Abstract
Toward the goal of monitoring activity of native mammalian promoters with molecular imaging techniques, we stably transfected DU145 prostate carcinoma cells with a fusion construct of enhanced green fluorescent protein (EGFP) and wild-type herpes simplex virus-1 thymidine kinase (HSV1-TK) as a reporter gene driven by the promoter for human elongation factor 1α (EF-1α-EGFP-TK). Using this model system, expression of EGFP was quantified by flow cytometry and fluorescence microscopy, while the HSV1-TK component of the reporter was quantified with 8-[3H]ganciclovir (8-[3H]GCV). As analyzed by flow cytometry, passage of EGFP-TK-DU145 transfected cells (ETK) in vitro resulted in populations of cells with high and low expression of EGFP over time. High and low ETK cells retained 23-fold and 5-fold more GCV, respectively, than control. While differences in uptake and retention of GCV corresponded to relative expression of the reporter gene in each subpopulation of cells as determined by both flow cytometry (EGFP) and quantitative RT-PCR, the correlation was not linear. Furthermore, in high ETK cells, net retention of various radiolabeled nucleoside analogues varied; the rank order was 8-[3H]GCV < 9-(4-fluoro-3-hydroxymethylbutyl)guanine ([18F]FHBG) ≈ 8-[3H]penciclovir (8-[3H]PCV) < 2′-fluoro-2′-deoxy-5-iodouracil-beta-
Introduction
Mouse models offer well-controlled experimental systems for studying normal and abnormal development and physiology. However, investigating gene expression in these models frequently requires histological analyses, precluding studies in which patterns of gene regulation in the same animal are monitored over time and/or in response to various stimuli. Thus, the kinetics, magnitude, and localization of gene expression remain poorly characterized in living animals, limiting our understanding of gene regulation in vivo.
Recent studies have shown that reporter genes can be detected in vivo with a variety of imaging modalities including radionuclide techniques with single-photon emission computed tomography (SPECT) or positron emission tomography (PET) [1–6], magnetic resonance (MRI) [7], [8], and optical approaches such as luminescence and near-infrared fluorescence (NIRF) [9], [10]. Because radiotracer methods offer tomographic localization, sensitive detection, and quantification of signals in living animals, SPECT or PET commonly have been used to interrogate gene expression in vivo, using enzymes such as herpes simplex virus-1 thymidine kinase (HSV1-TK) or receptors such as the dopamine-2 receptor as reporter molecules. Imaging of HSV1-TK typically has used model systems in which expression of the reporter gene is controlled by strong, constitutive viral promoters, such as cytomegalovirus (CMV) and Rous sarcoma virus (RSV). Although these systems have provided important “proof-of-principle” data for imaging gene expression with PET reporter systems, these viral promoters do not reproduce the strength and dynamic regulation of most mammalian promoters. Ultimately, a goal of molecular imaging is to detect, localize, and quantify in vivo changes in expression of reporter genes that are regulated by endogenous mammalian promoters.
Towards this goal, we constructed a reporter gene in which expression is driven by the promoter for human elongation factor 1α (EF-1α). EF-1α is a cofactor in eukaryotic protein synthesis and a component of the actin cytoskeleton. Likely an effector in metastasis, overexpression of EF-1α at the level of both protein and mRNA has been correlated with increased metastatic potential in mammary adenocarcinoma [11]. The promoter for EF-1α also has been exploited for stable expression of transgenes in T lymphocytes in the context of gene therapy [12]. To allow detection of a dual-function reporter gene by both radiotracer and fluorescence imaging techniques, we engineered a variant fusion protein of HSV1-TK and enhanced green fluorescent protein (EGFP) controlled by the EF-1α promoter. In DU145 prostate cancer cells stably transfected with the reporter gene (ETK cells), expression of EGFP-TK mRNA was quantified by RT-PCR (TaqMan), and function of the enzyme was detected with a radiolabeled nucleoside analog both in vitro and in vivo. Differential expression of EGFP fluorescence from the reporter gene within subpopulations of cells was also detected by flow cytometry, allowing us to analyze the relationship between amounts of mRNA and both activities of the fusion reporter construct. This model system established that activity of a human promoter can be detected and quantified in cultured cells and in mice using an EGFP–HSV1-TK reporter gene.
Materials and Methods
Cell Culture, EGFP–HSV1-TK Plasmid, Transfection
DU145 human prostate cancer cells were cultured in Earle's MEM, heat-inactivated 10% fetal bovine serum, 1% glutamine, and 0.1% penicillin/streptomycin in a 5% CO2 incubator at 37°C. Complementary DNA for HSV1-TK was amplified from the pPNT plasmid (gift of R. Mulligan) by polymerase chain reaction (PCR) using forward and reverse primers engineered to contain BsrGI and NotI sites, respectively (underlined): 5′-CTAGTA
DU145 cells were transfected with EF-1α-EGFP-TK using Superfect (Qiagen), and stable transfectants were both isolated (after 6–8 weeks) and maintained in media containing 0.25 μg/ml puromycin. Expression of EGFP in clonal cell lines was determined by fluorescence microscopy, and the clone with the highest initial fluorescence (ETK) was expanded and used for subsequent experiments.
Quantitative RT-PCR (TaqMan) Analysis
EGFP-TK mRNA levels were determined using the TaqMan 7700 sequence detection system (PerkinElmer Life Sciences) as we have described previously [14]. Sequences of RT-PCR primers (recognizing the TK component of the fusion transcript) were 5 -TGACAAGCGCCCAGATAACA-3′ and 5′-GCATGTGAGCTCCCAGCC-3′.
Flow Cytometry
Expression of EGFP-TK in transfected cells was determined by fluorescence-activated cell sorting (FACS; flow cytometry). Cells were harvested by trypsination, resuspended in growth medium, pelleted by centrifugation, and resuspended in phosphate-buffered saline with 1% serum. Cells were stored on ice in the dark until FACS analysis. Relative levels of EGFP were analyzed with a FACScan flow cytometer (Becton Dickinson) using a total of 10,000 events per sample. Prior to selected experiments (see Results), living ETK cells were sorted into populations with high and low expression of EGFP using the protocol described above to harvest cells, except that after sorting, cells were resuspended in normal growth medium.
Fluorescence Microscopy
DU145 cells in tissue culture were fixed and examined for expression of EGFP by fluorescence microscopy as described previously [15]. For analysis of EGFP in tumor xenografts, tumors were excised and snap-frozen prior to cryosectioning. Tumor sections were fixed in 4% paraformaldehyde before fluorescence microscopy.
Radiochemicals
8-[3H] Ganciclovir (8-[3H]GCV; 20.0 Ci/mmol), 8-[3H]penciclovir (8-[3H]PCV; 14.9 Ci/mmol), and 2′-fluoro-2′-deoxy-5-iodouracil-beta-
Radiotracer Accumulation Assays
Cellular accumulation of 8-[3H]GCV, 8-[3H]PCV, 2-[14C]FIAU, and [18F]FHBG was determined as previously described for radiotracer assays in cultured cells, except that incubations were performed in normal growth medium in a 5% C02 incubator at 37°C [18]. Radiotracers were typically present in the medium at 0.2–7 μCi/ml. Time periods of uptake and washout of radiotracer are indicated in the figures or legends. Data for cellular content of the nucleoside analogues were expressed as fmol (mg protein)−1 (nMo)−1, where nMo represents concentration of nucleoside in the extracellular space.
Tumor Xenografts and Biodistribution Studies
ETK and parental DU145 tumors were grown by injecting 3 × 106 and 2.5 × 106 cells of each cell line, respectively, into the right or left flanks of 6- to 7-week-old NCr nude mice (nu/nu; Tac: Cr: (NCr)-Hfhll nu ) (Taconic, Germantown, NY) as described previously [19]. Biodistribution studies with 8-[3H]GCV were performed 4 to 5 weeks after inoculation of cells. 8-[H]GCV (2 μCi) was injected intravenously by tail vein, and animals were sacrificed 1 hr after injection. Accumulation of radioactivity in the blood, heart, and excised tumors was determined by liquid scintillation counting as described previously [20]. Content of radiotracer in tumors and tissues was determined as percent injected dose (% ID) per gram and were expressed as ratios of radioactivity in tumor to blood or heart [21].
Statistical Methods
Data pairs were compared with Student's t test. Values ≤.05 were considered significant.
Results
We fused EGFP with HSV1-TK to create a dual-function reporter gene for monitoring gene expression with both fluorescence and radiotracer techniques. As a model system for detecting activity of an endogenous mammalian promoter, the reporter molecule was placed under control of the constitutively active EF-1α promoter, creating EF-1α-EGFP-TK. DU145 prostate cancer cells were transfected with EF-1α-EGFP-TK and were selected with puromycin to derive a stable clonal cell line (ETK) for use in further validation of both the EGFP and HSV1-TK components of the reporter gene. For all subsequent experiments, a frozen stock of ETK cells was thawed and expanded for 1 week prior to in vitro analysis.
We characterized the reporter gene by determining levels of EGFP by FACS in parental DU145 and ETK cells. Initially, a single population of ETK cells could be identified showing a high expression of EGFP compared with parental DU145 cells. However, after maintaining the transfected cells in culture under continuous selection for 6 weeks, two distinct populations of cells were detected by FACS analysis of EGFP. One population retained high levels of EGFP while expression of the reporter gene was reduced significantly in the second group of cells (Figure 1). Similar patterns of biphasic decay of expression of transgenes have been reported previously and may be due to loss of insert copies, methylation of promoter DNA, and/or deacetylation of histones [22], [23]. Thus, these data suggested that the two levels of expression could be exploited to characterize and validate the EGFP-TK reporter gene in relation to cellular subpopulations over time.

Expression of EGFP in DU145 (A) and ETK (B) cells after 6 weeks in culture. DU145 and ETK cells were analyzed for expression of EGFP by FACS, with relative amounts of EGFP on the x-axis versus cell number on the y-axis. Compared with parental DU145 cells, two predominant populations of ETK cells with high and low relative amounts of EGFP were identified.
To establish that activity of the EF-1α promoter could be detected with HSV1-TK, we incubated early-passage ETK cells and parental DU145 cells with 8-[3H]GCV, a nucleoside analog that is phosphorylated by viral but not endogenous mammalian TK. Molecules of GCV that are phosphorylated by HSV1-TK remain trapped within cells [24]; the nucleoside is not retained significantly within cells that lack the heterologous enzyme. ETK and parental cells were incubated with 8-[3H]GCV and cell-associated radiotracer was quantified at various time points throughout 2 hr (Figure 2). The content of GCV in ETK transfectants was significantly greater than control cells after 60 min of incubation with radiotracer and continued to increase at subsequent time points. After 2 hr, ETK cells accumulated approximately 2.5-fold more radiotracer than control DU145 cells (p < .001).

Uptake of 8-[3H]GCV in DU145 (⧫) and ETK (▪) cells. Cells were incubated with 8-[3H]GCV and cell-associated radiotracer was determined at various times between 30 and 120 min. Cell content of GCV in ETK transfectants was significantly greater than in DU145 cells at 60, 90, and 120 min. Each point represents n = 4 with error bars denoting ± SEM when larger than the symbol.
However, intravenous injection of radiolabeled nucleosides in vivo produces a rapid peak and subsequent decline in extracellular concentration of radiotracer as the nucleoside is cleared from blood [5]. Thus, continuous incubation of cells in vitro with constant amounts of extracellular GCV does not reproduce the situation in vivo. To more closely mimic in vivo changes in extracellular concentrations of radiotracer, we modified the in vitro protocol to include a subsequent incubation in media without 8-[3H]GCV to simulate both uptake and washout pharmacokinetic phases. We hypothesized that this protocol would amplify final differences in accumulation of GCV between ETK transfectants and parental DU145 cells because incubation without radiotracer should allow efflux of nonphosphorylated GCV from cells. Therefore, DU145 cells and ETK cells were incubated with 8-[3H]GCV for 2 hr and then transferred to radiotracer-free medium for various times (Figure 3). Prior to incubation in radiotracer-free medium, cells expressing the reporter gene contained only 1.6-fold more GCV than control. However, cell content of radiotracer rapidly decreased in DU145 cells during the washout period. Cell-associated GCV decreased by 85% in control cells after 30 min in radiotracer-free medium, and 95% was lost by the 1-hr time point. By comparison, content of GCV in ETK cells declined by only 12–25% during 2 hr of incubation without radiotracer. ETK transfectants had approximately 23-fold more GCV than control DU145 cells after the full 2-hr washout period (p < .001). These data demonstrate that differences between ETK and control cells are significantly enhanced by including a washout period in the assay, consistent with phosphorylation and trapping of GCV in cells that express the EF-1α-EGFP-TK reporter gene.

Retention of 8-[H]GCV in DU145 (⧫) and ETK (▪) cells. Cells were preincubated with 8-[3H]GCV for 2 hr, followed by incubation in radiotracer-free medium for various times as indicated (0 time represents the beginning of the washout period). GCV rapidly washes out of control DU145 cells, while most of the drug remains associated with ETK cells throughout the washout period. Each point represents n = 4 with error bars denoting ± SEM when larger than the symbol.
Using an uptake and washout protocol, we then investigated if cell accumulation of GCV in vitro was dependent on concentration of nucleoside analog in the extracellular medium. Cells were incubated for 30 min with 8-[3H]GCV at concentrations of GCV between 1 and 125 nM, followed by a 1-hr washout period without radiotracer (Figure 4). Data are presented as {fmol (mg protein)−1 (nMo)−1}−1 versus (nMo) which is essentially a Hanes–Woolf plot ([substrate]/velocity vs. [substrate]; [25]) wherein fmol (mg protein)−1 represents the velocity of TK-dependent cell uptake of nucleoside over 30 min and (nMo) represents substrate concentration in the extracellular spaces. In both ETK transfectants and parental DU145 cells, TK-dependent uptake of GCV was linearly dependent on extracellular GCV concentration (a flat Hanes–Woolf plot), although net content of nucleoside was approximately 20-fold greater in ETK than control cells at all concentrations (p < .025) (Figure 4). Thus, at the tested concentrations, no evidence was observed for saturation of the overall process of transport and phosphorylation of GCV in these cells. Note that data presented as fmol (mg protein)−1 (nMo)−1, when divided by cell water space [μl (mg protein)−1], yield an intracellular-to-extracellular ratio of nucleoside concentration at a given point in time. For typical cell volumes (~5μl (mg protein)−1 [26]), mean in-to-out ratios of 0.6 and 0.03 were calculated for ETK and control DU145 cells, respectively (30 min uptake and 60 min washout protocol). Similarly, in-to-out ratios of 1.1 and 0.04 were calculated for ETK and control DU145 cells, respectively, after a 60-min uptake and a 120-min washout protocol (Figure 3), indicating that the reporter gene was capable of concentrative tracer accumulation.
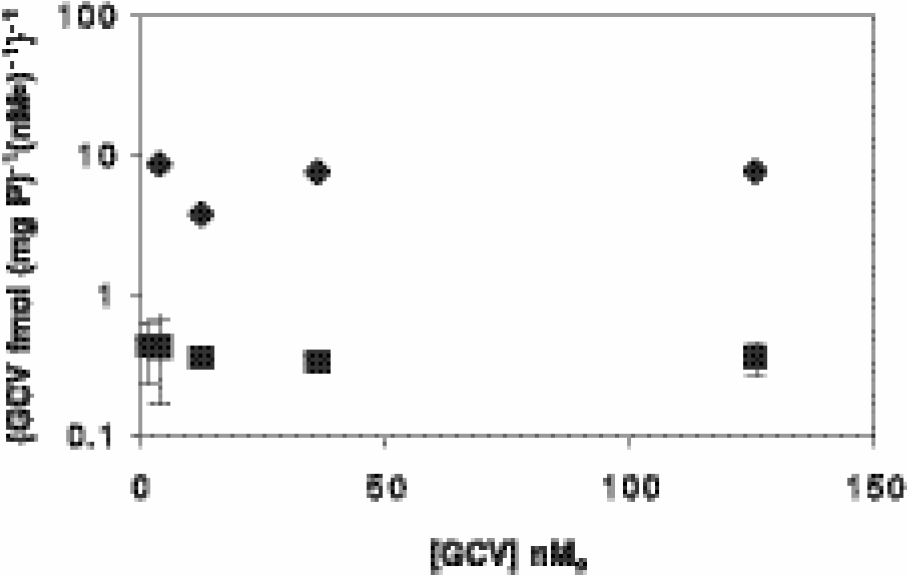
Hanes – Woolf plot ([substrate]/velocity vs. [substrate]) of the effect of extracellular concentration of GCV on net cell content of radiotracer. DU145 (⧫) and ETK (▪) cells were incubated with increasing concentrations of 8-[3H]GCV for 30 min, followed by 1 hr of incubation in radiotracer-free medium. Final cell-associated GCV content increased in direct proportion to extracellular concentrations of GCV (flat line) for both cell lines. Note that data are plotted on a log-linear scale. Each data point represents n = 4; bars represent ± SEM when larger than the symbol.
Exploiting the slow loss of reporter gene expression in a subpopulation of cells over time, we used FACS to sort ETK cells that had been cultured for 6 weeks after thawing into subpopulations with high and low levels of GFP. Relative expression of the reporter gene in each subpopulation of ETK cells and control DU145 cells was determined by quantitative RT-PCR (TaqMan). As expected, parental DU145 cells had negligible expression of EGFP-TK mRNA, while ETK-high cells expressed approximately 2-fold more mRNA for the reporter gene than ETK-low cells (Figure 5A). These same subpopulations of cells were then incubated with 8-[3H]GCV to measure activity of the HSV1-TK component of the reporter gene. Cells were incubated with radiotracer for various periods of time, followed by a 1-hr washout in medium without radiotracer to determine net retained GCV (Figure 5B). At all time points, the subpopulation with high expression of EGFP-TK accumulated significantly more GCV than cells with low levels of the reporter gene (p < .001). The maximum difference in accumulation of GCV between subpopulations of ETK cells was approximately 7-fold. GCV content in the low ETK cells was significantly greater than control only for uptake times greater than 1 hr. Dividing the slopes of retained tracer over time by cell water space yielded net uptake rate constants (apparent kin) of 0.032, 0.0048, and 0.0009 (min)−1 for high ETK, low ETK, and DU145 cells, respectively. While amounts of EGFP and function of HSV1-TK did not linearly correlate with expression of the reporter gene at the level of mRNA, the rank order of both reporter components always correlated with mRNA and thus, could be used to correctly identify relative differences in promoter activity and gene expression in this system.

Expression of EF-1α-EGFP-TK and accumulation of radiolabeled nucleoside analogues in subpopulations of ETK cells. ETK cells were sorted into populations with low and high amounts of EGFP relative to control cells. (A) Relative amounts of mRNA for EGFP-TK in DU145, ETK (low), and ETK (high) cells were determined by quantitative RT-PCR (TaqMan) analysis. (B) DU145 (⧫), ETK (low) (▴), and ETK (high) (▪) cells were incubated with 8-[3H]GCV for various periods of time, followed by a 1-hr chase in medium without radiotracer. Cell-associated radiotracer increased in populations of cells with relatively greater expression of the reporter gene. (C) Cell-associated radiotracer in DU145, ETK (low), and ETK (high) cells incubated with 8-[3H]GCV, 8-[3H]PCV, [18F]FHBG, or 2-[14C]FIAU for 1 hr followed by a 1-hr washout in medium without radiotracer. Data represent n = 4 for each time point; error bars represent ± SEM when larger than the symbol.
Several nucleosides have served as substrates for the HSV1-TK reporter system in vitro and in vivo [27]. To begin to characterize the DU145 model system in relation to these substrates, cells were incubated with various nucleoside analogues (8-[3H] GCV, 8-[3H] PCV, [18F]FHBG, and 2-[14C]FIAU) for 1 hr followed by a 1-hr washout in medium without radiotracer to determine net retained tracer (Figure 5C). Under this protocol, none of the tracers showed clear distinctions between control cells and low ETK cells, but all the tracers revealed readily observable enhanced retention in high ETK cells. For high ETK cells, FIAU showed the greatest uptake; in-to-out ratios were 1.3, 3.2, 2.2, and 11.1, respectively, for GCV, PCV, FHBG, and FIAU. However, the specific activity of 2-[14C]FIAU was the lowest among these test substrates.
To determine if the EF-1α-EGFP-TK reporter gene would function in vivo, we used a freshly thawed population of ETK cells to produce xenograft tumors in flanks of nu/nu mice. Parental DU145 cells were injected into the contralateral flank of each animal as a control. When palpable tumors were detected, we quantified biodistribution of 8-[3H]GCV in tumors and selected tissues 1 hr after tail vein injection of radiotracer. One hour after systemic tracer administration, accumulation of 8-[3H]GCV in ETK tumors was approximately 2.3-fold greater than in control DU145 tumors (p ≤ .001) (Figure 6A). The ratios of radioactivity in tumor relative to blood or heart were 1.4 ± 0.2 and 2.8 ± 0.2, respectively, for ETK tumors, and 0.5 ± 0.1 and 1.1 ± 0.1, respectively, for control DU145 tumors (p ≤ .001). We also qualitatively assessed expression of the reporter gene in tumor xenografts by fluorescence microscopy (Figure 6B). In ETK tumors, heterogeneous distribution and intensity of EGFP were observed throughout. Although foci of cells with bright fluorescence from EGFP were detected, most of the transfected cells did not express EGFP above background autofluorescence present in control DU145 cells. This expression pattern of EGFP in tumor specimens was consistent with the progressive extinction of the reporter gene that was observed in vitro (Figure 1 and Ref. [23]).

In vivo function of EF-1α-EGFP-TK reporter gene. (A) DU145 and ETK (high) cells were injected into flanks of nu/nu mice, and biodistribution of 8-[3H]GCV in 4–5-week tumor xenografts was determined 1 hr after intravenous injection of radiotracer. Net content of radiotracer per gram of tumor tissue was significantly greater in tumors expressing the reporter gene (n = 3). (B) ETK tumors were excised and analyzed for expression of EGFP by fluorescence microscopy (40x). Heterogeneous expression of EGFP was detected in the tumor specimen.
Discussion
In this study, we have established that activity of the promoter for human EF-1α can be detected both in vitro and in vivo using a fusion protein of EGFP and HSV1-TK. Although other fusions of EGFP and HSV1-TK have been described for molecular imaging or gene therapy [28], [29], previous investigations generally have used viral promoter and enhancer elements to control expression of the EGFP-TK fusion protein [29], [30]. Thus, the current data represent a first step toward detecting native mammalian promoters with HSV1-TK in living animals and humans. The combined EGFP–HSV1-TK reporter gene offers advantages of both optical and radiotracer techniques for monitoring promoter function in vivo. Using appropriately labeled nucleoside analogs, such as [18F]FHBG [17] or [124/131I]FIAU [1], enzymatic activity of HSV1-TK can be detected in vivo with PET or SPECT. Both imaging modalities allow three-dimensional localization of reporter genes, while PET provides the additional benefit of quantifying absolute accumulation of radiotracer in vivo with improved spatial and dynamic temporal resolution [31]. By including EGFP in the fusion gene, the reporter molecule can also link directly with techniques for detecting and localizing protein expression by fluorescence microscopy in vitro as well as in real time in vivo [32], [33]. Indeed, it was recently reported that activity of an artificially constructed cis-acting p53-specific enhancer containing 15 tandem repeats of paired p53 binding sites could be monitored in vitro and in vivo with a different HSV1-TK–EGFP reporter [34]. The relation between this artificial construct and imaging the endogenous enhancer element remains to be investigated.
Previous studies have quantified in vitro activity of HSV1-TK reporter genes by incubating cells in the continuous presence of radiolabeled nucleoside analog [5], [35]. Using this assay system, we detected only small differences in GCV content between ETK transfectants and control DU145 cells during the uptake period. However, differences between the cell lines were significantly increased when a subsequent washout period in isotope-free medium was included in the assay. After nucleoside transporter-mediated uptake into cells [36], GCV is converted to mono-, di-, and triphosphate forms by HSV1-TK and downstream kinases, resulting in trapping of phosphorylated metabolites within cells that express the reporter enzyme. Note that if nucleoside transporter-mediated uptake were rate limiting relative to the enzymatic turnover of HSV1-TK, then nearly all cell-associated radioactivity would represent phosphorylated nucleoside and little washout would be observed. On the other hand, if transport exceeded HSV1-TK capacity, then a sizable pool of radiolabeled, but nonphosphorylated, nucleoside would be present at the end of the uptake period, poised to efflux from the cells when placed in isotope-free medium. We postulate that inclusion of a washout period in the biochemical assay allowed nonphosphorylated nucleoside to be transported out of cells and focused on metabolites that were phosphorylated and retained within cells. Similar but variable washout trends have been observed with all tested nucleosides (data not shown). We also propose that this protocol more closely reproduces experimental conditions in vivo, in which intravenous bolus administration of a nucleoside analog produces a peak with a subsequent decline of extracellular radioactivity.
For the EF-1α promoter, we observed progressive extinction of the reporter gene in a subpopulation of these cells over time. As a result, two subpopulations of cells with differing amounts of mRNA for the reporter gene were produced. The mechanism of this extinction process was not studied, but we were able to use these subpopulations to show that differences in activity of a human promoter could be detected and monitored qualitatively and quantitatively with fluorescence and radiotracer techniques, respectively. Although mRNA levels for the reporter gene did not linearly correlate with protein amounts or activity in this model system, nonetheless, mRNA levels did correspond with the rank order of function of the EGFP-TK dual reporter. While these data differ from previous work with HSV1-TK as a reporter gene [5], discordance between levels of mRNA and protein has been reported in many other studies of reporter genes and endogenous proteins [37], [38]. Although these nonlinear results demonstrate a potential limitation of in vivo imaging of gene expression, curve fitting of data and nonlinear modeling of relationships could nonetheless enable quantification. In addition, as the field of molecular imaging progresses to analyzing changes in promoter activity in response to defined stimuli and interventions, appropriate controls will be needed to ascertain that changes in function of the reporter gene are due to experimental intervention and not to independent changes in promoter function. Alternatively, the phenomenon of extinction of a reporter transgene could be exploited to investigate mechanisms of silencing and chromatin remodeling in vivo.
Data from the current study demonstrate that further advances in reporter genes and radiotracers will be necessary to improve imaging of gene expression in vivo. Using reporter genes based on HSV1-TK, enzymes with enhanced phosphorylation of nucleoside analogs, such as described by Black et al. [39], have been shown to increase sensitivity for imaging by approximately 2-fold [40]. Potentially, mutants of HSV1-TK that confer even greater intracellular accumulation of radiolabeled nucleoside analogs may be identified through screening of additional clones. In preliminary reports, strength of the signal from reporter genes has also been amplified successfully using systems in which a promoter of interest is linked in trans to expression of a reporter molecule [41], [42]. Innovations in design and chemical synthesis of radiolabeled substrates for HSV1-TK can enhance sensitivity for imaging either alone or in combination with various mutant TK enzymes. In this regard, no consensus has been reached as to the optimal probe–reporter pair. For example, net uptake in vitro of [18F]FHBG is reported to be significantly higher than 8-[18F]PCV or [14C]FIAU in rat C6 cells transfected with wild-type or mutant sr39 HSV1-TK [43]. However, other investigators report that uptake of [124F]FIAU in vitro and in vivo is higher than [18F]FHBG or 9-[(3-fluoro-1-hydroxy-2-propoxy)methyl]guanine ([18I]FHPG) in RG2 cells or T1115 human glioblastoma cells stably transfected with wild-type HSV1-TK [27], [44]. In our DU145 transfectants containing wild-type HSV1-TK fused to EGFP, the rank order of net uptake was 8-[3H]GCV < [18F]FHBG
In summary, data from the current study demonstrate that a combined EGFP–HSV1-TK reporter can be used to identify and quantify function of a native human promoter by radiotracer and fluorescence techniques. Although activity of the reporter gene could be detected in vitro and in vivo, the signal-to-background ratio for the reporter gene decreased significantly in living animals. Studies are currently in progress to further improve probes and reagents for molecular imaging of gene expression, thus allowing expansion of the dynamic range of biological hypotheses that can be investigated in vivo.
Footnotes
Acknowledgments
This work was supported by NIH grants P20 CA86251 and P50 CA94056 (D.P.-W.) and the Association for the Cure of Cancer of the Prostate (J.M.). We thank Michael Welch and colleagues in the Washington University Research Resource for Isotope Research (R24 CA8630702) for production of 18F.
