Abstract
Background
Minimally invasive operative approaches for resection and thermal ablation (eg, microwave, radiofrequency) of hepatocellular carcinoma (HCC) have been successfully implemented over the last two decades, although identifying tumors can be challenging. Successfully performing laparoscopic ablation requires real-time visualization and ultrasonography skills for direct placement of the ablation probe.
Methods
In this study, we introduce a novel adjunct to ultrasound imaging for tumors located near or on the surface of the liver via intravenous delivery of indocyanine green (ICG) dye. Non-resectable lesion(s) not amenable to percutaneous ablation were considered for laparoscopic microwave ablation. Each patient initially received a dose of .3125 mg ICG via peripheral IV.
Results
A total of 17 patients were included. There was brisk uptake of ICG throughout the liver parenchyma in under 2 minutes in 15 of 17 patients; the remaining 2 required a second dose of ICG. In 14 cases, a hypo-fluorescent perfusion pattern in the tumor was clearly identified.
Discussion
Integrating ICG and fluorescent imaging provides a complementary adjunct to ultrasound in identifying HCC nodules. While previous applications of ICG typically require injections several days prior to surgery or segmental injections, this study demonstrates a novel real-time application of ICG to aid surgeons with various experiences in laparoscopic-assisted ablation procedures for HCC.
Key Takeaways
• Integrating ICG and fluorescent imaging provides a complementary adjunct to ultrasound in identifying HCC nodules • ICG dye can be used in real-time to aid surgeons with various experiences in laparoscopic-assisted ablation procedures for HCC
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common tumor and the second most common cause of cancer-related death according to the World Health Organization. 1 Over the last 20 years, the incidence of HCC has been rapidly rising in the United States, primarily related to hepatitis C infections and non-alcoholic fatty liver disease (NAFLD). The National Cancer Institute’s Surveillance Epidemiology End Results program estimated that the United States had 40 000 cases of HCC and 27 170 deaths due to HCC in 2016. The incidence of HCC is projected to continue to increase with the ongoing rise in chronic liver disease.1,2
In 2018, the American Association for the Study of Liver Disease evaluated guidelines for the diagnosis and treatment of HCC. Stage T1 and T2 includes a wide range of tumor sizes up to 5 cm in diameter, and the effectiveness of available therapies depends on tumor size, number, and location. Smaller single tumors (<2.5 cm) that are favorably located can be successfully treated with locoregional therapy (eg, resection, ablation), while bilobar tumors or centrally located tumors are usually not resectable due to the underlying chronic liver pathology. 1 The efficacy of ablation is limited for tumors larger than 3 cm, multifocal lesions, or tumors located near major vascular or biliary structures.
Thermal ablation encompasses a broad set of technologies that impact tissue temperatures of the tumor and its surrounding liver parenchyma thus inducing cell death. Heat induction modalities include radiofrequency (RFA) and microwave ablation (MWA).3,4 Since the 1990s, there has been growing enthusiasm for thermal ablation, with early generation devices based on RFA, followed by the development of microwave technologies.5,6 RFA uses a flux of high-frequency alternating current passing through an insulated needle shaft to an unprotected needle tip within the tumor, which dissipates into the surrounding tissue and subsequently generates a rapid vibration of ions in the tissue causing frictional heat. 6 This energy is conducted into the surrounding tissue in a predictable manner resulting in coagulative necrosis. However, the presence of a heat sink can occur near vessels, and this decreases the efficacy of the technique. RFA can also contribute to an increase in the patient’s core temperature, limiting the ability to achieve maximal tissue temperatures. 6 In contrast, MWA creates energy from the collision of rapidly oscillating water molecules at microwave energy levels to release heat and cause degeneration of the surrounding liver tissue. 5 Meta-analysis of RFA and MWA techniques show no significant differences in complete ablation, intrahepatic distant recurrence, or complications, though subgroup analyses show decreased rates of local tumor progression with MWA. 7
When considering minimally invasive or percutaneous approaches for ablation of HCC nodules, image-guidance is critical and may be provided by ultrasound, computed tomography, or MRI.4,5 Laparoscopic assistance has been incorporated as an adjunct in the ablation procedure for patients who are at higher risk for complications due to tumor location or comorbidities, including ascites, severe portal hypertension, or advanced cirrhosis.5,8 In laparoscopic-assisted ablation, the operating surgeon must acquire the skillset to perform intraoperative ultrasonography to identify target lesions which can be challenging as many surgeons have limited training and/or clinical experience with intraoperative ultrasonography.
ICG is a dye with multiple medical applications, including hepatobiliary surgical procedures for both open and laparoscopic procedures. 9 The use of ICG to identify metastatic tumors is based on retention of the dye within the tumor after it washes out from the normal liver parenchyma. This practice requires ICG administration 24-48 hours prior to the surgery which can be impractical for patients with resource limitations. Based on the differential vascular supply of the liver (arterial and portal) compared with tumor nodules (arterial only) we hypothesized that HCC nodules would have an early hypoperfusion pattern compared to the surrounding hepatic parenchyma and we proposed incorporating real-time intraoperative administration of ICG dye as an adjunct to identify tumors in the setting of cirrhosis.
Methods
This study was approved by the University of North Carolina at Chapel Hill School of Medicine Institutional Review Board with a waiver of informed consent. LI-RADS, the Liver Imaging Reporting and Data System, is the standard for diagnosing HCC and LI-RADS 5 is a diagnosis of hepatocellular carcinoma in the setting of cirrhosis. All patients with HCC and referred for locoregional ablation therapy at a single tertiary care center were screened for the presence of superficially located tumors. Patients were evaluated at a multidisciplinary conference where the determination was made that they were not candidates for resection or percutaneous ablation, either secondary to severity of portal hypertension, degree of hepatic dysfunction, or other comorbidities. When the lesion(s) was not amenable to percutaneous intervention due to tumor location on the surface of the liver, near the gallbladder or other visceral organ, or in an area that was otherwise not amenable to percutaneous ablation, they were referred for laparoscopic-assisted MWA and included in this study. Eligibility for the study required that the tumor(s) were in a subcapsular location based on MRI findings. Exclusion criteria were tumors that were located deeper than 1 cm from the surface of the liver or an allergy to iodine, shellfish, or ICG. Patient demographics were obtained from the electronic medical record. Child-Pugh and Model for End-Stage Liver Disease (MELD) scores were calculated to estimate the degree of hepatic dysfunction.
ICG-Guided Laparoscopic Ablative Surgery
All microwave ablations were performed under general anesthesia using a laparoscopic approach. A single 12 mm trocar was placed in an infraumbilical position, and this port was used for the laparoscopic ultrasound probe. One or two 5 mm trocars were placed in the peritoneal cavity to assist with targeting (Figure 1). Port placement for laparoscopic ablation procedures.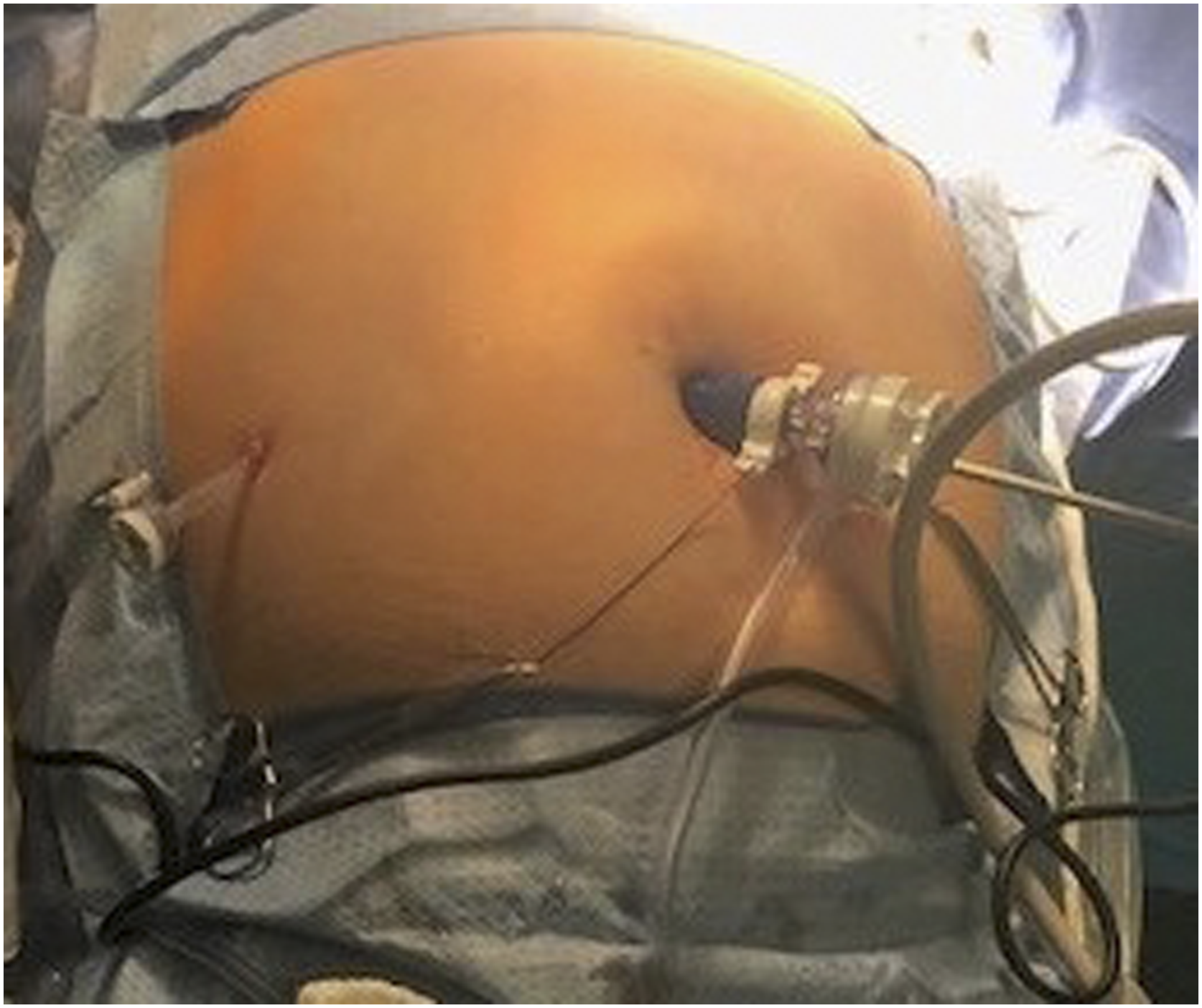
An intra-operative intravenous injection of ICG (Patheon Italia S.p.A.; Hub Pharmaceuticals LLC) was performed by suspending the ICG (25 mg vials) in sterile water creating a concentration of either 1.25 or 2.5 mg/mL stock solution. Each patient initially received a dose of .3125 mg ICG via peripheral intravenous access. Immediately after the dye was injected, fluorescent imaging was performed using the Pinpoint™ (Stryker Corporation, Kalamazoo, MI) fluorescence system for intra-operative laparoscopic imaging. The light source was a light-emitting diode (LED), and the detector was a charge-coupled device (CCD) camera that captured images. The fluorescence signals were sent to a digital video processor for display on a computer monitor. Real-time ICG interpretation was performed by assessing for early signal in the liver parenchyma followed by further perfusion across the liver. Attention was placed on the region of the liver that correlated with the location of the tumor on the preoperative MRI. If after 5 minutes the patient did not have uptake in the liver parenchyma, they received a second dose of .3125 mg ICG with ongoing liver surveillance.
A 2.45 GHz, 14 G microwave antenna (Emprint, Covidien, Boulder, CO) was inserted into the peritoneal cavity through a skin incision. Using .9% NaCl infusion to cool the antenna, the generator was set between 75 and 100 watts (W) for 5-10 minutes, based on the projected size of the ablation zone. Ablations were performed with the aim of creating a margin of approximately 5 mm around the tumor using real-time ultrasonographic monitoring. Additional ablation cycles were performed when the initial ablation zone did not fully cover the tumor. For superficial or exophytic lesions, an initial 5-minute ablation at 50 W was performed to prevent disruption of the liver/tumor capsule, followed by an ablation cycle between 75 and 100 W. Track ablation was performed by setting the ablation device at 100 W while slowly withdrawing the antenna and ablating every centimeter of the needle track for 10 seconds.
Statistical Analysis
Descriptive statistics were used to examine baseline characteristics of participants. Categorical variables were summarized with N (%) and continuous variables were summarized with mean (standard deviation, SD). No additional statistical analyses were conducted with statistical software for this study.
Results
Patient Clinical Characteristics.
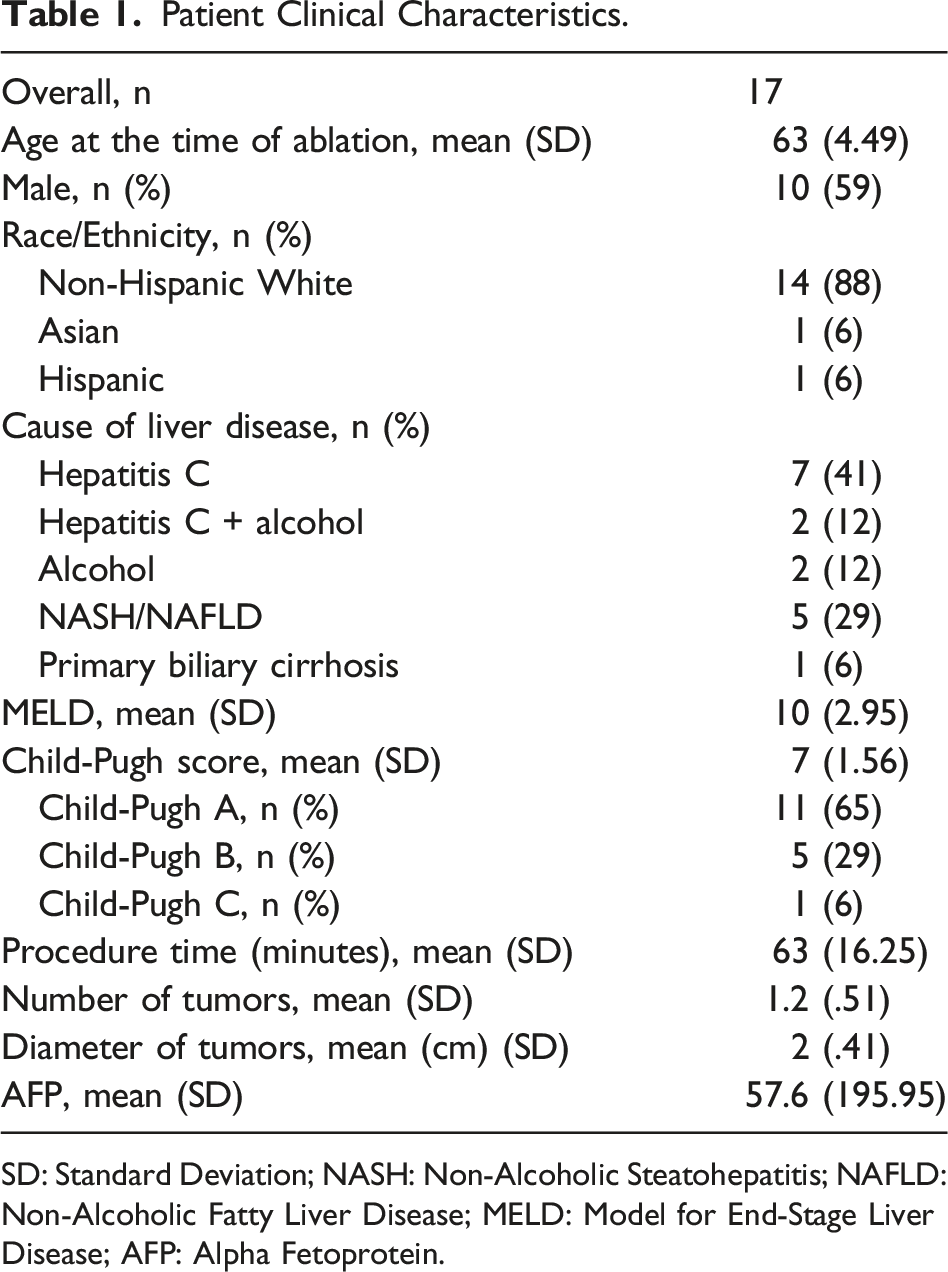
SD: Standard Deviation; NASH: Non-Alcoholic Steatohepatitis; NAFLD: Non-Alcoholic Fatty Liver Disease; MELD: Model for End-Stage Liver Disease; AFP: Alpha Fetoprotein.
All operations were successfully completed, and no perioperative complications occurred. The mean operative time was 63 minutes. Mean tumor diameter was 2 cm and mean number of tumors treated per patient was 1.2. Monitoring of ICG dye perfusion throughout the liver was performed in real-time. Hepatic uptake and tissue perfusion was noted in all patients. There was brisk uptake of the dye throughout the entire liver in under 2 minutes in 15 of 17 patients (88%) while two patients required a second dose of ICG.
ICG dye binds to hepatocytes and we anticipated a hypoperfusion imaging pattern of HCC nodules due to their distinct blood supply compared to the surrounding hepatocytes. Figure 2 is a representative example of a patient with macronodular cirrhosis demonstrating a hypo-fluorescent perfusion pattern of a HCC nodule compared to the surrounding hepatic parenchyma. Based on the hypo-fluorescent pattern detected on the CCD, the surgical team used ultrasonography to further identify the dimensions of the tumor nodule and guide placement of the ablation antenna. In three cases, there was brisk, hyperintense fluorescence throughout the hepatic parenchyma with no hypo-perfusion pattern in the tumor. Intraoperative image demonstrating cirrhotic liver with indocyanine green uptake contrasted with hypoperfusion of hepatocellular carcinoma nodule.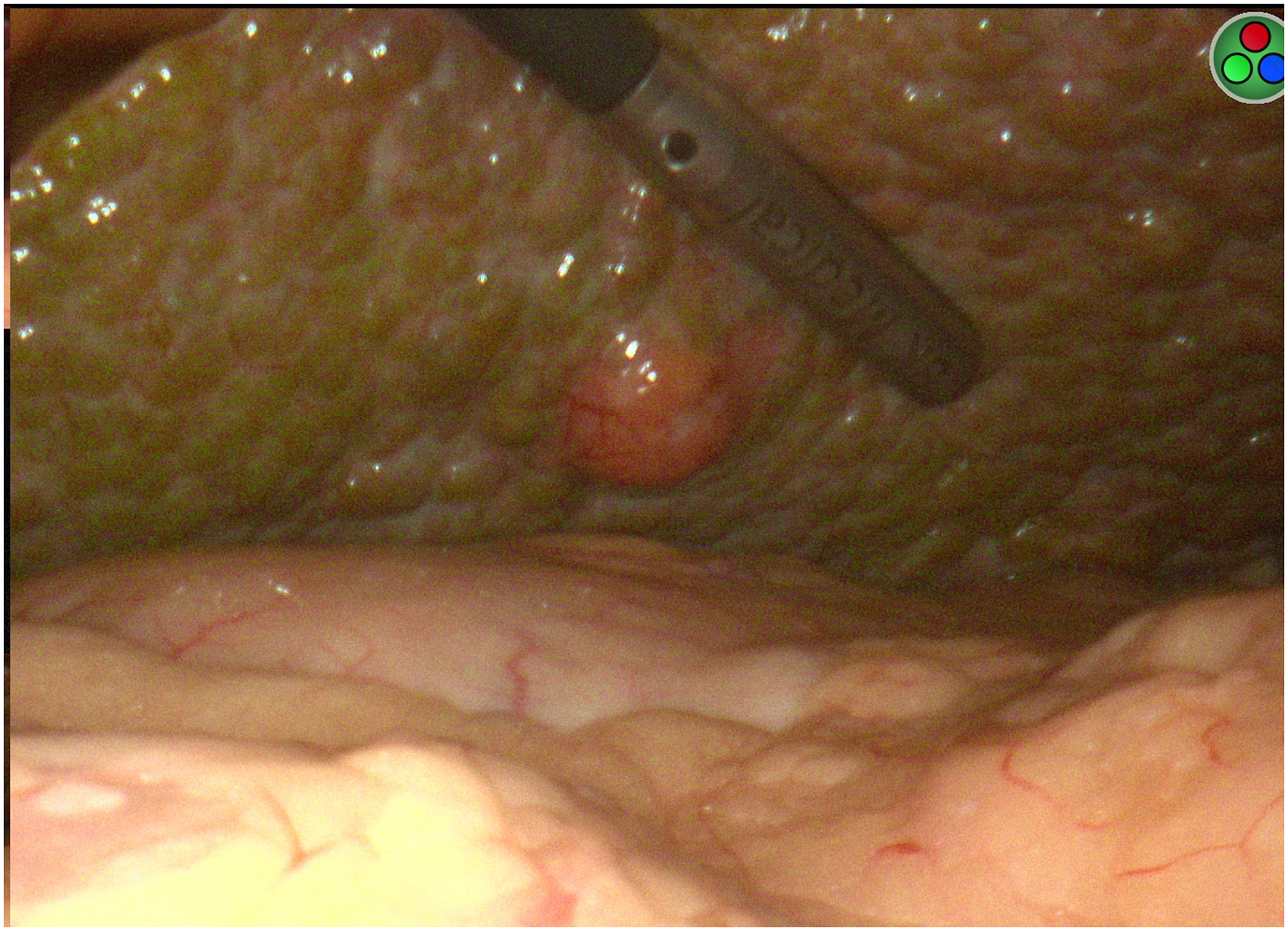
Discussion
This study demonstrates a novel approach of incorporating ICG fluorescence as a real-time adjunct when treating HCC with laparoscopic-assisted ablation. ICG consists of a negatively charged, water soluble polymethine tricarbocyanine dye which binds to albumin in the blood. After ICG binds to albumin, it is taken up by hepatocytes and subsequently excreted in bile. The technique is based on the principle that ICG binds plasma proteins, and that protein-bound ICG emits light with a peak wavelength of about 830 nm when illuminated with near-infrared light.10,11 ICG has been successfully used to identify small, difficult-to-visualize tumor nodules, given the dye is retained in tumor nodules beyond the time from which it has been excreted from normal hepatocytes. 12 ICG dye also has expression patterns based on the morphology and gene expression of the tumors. 13 It was previously demonstrated that poorly differentiated HCC nodules may not accumulate ICG, thus appearing as hypo-fluorescent regions in the liver. 12 Technical limitations of ICG included a tissue penetration depth of ≤10 mm and that published studies were based on administration of dye up to 1 week prior to surgery or required intra-operative segmental portal vein injection, which could be technically challenging.
The adoption of laparoscopic-assistance MWA for liver tumors is an approach that allows visual evaluation of the entire liver without the limitations associated with percutaneous-only approaches for locoregional therapies. With direct ultrasonography and visualization the microwave antenna can be seen entering the liver; this approach allows the surgeon to minimize risk of injury to surrounding organs while safely treating the HCC nodule.3,5,8,14,15 One challenge of laparoscopic-assisted thermal ablation involves identifying the tumor in the setting of a nodular/cirrhotic liver. Patients with macronodular cirrhosis have regenerative nodules throughout the liver parenchyma and identification of the suspicious nodule requires targeted intra-operative ultrasound based on preoperative cross-sectional imaging. The techniques of intra-operative ultrasonography are complex and can be difficult without proper training and familiarity with laparoscopic-assisted ultrasound.
Utilization of ICG dye in cirrhotic livers is not well-established, but several groups have been incorporating ICG dye to evaluate hepatic function in the setting of portal hypertension and liver disease.16,17 In some studies, ICG has been used as an indicator of severe portal hypertension for patients categorized as Childs A or B. 17 ICG has an LD50 (lethal dose) of 50-80 mg/kg and a standard dose of less than 2 mg/kg, making it virtually nontoxic.18,19 ICG is also excreted in the bile, so dosing adjustments are not needed based on a patient’s renal function. The current study takes advantage of the differential uptake and expression of ICG dye in the hepatic parenchyma vs tumor nodules as a means to augment the established approach of laparoscopic-assisted MWA and improve identification of suspicious nodules. This technique is especially valuable for tumors that are partially exophytic as ICG dye expression can help delineate tumor borders. 20
To our knowledge, this is the first study to use real-time ICG in laparoscopic HCC ablation surgery. Limitations of this study include the small sample size and the reliance on a differential hypoperfusion pattern in the tumors following ICG administration. While it could be argued that a superficial tumor can be visualized without an augmented approach, modalities that increase confidence and ready identification of the targeted lesion are very useful in practice.
In conclusion, ICG should be considered as a tool to help distinguish HCC nodules from non-cancerous nodules in a cirrhotic liver. Most applications of ICG require injections several days prior to surgery, necessitating that a patient have an extra visit, or segmentally injecting ICG into a vessel, making it technically challenging for the surgeon. This study describes a novel application that can aid surgeons with variable levels of experience in an effort to achieve complete treatment in a safe and efficacious manner.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The clinical data used to support the findings of this study are available from the corresponding author upon request.
