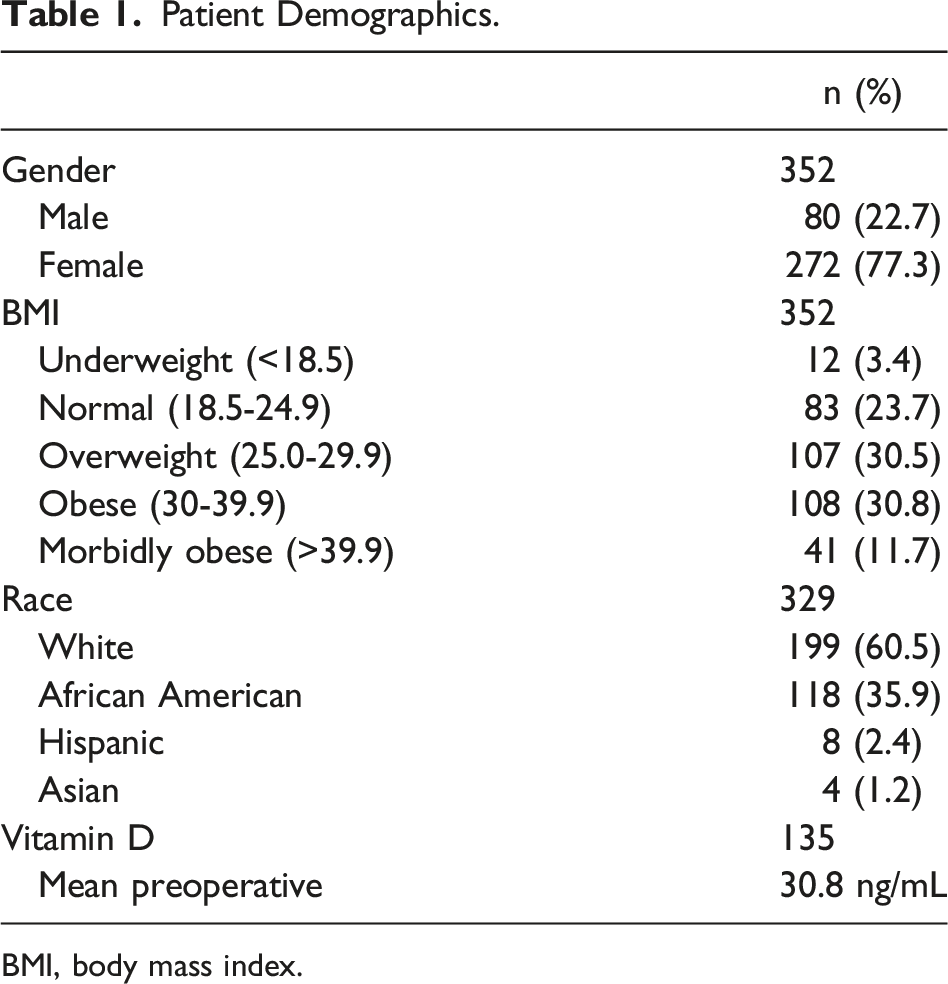
BMI, body mass index.
PTH Values and Hypocalcemia Symptoms by BMI Category and Race.
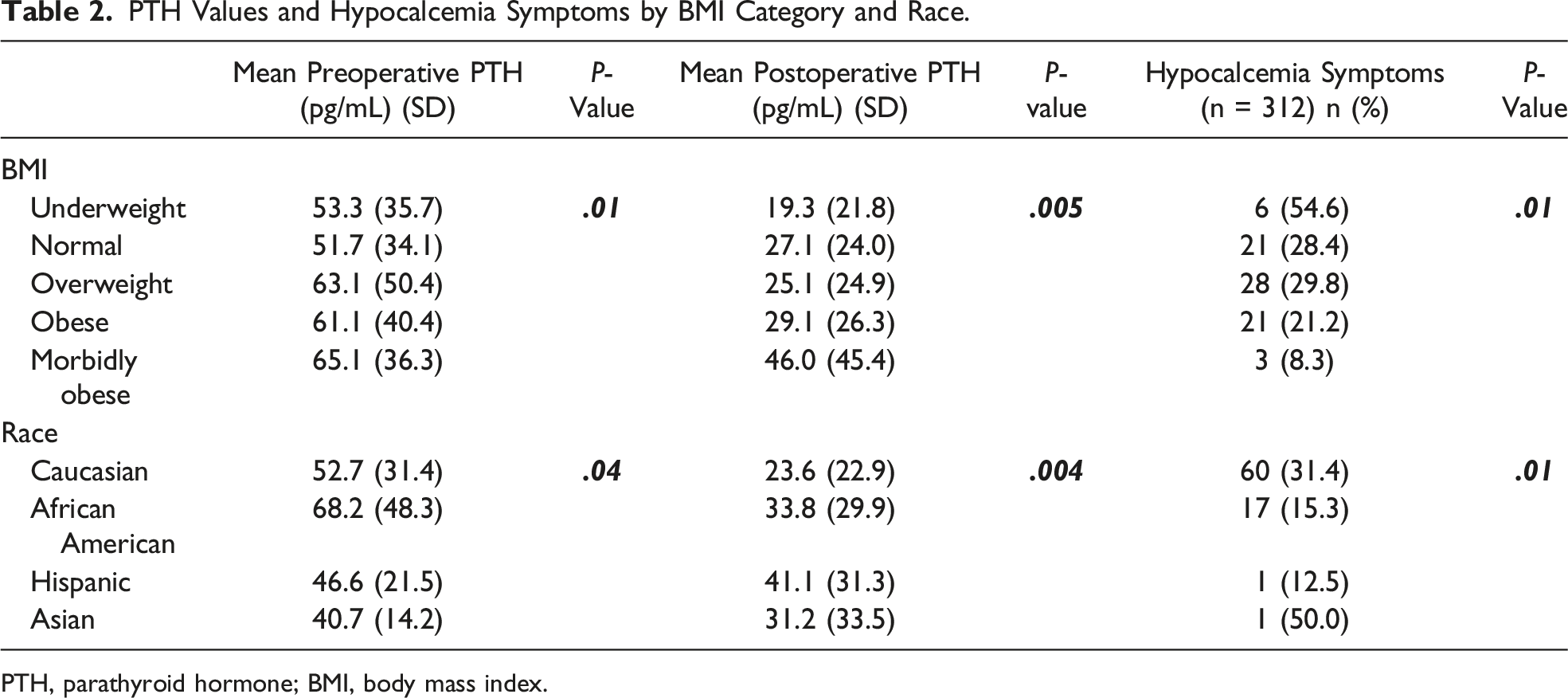
PTH, parathyroid hormone; BMI, body mass index.
The mean number of parathyroid glands visualized during the operation was 3.34 (SD +/− .84) with 53.3% having all 4 glands visualized. One-fifth (n = 71) of patients underwent parathyroid autotransplantation, with 80% (n = 57) of these being a single gland transplanted, 17% (n = 12) with two glands, and 3% (n = 2) with three glands transplanted. Neither the number of parathyroid glands visualized nor autotransplanted were associated with postoperative PTH values (P = .28, .23, respectively). Indication for surgery was also not a significant predictor of postoperative hypoparathyroidism, with it being observed in 37% (n = 10) of patients with an enlarging gland, 31% (n = 27) with nodules, 35% (n = 14) with malignancy, and 26.8% (n = 40) with Graves’ disease (P = .74). On bivariate analysis, examination of individual surgeon influence of postoperative PTH identified no difference in preoperative PTH values (P = .46), but a significant relationship with postoperative PTH (P = .03) was found.
Lower preoperative PTH values were associated with an increased risk of postoperative hypoparathyroidism (P = .01), while increasing BMI was significantly associated with higher mean postoperative PTH levels (19.3 vs 46.0 pg/mL in the BMI < 18.5 vs > 40 group, respectively, P = .001) (Table 2). Examining BMI and PTH as continuous variables identified a weakly positive, statistically significant correlation between BMI and postoperative PTH (rho = .20, P = .01, Figure 1). This relationship was still statistically significant with outliers removed (P = .005). Body mass index category was also significantly associated with patient-reported symptoms of hypocalcemia, with these occurring in over 50% of the lowest BMI group compared to only 8% of the highest BMI group (Table 2). Scatter plot of continuous BMI and postoperative PTH values. PTH, parathyroid hormone; BMI, body mass index.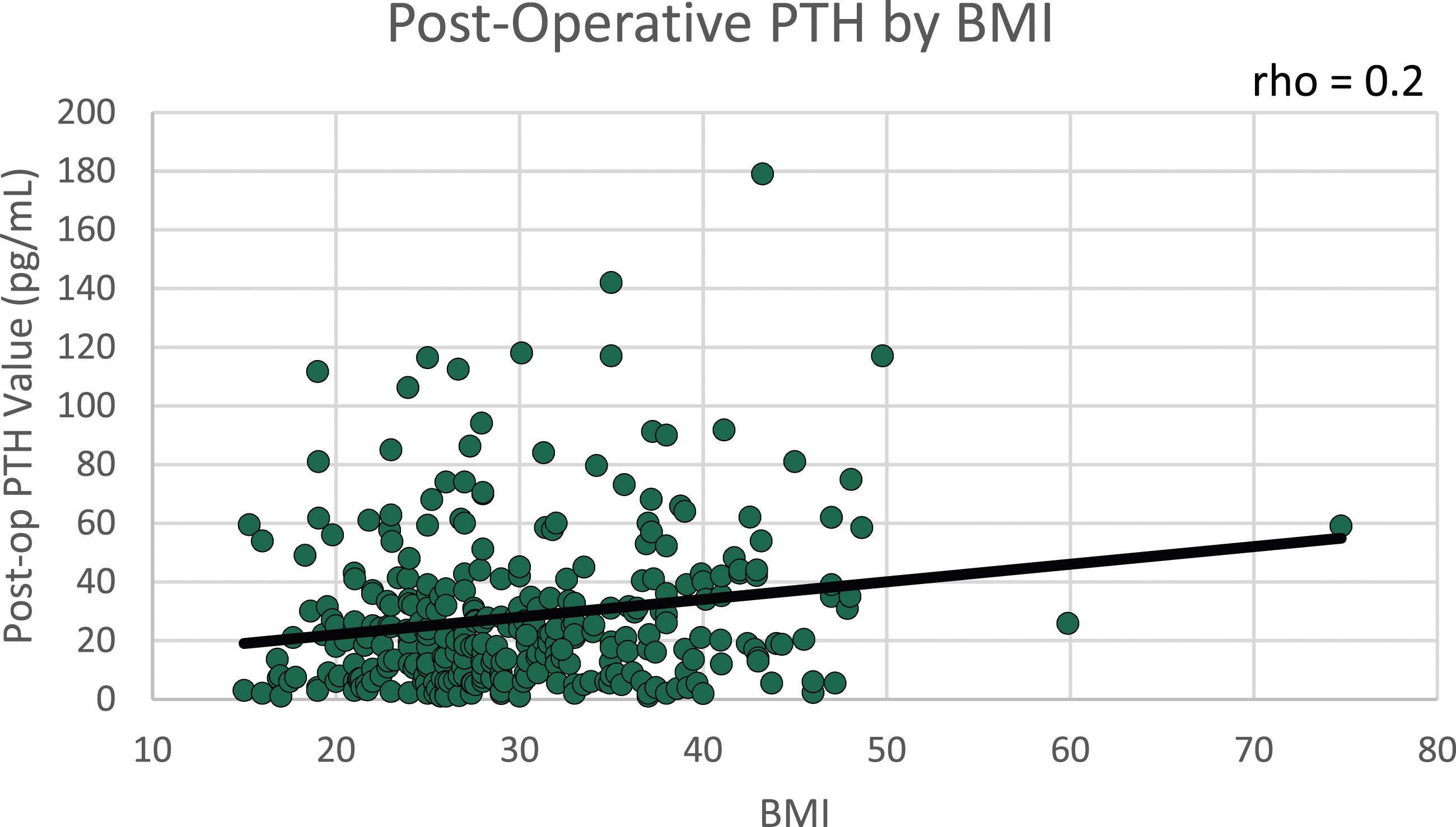
Patient race was significantly associated with preoperative PTH, postoperative PTH, and reported hypocalcemia symptoms (P = .03, P = .004, and P = .012, respectively), with Hispanic and African American patients having higher mean pre- and postoperative PTH values (Table 2). Caucasian and Asian patients were most likely to report hypocalcemia symptoms at 31% and 50%, respectively, though 2 Asian patients had missing hypocalcemia data. Despite the relationship between both BMI and race with postoperative PTH, there was no significant relationship identified between BMI and race themselves to explain the association with postoperative PTH (P = .14).
Multivariable Analysis of Postoperative PTH and Hypocalcemia Symptoms.
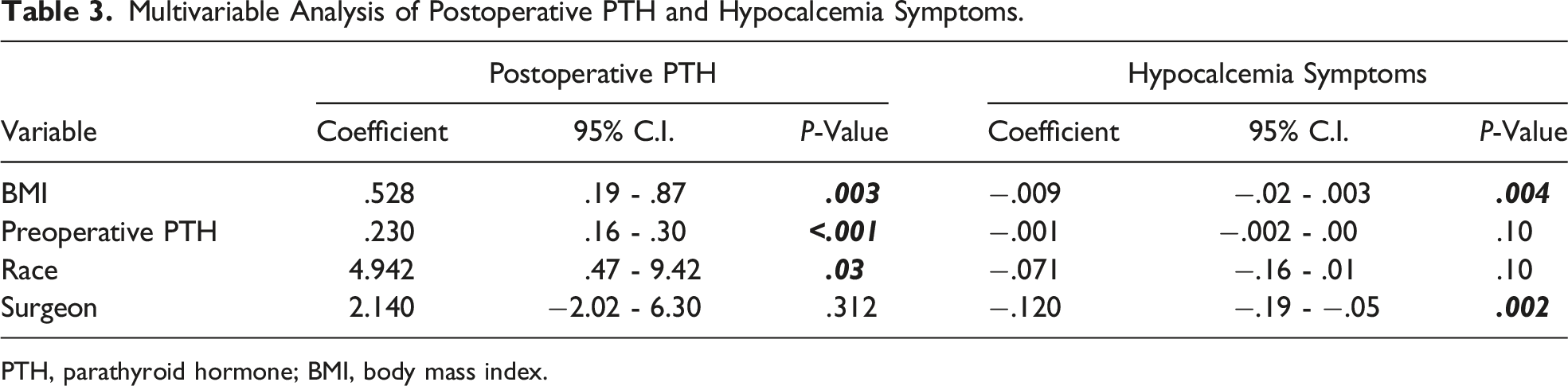
PTH, parathyroid hormone; BMI, body mass index.
Discussion
This study demonstrates a principle known as the obesity paradox—that higher BMI can be associated with a positive health outcome such as in this case, that a higher patient BMI can be protective against postoperative hypoparathyroidism. Our findings demonstrate that increasing BMI was significantly associated with higher mean postoperative PTH levels. This relationship was significant when BMI was analyzed both as a continuous variable as well as when it was grouped into nutritional status groups based on World Health Organization classification. 14 Though no BMI category had a mean postoperative PTH value that was true hypoparathyroidism (PTH < 12), the lowest BMI groups had the lowest average PTH values suggesting they would be at the highest risk of developing postoperative hypoparathyroidism.
The known relationship among BMI, vitamin D, and PTH in the absence of any pathology9,11 is typically explained by higher levels of the adipose tissue that can sequester large amounts of vitamin D, leading to a circulating vitamin deficiency and subsequently raising levels of PTH. 11 Interestingly, our study shows a lack of statistical significance when looking at preoperative vitamin D levels with pre- or postoperative PTH values, though this data point was missing for a large number of patients. While this could be attributed just to missing data, this could also imply that an additional process is leading to the association we have seen in our study between BMI and postoperative PTH values. Remer and colleagues 15 recently noted a related relationship in their study of total thyroidectomy patients showing that obese patients had lower rates of transient hypocalcemia, though they did not specifically study the relationship with PTH. The results shown here counter statements suggesting that a higher BMI can increase a patients’ risk of hypoparathyroidism in the postoperative period. 16
When looking at race, African Americans are also reported to have an increased risk of vitamin D deficiency, which is also associated with an elevation in PTH.17,18 Though our data showed no significance between preoperative vitamin D levels and race, the relationship between race and preoperative PTH was significant with African Americans having the highest mean preoperative PTH values. Race also had a significant relationship with postoperative PTH, with Hispanic and African American patients having the highest means. On multivariable regression, higher preoperative PTH, non-white race, and increasing BMI were all independent predictors of postoperative PTH values. With race and BMI being contributing factors to preoperative PTH levels, it was not surprising that preoperative PTH was also an independent predictor.
Given that hypoparathyroidism is often associated with hypocalcemia, we also assessed the relationship among patient BMI, race, and hypocalcemic symptoms. Since most of our patients are discharged on the day of surgery, we do not have information regarding early postoperative calcium levels, and these data are patient-reported symptoms alone. While it was expected that patients with lower BMI more often reported hypocalcemia, consistent with our findings about low BMI and hypoparathyroidism, we also found hypocalcemia symptoms were not significantly associated with patient race or preoperative PTH level. Since these are patient-reported data, this may be part of the explanation for why surgeon was a predictive factor but race was not—as both of these are social constructs. This is certainly an area in need of further study, particularly given increasing utilization of patient-reported outcomes in the medical literature.
As with all retrospective studies, no causal inferences can be made, and our findings must be interpreted in light of the limitations in methodology. This is a single-center experience with surgery performed by high-volume endocrine surgeons with a relatively small sample size. Selection bias of those chosen for surgery may play a role, though the study included a high percentage of patients with Graves’ disease who have been demonstrated to be at a risk for hypoparathyroidism,19,20 so this may help with elucidation of these relationships. As a retrospective study, some potentially important variables may have been omitted, as well as the sample being whittled down due to missing data points, including that vitamin D levels are not routinely obtained by all high-volume thyroid surgeons at our institution. Preoperative DEXA scans are also not routinely performed in our patient population in an effort to prevent poor utilization of resources and mitigate unnecessary costs. When trying to stratify the significance of BMI to neck surgery, we acknowledge that neck circumference is an important aspect to surgical planning—and potentially to technical difficulty leading to increased risk. We also acknowledge that BMI is not the optimal value for measuring adiposity but was felt to be a more reliable marker than weight alone given that percentage body fat and neck circumference are not measured at our institution. These will serve as important mechanisms to enhance the validity of these findings in future. These considerations all potentially limit the generalizability of study findings.
The thought that higher BMIs offer a protective effect against the development of postoperative hypoparathyroidism could have important clinical implications. Surgeons may need to alter their techniques to enhance parathyroid protection or consider parathyroid autotransplantation more aggressively in patients with lower BMIs. Also, this could influence postoperative calcium protocols so that patients may be weaned more aggressively if they have a higher BMI. This early investigation into the relationship of BMI and postoperative hypoparathyroidism could serve as a roadmap for additional, prospective studies.
Conclusions
Patients with higher BMIs have relative protection from postoperative hypoparathyroidism independent of other factors including surgical indication, number of parathyroid glands visualized during surgery, or vitamin D levels. Preoperative PTH values, BMI, and race were all independent predictors of postoperative PTH levels on multivariable analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
Data were presented at the American Association of Endocrine Surgeons Annual Meeting, Cleveland, Ohio, May 2022.
Study Type
Retrospective cohort study.
