Abstract
Background
Virtual Reality (VR) is used as an effective tool for distraction and as an adjunct for pain management. This study was conducted to compare VR to standard iPad use after surgery and examine its effect on pain score and opioid consumption.
Methods
This was a randomized controlled study, with stratification by surgery type, age group (7-12yo, 13-18yo) and gender. Pain and anxiety were assessed with validated scales (STAI, FACES, VAS, FLACC) and outcomes were compared between each group.
Results
50 of the 106 enrolled patients used the VR device. After adjusting for age, gender, and STAI, patients had a decreased FLACC score while using the VR device compared to the iPad group (odds ratio 2.95, P = .021). The younger patients were found to have lower FLACC scores while using the VR device (odds ratio 1.15, p=0.044); this finding was most significant when patients used the VR device for 20-30 minutes (odds ratio 1.67, P = .0003). Additionally, after adjusting for treatment group, gender, and STAI, the younger patients had higher odds of withdrawal or exclusion from the study (odds ratio 1.18, P = .021). No significant difference in opioid consumption between the groups was found.
Discussion
Virtual reality was well tolerated and more effective in decreasing pain during the immediate postoperative period than iPad use. Despite a slightly higher withdrawal rate, younger patients benefited more from the intervention.
Introduction
Previous studies have shown that when distraction is combined with medication, postprocedural pain, anxiety, and satisfaction are all improved versus treatment with medications alone.1-3 Immersive distraction can provide an alternative, non-invasive method for pain management through downregulation of pain signaling, redistribution of attention, concentration, and emotional investment, redirecting neural signals and decreasing the patient’s perception of pain and anxiety, such as in dressing changes in pediatric patients.4,5 Underlying this relationship between anxiety and pain is the mechanism of anxiety modulation of pain perception, which can promote hyperalgesia. 6 A small retrospective study showed that VR use reduced pain and opioid need in a select population, 7 but this and other VR studies have had limited scopes of eligible procedures and small sample sizes.
No prior study has examined VR in the immediate postoperative period. The fully immersive environment of VR facilitates a feeling of presence in the virtual world, 8 disrupting awareness of and sensitivity to stressors. The use of VR has been suggested to be particularly effective in the pediatric population, as the younger demographic often becomes more engaged and captivated by imaginary spaces. 9 Virtual reality is an economically feasible and familiar option for children, with a variety of applications that target a range of interests. 10 A VR application with a peaceful environment, calming audio, and slow-paced movements was intentionally chosen for this study to minimize adverse effects such as dizziness and nausea. Nature Treks VR (Greener Games Ltd.; Shropshire, England) fulfilled these criteria with its wide array of calming environments for immersive exploration. 11 In this study, we hypothesized that the use of the VR would be an effective method to improve pain scores and reduce opioid consumption after surgery, observed in a postoperative anesthesia care unit (PACU).
Methods
Participants
This study was reviewed and approved by the Institutional Review Board (IRB) of the University of North Carolina at Chapel Hill (IRB number 18-0658). Participants were English-speaking patients ≤18 years old undergoing pediatric surgery at UNC Children’s Hospital. UNC Children’s Hospital is a quaternary academic medical center performing over 9000 anesthetics per year at its main campus. Enrolled patients were undergoing one of four surgeries: general (including urologic and plastic), spine, other orthopedic, or burn (<10% total body surface area). Patients were excluded if they had a history of seizures, developmental delay, uncorrected vision or hearing loss, head or neck surgery, or with chronic pain requiring the daily use of opioids for more than two weeks prior to the procedure. Study personnel consisted of four research assistants who met regularly to review the processes to decrease variability with study participant interactions.
Procedures
Consent
Study personnel approached patients and their families in the surgical waiting room to determine interest and eligibility. If interested, the remainder of the conversation and consent process was conducted in a private room. Informed consent and assent were then obtained from the guardian and patient.
Randomization
Following consent, patients and guardians completed baseline measures about preprocedural pain and anxiety. Patients self-reported pain levels using the Wong-Baker FACES Pain Rating Scale (FACES) 12 and anxiety using the Spilberger State Trait Anxiety 6-question Short Form (STAI) scale. 13 Guardians completed a measure of the patient’s pain using the Visual Analog Scale (VAS). 14 Last, the study personnel completed a Face, Legs, Activity, Cry, Consolability (FLACC) assessment. 15 Standard pre-op vitals were collected, and patients were randomized by block randomization in REDCap, a secure web-based platform for building and managing online databases and surveys. The randomization scheme was stratified by surgery type and age (7-12 yo and 13-18 yo). Patients were assigned to one of two arms: the Standard of Care (SOC) device (iPad with preloaded applications) or the VR device. Patients and guardians were unaware of the other arm of the study until PACU discharge. After randomization, patients were allowed 5-10 minutes to familiarize themselves with the operations of their assigned device prior to surgery.
Outcome Measures
The primary endpoint of the study was patient- and caregiver-reported pain scores. The validated scales were repeated in the PACU to assess patient anxiety and pain prior to the intervention. Upon sufficient alertness, the device was presented to the patient. During device use, standard vital signs were collected and repeated in 10-minute intervals concurrently with FLACC assessment, for a total of 30 minutes (Baseline FLACC, FLACC1, FLACC2, and FLACC3). If a patient discontinued use of the device prior to the end of a 10-minute interval, vital signs and a FLACC assessment were collected at the latest point in the interval. Qualitative outcomes were collected through patient and caregiver surveys at the completion of the intervention.
Intervention
Device Utilization
Study personnel facilitated device use upon emergence from anesthesia and completion of postoperative pre-intervention assessments. To avoid potential unwanted side effects such as nausea and dizziness, both devices were vetted by the UNC Child Life team and limited to 30 total minutes of use. Patients could stop using the device at any point during the intervention and could elect to resume using the device at any point within 30 minutes from the beginning of the intervention. Patient refusal of the device in the PACU three times, or delayed emergence from anesthesia after PACU arrival in excess of 90 minutes, resulted in mandatory withdrawal. Upon study completion, patients and guardians completed a survey about their experience with their assigned device and general feedback was solicited.
Devices
Standard of Care
Patients at our institution are offered an iPad 5 (Cupertino, CA), or newer, for postoperative distraction. These tablet devices are preloaded with educational games approved by UNC Child Life Specialists to appeal to a wide range of ages and interests.
Virtual Reality
The Oculus Go (Irvine, CA) headset was preloaded with the Nature Treks VR application, which provides immersive audio-visual environments to explore with different settings ranging from outer space to the deep sea. 10 The funding for the headsets and the application was provided by the UNC Department of Anesthesiology Research Department. Of note, the iPad, Oculus Go, and the Nature Treks VR application are not FDA approved for this purpose, as was outlined and disclosed in the IRB application.
Follow-Up
Follow-up extended through a 10-day period, consisting of a survey administered to guardians or caregivers, over the phone or by email, both 2-3 days and 7-10 days postoperatively. This was done with the Post Hospitalization Behavior Questionnaire for Ambulatory Surgery (PHBQ-AS) survey, a validated questionnaire used to assess post-hospitalization behavioral changes in children. 16
Statistical Analysis
The software used for data analysis was R version 3.6.0. The chi-square test was used for all categorical variables. ANOVA was used for numeric variables, and the Wilcoxon rank-sum test was used for the PHBQ-AS analysis.
Results
Patient Demographics
Patient Demographics.
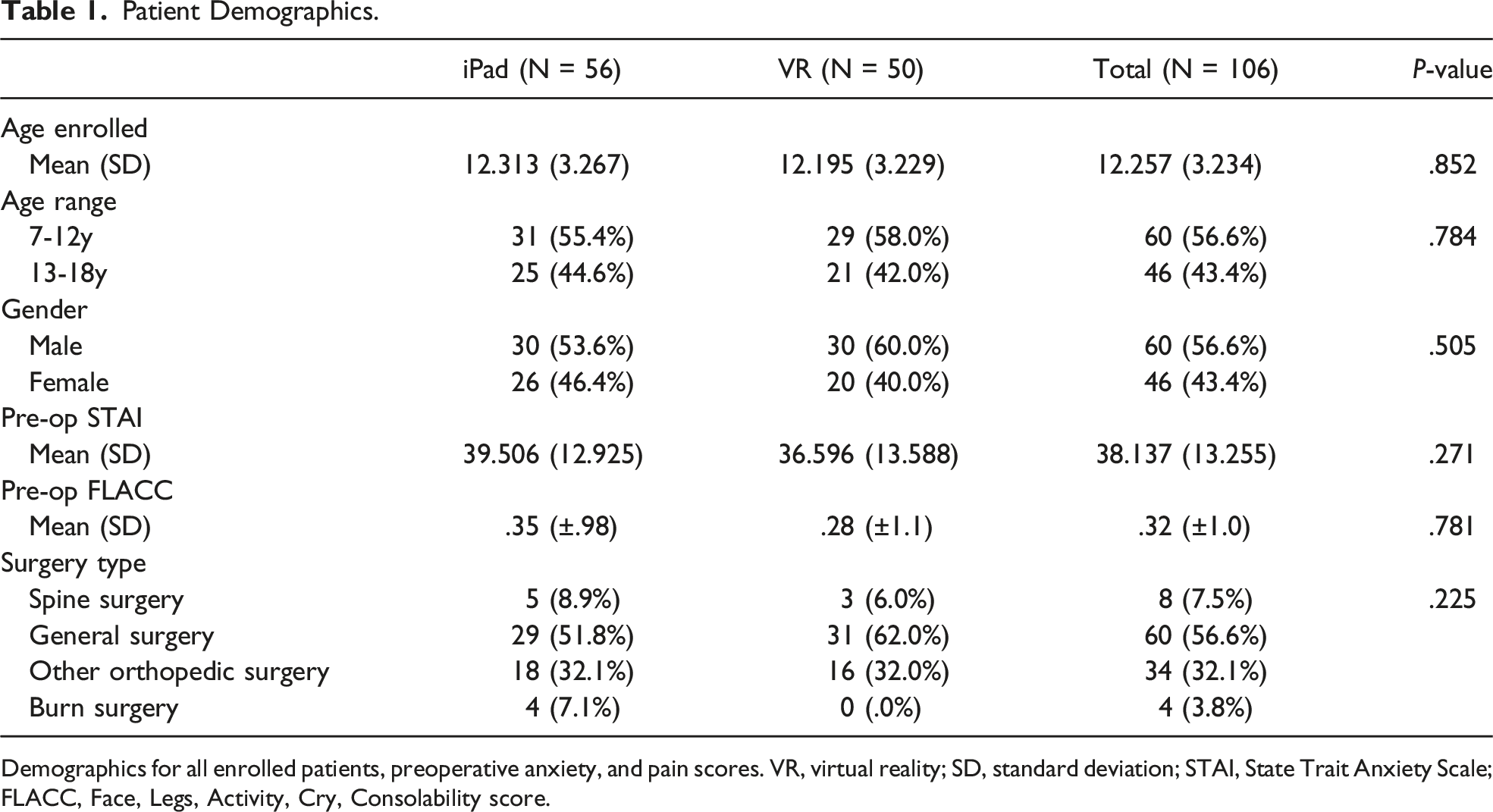
Demographics for all enrolled patients, preoperative anxiety, and pain scores. VR, virtual reality; SD, standard deviation; STAI, State Trait Anxiety Scale; FLACC, Face, Legs, Activity, Cry, Consolability score.
Postoperative
Study Completion Data Without Stratification.
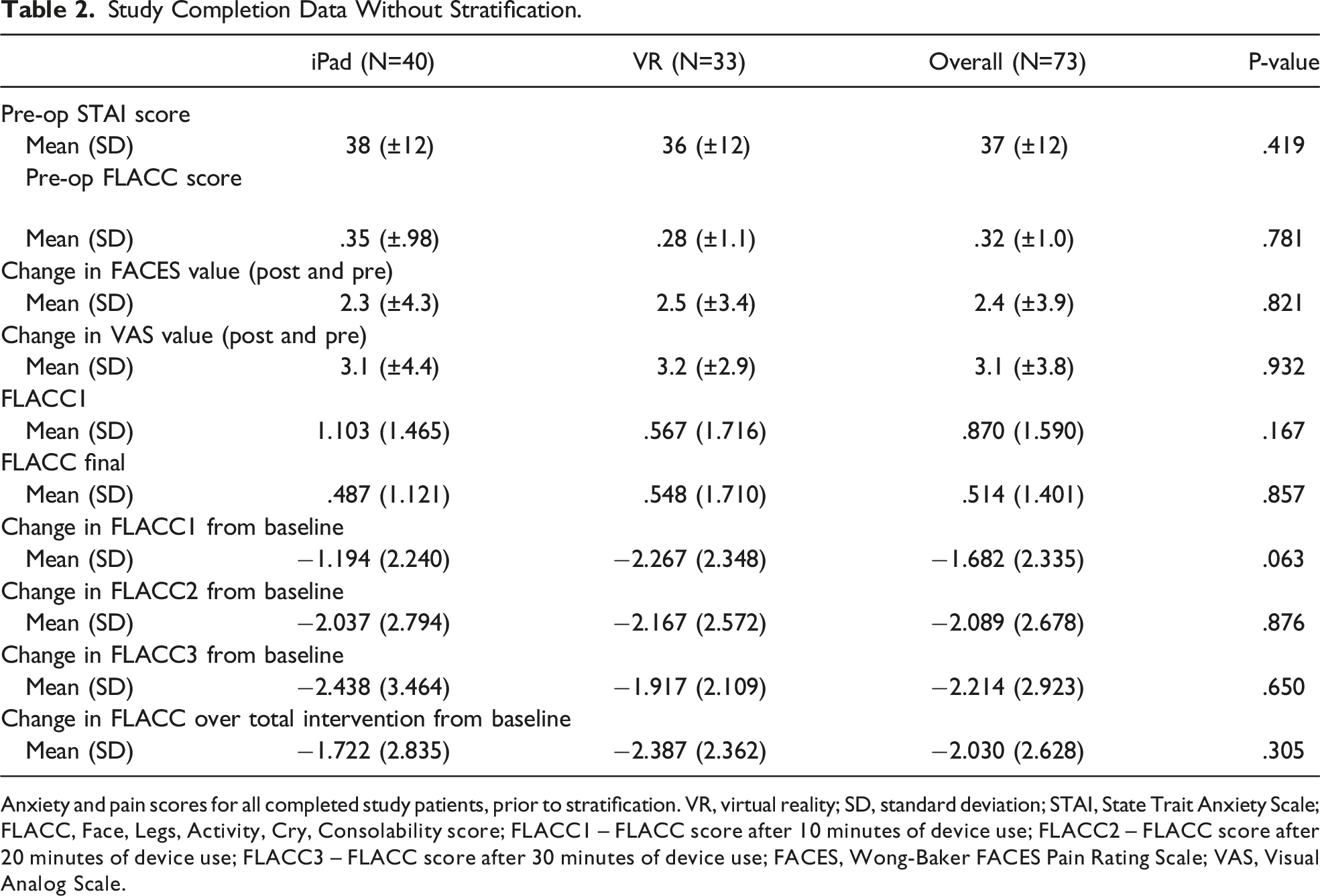
Anxiety and pain scores for all completed study patients, prior to stratification. VR, virtual reality; SD, standard deviation; STAI, State Trait Anxiety Scale; FLACC, Face, Legs, Activity, Cry, Consolability score; FLACC1 – FLACC score after 10 minutes of device use; FLACC2 – FLACC score after 20 minutes of device use; FLACC3 – FLACC score after 30 minutes of device use; FACES, Wong-Baker FACES Pain Rating Scale; VAS, Visual Analog Scale.
Opioid Use.
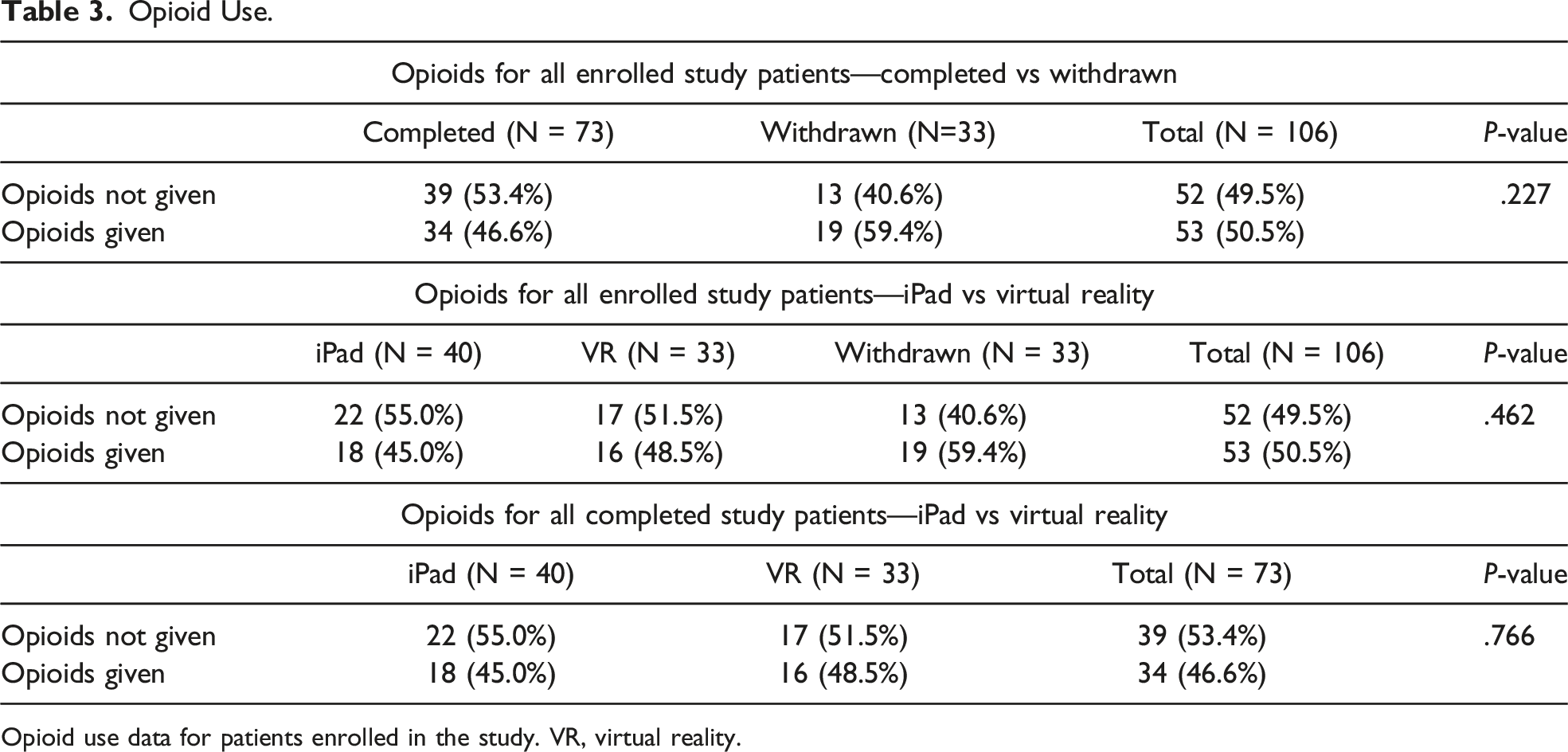
Opioid use data for patients enrolled in the study. VR, virtual reality.
FLACC Score
Regression Analysis: Adjusted Odds Ratios and P-Values.
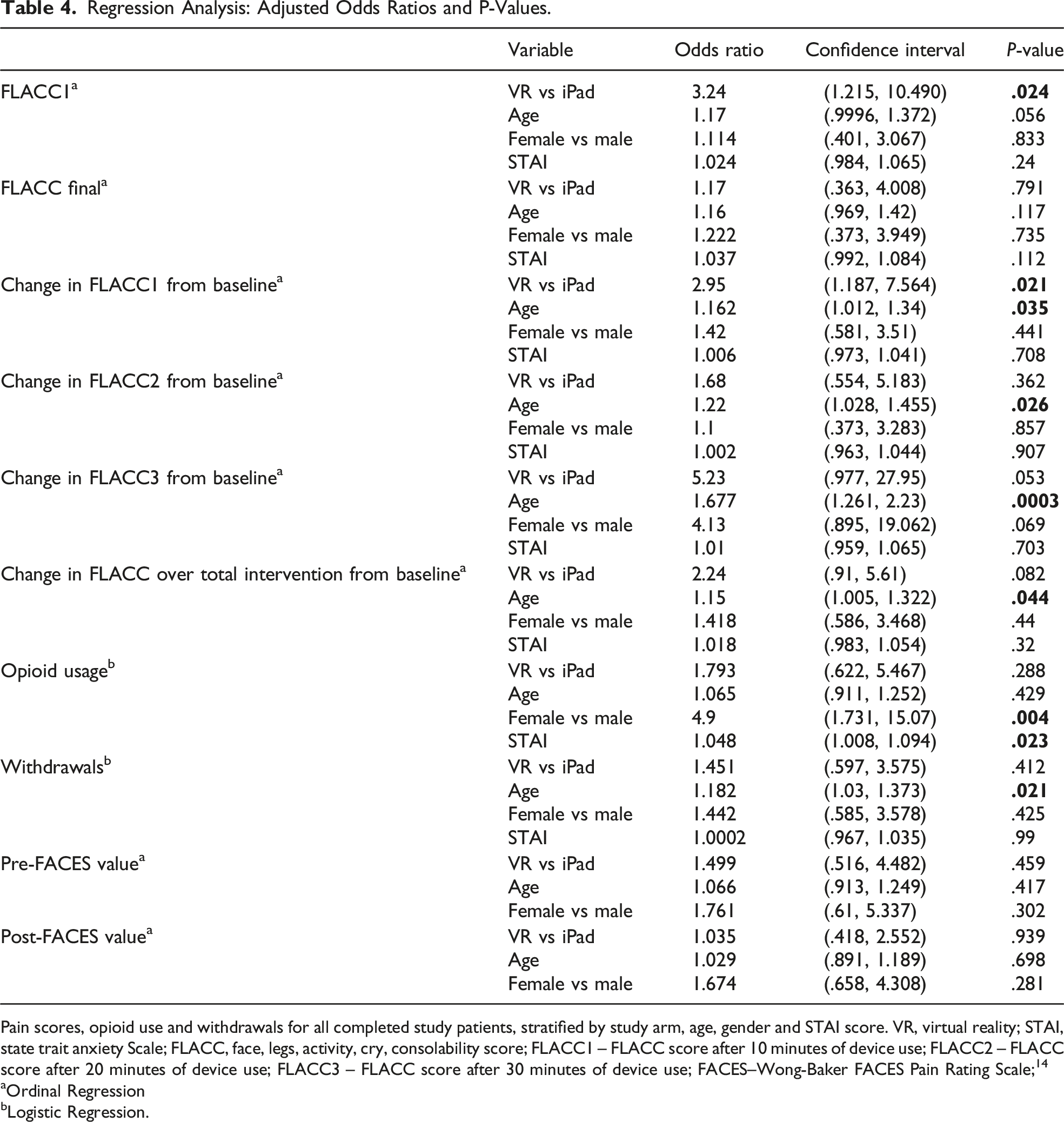
Pain scores, opioid use and withdrawals for all completed study patients, stratified by study arm, age, gender and STAI score. VR, virtual reality; STAI, state trait anxiety Scale; FLACC, face, legs, activity, cry, consolability score; FLACC1 – FLACC score after 10 minutes of device use; FLACC2 – FLACC score after 20 minutes of device use; FLACC3 – FLACC score after 30 minutes of device use; FACES–Wong-Baker FACES Pain Rating Scale; 14
aOrdinal Regression
bLogistic Regression.
Withdrawals
There was no statistically significant difference in withdrawal percentage between the two study arms. After adjusting for treatment group, gender, and STAI, the odds of withdrawal for younger patients are 1.18 that of older patients (P = .021; CI, 1.0 to 1.4; Table 4). While difficult to track, there seemed to be great variability in nursing and caregiver desire to engage and encourage younger patients to start using the devices, compared to older patients. Parents of the younger patients seemed more eager to make decisions about length of use of the devices based on subjective discomfort or enjoyment. For patients that had prolonged emergence from anesthesia, data were unable to be gathered, thereby excluding them from the analysis.
Gender
Patients were able to ask for pain medications, but to help minimize subjectivity, opioid medications were offered to all patients with pain scores >4 (0-10 scale), FLACC score >4 (0-10 scale), or FACES score >2 (0-10 scale), correlating with moderate-to-severe pain, which are routine criteria for postoperative as-needed opioid administration orders. There were no statistically significant differences when comparing age, gender, or STAI between the two groups, and there were no significant differences in pain scores between males and females.
Post Hospitalization
The PHBQ-AS score at 72 hours showed no difference in iPad and VR group (P = .409). Unfortunately, 74 patients were lost to follow-up in the 2-3-day follow-up period and 86 were lost to follow-up in the 7-10-day period. Despite this, there was a significant statistical difference in the PHBQ-AS score between the groups at 7-10 days (P = .006).
Discussion
The VR headset was more effective in decreasing pain scores in the PACU during the immediate postoperative period. In this cohort, younger patients benefited more from intervention and saw the greatest and most significant decrease in FLACC score when the device was used for 20-30 minutes. The increased odds of withdrawal for younger patients suggests that despite evidence that younger patients benefit more from using VR in the immediate postoperative period, more barriers exist for them to consistently use the device. This outcome highlights the importance of guardian and nursing engagement in facilitating non-pharmacological methods in the PACU. The study team collected qualitative data for each intervention, and it was widely documented that children were more receptive to engaging with the device when paired with guardians and providers who reminded and encouraged them to do so.
While variability of parental and nurse engagement is a limitation of this study, it reflects the varied engagement that would occur if VR were translated to standard of care in a PACU. Due to the broad inclusion and exclusion criteria, as well as the decision to not standardize the medical team or caregiver contact in the PACU, these data model a hypothetical implementation of VR as distraction therapy in a large, high-flow pediatric PACU. Due to the relatively large size of the randomized cohort, this variability should not skew distribution of data in either arm.
There was no significant difference between the two arms for length of PACU time until readiness for discharge, measured from initial Aldrete score until bedside nursing evaluation of an Aldrete of 9 or 10. 17 No adverse events were reported during any phase of this study. Because many patients were lost to follow-up, these results only represent a small fraction of the total enrollment population. Despite this, the lack of adverse events in the 10-day post-discharge period provides important insight into the safety of using VR in the immediate postoperative period. This highlights the potential need for further, and more focused, exploration of the utility of VR postoperatively in pediatrics.
The Centers for Disease Control reports that half of opioid overdoses are a result of prescription medications. 4 This potential was supported by prior research that suggested the use of VR in tandem with analgesics is a more effective method of pain management than analgesic methods alone, in the pediatric acute burn patient demographic. 18 Implementation of VR as a clinical distraction intervention can be beneficial for patient care by enhancing the clinical experience through the reduction of procedural anxiety and pain perception, but additional follow-up studies need to be done to determine if VR use can help to decrease the overuse of opioids postoperatively.
Limitations
Randomized trials typically account for bias and other confounding factors. Given that our study did not have a power calculation for determining the most appropriate study size, and that this study was prematurely ended due to COVID-19 (106 enrolled, goal of 150), bias may have been introduced at various stages of the study. The study was voluntary, so self-selection bias may have influenced enrollment. High anxiety patients and families may have been less likely to electively participate in this study. The self-reporting of anxiety and pain scores may vary by child, given their previous exposure to pain, developmental upbringing, and several confounding social factors. Since assessing and measuring anxiety and pain levels was part of data analyses, this may have presented influencing factors to the sample population. For these concerns, the VAS was given to guardians (pre-op and post-op) to more accurately determine and confirm self-reported scores of the patients. Also, the FLACC assessment collected by the research assistants uses objective, well-defined categorical pain scales.
Multiple research assistants conducted the study, and despite all having received the same study preparation and training, this may have introduced bias at any stage of the research. Similarly, the study did not control which nurses received the research patients upon entry to PACU. Anxiety and pain can be altered by a patient’s comfort within their clinical environment and care providers, thereby influencing our results. However, this study attempted to reduce biases by proper training of nurses and research assistants.
Given this study’s broad scope, we could not determine which surgery subset benefits most from the intervention. We currently have a more focused investigation underway for adolescents with idiopathic scoliosis undergoing posterior spinal fusion, comparing the VR device to SOC and evaluating pain scores and opioid use postoperatively. Future investigations may require a limited group of trained nurses caring for patients undergoing a single type of surgery.
Footnotes
Acknowledgments
We would like to acknowledge the UNC Child Life Team, as they were instrumental in providing support for our research assistants. We also would like to recognize the UNC Department of Anesthesiology Research Department for the financial and statistical support provided.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
